Abstract
Congenital nasal pyriform aperture stenosis (CNPAS) is a rare anomaly causing respiratory distress in newborns. While the primary surgical technique is well established, the timing of the removal of the stents and the management of restenosis remain a matter of debate. We report a case of a female newborn affected by CNPAS with the recurrence of respiratory distress after primary surgery due to the early removal of nasal stents, causing an overgrowth of granulation tissue. This report notes that restenosis was successfully managed by repeating the procedure over a 14-day period, with soft polyvinyl chloride uncuffed tracheal tubes acting as nasal stents.
Introduction
Congenital nasal pyriform aperture stenosis (CNPAS) is a rare cause of respiratory distress in newborns, characterized by deficient development of the primary palate and bony overgrowth of the nasal processes of the maxilla. 1 It is often associated with other midline defects (solitary median maxillary central incisor, hypotelorism, holoprosencephaly, pituitary dysfunction).2,3 Failure of primary surgery is often due to the overgrowth of soft inflammatory tissue, which causes restenosis.4,5 Given the rarity of the disease, the management of failure after primary surgery is not well established. We report a case of a female newborn with isolated CNPAS treated surgically at 1 month of life. Early removal of nasal stents allowed granulation tissue growth and a recurrence of respiratory distress. This report highlights the importance of the timing of nasal stenting after primary surgical treatment, as early removal can lead to restenosis, and we report a successful possible treatment of restenosis based on the use of soft stents.
Case Report
Born at term from regular pregnancy, a female newborn was admitted to our Neonatal Intensive Care Unit (NICU) immediately after birth because of respiratory distress, desaturation, and cyanosis occurring during feeding and reverted by crying. Clinical examination revealed a barrel chest with intercostal indrawing during feeding and hypoplasia of the upper lip frenulum (Figure 1A). Nasal fibroendoscopy showed stenosis of the anterior portion of the nasal cavities (Figure 2), with failure to pass a 2.9 mm endoscope. A computed tomography scan of the nose and sinuses showed pyriform aperture stenosis and a solitary median maxillary central incisor (Figure 3). The baby was unable to feed sufficiently because of respiratory distress occurring during feeding. Conservative treatment was attempted with airway measures (Figure 1B), and topical corticosteroid and decongestant drops were applied. The nasogastric feeding tube was positioned to pass through the oral cavity. However, failure to thrive required surgery at 1 month of age. The preanesthetic routine examination was normal. The baby had no other associated congenital abnormalities, and her family had no familiar history of unexplained death under anesthesia, anesthetic complications, or neuromuscular disease. Anesthesiologic management began with preoxygenation of the newborn; the induction was realized by mask with oxygen and sevoflurane. After intravenous access was established, fentanyl 1 μg/kg was given, followed by orotracheal intubation, keeping spontaneous breathing associated with lidocaine topical anesthesia application to the pharyngeal and laryngeal mucosa. Dexamethasone 0.2 mg/kg and antibiotic prophylaxis were also administered. The anesthesia was maintained with 2.5% to 3% concentrations of sevoflurane and fentanyl. Intraoperative monitoring included pulse oximetry, capnography, concentration of inhaled and exhaled gases, noninvasive blood pressure, ECG, glucose level, and core body temperature. The intrasurgical infusion rate was 10 mL/kg/h, and isotonic balanced solution with glucose (2%) was used. A sublabial approach was performed (Figure 1C) as reported by Wood et al, 2 exposing the inferior and lateral pyriform aperture. An otologic 2.0 mm diamond burr was used to drill the lateral nasal wall to widen the pyriform aperture. A 3.0 mm endotracheal tube (ShileyTM 3.0 uncuffed neonatal/pediatric tracheal tube) was placed in each nasal cavity as a stent (Figure 1D). The operation was performed with no complications. She was transferred to the NICU. Tracheal extubation was performed without issue. The nasal stents had to be removed 4 days later because of the formation of pressure ulcers at the ala of the nose. Granulation tissue overgrowth in the nasal cavities caused relapse of respiratory distress 10 days after stent removal. A second surgery with the same anesthesiologic schedule was required to position nasal stents made of soft polyvinyl chloride (Portex® 3.0 uncuffed neonatal/pediatric tracheal tube). The tracheal tube was removed immediately at the end of the procedure, and the nasal stents were maintained for 2 weeks without complications. The patient remained hospitalized for an additional 15 days after stent removal for clinical monitoring. At the 2-month follow-up, the child showed good healing of the surgical site with complete relief from symptoms.
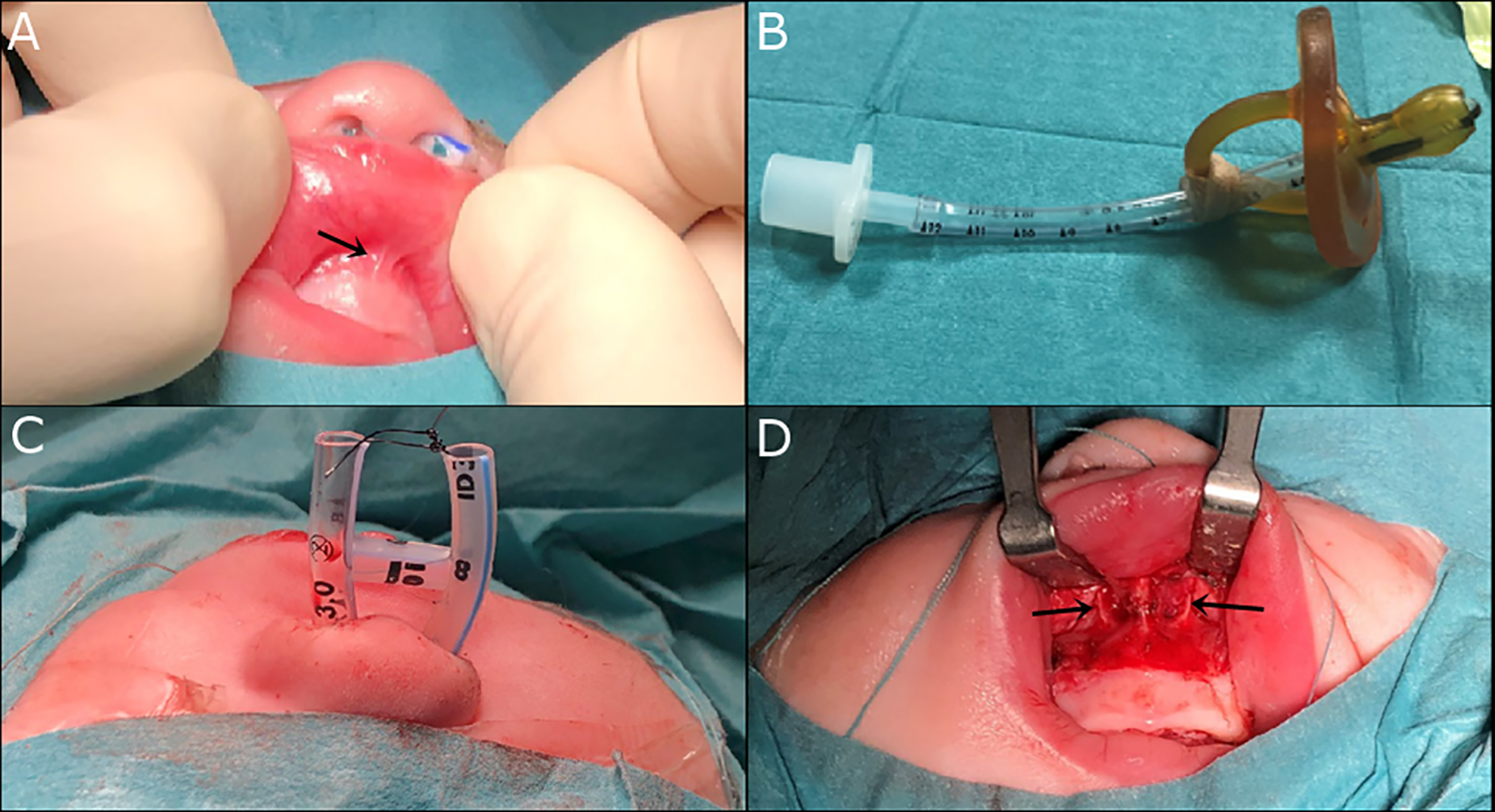
Clinical pictures. A, Hypoplasia of the upper lip frenulum (arrow). B, Pharyngeal tube inserted in a pacifier (McGovern nipple). C, Sublabial surgical approach with incision of the mucosa and exposure of the pyriform aperture and the nasal processes of the maxilla (arrows). D, Endotracheal 3.0 mm tubes are placed in the nasal cavities, cut and fixed to each other to avoid accidental removal.
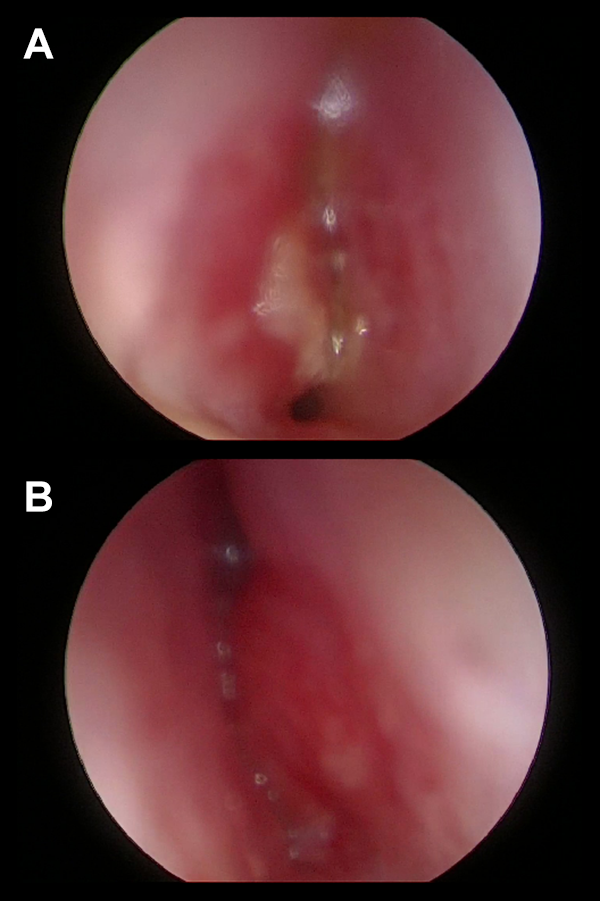
Fibroendoscopy of the anterior nasal cavities. A, Right side. B, Left side.
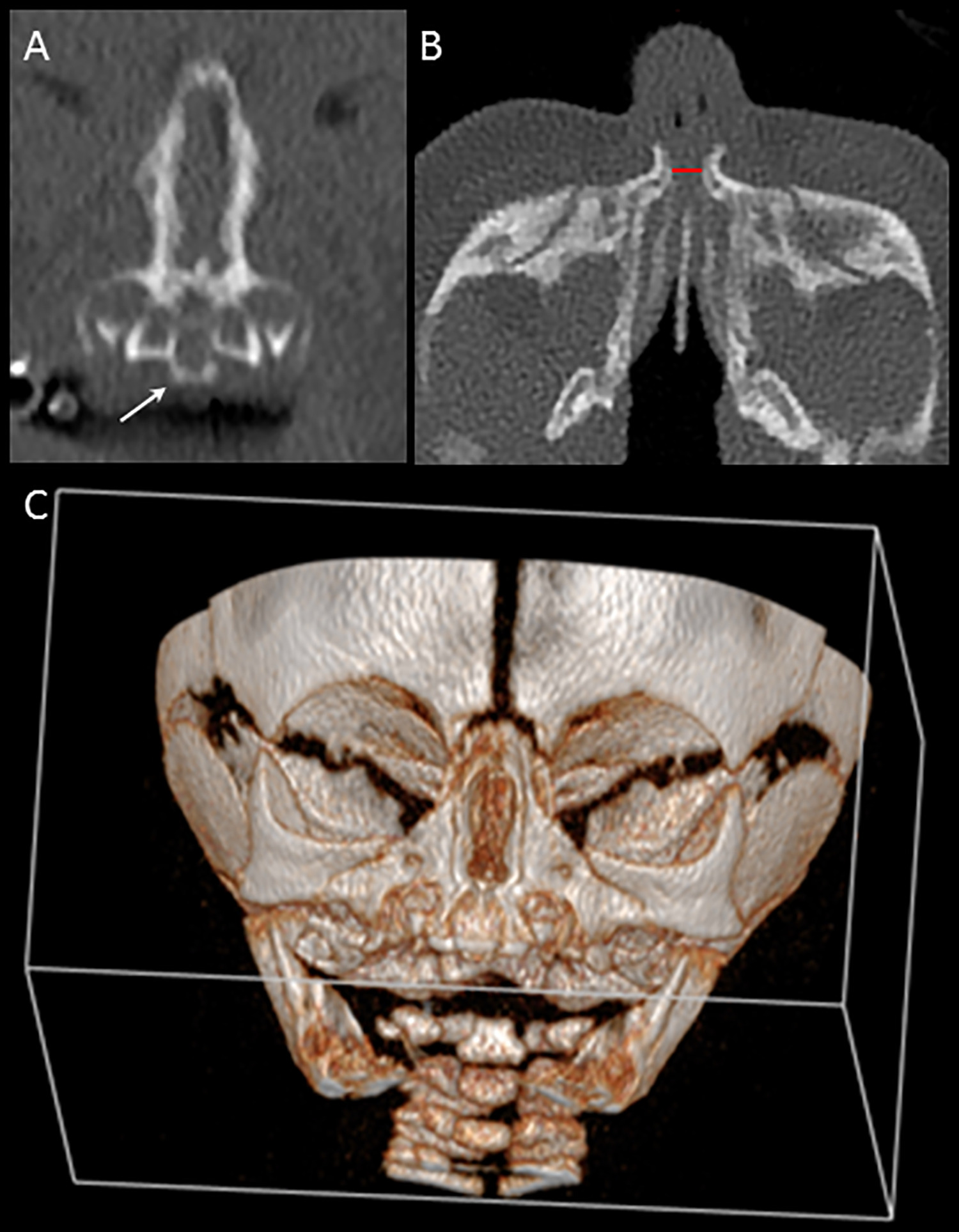
Computed tomography (CT) scan of the nose and sinuses. A, Coronal section. The arrow indicates the solitary median maxillary central incisor. B, Axial section. The nasal pyriform aperture maximum diameter is 3.4 mm measured at the level of the inferior meatus (red line). C, 3D-reconstruction.
Discussion
Failure to pass a nasogastric tube through the nasal cavities could suggest choanal atresia or CNPAS. An obstruction of the anterior portion of the nasal cavity (≤ 1 cm from the vestibule) is predictable for CNPAS. 3 A radiologic finding of pyriform aperture width < 11 mm (at the level of the inferior meatus in axial section) in a term neonate confirms the diagnosis of CNPAS. 6 First-line conservative treatment should be attempted for a minimum of 15 days, adopting airway measures, frequent nasal irrigations, and application of intranasal topical steroids and decongestants. 7 The surgical procedure consists of a sublabial approach with exposure of the pyriform nasal aperture. 2 Care should be taken in drilling the nasal processes of the maxilla not to injure the lacrimal system, tooth buds, nasal mucosa, or inferior turbinate. Contact between the tubes and the posterior wall of the oropharynx should be avoided. Pressure ulcers at the ala of the nose or columella are possible complications of nasal stenting; therefore, daily inspection of the nose is required. The tubes should be handled as little as possible to avoid microtraumatism, which could facilitate granulation tissue growth. Regular aspirations through the tubes are needed to prevent their occlusion with secretions. While the technique in primary surgery is well established,2,6 the timing of stent removal remains a matter of debate. Leaving the tubes in place for too short a time allows soft tissue overgrowth with restenosis. On the other hand, a long permanence can lead to pressure ulcers and risk of infection. In the second surgery, we used tubes made of a softer material (soft PVC) to reduce the risk of pressure ulcers. After a 14-day period of nasal stenting, no complications were registered, and the tubes were successfully removed. The good result persisted at the 2-month follow-up. We agree that 2 weeks of stenting after primary surgery could be a good compromise. This report notes that postoperative granulation tissue formation with restenosis in CNPAS could be successfully managed by a 14-day period of nasal stenting. The correct timing of nasal stenting in the treatment of CNPAS is essential for success after primary surgery, as early removal could lead to soft tissue overgrowth with restenosis, and excessive persistence of nasal stents can cause pressure ulcers in the ala of the nose. Failure after primary surgery due to granulation tissue formation could be successfully managed by a 2-week period of nasal stenting. For the choice of nasal stents, soft materials (eg, soft PVC) could allow atraumatic insertion and reduce the risk of pressure ulcers.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
