Abstract
Objectives:
The aim of this study was to compare the efficacy of bioabsorbable steroid-eluting sinus stents versus absorbable Nasopore packs after endoscopic sinus surgery (ESS) for the treatment of chronic rhinosinusitis (CRS).
Methods:
One hundred eighty-one patients with CRS who underwent ESS were randomly assigned to receive a steroid-eluting sinus stent in one ethmoid sinus cavity, whereas the contralateral control side received a Nasopore pack. Endoscopic evaluations were performed 14, 30, and 90 days after the ESS. Postoperative intervention, polyp formation, adhesions, and middle turbinate (MT) position were assessed as efficacy outcomes.
Results:
The stents were successfully deployed in all 181 sinuses. Thirty days after the ESS, the stents significantly reduced the need for surgical intervention compared to the Nasopore (P < .0001). The percentage of cases with polyp formation was significantly lower on the stent sides compared with the Nasopore sides (P < .0001) at 14, 30, and 90 days after ESS. The percentage of severe adhesion was significantly lower on the stents sides than on the Nasopore sides at postoperative day 90 (P = .0003), whereas they were not significantly lower at postoperative days 14 and 30. There were no significant differences between the stent sides and the Nasopore sides regarding the frequency of MT lateralization at all end points. No device-related adverse events occurred.
Conclusions:
Our study demonstrated significant improvement in the early postoperative outcomes by reducing the need for postoperative surgical intervention and polyp formation using steroid-eluting stents when compared with absorbable Nasopore packs. The steroid-eluting sinus stents and the Nasopore packs were each effective in preserving the ethmoid sinus patency and in preventing MT lateralization. A further prospective cohort study with long-term postoperative outcomes is warranted.
Introduction
Given the nature of chronic rhinosinusitis (CRS) as a chronic condition, the primary goal of medical and surgical treatments for CRS is to achieve and maintain clinical control. 1 Apart from endoscopic sinus surgery (ESS) improves ventilation and drainage, it gives better access of local anti-inflammatory treatment if anti-inflammatory treatment alone is insufficient. The maintenance anti-inflammatory treatments after ESS were recommended is to achieve and maintain clinical control of the chronic disease. 1 The inadequate surgery and poor adherence to medical management often contribute to the failure of primary ESS. Continued use of postoperative intranasal corticosteroids has been shown to improve the endoscopic scores. One large retrospective patient-based study showed that the topical corticosteroids were prescribed at a low rate and it indicates that many patients do not use the local corticosteroids. 2 Hence, the steroid-eluting stent maybe a solution for the problem of compliance.
Bioabsorbable steroid-eluting sinus stents are designed to insert into the sinuses following an ESS to preserve sinus openings during the postoperative healing period. The slow release of corticosteroids aims to decrease mucosal inflammation and edema and to expedite wound healing.3-6 Currently, there are 2 steroid-eluting (mometasone furoate) stents, including the PROPEL implants (Intersect ENT) and the BISORB Drug-Eluting Sinus Biopolymer Stent System that are approved by the US Food and Drug Administration5-7 and the China Food and Drug Administration (CFDA), 3 respectively. These 2 sinus stents are designed for placement in the ethmoid or frontal sinuses after an ESS.
Previous randomized clinical trials (RCTs) have demonstrated that PROPEL steroid-eluting stents may improve surgical outcomes when compared with non-steroid-eluting stents by maintaining the patency of the ethmoid and frontal sinuses, reducing inflammation, polyp formation, and mucosal adhesion.8-12 Interestingly, Rawl et al 11 showed no significant improvement in postoperative outcomes using PROPEL steroid-eluting stents when compared with nonabsorbable packs. European Position Paper on Rhinosinusitis and Nasal Polyps 2020 (EPOS 2020) revealed that the postoperative placement of steroid-eluting stents compared to placebo does not have an influence on symptomatology but has a significant effect on endoscopic scores. 1 The EPOS 2020 could not advise about the use of steroid-eluting stents following ESS because of steering group felt the quality of the evidence was low. 1 However, International Consensus Statement on Allergy and Rhinology: Rhinosinusitis recommended that steroid-eluting stents are optional for the ESS. 13 Our previous Cochrane review revealed that there was no evidence from suitable RCTs to show that one steroid-eluting sinus stent is superior to other absorbable nasal packs. 4 Hence, the purpose of this study was to compare the efficacy of bioabsorbable steroid-eluting sinus stents versus absorbable Nasopore packs after ESS for the treatment of CRS.
Methods and Materials
Study Design
This study was approved by the institutional review board of the Nose and Throat Hospital, Fudan University and the Beijing Tongren Hospital, Capital Medical University. This study was registered in the Chinese Clinical Trial Registry (http://www.chictr.org.cn/abouten.aspx) center of the World Health Organization (NO.ChiCTR-IIR-17013832). This is a prospective, multicenter, randomized, single-blind, controlled clinical trial. This study was conducted at 8 medical centers enrolling consecutive patients between September 2014 and June 2016 in China. This study used a within-patient or intrapatient control method.
Patients
Patients with CRS and no nasal polyps and patients with CRS and with nasal polyps who underwent ESS were enrolled in this study. Chronic rhinosinusitis was diagnosed based on the EPOS 2020 criteria 1 :
Inclusion and Exclusion Criteria
Inclusion criteria included: (1) Patients aged 18 to 65 years, males or nonpregnant females; (2) Patients or their statutorily authorized representatives must be able to understand the purpose and procedures of the trial and voluntarily sign the informed consent form; (3) Patients must meet the diagnostic criteria of CRS, diagnosed with bilateral CRS, confirmed by CT examination of both a Lund-Mackay score of 26 points.
Exclusion criteria included: (1) A known allergic reaction or contraindication to the device material and its degradation products (mometasone furoate and polylactic acid lactide); (2) A patient needing long-term oral hormones; (3) A patient receiving immunosuppressive therapy or is known to have an immunosuppressive or autoimmune disease; (4) Patients with diabetes, glaucoma, or ocular hypertension; (5) Patients with acute bacterial sinusitis or acute fungal sinusitis.
Sample Size Calculation
According to a similar previous study 9 and based on our previous work, we deemed the necessary sample size for α = .05 and odds ratio = 2.2, to be approximately 163 patients. To rule out the possibility of a patient lost to follow-up or a missing data value, we added an additional 10% of cases to our target calculations. Eventually, the sample size target was determined to be 180 cases.
Intervention
The BISORB bioabsorbable steroid-eluting sinus stent (Puyi (Shanghai) Biotechnology Co., Ltd, China) was used for this study. The prior animal (rabbit) study showed these stents were safe, biocompatible and does not lead to mucosal inflammatory response at 7, 14, 30, 45, and 90 days after implantation of the stents (unpublished data by Dehui Wang, MD. It was approved by the CFDA in 2017 (20173460679). This product was also internationally approved, and bibliographic data are available on the Espacenet website (BR112014028185 Implanted System for Treating Sinusitis or Allergic Rhinitis), and is priced at $600 [USD 600]. This sinus stent is intended for use in adult patients with CRS with the dual benefits of mechanical spacing and anti-inflammatory pharmacotherapy. The stent is composed of a bioabsorbable polylactide-co-glycolide polymer and is coated with 652 µg of a corticosteroid (mometasone furoate). The corticosteroid is released in a controlled fashion over approximately 30 days into the surrounding mucosa. The dosage of released each day is 20 to 25 µg of mometasone furoate. This stent is designed to maintain the sinus opening after sinus surgery and offers the benefits of anti-inflammatory pharmacotherapy. The detail of drug elution profile is in Supplementary Table 1.
Surgical Procedures
Senior surgeons from 8 medical centers performed the ESS procedures. All ESS with ethmoidectomy were performed bilaterally under general anesthesia. An intrapatient control design was used in this study. Random allocation (randomization) was conducted using a randomized number (generated by SAS 9.4) contained in an envelope. The treatment-side sinus cavity received one bioabsorbable steroid-eluting sinus stent, whereas the contralateral side received the Nasopore pack (Stryker Corp) in a randomized fashion at the end of the ESS. Nasopore is a fully synthetic absorbable packing material and has been studied extensively. No steroids were allowed to add to the Nasopore packs in this study. Postoperative videos were analyzed by independent experts and clinical investigators.
Perioperative Care and Follow-Up
There were no restrictions on preoperative oral or intranasal steroid use. For the stent sides, no additional stents were placed into the frontal and maxillary sinuses. No other packing materials were inserted into the stents. After the ESS, a 7-day course of antibiotics was required. Nasal saline irrigation and oral mucolytics were routinely used during the follow-up period. Intranasal steroid sprays were allowed starting 30 days after the ESS.
The use of saline sprays or irrigation was permitted as needed both before surgery and during follow-up. The use of saline spray or irrigation in the study varied according to physician preference.
All patients required clinic visits for endoscopic examination at 14, 30, and 90 days after the ESS. Endoscopic debridement was permitted during the clinic visits. The videos of the endoscopic examinations were recorded at 14, 30, and 90 days after the ESS and were provided to the panel of reviewers and/or clinical investigators for review, evaluation, and grading. The independent reviewers of the panel are blinded to the type of packing materials as well as to the patients. Telephone contacts only occurred at 180, and 365 days after the ESS.
Efficacy Outcomes
Rate of Postoperative Surgical Interventions (Debridement)
The primary outcome of this study was the rate of postoperative interventions within 30 days after ESS. The endoscopic findings were evaluated by 3 independent reviewers and clinical investigators. The following endoscopic grading system was used for the evaluation: For polypoid tissue, 0 = none; 1 = minor polypoid tissue within the middle meatus; 2 = multiple polypoid tissue within the middle meatus; and 3 = polypoid tissue beyond the middle meatus. For middle turbinate (MT) lateralization, 0 = medialized; 1 = normal; 2 = partially lateralized; and 3 = completely lateralized. For adhesion, 0 = none; 1 = mild; 2 = moderate; and 3 = severe. The surgical intervention was recommended when the at least one following endoscopic scores over 2 or 3: (1) polypoid tissue ≥2; (2) adhesion scores were ≥2; and (3) the MT lateralization score was ≥3. Polyps, MT lateralization, and adhesion scores were also independently analyzed at 14, 30, and 90 days after ESS.
Safety Outcomes
The intraocular pressure (IOP) was measured using Tono-pen tonometry (Reichert Inc) at baseline and at 14, 30, and 90 days after the ESS. Dilated slit lamp examination of the crystalline lens was also performed at baseline and at 14, 30, and 90 days after the ESS. Examinations were performed by ophthalmologist. Additionally, adverse events were recorded at 14, 30, 60, 90, 180, and 365 days after the ESS via either a clinic visit or by telephone contact.
Statistical Analysis
The data were collected from all of the medical centers and analyzed by independent biostatisticians. Statistical analyses were performed using SAS software (version 9.4; SAS Institute, Inc). The within-patient or intrapatient comparison was used for all statistical comparisons. The McNemar test was used for testing on a 2 × 2 contingency table, arising from the intrapatient or within-patient control design, in which patients served as their own control. The need for postoperative interventions was analyzed using the McNemar test. All P values were 2 sided and significant differences were defined as those having a P < .05.
Results
Patient Enrollment and Characteristics
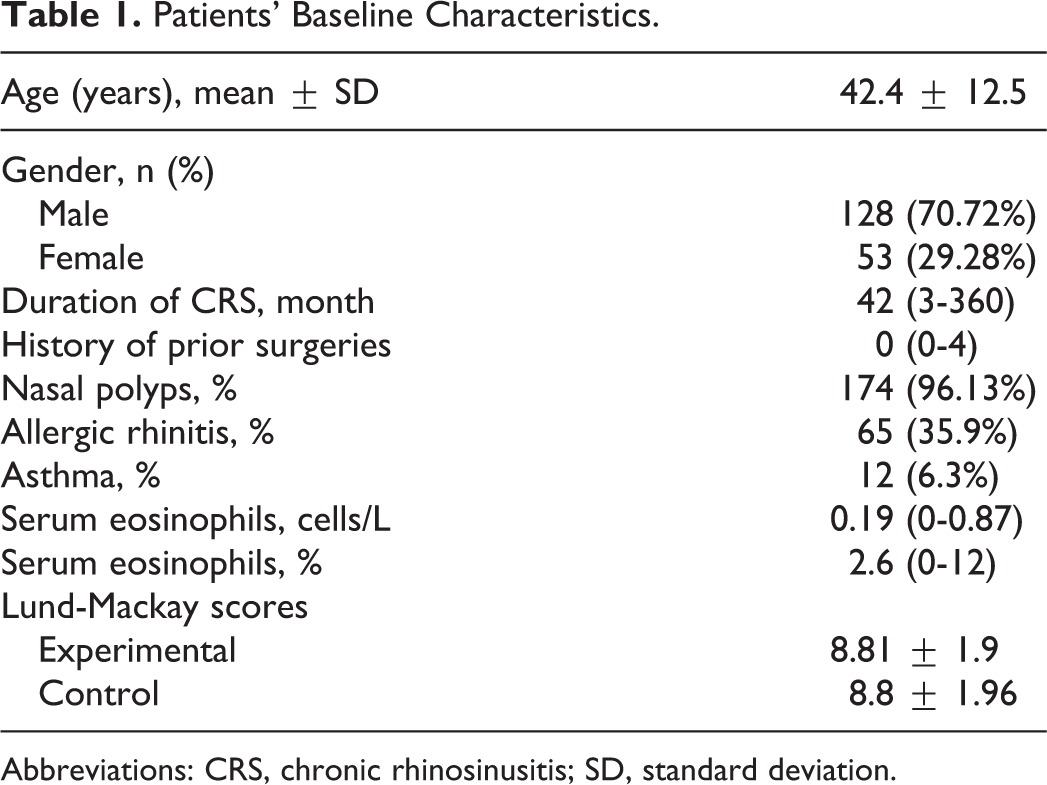
The patients’ characteristics are summarized in Table 1. A total of 187 patients consented to participate in the study from September 2014 to August 2015. A total of 183 patients met all eligibility criteria and were randomly assigned to receive a steroid-eluting sinus stent in one of the ethmoid sinuses and a Nasopore pack in the contralateral control side (Figure 1). A total of 181 patients completed the follow-up visits for the first 30 days and 175 patients completed the follow-up visits for the first 90 days. The mean age of patients was 42.4 ± 12.5 years, with 128 (70.72%) males and 53 (29.28%) females. The mean Lund-Mackay scores at baseline were 8.81 ± 1.9 on the stent side and 8.8 ± 1.96 on the Nasopore control side (Table 1).
Patients’ Baseline Characteristics.
Abbreviations: CRS, chronic rhinosinusitis; SD, standard deviation.
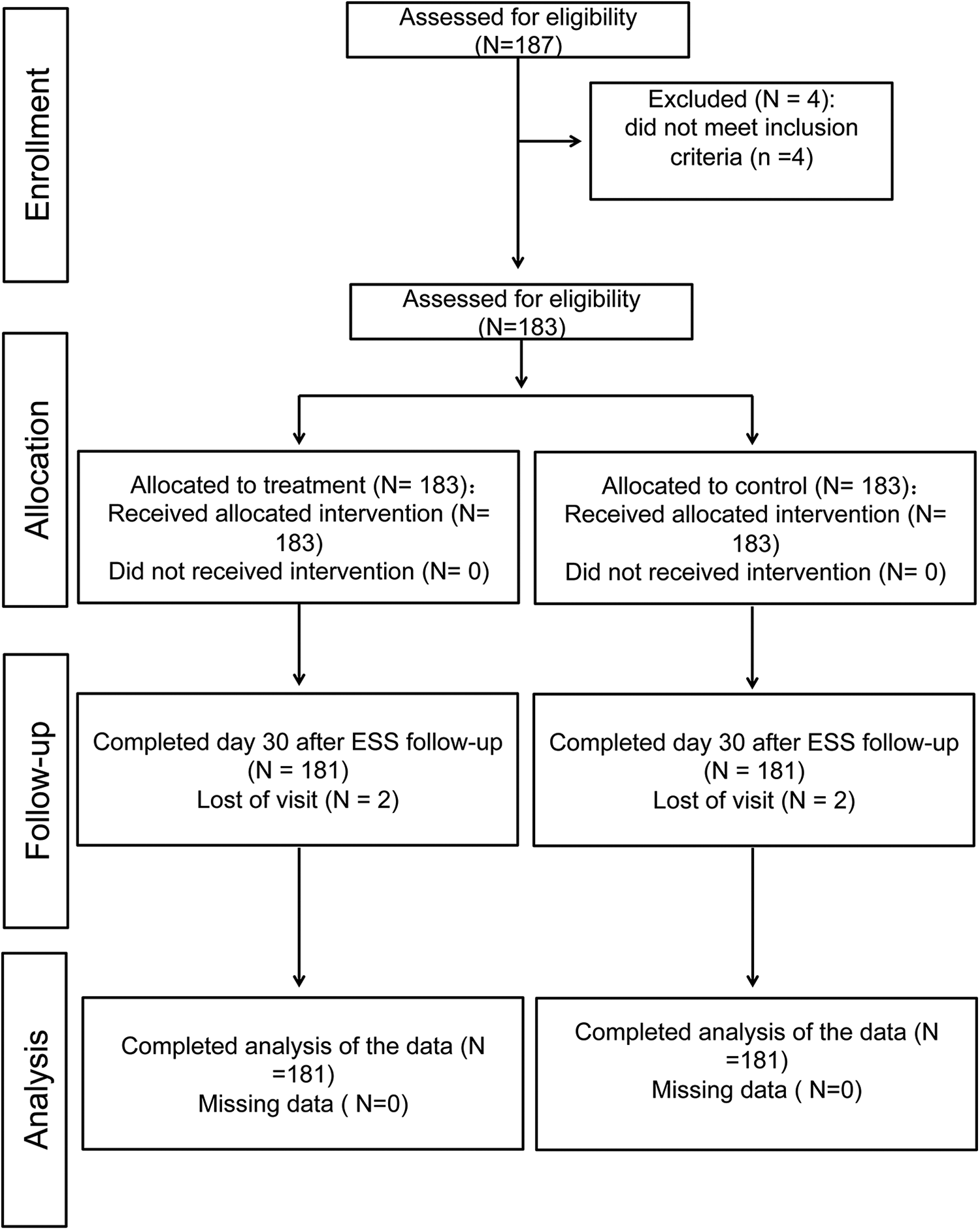
Consort diagram.
All patients underwent ESS, and no complications occurred. No adverse events occurred during the follow-up period.
Data Extraction and Accounting
On postoperative day 30, video endoscopies from all 181 patients were evaluated by an independent reviewer and a clinical investigator for the efficacy outcomes (Figure 2). On postoperative day 90, video endoscopies from all 181 patients were evaluated by the clinical investigator for the efficacy outcomes, whereas 57 of 181 patients could not be evaluated because the videos were not recorded during endoscopic examination (Figure 2).
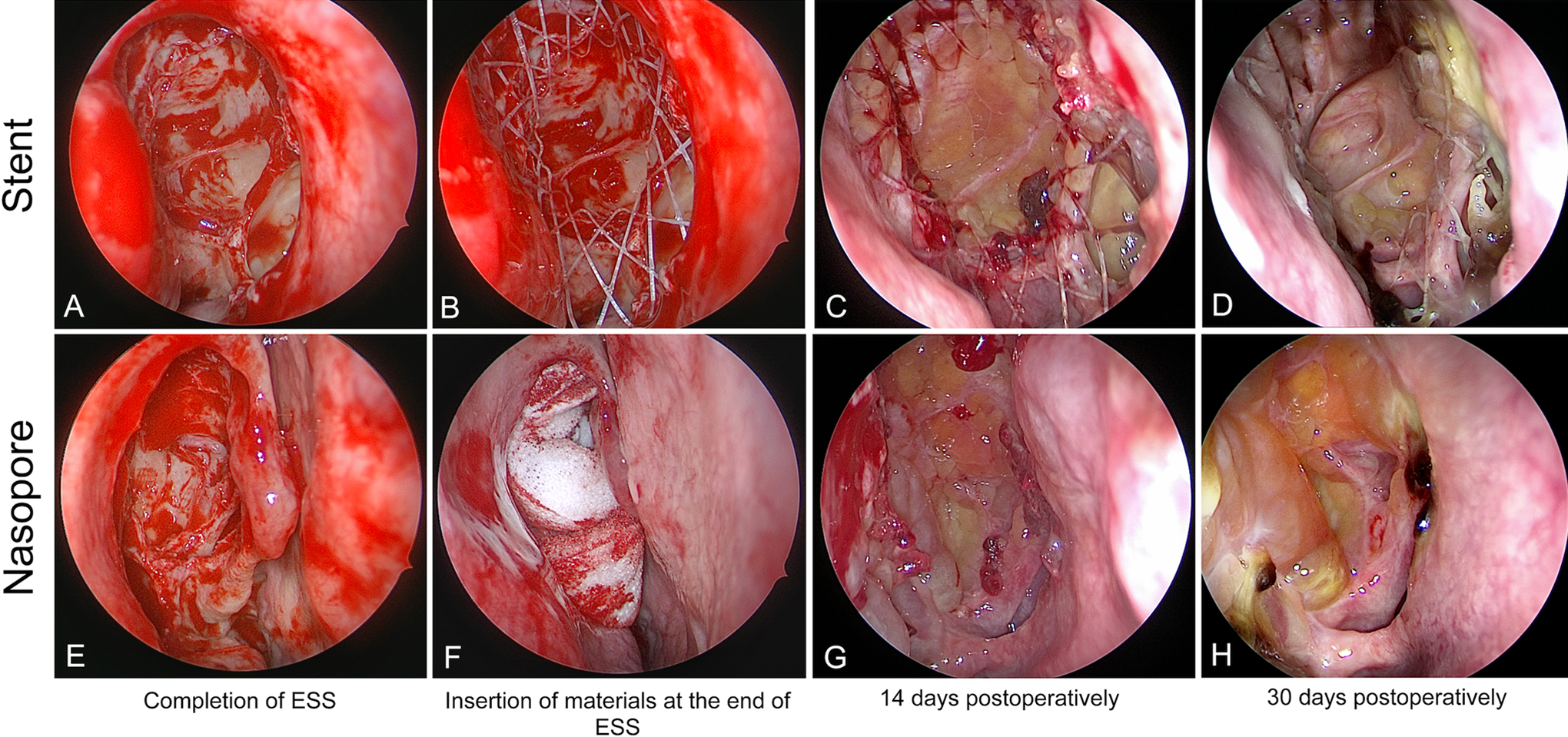
Endoscopic photographs from one study patient (A, E) show the ethmoid sinuses after endoscopic sinus surgery (ESS; postoperative day 0). (B) Steroid-eluting sinus stent placed in the left postoperative ethmoid sinus cavity and (F) Nasopore placed on the right side (postoperative day 0). (C, G) The same ethmoid sinuses on postoperative day 14. (D) Steroid-eluting sinus stent on the left at postoperative day 30. (H) Nasopore on the right at postoperative day 30.
Efficacy Outcomes
Need for Postoperative Surgical Interventions
On postoperative day 30, the efficacy outcome assessed by the clinical investigators showed that the percentage of patients who needed postoperative interventions in the ethmoid sinus was significantly lower on the steroid-eluting sinus stent sides at 14.38% and 33.7% compared with the Nasopore sides at 75% and 66.3%, respectively (both P < .0001; Table 2). The need for postoperative interventions on postoperative days 14 and 90 was not evaluated.
Endoscopic Efficacy Results at 30 and 90 Days After Surgery.
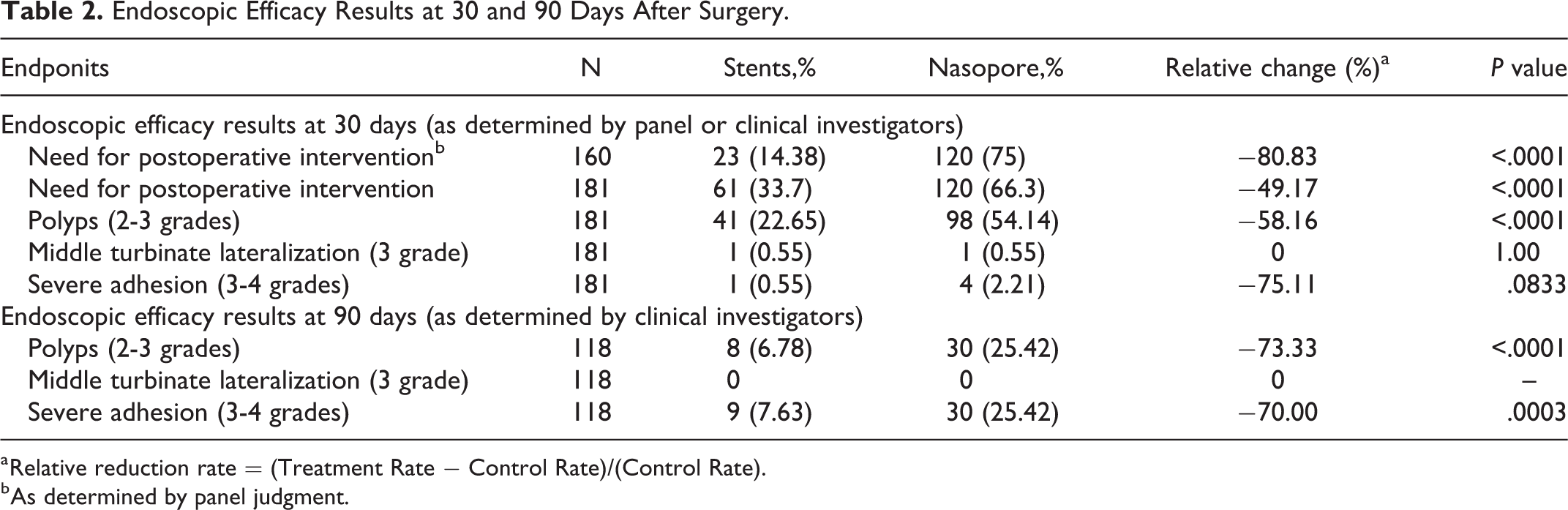
a Relative reduction rate = (Treatment Rate − Control Rate)/(Control Rate).
b As determined by panel judgment.
Endoscopic Evaluation
On postoperative days 14, 30, and 90, the percentage of cases with polyp formation was significantly lower on the steroid-eluting sinus stent sides at 16.57%, 22.65%, and 6.78%, respectively, compared with the Nasopore sides at 44.75%, 54.14%, and 25.42%, respectively (both P < .0001; Tables 2 and 3).
Endoscopic Efficacy Results at 14 Days as Determined by Clinical Investigators.
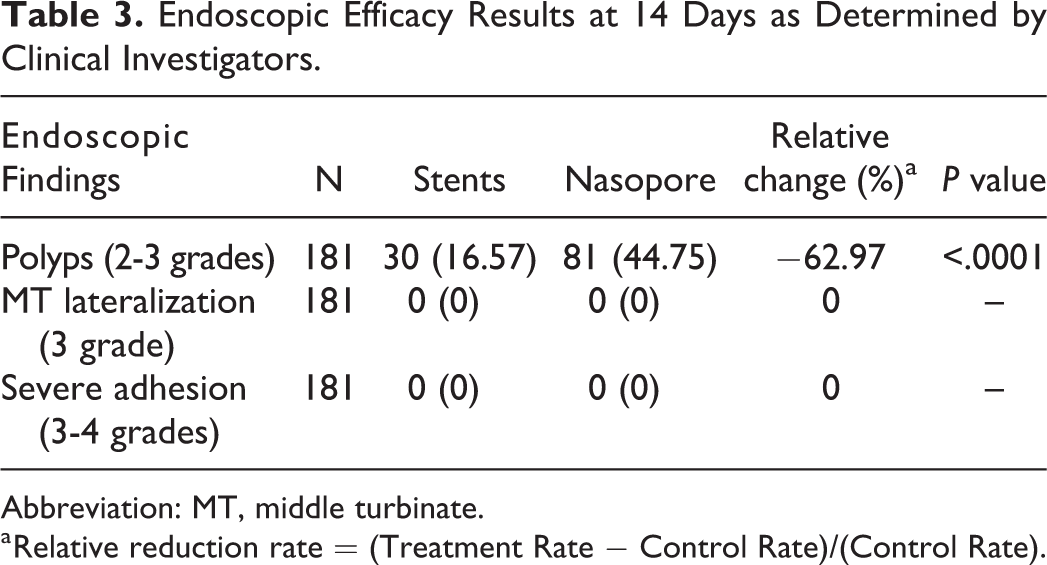
Abbreviation: MT, middle turbinate.
a Relative reduction rate = (Treatment Rate − Control Rate)/(Control Rate).
For the percentage of MT lateralization (grade 3), no significant difference was found between the steroid-eluting sinus stent sides and the Nasopore sides at all end points (Tables 2 and 3).
The percentage of severe adhesion (grades 3-4) was significantly lower on the steroid-eluting sinus stent sides than on the Nasopore sides at postoperative day 90 (7.63% vs 25.42%; P = .0003), whereas they were not significantly lower at postoperative days 14 and 30 (Tables 2 and 3).
Safety Outcomes
There were no significant differences in IOP (Table 4) and lens opacities between the baseline and at postoperative days 14, 30, and 90 on the 2 sides.
Preoperative and Postoperative Intraocular Pressure.
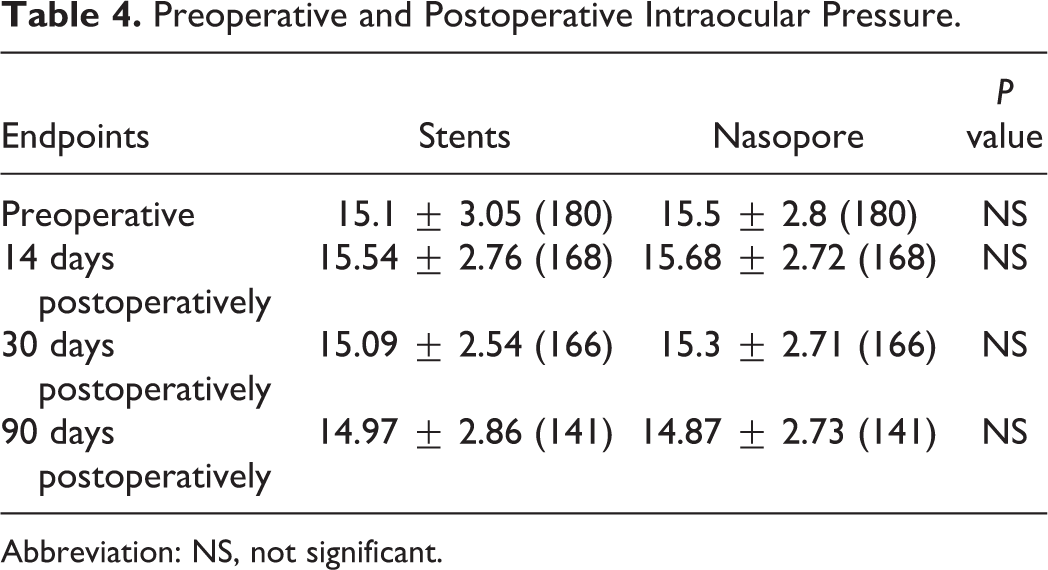
Abbreviation: NS, not significant.
There were 5 and 21 adverse events that were judged by clinical investigators as having an indeterminate and unrelated relationship to the sinus stent and steroids from the sinus stent (nasal pain, nose bleeding, headache, acute rhinosinusitis, etc).
Discussion
Our present study showed that the percentage of patients requiring postoperative interventions in the ethmoid sinus was significantly lower on the BISORB steroid-eluting sinus stent sides than on the absorbable Nasopore pack sides at days 30 after ESS. The evidence of the previous 2 RCTs that PROPEL steroid-eluting stents could reduce the need for postoperative interventions was supported or confirmed by our present study.9,10 Interestingly, previous studies showed the percentage of need for postoperative interventions (62.7% and 46.9%) that judged by independent reviewers was higher than that (41.8% and 31.4%) judged by clinical investigators.9,12 Similarly, the percentage of need for postoperative interventions (75%) judged by independent reviewers were higher than that (66.3%) judged by clinical investigators. The fact should consider that the study was not really blinded because the reviewer could recognize which side had the stent on 30 days postoperatively. As we know, postoperative severe new polyp formation and adhesion require postoperative intervention. We should point out that the anti-inflammatory effect of the steroid-eluting sinus stents could reduce the need for postoperative interventions by reducing the underlying inflammatory load that leads to mucosal edema and polyp formation.
Evidence from previous 2 RCTs showed that PROPEL steroid-eluting sinus stents are more effective in reducing polyp formation than non-steroid-eluting sinus stents 30 days after ESS.9,10 This finding was partially echoed in our present study which showed that the percentage of polyp formation was significantly lower on the BISORB steroid-eluting sinus stent sides compared with the absorbable Nasopore pack sides at 14, 30, and 90 days after ESS. These data indicate that steroid-eluting sinus stents are more effective in reducing polyp formation during the first 3 months of the postoperative period. Despite the fact that the steroid-eluting stents dissolved 30 days after placement into the sinuses, we postulated that the effects of the anti-inflammatory agents may last for 90 days or 3 months.
Nasal packing or stents are also used for internal stabilization after operations involving the cartilaginous-bony skeleton of the nose. 14 It is not surprised to found that there was no significant difference between steroid-eluting stent sides and the Nasopore sides in terms of the percentage of MT lateralization at all end points. Similarly, one RCT was performed by Rawl et al 11 comparing nonabsorbable Merocel packs to PROPEL steroid-eluting absorbable stents as middle meatal spacers after ESS in patients with CRS. This study showed no statistically significant difference in MT lateralization in favor of a steroid-eluting sinus stent compared to nonabsorbable-gloved Merocel pack. The findings of our present study indicated the steroid-eluting sinus stent does not confer the more beneficial effects over absorbable Nasopore packs in terms of internal stabilization.
The steroid-eluting sinus stents have been shown to be a safe material that inserts into the sinus after ESS by numerous RCTs8-10,12,15,16 and a previous retrospective study. 3 Our results are consistent with the findings of previous RCTs.8-10,12 In our present study, there were no related adverse events (ie, crusting, nose bleeding) and no clinically significant changes from baseline in terms of IOP. Our study showed that BISORB steroid-eluting sinus stents were safe and effective in improving the postoperative outcomes.
Limitations
We cannot deny our present study has some limitations. The independent reviewers and clinical investigators could tell the difference between the stents and the Nasopore packs within the nose cavities within 30 days postoperatively. That is the reason we invited independent reviewers to evaluate the outcome measurements on 30 days postoperatively but not 90 days postoperatively. Additionally, we failed to investigate whether the proportion of patients requiring revision ESS was reduced by using the steroid-eluting sinus stents. A further prospective cohort study with long postoperative outcome is needed.
Conclusion
Our study demonstrated significant improvement in the early postoperative outcomes by reducing the need for postoperative surgical intervention and polyp formation using the steroid-eluting sinus stents when compared with the absorbable Nasopore packs. The question from our previous Cochrane systematic review has been answered by the findings from our present RCT. The evidence from this RCT demonstrates that the steroid-eluting sinus stents are superior to the absorbable Nasopore packs. Steroid-eluting stents and the Nasopore packs were each effective in preserving the ethmoid sinus patency and preventing MT lateralization. A further prospective cohort study with long-term postoperative outcomes is warranted.
Supplemental Material
sj-pdf-1-ear-10.1177_0145561320947632 - Comparison of Bioabsorbable Steroid-Eluting Sinus Stents Versus Nasopore After Endoscopic Sinus Surgery: A Multicenter, Randomized, Controlled, Single-Blinded Clinical Trial
sj-pdf-1-ear-10.1177_0145561320947632 for Comparison of Bioabsorbable Steroid-Eluting Sinus Stents Versus Nasopore After Endoscopic Sinus Surgery: A Multicenter, Randomized, Controlled, Single-Blinded Clinical Trial by Zhenxiao Huang, Bing Zhou, Dehui Wang, Hongrui Zang, Huankang Zhang, Huan Wang, Shenqing Wang, Lei Cheng, Jinrang Li, Wenying Wu, Huifang Zhou and Huili Wu in Ear, Nose & Throat Journal
Footnotes
Authors’ Note
Informed consent was obtained from all individual participants included in the study.
Acknowledgments
The authors thank Puyi (Shanghai) Biotechnology Co., Ltd, China, for supporting this trial. The authors thank the following independent reviewers: Jianbo Shi, the First Affiliated Hospital, Sun Yat-sen University, Lei Chen, Chinese PLA General Hospital, and Wei Lv, Peking Union Medical College Hospital.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This study was supported by Puyi (Shanghai) Biotechnology Co., Ltd and National Natural Science Foundation of China (NO.81770977).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
