Abstract
Mammary analogue secretory carcinoma (MASC) of the salivary gland is a rare tumor that was first described by Skalova et al in 2010, and since then, only a few hundred cases have been reported in the literature. Prior to Skalova’s report, MASC was histologically misclassified as acinic cell carcinoma (ACC), pleomorphic adenoma, mucoepidermoid carcinoma, or adenocarcinoma, not otherwise specified. Mammary analogue secretory carcinoma has a low incidence rate overall, accounting for less than 0.3% of all salivary gland tumors. Histopathologic and cytogenic analysis of MASC is identical to secretory carcinoma of the breast, leading to the proposed name by Skalova. The purpose of this case presentation is to describe an atypical presentation of MASC, to compare this case with the classic description of MASC, and to contrast the various features of MASC to ACC in order to improve the accuracy of future diagnoses and help guide treatment.
Keywords
Introduction
Mammary analogue secretory carcinoma (MASC) of the salivary gland is a rare tumor that was first described by Skalova et al in 2010, and since then, only a few hundred cases have been reported in the literature. 1 Prior to Skalova’s report, MASC was histologically misclassified as acinic cell carcinoma (ACC), pleomorphic adenoma, mucoepidermoid carcinoma, or adenocarcinoma, not otherwise specified.2-4 Mammary analogue secretory carcinoma has a low incidence rate overall, accounting for less than 0.3% of all salivary gland tumors. 5 Histopathologic and cytogenic analysis of MASC is identical to secretory carcinoma of the breast, leading to the proposed name by Skalova. 1 The purpose of this case presentation is to describe an atypical presentation of MASC, to compare this case with the classic description of MASC, and to contrast the various features of MASC to ACC in order to improve the accuracy of future diagnoses and help guide treatment.
Case Report
The patient provided written informed consent for his information and images to be published. A 21-year-old male presented to the otolaryngologist due to a bleeding lesion on the buccal mucosa of the upper lip near the base of the gingiva. He described that the lesion started as a small pea-sized mass and eventually grew to a size that impeded speech and lip movement. The mass was present for 5 years before the patient received further evaluation (Figure 1). The patient was otherwise healthy and had no significant past medical, family, or social history.
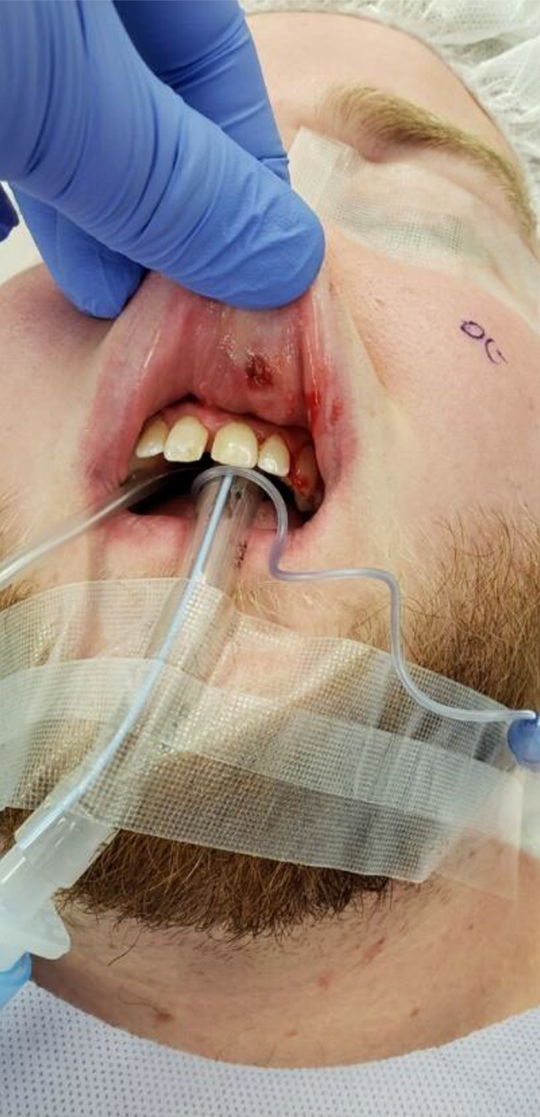
Enlarging mass on buccal mucosa of the upper lip near the base of the gingiva.
Punch biopsy of the tumor was taken and sent for analysis. Diagnosis of MASC was made via histology and immunohistochemistry. Histological analysis of the tumor showed an infiltrative neoplasm (Figure 2) composed of epithelioid cells with abundant vacuolated eosinophilic cytoplasm (Figure 3). Cystic and tubular structures were present with deposition of eosinophilic luminal material and mucin production (Figure 4). Many cells showed clear nuclei; however, cells with prominent central nucleoli were also seen. Immunohistochemistry performed showed that lesional cells were positive for S-100, CK7, SOX-10, GATA-3, mammaglobin, and mucicarmine (Figure 5). Tumor cells were negative for p63, CD68, and TTF-1.
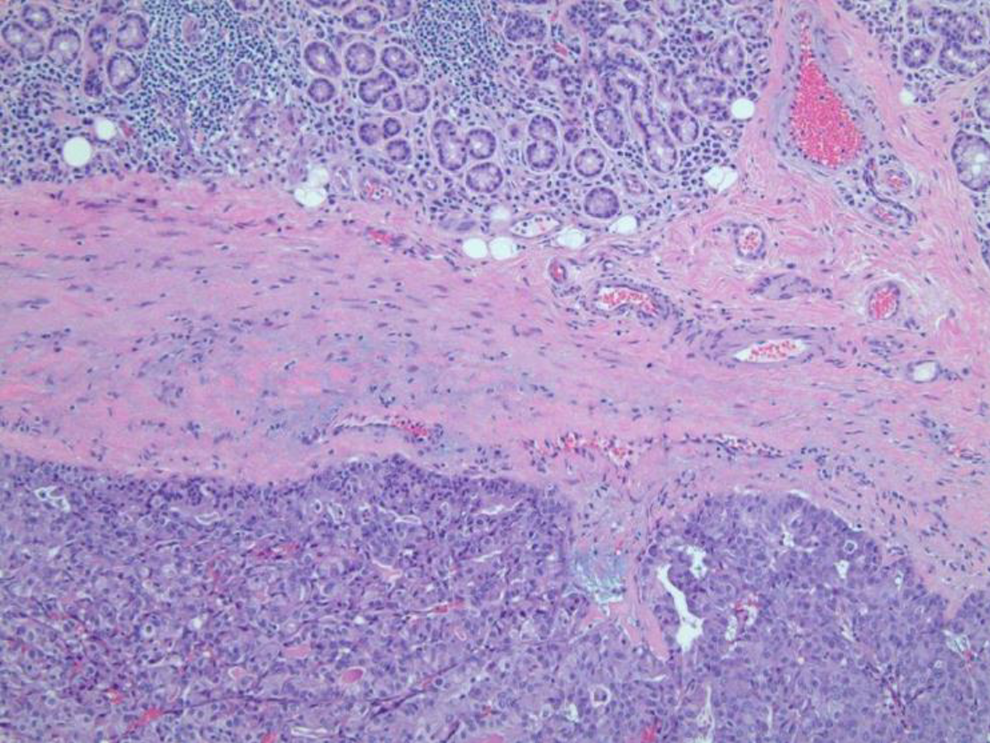
Tumor with adjacent salivary glands.
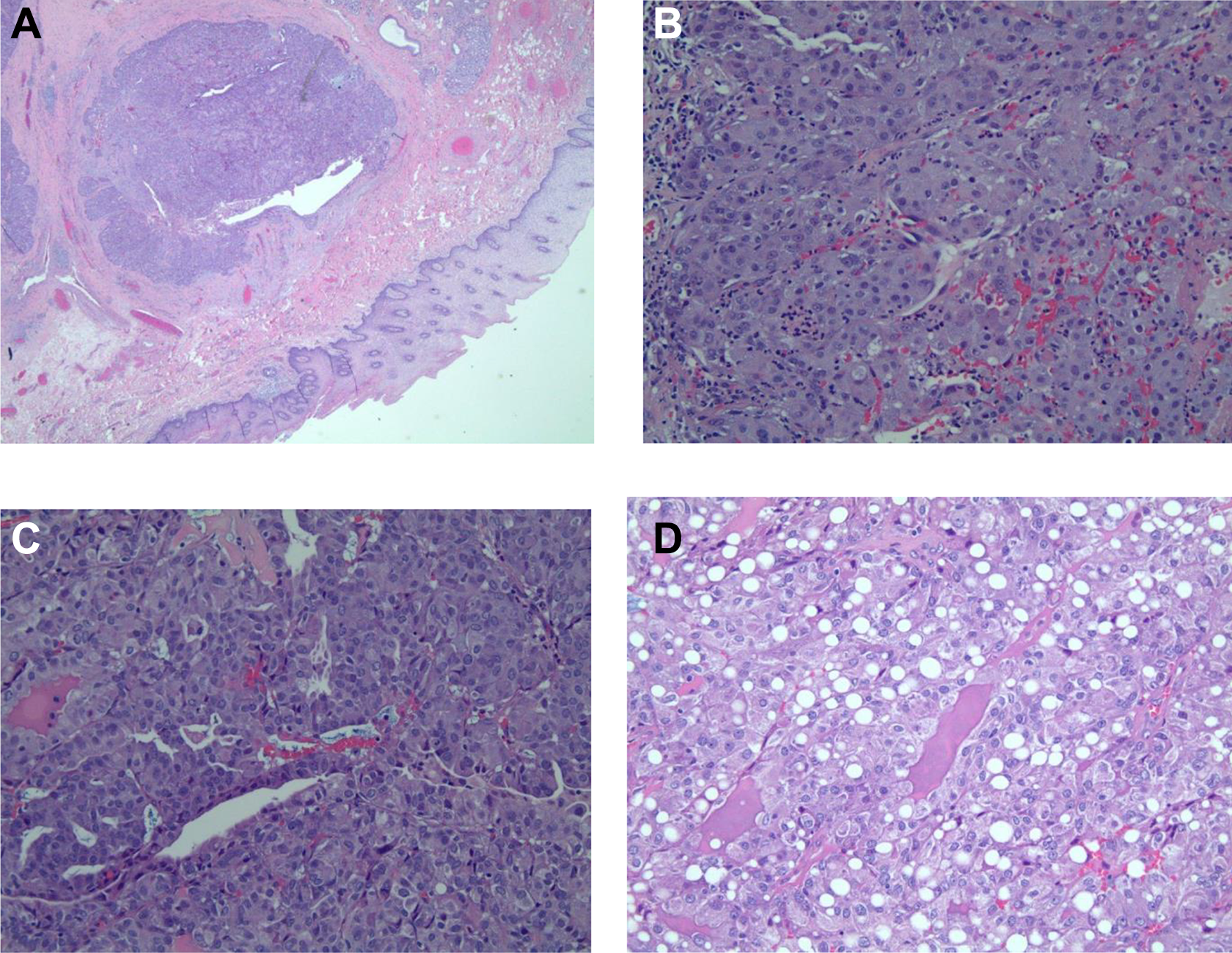
A, Low magnification. B and C, High magnification. D, Vacuolated area.
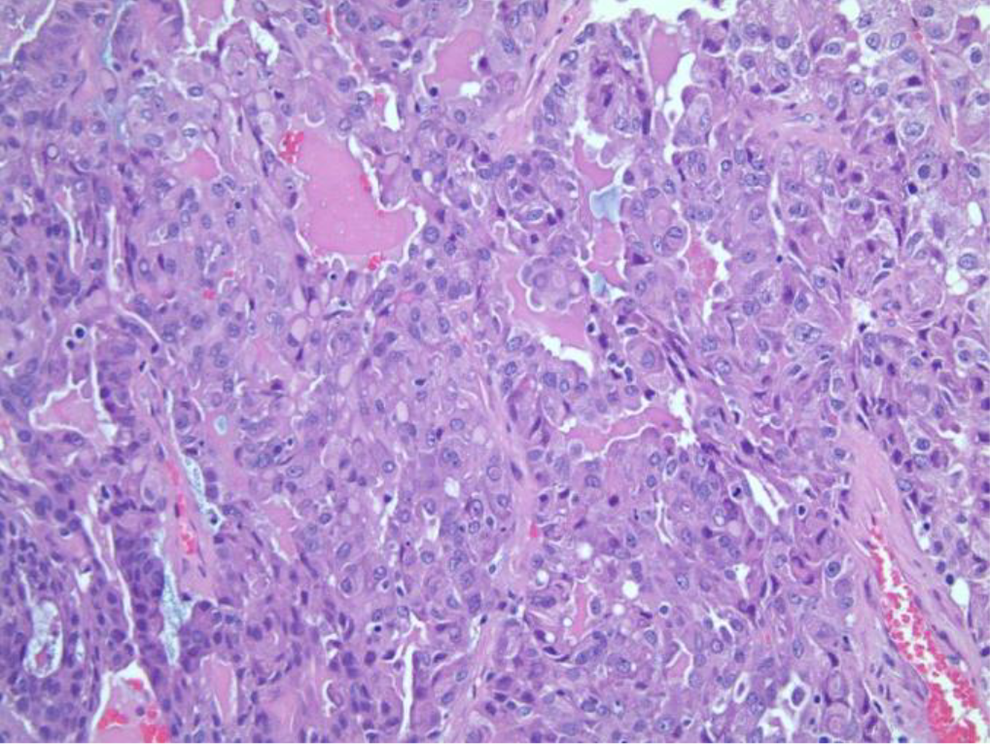
Eosinophilic secretions.
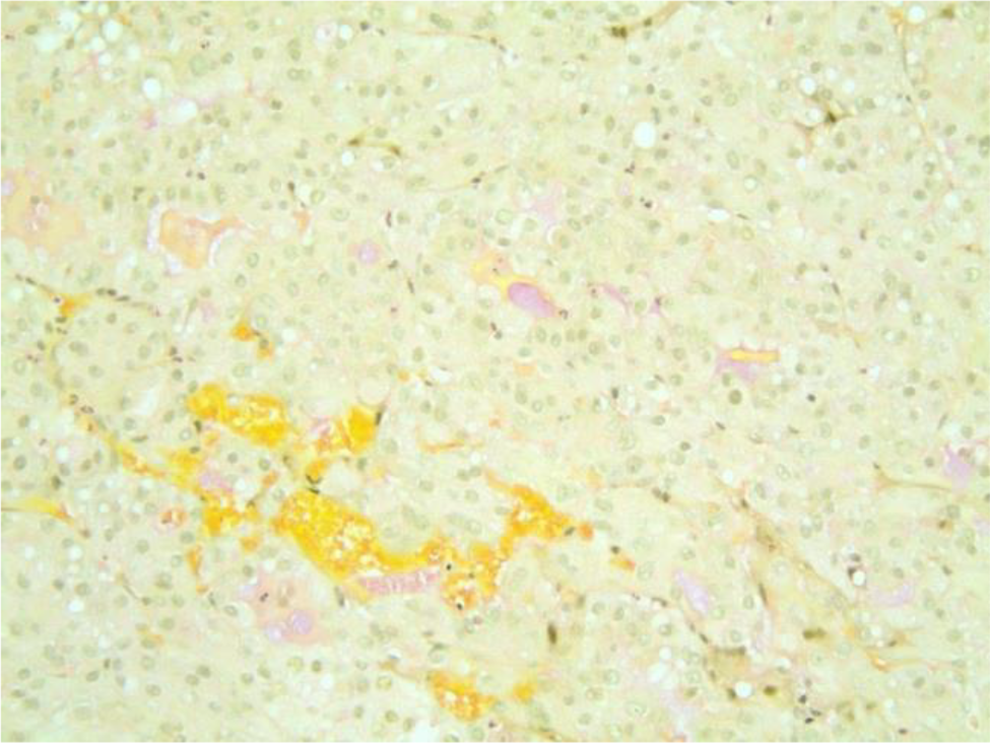
Mucicarmine stain.
After confirmation of MASC, a computed tomography scan was obtained to rule out distant metastasis, which returned with negative results. The tumor was excised completely and clear margins were confirmed with intraoperative frozen pathological analysis (Figure 6). The defect in the buccal mucosa was reconstructed with a muscle advancement flap in order to maintain oral competence. The patient returned for follow-up at 3-month intervals to assess for recurrence and has been tumor-free for over 1 year (Figure 7). The patient will continue to be closely monitored for recurrence.
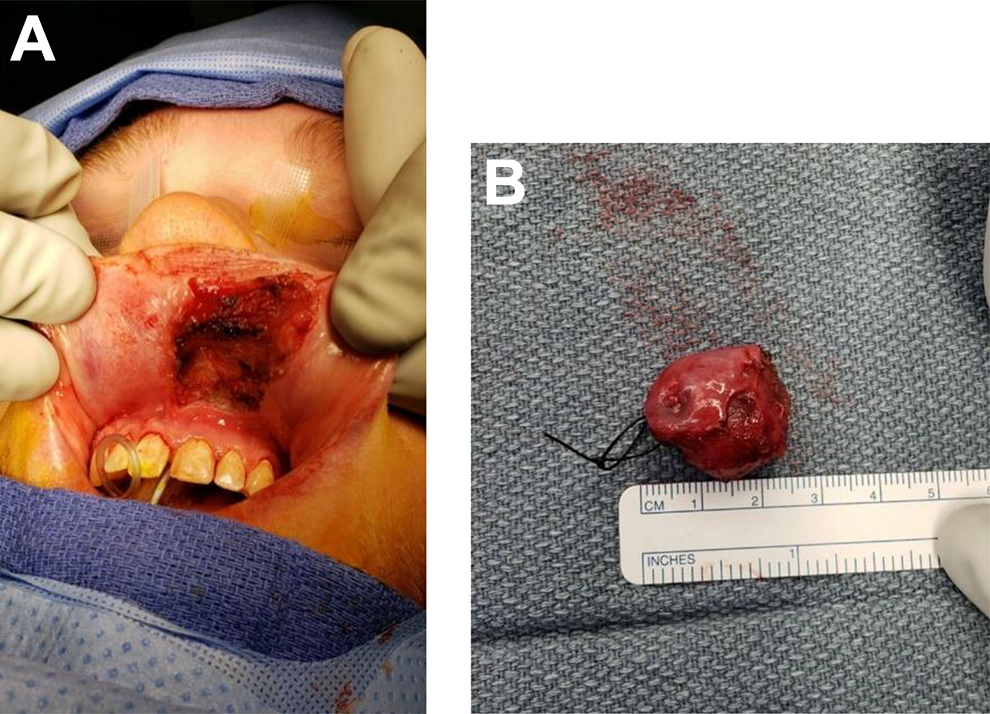
A and B, Complete excision with clear margins.
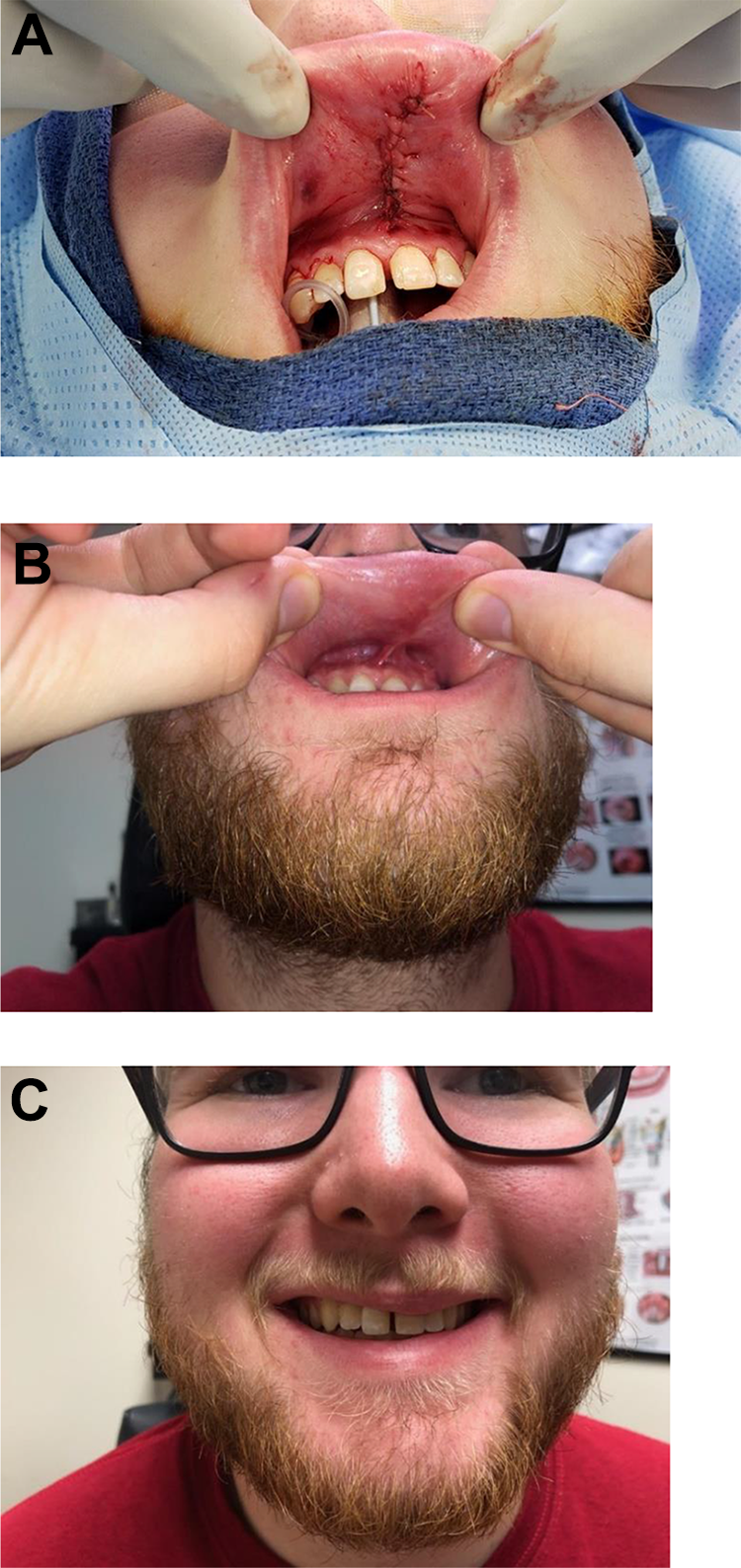
A, Muscle advancement flap reconstruction. B and C, Three-month follow-up.
Discussion
The case above presented as an atypical variant of MASC relative to its classical description. This, along with the rarity of MASC in general, provided a challenge in reaching the correct diagnosis and creating an appropriate treatment plan for the patient. Potential lack of recognition and misdiagnosis could have harmful outcomes on future patients with MASC. For this reason, we have decided to expand upon the primary similarities and differences between MASC and ACC in order to aid in the recognition of this commonly misclassified tumor.
Presentation
Mammary analogue secretory carcinoma most commonly occurs in the parotid gland but has been reported to occur in minor salivary glands, palate, buccal mucosa, base of the tongue, and lips.1,2 The average age of presentation of the tumor is in the fourth or fifth decade, with only a few cases being reported in children and adolescents, and is slightly more prevalent in men.2,3,6 Mammary analogue secretory carcinoma commonly presents as a slow growing, painless mass. The size of the mass ranges from 0.2 to 5.5 cm, with an average size of 1.7 cm. 4
The first differing feature in our case is the patient’s age at presentation. As mentioned previously, the average age of presentation of MASC is in the fourth of fifth decade of life, while this patient was only 21 years old and already had the tumor for 5 years. The second atypical feature, and what makes this case so unique, is the location of the mass. Mammary analogue secretory carcinoma is traditionally thought to occur in the major salivary glands. However, this occurrence in a minor salivary gland in the mucosa of the lip is less common.
Acinic cell carcinoma is a significantly more common salivary gland tumor than MASC, accounting for approximately 17% of primary salivary gland malignancies. When in the head and neck region, ACC is predominately found in the parotid gland, although it has been reported in the submandibular and minor salivary glands. Acinic cell carcinoma is also known to be found in the pancreas, lungs, and prostate gland. Acinic cell carcinoma is slightly more common in women than in men, and the average age at diagnosis is in the fifth decade of life. There is a moderate genetic predisposition for ACC and risk associated with prior radiation exposure.7-9
Histopathology
On gross examination, MASC appears as a solitary and well-circumscribed tan-pink or brown mass with rubbery texture, as demonstrated in our case (Figure 6B). The main histological features of MASC include combinations of microcystic, papillary cystic, tubular, and/or solid patterns of architecture. Tumor cells are low grade and contain eosinophilic vacuolated cytoplasm and pale nuclei with low mitotic activity. Necrosis and perineural invasion are uncommon. Mammary analogue secretory carcinoma is frequently noted to have eosinophilic colloid-like secretions.4,10-12 While the cells in our biopsy appeared to be more oncocytic than in the classically described histological features, the same cellular architecture and defining characteristics of MASC were still present.
Acinic cell carcinoma usually present grossly as a solitary and encapsulated, soft gray-white mass. In recurrent cases, the tumor may be unencapsulated and may contain areas of necrosis. Acinic cell carcinoma histologically resembles normal serous acinar salivary gland cells, with basophilic cytoplasm, normally growing in a solid pattern with fine serous cytoplasmic granules. However, growth patterns of ACC are diverse and include microcystic, papillary cystic, follicular, vacuolated, clear, and nonspecific glandular, among others. Invasion of the capsule and calcifications are commonly seen. Unlike MASC, ACC may display frequent mitosis and areas of perineural invasion in more aggressive cases.9,13
Cytogenic Analysis
Cytogenic analysis was used to determine that MASC had a fusion of the ETV6-NTRK3 genes, and the t(12;15)(p13;q25) translocation became gold standard for diagnosis. This fusion gene codes for a chimeric tyrosine kinase, which plays a role in oncogenesis and carries risk for high-grade transformation. The same translocation is found in secretory carcinoma of the breast. 1 This ETV6-NTRK3 translocation may be detected by fluorescent in situ hybridization or by reverse transcription polymerase chain reaction. 4 One study performed using this method identified 79% of parotid gland tumors previously diagnosed as ACC were actually MASC. 3 However, there are recent reports revealing other translocation partners for ETV6, including RET and MET, which suggest that the molecular diagnostic criteria should be modified.14,15
Large studies performed to analyze ACC of the parotid gland detected genetic alterations or deletions at chromosomes 4p, 5q, 6p, 6q, and 17p. These findings suggest the involvement of tumor suppressor genes in the oncogenesis of the ACC tumors.9,16
Immunohistochemistry and Histopathology
Due to the cost of cytogenic testing, immunohistochemical staining is more commonly used in the diagnosis of MASC. Mammary analogue secretory carcinoma is characterized by staining strongly for S-100 and vimentin, which are negative in ACC. Mammary analogue secretory carcinoma is usually also positive for a number of other stains including mammaglobin, mucicarmine, MUC1, MUC4, CK7, CK8, CK18, CK19, and STAT5a. The colloid-like secretions in MASC are Periodic Acid-Schiff positive and diastase resistant.1,10,12 While ACC is positive for DOG1, Bcl-10, trypsin, and chymotrypsin, MASC is negative for these stains.7,17 A study done found that adipophilin expression is strong and diffuse in MASC, while it is patchy and present in only small amounts in ACC. 12 This was the main method of diagnosis in our case. As described in the case presentation, the characteristic immunohistochemical stains were displayed in our biopsy.
Prognosis and Treatment
Since MASC is still relatively new and underreported in the literature, data investigating long-term follow-up are minimal and continue to be collected and analyzed in order to make accurate predictions regarding long-term survival and give definitive guidelines for management.6,10 Data regarding ACC in parotid glands are historically well known but may be skewed due to a subset of patients diagnosed with ACC actually having MASC that was misclassified. However, the impact of this misclassification on the data is likely minimal due to how rare MASC is.3,10
Mammary analogue secretory carcinoma is generally low grade and indolent but has the capacity to behave aggressively, metastasize to regional lymph nodes, and recur after treatment.1,3 It has a favorable prognosis overall, but the presence of necrosis is an indicator for a more aggressive tumor and poorer outcomes.2,5 Small studies have been performed that did not find statistically significant differences between MASC and ACC in terms of lymph node involvement at the time of diagnosis and disease-free survival time after treatment.10,18 One study by Chiosea et al found that the rate of lymph node metastasis for patients with MASC was 17.6% (6/34) compared to 7.9% (3/38) in ACC. The average disease-free survival time, including death or recurrence, after treatment for MASC and ACC was 92 months and 121 months, respectively. 2 It seems that MASC and ACC have similar survival rates, with 5-year and 10-year survival for ACC estimated to be 91% and 88%, respectively. 9
As data are limited, and MASC was treated traditionally as ACC, it is currently safe to assume that MASC should continue to be treated as ACC until more studies investigating treatments and outcomes have been conducted.6,10 The current standard of care for MASC is radical surgical resection with preservation of nearby nerves. Postoperative radiotherapy (PORT) is recommended for patients with incomplete resection, close margins (<5 mm), tumors with perineural invasion, tumors classified as T3 to T4, or cervical lymph node metastasis. Data have been shown to improve long-term outcome in these patients after receiving PORT.19,20 In aggressive cases following surgical resection, if there is a confirmed ETV6-NTRK3 or ETV6-MET translocation, tyrosine kinase inhibitors or c-Met inhibitors are potential therapeutic options that should be considered.15,21
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
