Abstract
Combined small-cell carcinoma and squamous cell carcinoma of the larynx is an exquisitely rare and underreported primary tumor the head and neck region, with an English literature review revealing only 17 documented cases. There is limited information on how best to treat these patients oncologically, given the low number of reported cases. A subset of the reported cases also detail a unique local spread of this combined carcinoma, further obscuring the clinical picture of these patients. Here, we detail an 18th case, with nodal metastasis of only one component of the primary tumor, and discuss the published literature surrounding this etiology.
Introduction
Laryngeal cancer is the third most common malignancy seen in the head and neck region worldwide, with a male to female ratio of approximately 7:1. 1 A vast majority of these tumors are of squamous cell origin, typically cited between 85% and 95%, with the glottis being the most common site of involvement. Despite this, there are a multitude of other neoplasms that can affect the larynx, including sarcomas, minor salivary gland neoplasms, and neuroendocrine neoplasms (NENs). A recent study found that non-squamous cell carcinomas (SCCs) represent approximately 1% of all malignant laryngeal cancers and that NENs are the most common of those non-SCCs, representing approximately 37% of those cases. 2 Small-cell neuroendocrine carcinoma (SCNEC) of the larynx is considered the most aggressive and malignant and second most common of all neuroendocrine tumors.
Small-cell neuroendocrine carcinomas are more common in males and typically arise in the supraglottis. Sometimes, these tumors also have an associated squamous cell component and are categorized as combined or composite carcinomas. These are exceedingly rare in the literature, with approximately 17 cases reported to date. 3 The majority of these reported cases have been of males in the sixth and seventh decades, with some reported cases showing unique metastatic spread of the individual components of the tumor. 3 Current literature reveals sparse data on how these patients have been managed as well as how the pathologic diagnosis of these tumors can be exceedingly difficult given their rarity.
Here, we present a case of combined small-cell carcinoma and SCC of the supraglottis in a male patient in the sixth decade of life, where the patient was found to have positive nodal disease in the neck with only one component of the carcinoma. Furthermore, the literature is reviewed, and the management of this patient is discussed.
Case Report
A 59-year-old male with a past medical history significant for multiple cardiopulmonary arrests, cardiomyopathy, diabetes, and chronic obstructive pulmonary disease (COPD) presented to a community-based hospital complaining of a left-sided neck mass that has been present for approximately one month. The patient also had associated severe dysphagia and odynophagia, in addition to shortness of breath that he attributed to his long-standing history of COPD, but denied dysphonia. The patient had an approximate 100-pack-year history of smoking, stated that he had quit 2 years prior to presentation, and denied any significant alcohol use. He also admitted to daily marijuana use. In addition, there was associated unexplained weight loss, despite recent increased oral intake.
Physical examination revealed a large left-sided neck mass in level 2 of the neck that was firm and nontender to palpation. Several other lymph nodes were noted along the left cervical chain. Flexible nasolaryngoscopic examination revealed a thickened and erythematous left aryepiglottic fold and laryngeal surface of the epiglottis, with no discrete mass appreciated. No other masses or lesions were identified in the upper aerodigestive tract, and the vocal cords were found to be fully mobile bilaterally. A computed tomography (CT) scan of the neck showed a dominant left-sided lymph node in the jugulodigastric region measuring 4.0 × 3.2 × 2.4 cm, as well as asymmetric thickening of the left aryepiglottic fold measuring 1.4 × 1.3 cm.
Decision was made to perform a fine needle aspiration of the left-sided lymph node, and after pathologic analysis, the patient was deemed to have a poorly differentiated carcinoma with no further diagnosis made secondary to inadequate specimen. Before a second attempt at fine needle aspiration could be made, the patient left the hospital against medical advice. He then returned to the hospital approximately one month later for similar complaints, at which time he was brought to the operating room for direct laryngoscopy with biopsy as well as repeat fine needle aspiration.
Microscopic analysis of the specimens showed that the biopsy of the aryepiglottic fold demonstrated 2 distinct histologies, with the predominant histology consistent with basaloid SCC, confirmed with positive immunohistochemical staining for CK5/6 and p63 (Figure 1). The other component was consistent with SCNEC, confirmed with positive staining for synaptophysin (Figure 1). Both components stained positive for p16. Immunohistochemical staining of the fine needle aspirate showed positivity for CK88 and synaptophysin, consistent with SCNEC (Figure 2). The basaloid SCC component of the tumor, confirmed by the lack of CK5/6 positivity, was not present in the lymph node sampling (Figure 2).
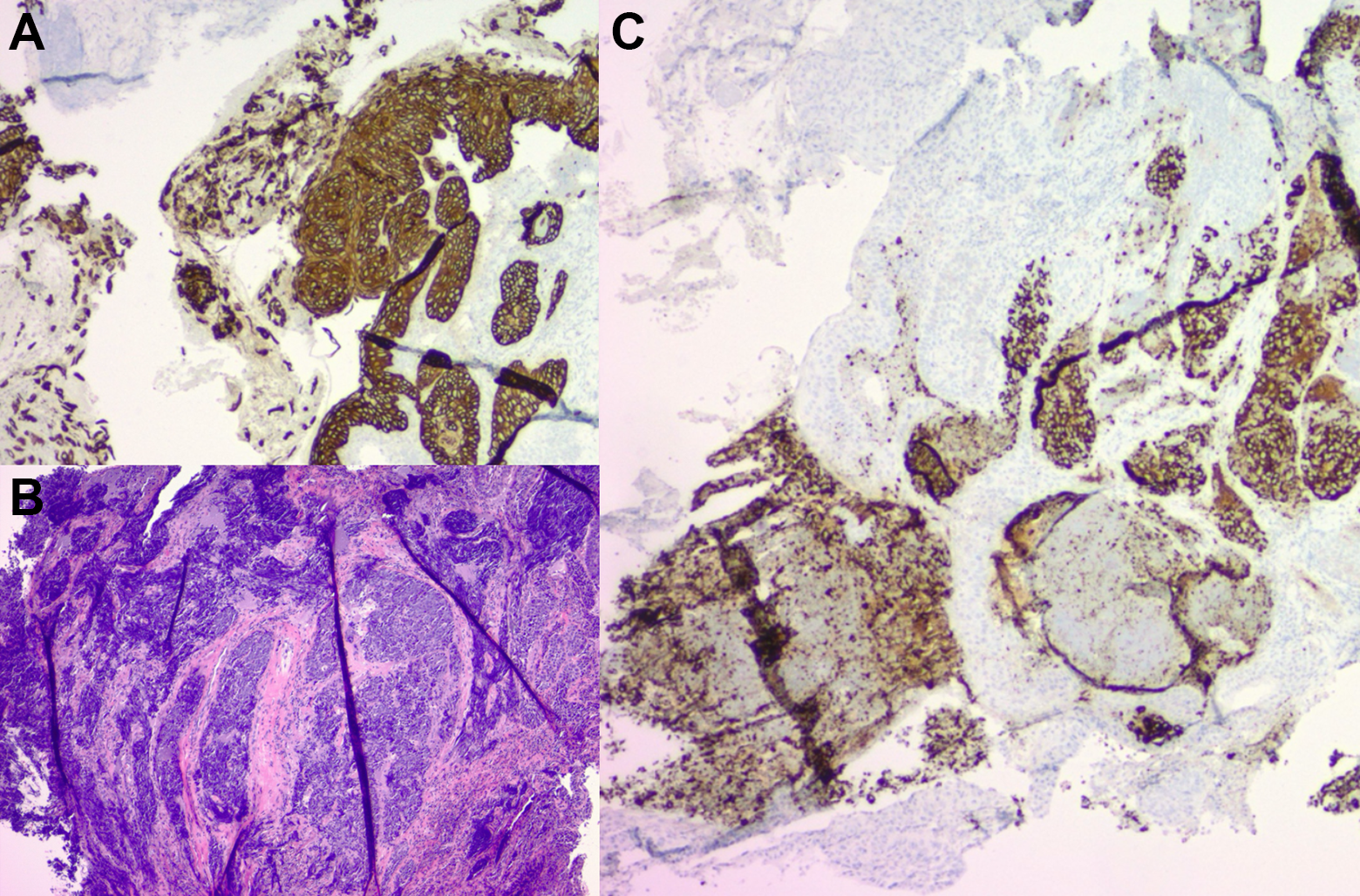
Biopsy of left aryepiglottic fold. Panel A (top left): Immunohistochemical staining of left aryepiglottic fold specimen with CK5/6 showing strong positivity. Panel B (lower left): Hematoxylin and eosin (H&E) stain of left aryepiglottic fold biopsy showing both squamous and small-cell components. Panel C (right): Immunohistochemical staining of left aryepiglottic fold specimen with synaptophysin showing strong positivity.
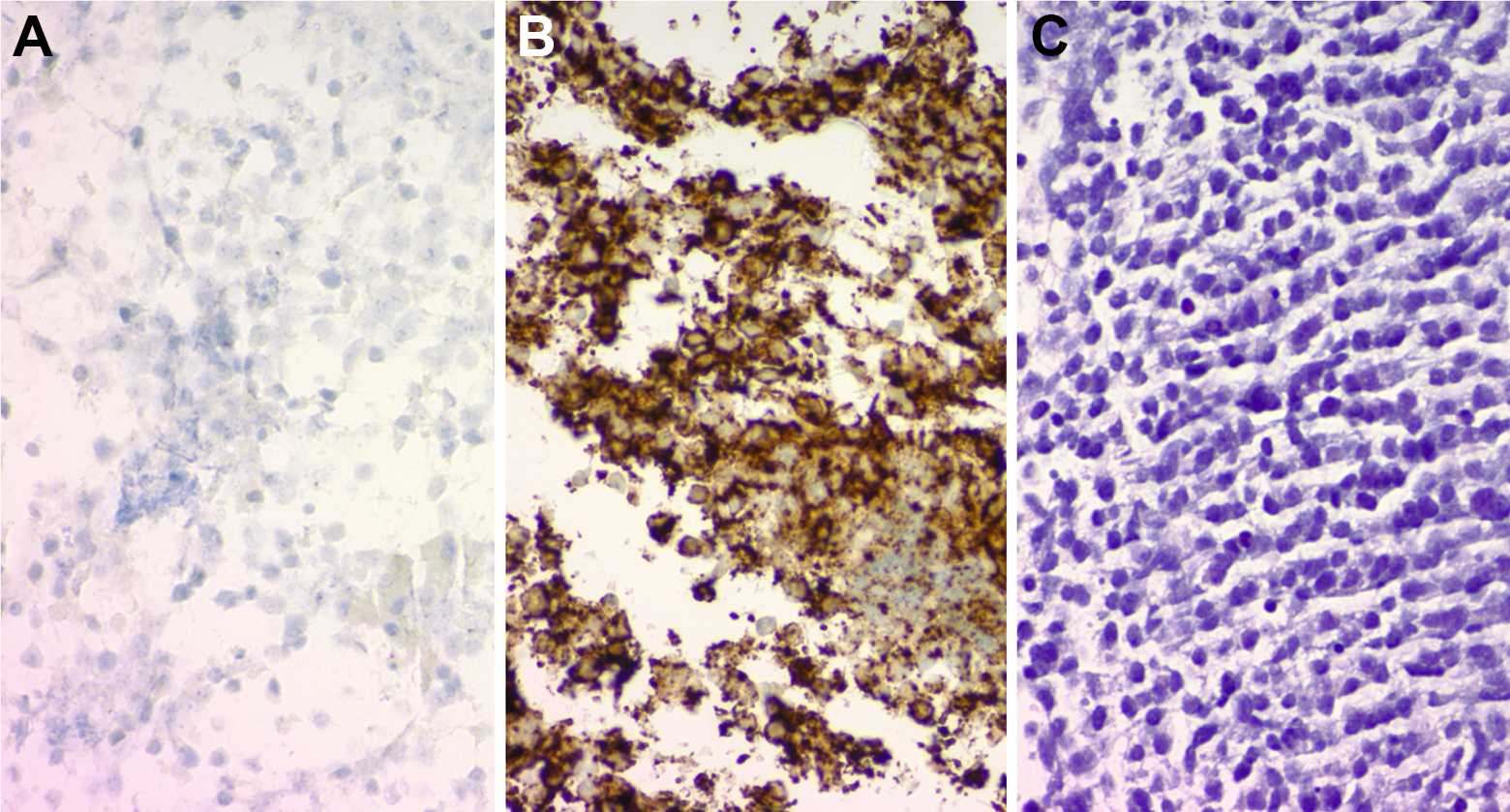
Biopsy of left level IIA lymph node. Panel A (left): Immunohistochemical staining of left level IIA lymph node with CK 5/6 showing no response. Panel B (center): Immunohistochemical staining of left level IIA lymph node with synaptophysin showing strong positivity. Panel C (right): Hematoxylin and eosin (H&E) stain of the left level IIA lymph node showing small-cell component.
After consultation with both medical oncology and radiation oncology, the patient decided to proceed with chemotherapy alone, despite recommendation to undergo dual therapy, and was started on carboplatin and paclitaxel therapy. The patient has admittedly missed several chemotherapy appointments, and a recent CT scan of the neck, performed approximately 3 months after diagnosis, has showed interval enlargement of the supraglottic mass as well as the left neck adenopathy.
Discussion
Small-cell neuroendocrine carcinomas of the larynx are exceedingly rare, most frequently cited as comprising around 0.5% of all laryngeal tumors. The combined subtype including a squamous cell component is considered to be even rarer. Studies have suggested that the histogenesis of this combined tumor may be explained by one common cell that differentiates into divergent pathways, one with Kulchitsky cells, and the other with squamous cells. 4 To date, an English literature review revealed only 17 published cases, with ours being the 18th. A majority of the reported cases, including ours, involved the supraglottic region. 3 These tumors can be difficult to diagnose as a thorough pathologic analysis of the specimen as well as meticulous immunohistochemical staining must be performed for an accurate diagnosis. This may account for the sparsity of literature on this tumor.
The first case of combined small-cell carcinoma and SCC of the larynx was described and reported in 1985 and later coined “combined small-cell carcinomas of the larynx.” 5 This article was the first to describe the presence of 2 distinct cell lineages, both small cell and squamous cell, growing synchronously in the same laryngeal tumor. A more recent study by Jaiswal and Hoang described a combined small-cell carcinoma of the larynx diagnosed in a 41-year-old male in the glottic region. 6 Barbeaux et al published a study describing 2 patients diagnosed with combined small-cell carcinoma and SCC, one diagnosed in the glottis and the other found in the subglottis. 7 A most recent case published by Aggarwal et al detailed a combined small-cell carcinoma and SCC diagnosed in the supraglottis and glottic regions. 3 Review of the cases reported in the literature revealed that a majority of the nodal disease was found to be of the small-cell component, which aligns with what we found in our case. 7 Further analysis revealed that the clinical course appeared to be rapidly progressive, with the majority of patients dying within 2 years of diagnosis.7,8
The sparsity of literature describing this entity may be related to its relative difficulty in diagnosis; however, the diagnosis itself is entirely dependent on meticulous immunohistochemical staining. A recent study sought to update the current literature on most frequently used immunohistochemical stains for head and neck cancers and found that for basaloid SCC, the most applicable immunohistochemical stains were p63 and CK5/6. 9 Both of these immunohistochemical stains were found to be positive in our specimen. The authors also stated that, although p63 is also positive in adenoid cystic carcinoma, the staining patterns between the 2 are remarkably different, allowing for further differentiation. 9 This finding was further confirmed in another study, where 100% of keratinizing SCCs examined in nonoropharyngeal subsites showed positivity for CK 5/6. 10 Another recent study reviewing the staining characteristics of NENs again demonstrated that the most reliable stains are typically synaptophysin and chromogranin A, of which our specimen stained positive for synaptophysin. 11 The article also mentioned that synaptophysin and chromogranin A are positive immunohistochemical stains in NENs arising from anywhere in the body and that the neuroendocrine tumors arising in the head and neck region are of epithelial origin, which is why our specimen also stained positively for CK88. 11 Of note, however, another study demonstrated that the 2 most common immunohistochemical stains for NENs, synaptophysin and chromogranin A, are only present in head and neck specimens 41% and 18% of the time, further complicating the diagnosis of these tumors. 12
The patient presented in this article illustrates the difficulty in obtaining a clearly delineated diagnosis, as the initial fine needle aspiration revealed inconclusive results and the repeat fine needle aspiration, as well as laryngeal specimen, required detailed immunohistochemical staining. During his initial post diagnosis period, the patient was counseled to undergo combined treatment with chemotherapy and radiation, although he opted for chemotherapy alone. Initiation with carboplatin and paclitaxel was based on the oncologist’s experience treating small-cell lung cancer, although concurrent treatment with radiation most likely would have resulted in better local control. The patient also declined to undergo positron emission tomography staging for metastatic disease, further obscuring his overall oncologic presentation, as study by Ferlito et al detailed that more than 90% of patients with small-cell carcinoma of the larynx develop distant metastases. 13 A repeat CT scan of his neck performed approximately 3 months after diagnosis showed interval increase in the aryepiglottic mass, as well as increase in cervical adenopathy, indicating that chemotherapy alone was likely not adequate for treatment of combined small-cell carcinoma and SCC of the larynx in his case. Of course, further obscuring his treatment regimen is the fact that he admitted to multiple missed chemotherapy appointments. The study published by Barbeaux et al included 2 patients, both of whom underwent concomitant radiation with platinum-based chemotherapy and concluded that this is likely the best approach for nonmetastatic disease. 7 That study also concluded that the radiation dose should not be reduced beneath 70 Gy, in order to achieve local control of the squamous cell component.
Our study looked to add to the relative sparsity of literature describing this rare tumor of the larynx, as well as detail the treatment plan that the patient has underwent to date. The patient presented in the study emphasizes the necessity of detailed immunohistochemical staining of head and neck tumor specimens, in addition to the proposed need for concomitant chemotherapy and radiation treatment. Further investigation into the radiosensitivity of these tumors and the optimal chemotherapeutic regimen is required, although given their rarity, these data may be difficult to obtain.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
