Abstract
Objective:
To investigate the association between genetic polymorphisms and sudden sensorineural hearing loss (SSNHL). Most of the SSNHL cases still remain idiopathic, and several etiopathogenetic hypotheses, including a genetic predisposition, have been proposed.
Methods:
A literature review was conducted using different databases: Medline/PubMed, EMBASE, and CINAHL, according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. All databases have been searched from May 2016 to April 2020.
Results:
Genetic susceptibility could represent a key element in the pathogenesis of SSNHL. A number of genetic polymorphisms related to (1) inner ear microvascular disease and endothelial dysfunction and (2) to inner ear oxidative stress and inflammation have been addressed in the current literature.
Conclusions:
The potential identification of a genetic profile related to SSNHL could provide a more accurate prognostic evidence of idiopathic SSNHL (ISSNHL), offering to the patients not only early-prevention strategies but eventually information on various inheritance modalities.
Keywords
Introduction
Sudden sensorineural hearing loss (SSNHL) is defined as a unilateral or bilateral sensorineural hearing defect of ≥30 dB in at least 3 contiguous frequencies, within a time window of 72 hours. 1 In the SSNHL assessment, it is always necessary to rule out possible causes such as acoustic neuroma, stroke, malignancies, noise exposure, and ototoxic drugs 1 ; however, more than 90% of SSNHL cases still remain idiopathic since a specific etiology cannot be identified. 1
Several pathophysiological theories of idiopathic SSNHL (ISSNHL) have been proposed, such as inner ear viral infection, 2,3 inner ear vascular damage, 4 endolymphatic hydrops, destruction of the cochlear labyrinthine membranes, 5,6 immune-mediated disorders, 7,8 and electrolyte imbalance of intracochlear fluids. 9
Various genetic factors leading to SSNHL have also been addressed, 10 although there is little evidence in the literature for a genetic origin of SSNHL. 11 However, the influence of some genes, such as methylenetetrahydrofolate reductase, 12,13 protein kinase C eta, 14 complement factor H 15 , and lymphotoxin α, 16 on the SSNHL onset has been already reported. The majority of data in the literature refers to genes related to inner ear microvascular disease and/or endothelial dysfunction or to inner ear inflammation and/or oxidative stress. 10 The present article aimed to review the current literature and to investigate the association between genetic polymorphisms and SSNHL.
Methods
The PubMed, Embase, and Cinahl databases were searched from May 2016 to April 2020. Full-text articles were obtained in cases where the title, abstract, or key words suggested that the study may be eligible for the present review. The search was conducted according to Preferred Reporting Items for Systematic Reviews and Meta-Analyses criteria/guidelines (http://www.prisma-statement.org/): It was carried out independently and was restricted to English language papers. Additional relevant papers for the review were also identified from references in the published literature.
For the database queries, the following medical subject heading terms were used: inner ear; ISSNHL; SSNHL; genetic polymorphisms; mutation; genetic association; genetics.
The queries resulted in a total of 25 papers. The literature selection process is depicted in detail in Figure 1. After the application of the exclusion criteria (see paragraph below), the total number of papers was reduced to 18.
Several notes for the reader regarding the selection process (please consult Figure 1):
For the first step, the inclusion criteria were defined as papers from clinical series and reviews. The exclusion criteria were defined as: (1) not availability of a full text; (2) manuscripts not in the English language; and (3) case reports. For the second step, the inclusion criteria were defined as: (1) papers from clinical series, with an adequate number of assessed patients (n > 20); (2) review papers published in relevant journals showing rigorous methods and a rigorous reporting of results.
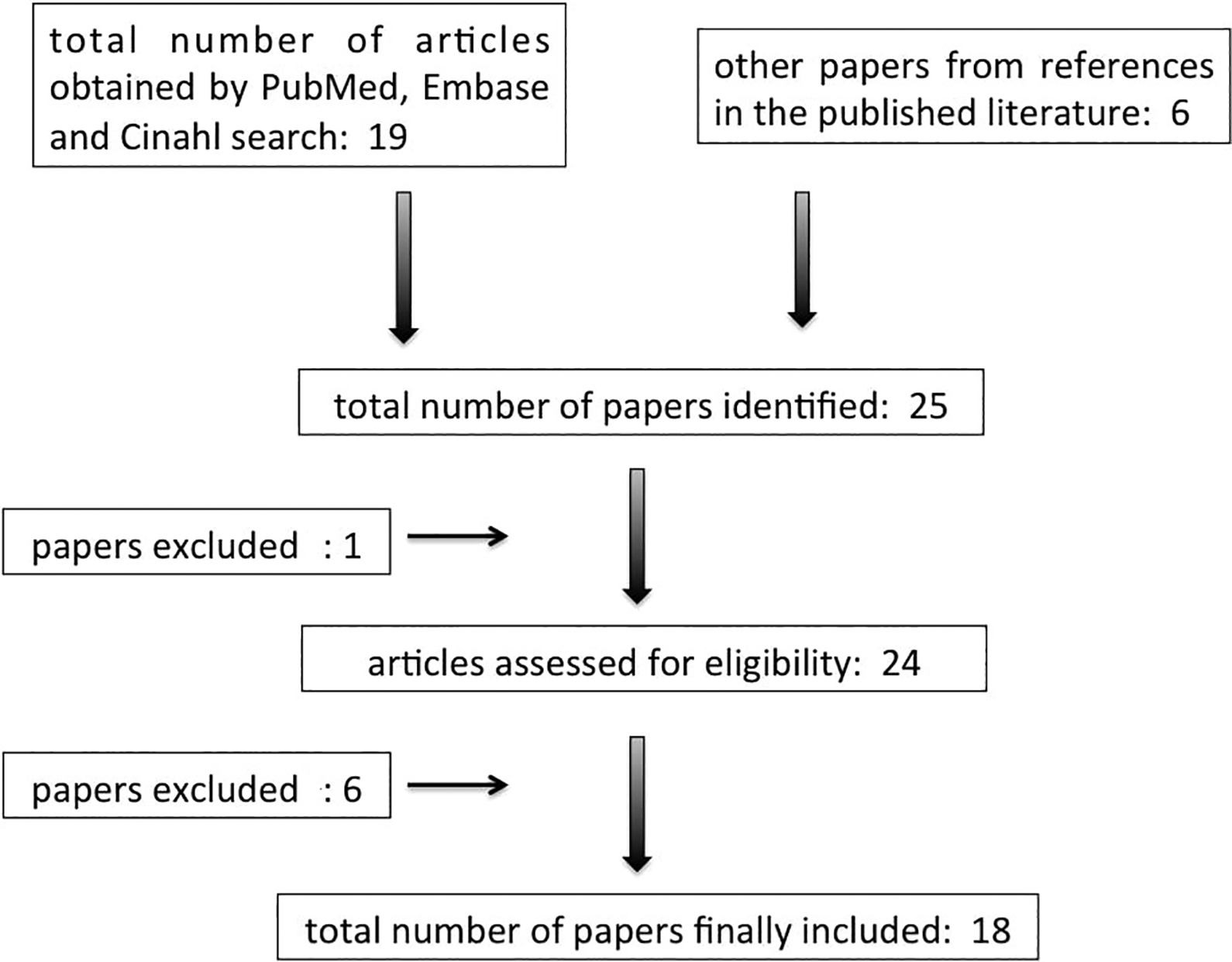
Literature evaluation and selection, according to PRISMA criteria (http://www.prisma-statement.org/). See the text for additional information on the inclusion criteria. PRISMA indicates Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Results
Genetic Polymorphisms and Inner Ear Microvascular Disease and/or Endothelial Dysfunction
Microvascular diseases may hamper and damage the inner ear, due to its high metabolism-dependent function and its terminal blood flow, supplied by the labyrinthine artery. 17 Endothelial factors (both pro-aggregants and anti-aggregants) may play a central role in the cochlear homeostasis. 18 Among the ISSNHL pathophysiological hypotheses of the inner ear a vascular damage, a micro-thromboembolism, a micro-hemorrhage, and a vascular spasm of the labyrinthine artery have been proposed. 19
In their case–control study, Uchida et al 20 investigated the relationship between the onset of SSNHL and endothelin-1 (EDN1), which is one of the main endothelial mediators showing a vasoconstriction function. Data in the literature report that several EDN1 polymorphisms are correlated with numerous disorders such as thrombotic diseases, including atherosclerosis. 21 Uchida et al suggested that the recessive genotype of EDN1 p.Lys198Asn polymorphism was significantly associated to SSNHL onset and hearing threshold level. 20 The recessive EDN1 p.Lys198Asn homozygosis has been reported to correlate with an increased SSNHL risk and a mild severity of the disease.
Endothelial nitric oxide synthase (eNOS) produces nitric oxide (NO) inducing a vasodilation of the smooth muscle cells; the eNOS p.Glu298Asp single-nucleotide polymorphism is correlated to cardiovascular diseases. 22,23 Yazdani et al 24 found a significant association between eNOS and SSNHL in a case–control study, suggesting that NO plays a crucial role in cochlear neurotoxicity.
Castiglione et al 25 hypothesized a stria vascularis mechanism, related to the supplementation of divalent ions into the endolymphatic space, which affects the volume and the acidification of the endolymph. In particular, the authors focused on the Fe2+ metabolism and showed a significant association between ISSNHL and the c.−8C>G polymorphism of Ferroportin gene 1 (FPN1). Their data show that patients carrying this polymorphism have an increased risk of developing this disease in adulthood. 25,26
Genetic Polymorphisms and Inner Ear Inflammation and/or Oxidative Stress
Oxidative stress or a cascade of inflammatory events directly involving the hair cells, should be assessed as a possible pathogenetic mechanism leading to ISSNHL. The latter stems from the generation of reactive oxygen species (ROS), which induce apoptosis into inner ear hair cells. 27
The association between genetic polymorphisms of prothrombotic and inflammation mediator genes and SSNHL has been studied. 12 Genes such as superoxide dismutase 1 (SOD1) 28 and factor V Leiden 12 have been assessed as possible factors involved in the pathogenesis of SSNHL. Superoxide dismutase is an important protective antioxidant system of the eukaryotic and of the inner ear cells, providing protection against ototoxic substances. 29,30 Superoxide dismutase genetic alterations have been linked to several diseases. Kitoh et al 28 investigated the correlation between a specific polymorphism of SOD1 and suggested that SOD1 rs4998557 could be associated to SSNHL.
Teranishi et al 31 investigated the association between the risk of SSNHL and 5 oxidative stress-related genetic polymorphisms, in a case–control study. They assessed the relationship between the recovery of hearing loss and the oxidative stress-related polymorphisms in SSNHL patients. The T allele of paraoxonase 1 (PON1; rs854560) was more frequent in SSNHL cases presenting a good hearing level recovery, compared to cases where patients recovered less. 31
The V Leiden factor is a variant of the human V factor, associated to a hypercoagulability state and thromboembolic disorders. 32 The meta-analysis performed by Shu et al 33 on the association between the V Leiden factor p.Arg534Gln mutation and SSNHL in an Italian population, did not show any significant results. The evidence about the real role of V Leiden factor polymorphisms on the SSNHL susceptibility remains to be elucidated.
Methylenetetrahydrofolate reductase is involved in the transformation process of homocysteine into methionine 34 ;the MTFHR p.Ala222Val mutation decreases the enzyme’s function and the homozygosis status represents the most common genetic cause of hyperhomocysteinemia, 35 a condition predisposing thrombotic events. In a recent case–control study in an Iranian population, Hamidi et al 36 concluded that the MTFHR p.Ala222Val polymorphism has a significant impact on SSNHL development.
Koide et al 37 reported a significant association between the mitochondrial uncoupling protein 2 (UCP2) polymorphism and the risk of SSNHL onset. UCP2 plays an important protective role against free radicals in the inner ear. Manche et al 38 has reported a correlation between UCP2 rs660339 polymorphism and presbycusis.
Polymorphisms of inflammation-related genes have been widely investigated in patients, affected by SSNHL, as possible pathogenetic factors. According to the Merchant’s stress response theory, 39 the aberrant activation of intracellular oxidative pathways in the inner ear’s neuroepithelium by inflammatory cytokines may hamper the cochlear homeostasis and cause SSNHL. Interleukins (ILs) are cytokines involved in the immune system activation and regulation, while adhesion molecules, such as intercellular adhesion molecule-1 (ICAM-1), are involved in the inflammatory response. Tian et al 40 conducted a case–control study investigating the role of IL-6 and ICAM-1 polymorphisms in SSNHL. They suggested that the IL-6 c.572C>G polymorphism is significantly related to the prevalence of SSNHL; furthermore, the combined presence of both IL-6 c.572C>G and ICAM-1 p.Lys469Glu polymorphisms was found to be significantly associated to an increased risk of SSNHL development.
Genetic polymorphisms have been investigated as possible prognostic factors after oral steroid therapy. 41 Kitoh et al 41 analyzed the correlation between genes related to oxidative stress or steroids receptors and ISSNHL hearing prognosis. They found a relationship between the presence of glutathione-disulfide reductase (GSR) rs2251780, rs3779647, nitric oxide synthases 3 (NOS3) rs1799983 polymorphisms, and a poor steroid-therapy outcome. The genetic polymorphisms reviewed in this study are summarized in Table 1.
Main Polymorphisms Involved in the Pathogenesis of ISSNHL.

Abbreviation: SSNHL, sudden sensorineural hearing loss.
Discussion
Sudden sensorineural hearing loss should be considered a multifactorial disease, due to a combination of environmental and genetic factors. 28 Data in the current literature show that several genetic polymorphisms correlated with oxidative stress, inflammation, thrombosis, and blood vessel permeability are associated to SSNHL predisposition and development. 10,12,20,24,28,36,37,40,41 On the basis of this evidence, it could be hypothesized that genetic factors could play a role in maintaining the delicate balance between ROS and antioxidants, pro-inflammatory and anti-inflammatory factors, pro-aggregants, and antiaggregants; since the balance among these factors is important for the inner ear homeostasis, their disruption could be a factor contributing to the ISSNHL onset. Furthermore, a possible link between SSNHL and major cardiovascular diseases, such as stroke and myocardial infarction, has been previously highlighted. 42 -44 Thus, investigating the family medical history, kinship degree, and phenotype should always be pursued as a possible helpful diagnostic tool 45 ; moreover, a careful investigation of cardiovascular risk factors among patients affected by SSNHL should always be recommended.
The data in the literature do not elucidate whether a genetic evaluation could be considered valuable in the clinical assessment of ISSNH; genetic factors should be investigated as possible etiopathogenetic causes, particularly in the presence of a positive familiar history for ISSNHL, in bilateral ISSNHL and in pediatric patients. 1,46,47
Genetic polymorphisms have also been shown to be related to hearing prognosis. 41 The data in the literature suggest that some genetic factors could confer a resistance to treatment, particularly to steroids. 34 The identification of the ISSNHL susceptibility genes could be important in the prediction of the treatment outcome and possibly could help physicians in choosing the most suitable therapeutic option (ie, tailored therapy). 10
The fact that the majority of the available studies in the literature used small sample sizes to assess specific ethnic groups suggests that additional research is needed to elucidate specific arguments in order to better understand the relationship between ISSNHL and genetic polymorphisms. Nowadays, it is only possible to speculate on the clinical significance of genetic polymorphisms in patients affected by SSNHL: Further studies are necessary in order to identify reliable genetic risk factors for SSNHL predisposition.
Finally, the possibility to recognize patients with genetic risk factors for SSNHL could be advantageous in planning effective preventive measures and intervention strategies.
Conclusion
Genetic susceptibility could represent a key element in the pathogenesis of SSNHL. Presently, the genetic evaluation of SSNHL could be included only in the clinical assessment of selected cases (ie, positive familial history for ISSNHL, bilateral and idiopathic SSNHL, etc).
The potential identification of a genetic profile correlated to SSNHL and the consequent genetic counseling could: (1) provide the patient with additional prognostic evidence on ISSNHL, and treatment outcomes; (2) offer early prevention strategies; (3) provide the patient with information about inheritance modalities; (4) and reduce the patient’s anxiety and frustration.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
