Abstract
There is a high prevalence of dysphagia in patients with neuromuscular diseases and stroke, and consequences can be profound. However, the correlation of dysarthria and oral-oropharyngeal dysphagia remains unclear. This review aimed to define the clinical co-presentation of dysarthria and dysphagia in this population. A PubMed search to identify literature on the prevalence of dysarthria and dysphagia was systematically conducted in the English language literature since 1995. Subjective and objective outcomes instruments were identified for both dysarthria and dysphagia. Studies that included prevalence and co-presentation were included. Inclusion and exclusion criteria were applied according to the Preferred Reporting Items for Systematic Reviews and Meta Analyses (PRISMA). Of the 1,056 articles identified in the search, 20 articles met the search criteria. An additional 4 articles were examined for a total of 24 articles for analysis. Dysarthria and dysphagia were found to be highly prevalent among patients with neuromuscular disease (NMD). Overall, there was a higher prevalence of dysarthria than dysphagia. Of those patients with dysphagia, some reports estimate 76-90% of patients with NMD also had dysarthria. Dysarthria is a strong clinical clue to the presence of dysphagia. Existing subjective questionnaires may not reveal the presence of oropharyngeal dysphagia, but objective measures are more revealing. Further study to correlate the degree of dysarthria and severity of oral-oropharyngeal dysphagia are warranted.
Keywords
Introduction:
Dysarthria and dysphagia are common in neuromuscular diseases 1 , but the relationship between the two conditions has not been prominently explored. Dysarthria is difficultly in oral articulation and motor speech function. In addition to impaired communication, dysarthria is characterized by poor voluntary oromotor control, so it is not surprising to frequently observe drooling of saliva in this population. Also, one would expect inability to process a food bolus and trigger a normal reflexive oropharyngeal swallow mechanism. In addition to declining quality of life associated with dysarthria and dysphagia, there is an increased risk of aspiration and its devastating consequences 2 .
Both dysarthria and dysphagia are prominent in cerebrovascular diseases and stroke, traumatic brain injuries (TBI), and neuromuscular diseases such as Amyotrophic Lateral Sclerosis (ALS), Multiple Sclerosis (MS), Myasthenia Gravis (MG), and Parkinson’s disease (PD). 3 These neuromuscular diseases are usually characterized by slow decline in coordinated muscle function that progressively deteriorates over time. There are challenges to draw a relationship between dysarthria with poor oromotor control, and oral-oropharyngeal dysphagia, starting with the measurements of dysarthria and dysphagia severity. Outcome measures have been assessed differently in much of the literature to include 1) subjective (self-reported questionnaires), 2) observed measures (as reported by a skilled clinician observing the patient), and 3) objective measures (quantifiable, for example on a biomechanical test). There are also 6 subtypes of dysarthria which have different features 4 : flaccid (bulbar palsy), hypokinetic (PD), hyperkinetic (dystonia), spastic (pseudobulbar palsy), ataxic (cerebellar disorders), and mixed (ALS).
The purpose of this systematic review is to examine the literature on dysarthria as it relates to dysphagia, with the hypothesis that dysarthria is a strong predictor for the presence of dysphagia. Our ultimate goal is to have the ability to use a dysarthria index to help screen for the presence of oral-oropharyngeal dysphagia, thus the patient’s risk for aspiration or nutritional deficiency.
Methods:
A comprehensive literature review was conducted in the PubMed database from June 1995 to June 2015 on all articles pertaining to dysarthria and dysphagia. The systematic literature review was completed in accordance with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. 5 Search inclusion terms for dysarthria were dysarthria, motor speech, articulation, and anarthria. Search inclusion terms for dysphagia were dysphagia, odynophagia, globus, deglutition, and swallowing. Individual search terms were linked with an “OR” statement, and the two searches were linked with an “AND” statement. Search criteria were limited to “Title/Abstract” content.
Exclusion criteria were articles published in languages other than English, articles published outside the 20-year timeframe (prior to June 1995), and articles not relevant to the relationship between dysarthria and dysphagia. Review articles, commentaries, and single-case studies were not considered. Additional articles were identified from the references of the articles in the initial search. A single author performed the initial screening of records that met inclusion and excluding those that met exclusion criteria which were straightforward. All 3 authors performed the critical review of the 24 articles as the subject of our analysis.
Results:
The initial PubMed search identified 1056 articles. There were 4 additional records identified from the references of relevant sources that were not in our original search. A total of 1060 records were screened for this literature review. 239 records were excluded for being published in a language other than English. 145 records were excluded for being published outside the 20 year timeframe (June 1995 to June 2015). 676 records were assessed for eligibility in this review in which 652 were excluded for reasons related to other inclusion and exclusion criteria. Exclusion criteria were straightforward and were able to be determined directly from the abstracts. In those cases where it was not obvious whether it was a review article, commentary or single case study, the paper was pulled to confirm inclusion or exclusion. A final 24 articles remained for analysis. This process is shown in Figure 1 of the PRISMA flow diagram.
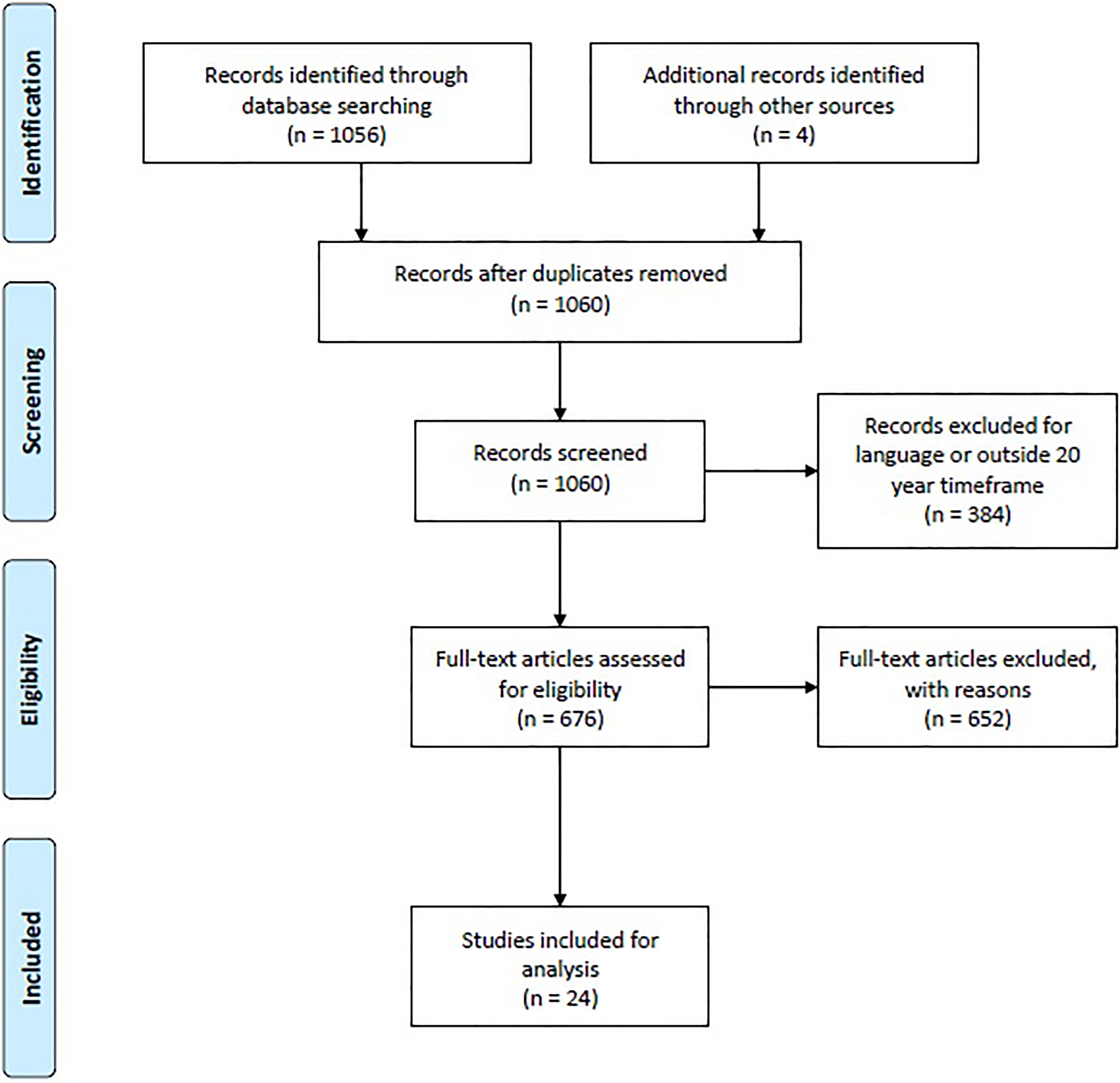
PRISMA Flow Diagram.
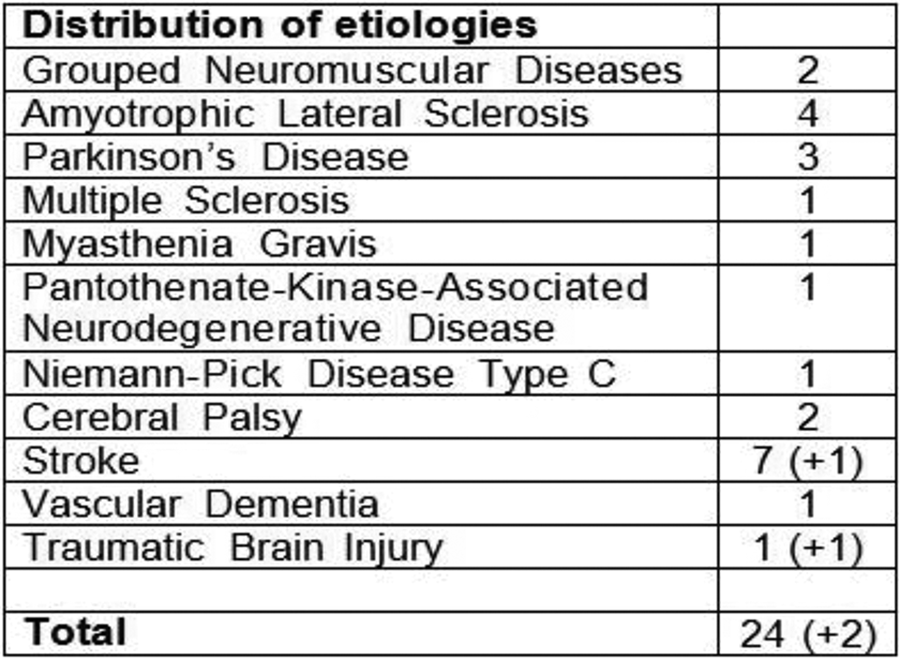
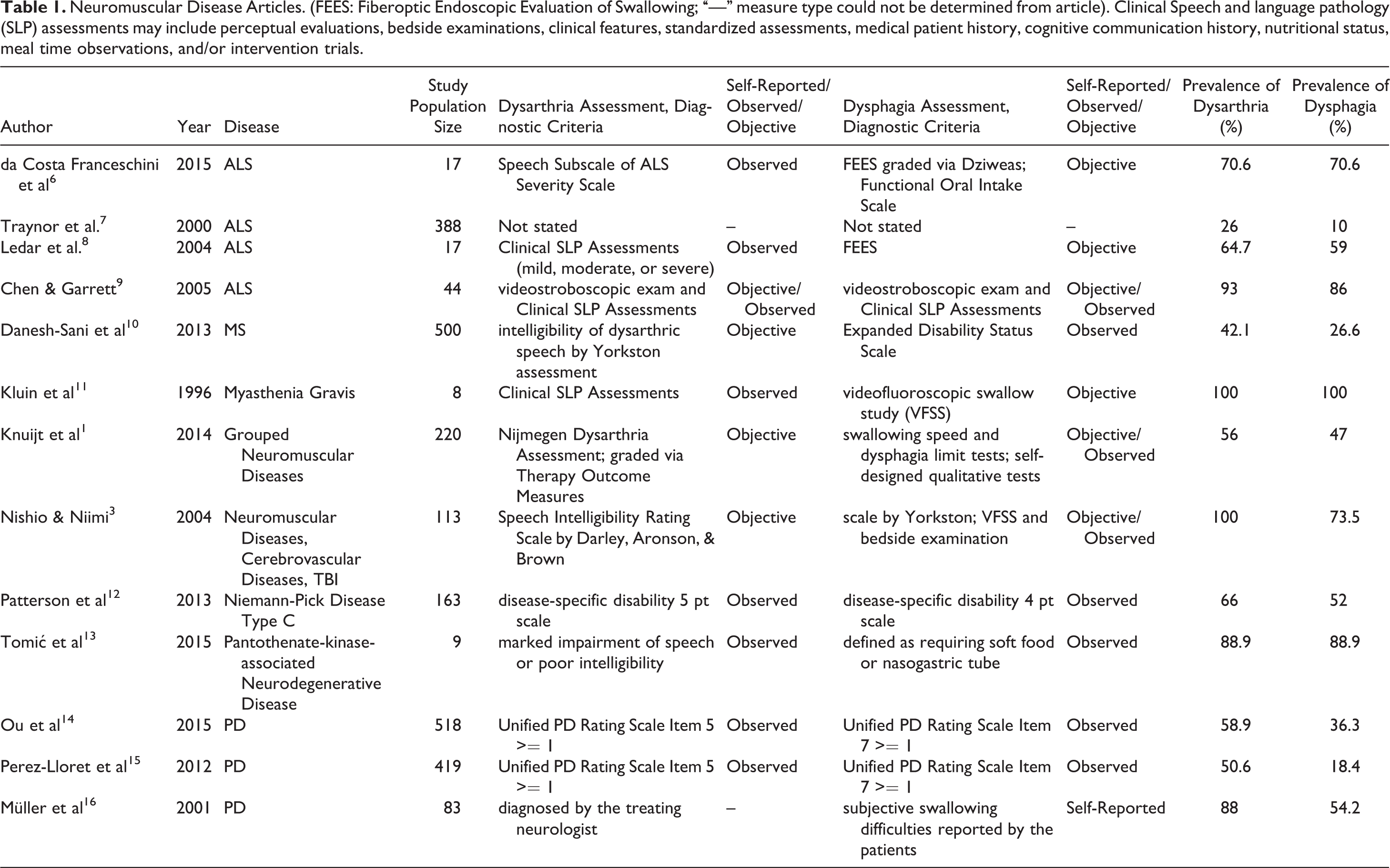
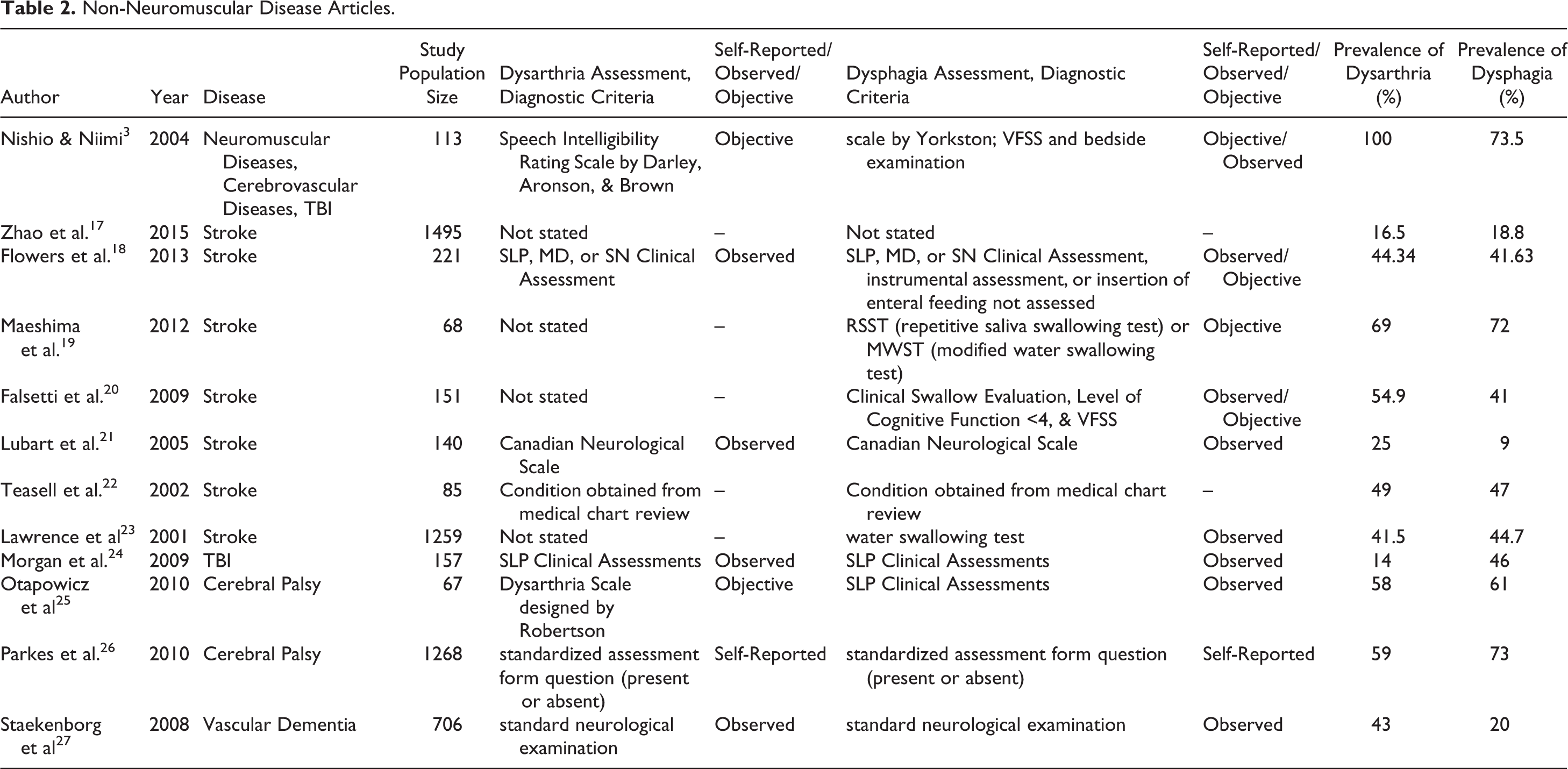
The 24 articles that met all inclusion and exclusion criteria are summarized and categorized in Table 1 for 13 articles on neuromuscular diseases, and in Table 2 for 12 articles on cerebrovascular diseases, TBI, and Cerebral Palsy. One of the articles is listed under both tables because it covered neuromuscular diseases, cerebrovascular diseases, and TBI. 5 Author, year, disease, study population size, dysarthria measure, dysphagia measure, prevalence of dysarthria, and prevalence of dysphagia were recorded. We organized the articles into two separate tables because the etiology of neurodegenerative diseases and cerebrovascular diseases like stroke can differ greatly. Table 3 shows the distribution of diseases of the 24 articles in the review. 12 articles are on cerebrovascular diseases and Cerebral Palsy.
Neuromuscular Disease Articles. (FEES: Fiberoptic Endoscopic Evaluation of Swallowing; “—” measure type could not be determined from article). Clinical Speech and language pathology (SLP) assessments may include perceptual evaluations, bedside examinations, clinical features, standardized assessments, medical patient history, cognitive communication history, nutritional status, meal time observations, and/or intervention trials.
Non-Neuromuscular Disease Articles.
Specific diseases in the articles for analysis.
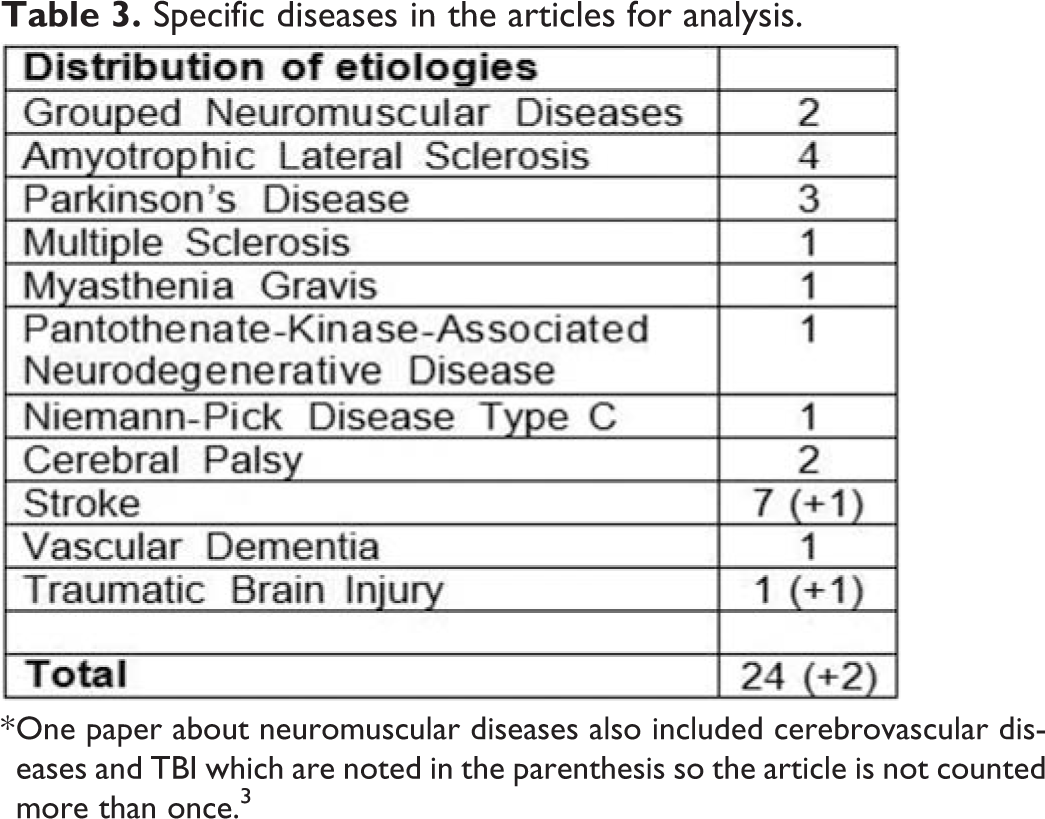
* One paper about neuromuscular diseases also included cerebrovascular diseases and TBI which are noted in the parenthesis so the article is not counted more than once. 3
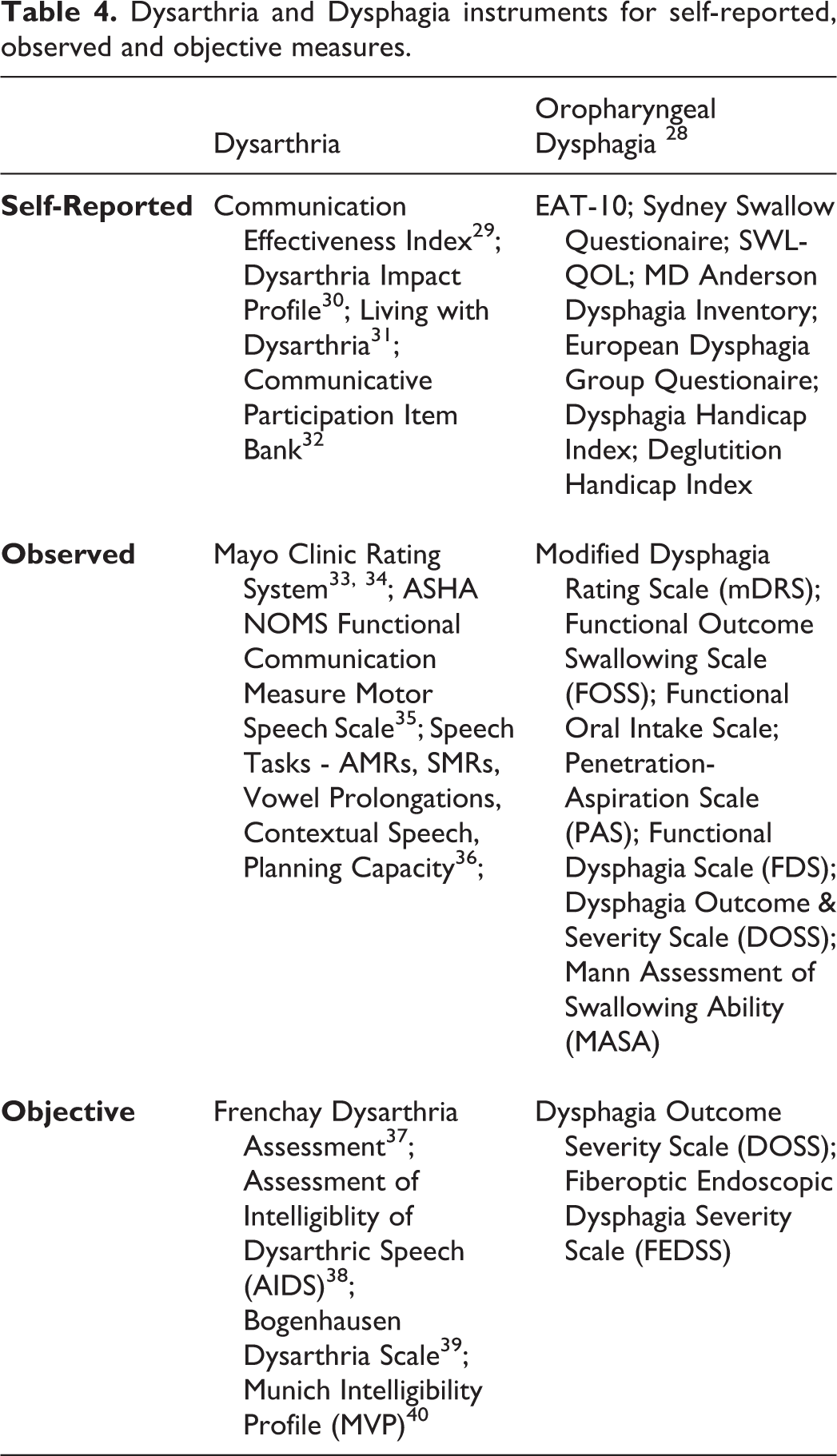
Dysarthria and dysphagia measures for each article were classified as self-reported, observed, or objective based on information in the methods section of each study. Vague descriptions of measures such as “obtained from medical chart review” or “detected by clinical examination” were classified as an observed measure, although these did not appear to be quantifiable by validated assessment instruments. Table 4 shows different measures for dysarthria and dysphagia that were encountered in our review and categorized as self-reported, observed, or objective (adapted in part from 28 ). In the 24 articles taken for review, 15 had observed dysarthria measures, 5 had objective measures, and 4 did not state how the study determined dysarthria. For the dysphagia measures, 1 article had self-reported measure, 10 articles had observed measures, 10 objective, and 3 were not stated.
Dysarthria and Dysphagia instruments for self-reported, observed and objective measures.
Based on the diversity of study designs, a proper meta-analysis could not be performed. In the neuromuscular disease population, study population size ranged from n=9-500 with prevalence of dysarthria ranging from 26-100% (mean 63%), and the prevalence of dysphagia ranging from 10-100% (mean 55%). In the predominantly non-neuromuscular disease group, study size ranged from n=68-1259, with a prevalence of dysarthria ranging from 14-100% (mean 57%), and a prevalence of dysphagia ranging from 9-73% (mean 41%). These broad estimates do not give weight to the different articles’ sample sizes and measure criteria. Also, while they documented co-prevalence in all disease states investigated, there were no studies which quantified or examined stronger association or causality between dysarthria and dysphagia.
Discussion:
There is a familiar and paradoxical scenario with end-stage neuromuscular disorders patients, which is accelerated over the similarities with the geriatric population. 41,42 Slowing deglutition leads to impaired nutrition, battling a negative nitrogen balance with the consequences of sarcopenia leading to “presbepharynges.” Without aggressive nutrition and hydration the process of functional decline continues, yet forced or aggressive attempts at swallowing increase the risk of aspiration.
Infection and respiratory failure leading to hospitalization can be devastating and may predispose a terminal event. Patients are generally sedentary during their hospitalization, and concern for further aspiration results in limiting oral intake (such as medications vitals for agility in patients with PD). Constricted movement contributes to decreased pulmonary ventilation and clearance, as well as physical deconditioning. While the literature is not clear, it is common for patients with neuromuscular disease and movement disorders to be prone to non per os (NPO) status, feeding gastrostomy tube insertion, and discharge to an extended care facility rather than to home. Presently, recommendations for NPO status or feeding tube insertion are controversial in the presence of advanced neuromuscular disease or cognitive deficit, and this was not able to be determined from the present review. 43
The idea of using an observed measure for dysarthria as a predictor of oral and oropharyngeal dysphagia is appealing, especially in the high-risk population of patients with neuromuscular diseases where a “sentinel indicator of decline” may be recognized. 2 Screening for dysphagia and aspiration in this group with self-reported surveys and instruments is potentially unreliable for a number of reasons, 44 -47 to include fear of not being allowed to eat, ensuing recommendations for feeding tube insertion with all of its sigma and potential consequences, and simply not being aware of that their swallowing and nutrition status is abnormal.
In our study, dysarthria appears to be a strong clinical indicator for the presence of dysphagia, and the severity of dysarthria corresponds to severity of dysphagia in most articles. Although there were no studies that demonstrated the natural decline of dysarthria and dysphagia together over time, some studies did hint at some relationship that was unable to be substantiated. In one study, the prevalence and severity of dysphagia varied depending on the type of dysarthria. 3 The more severe conditions of dysphagia were linked to dysarthria associated with bilateral impairment of upper or lower motor neurons such as flaccid, spastic, and mixed dysarthria. The idea that dysarthria and dysphagia could be correlated based on degree of severity was also supported in a 2010 study where the most severe dysarthria cases were associated with oropharyngeal dysphagia. 25
Among the 24 articles in this literature review, there was no prospective systematic approach and little uniformity in the specific dysarthria and dysphagia measures used. There were 7 articles that did not specify how dysarthria and/or dysphagia were diagnosed or what criteria were used to determine the symptom. Of the 24 studies, only 1 study had formal objective and subjective measures for both dysarthria and dysphagia. 9 Overall, informal observed measures for dysarthria (11 out of the 24 articles) and dysphagia (10 out of the 24 articles) were more commonly used than any other types of measures. Formal universal objective and subjective grading systems are needed for more accurate and consistent reporting in future studies. This literature review found that the most common used measures tended to be subjective and thus may not reveal the true presence of dysarthria and/or oropharyngeal dysphagia by leading to underreporting.
In a study on bulbar-onset ALS patients, the presence of dysarthria was discovered to be 8 times more common than dysphagia as an initial symptom. 7 The current literature is not clear on why dysarthria appears more often than dysphagia in neuromuscular diseases but possible hypotheses could be 1) dysarthria is easier to detect or dysphagia is harder to diagnosis, 2) dysarthria naturally precedes the development of dysphagia, or 3) there are compensatory mechanisms for swallowing which makes the prevalence of dysphagia appear lower than its true rate.
If dysphagia was a natural progression from dysarthria, it could be expected that this might be why dysarthria prevalence rates are higher. In a 2001 Parkinson’s study, researchers found the latencies to dysarthria would precede dysphagia. 16 The results suggest that if dysarthria occurs before the onset of dysarthria then dysarthria could be used as an early predictor for dysphagia. If this is the case, then we should expect to see most dysphagia patients with dysarthria. In the articles that specify concurrent presenting symptoms, the percentage of patients with dysphagia that also had dysarthria ranged from 66 to 90%. 1,6,15,18,20,24 This finding supports the idea that dysarthria could precedes the development of dysphagia.
There are limitations of the existing literature to include inconsistency of outcomes measures used for both dysarthria and dysphagia, limited studies which correlate subjective with observed or objective measures, and no longitudinal studies which show correlation over time. With a series of studies demonstrating high-levels of prospective evidence with proper statistical power, a quality checklist for health care intervention studies would have been a valuable tool to include grades on study validity, bias, confounding and power. Unfortunately the 24 studies identified were cross sectional observational studies that did not qualify for level 1 or 2 prospective evidence. Also, our review essentially identified “co-prevalence” in all the studies rather than causality. Although systematic in our approach at reviewing the present fund of knowledge, these limitations precluded us from performing a proper meta-analysis.
There is a hint that dysarthria precedes the development of dysphagia and that their severity are correlated to each other. A better defined relationship between dysarthria and dysphagia could lead to the utilization of a dysarthria scale that measures the risk for oropharyngeal dysphagia. Thus, earlier interventions could be provided that would allow for better management of speech and swallowing outcomes. Further investigation to correlate subjective, observed and objective measures in a longitudinal study would enable a better understanding of these relationships, and facilitate a more robust screening tool for aspiration risk in patients with neuromuscular disease.
Conclusions:
Dysarthria is a strong clinical clue to the presence of dysphagia in neuromuscular diseases. While this review highlights some of the serious limitations in the dysarthria and dysphagia literature, we have opened the discussion on how the two disorders are connected. A more formal screening tool for aspiration risk would be highly valuable as it relates to dysarthria and oral-oropharyngeal dysphagia in this patient population.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.