Abstract
Objective:
Head and neck cancer often accompany a synchronous secondary primary lesion in the digestive tract. The aim of this study was to compare detection rates between positron emission tomography–computed tomography (PET-CT) and esophagogastroduodenoscopy (G-fiber) or colonoscopy (C-fiber) in the initial staging and to analyze risk factors for premalignant, malignant, and total synchronous secondary primary lesions.
Methods:
A total of 739 patients with head and neck cancer who underwent PET-CT, G-fiber, or C-fiber were analyzed retrospectively.
Results:
Positron emission tomography–CT did not definitely detect any premalignant synchronous secondary primary lesions (0 [0%] of 739) but definitely detected 10 malignant synchronous secondary primary lesions (10 [1.35%] of 739). Esophagogastroduodenoscopy or C-fiber detected all 20 premalignant synchronous secondary primary lesions (20 [2.71%] of 739) and all 37 malignant synchronous secondary primary lesions (37 [5.00%] of 739). The patients with nasopharynx cancer tended to have premalignant synchronous secondary primary lesions (odds ratio [OR]: 3.793; 95% CI: 1.414-10.171; P = .008). Those with distant metastasis tended to have premalignant (OR: 4.743; 95% CI: 1.508-14.916; P = .009), malignant (OR: 3.803; 95% CI: 1.486-9.731; P = .005), and total synchronous secondary primary lesions (OR: 2.753; 95% CI: 1.159-6.538; P = .022).
Conclusions:
Premalignant or malignant synchronous secondary primary lesions that were not definitely detected by PET-CT could be found in the endoscopic examination.
Keywords
Introduction
Patients with head and neck squamous cell carcinoma (HNSCC) often have a second primary lesion (SPL) in the upper aerodigestive tract. Historically, HNSCC has been associated with tobacco and alcohol abuse. These carcinogens interact with the entire upper aerodigestive tract, placing the entire area at risk of the development of an SPL. 1 Second primary lesion is classified into synchronous and metachronous SPL. Synchronous and metachronous SPL are identified within and after 6 months of the index tumor, respectively. 2
Screening for synchronous SPL (SSPL) in patients with HNSCC is important. Evaluation of the upper aerodigestive tract with oral inspection, direct laryngoscopy, esophagoscopy, bronchoscopy, or some combination of these procedures, called pan-endoscopy, is essential in the initial staging of HNSCC.3-5 However, the roles of computed tomography (CT) and positron emission tomography (PET) have received more attention recently. In addition, some authors reported that the incidence of SSPL appears to be decreasing. They performed a retrospective study to examine data from the National Cancer Institute Surveillance, Epidemiology, and End Results registry from 1979 to 2008 and found that the incidence rate of SPL decreased over this period. 6 In fact, several studies have confirmed that PET-CT outperforms traditional pan-endoscopy for the detection of SPL, with the additional benefit of detecting diseases in locations outside the upper aerodigestive tract, such as the lung and liver. Therefore, some authors have criticized the use of routine pan-endoscopy in the initial staging of HNSCC.3,7-10
However, by using esophagogastroduodenoscopy (G-fiber) or colonoscopy (C-fiber), we have occasionally encountered cases of premalignant or malignant SSPL in the digestive tract, beyond the head and neck area, which were not clearly visible on PET-CT. Thus, this study aimed to compare the detection rate for premalignant and malignant SSPLs between PET-CT and G- or C-fiber. In addition, we aimed to analyze risk factors for premalignant, malignant, and total SSPLs.
Ethical Considerations
This study was reviewed and approved by our institutional review board (IRB No. 2018AS0213). The need for informed consent was waived because we reviewed the medical records of all the patients retrospectively.
Methods
Patients
We included 807 patients with HNSCC confirmed by primary site biopsy between 2007 and 2016 in Korea University Medical Centers. Fifty-four of the patients did not undergo C-fiber. Nine of the patients did not undergo G-fiber, and 5 did not undergo PET-CT. Thus, 68 patients were excluded. Finally, 739 patients were enrolled. All the enrolled patients with HNSCC underwent PET-CT, G-fiber, or C-fiber (Figure 1). If SSPL is suspected during G- or C-fiber, biopsy was performed at the digestive tract lesion. Patients with squamous cell carcinoma in the paranasal sinus, nasopharynx, oral cavity, oropharynx, hypopharynx, and larynx were included. However, lymphoma, salivary gland, and thyroid cancers were not included because of their varied pathologies, unlike squamous cell carcinoma. This study analyzed cases of premalignant or malignant SSPLs in the digestive tract. The study did not include SSPLs in locations outside the digestive tract, such as the liver and lung lesions. Hereinafter, we use SSPL to refer to those in the digestive tract.
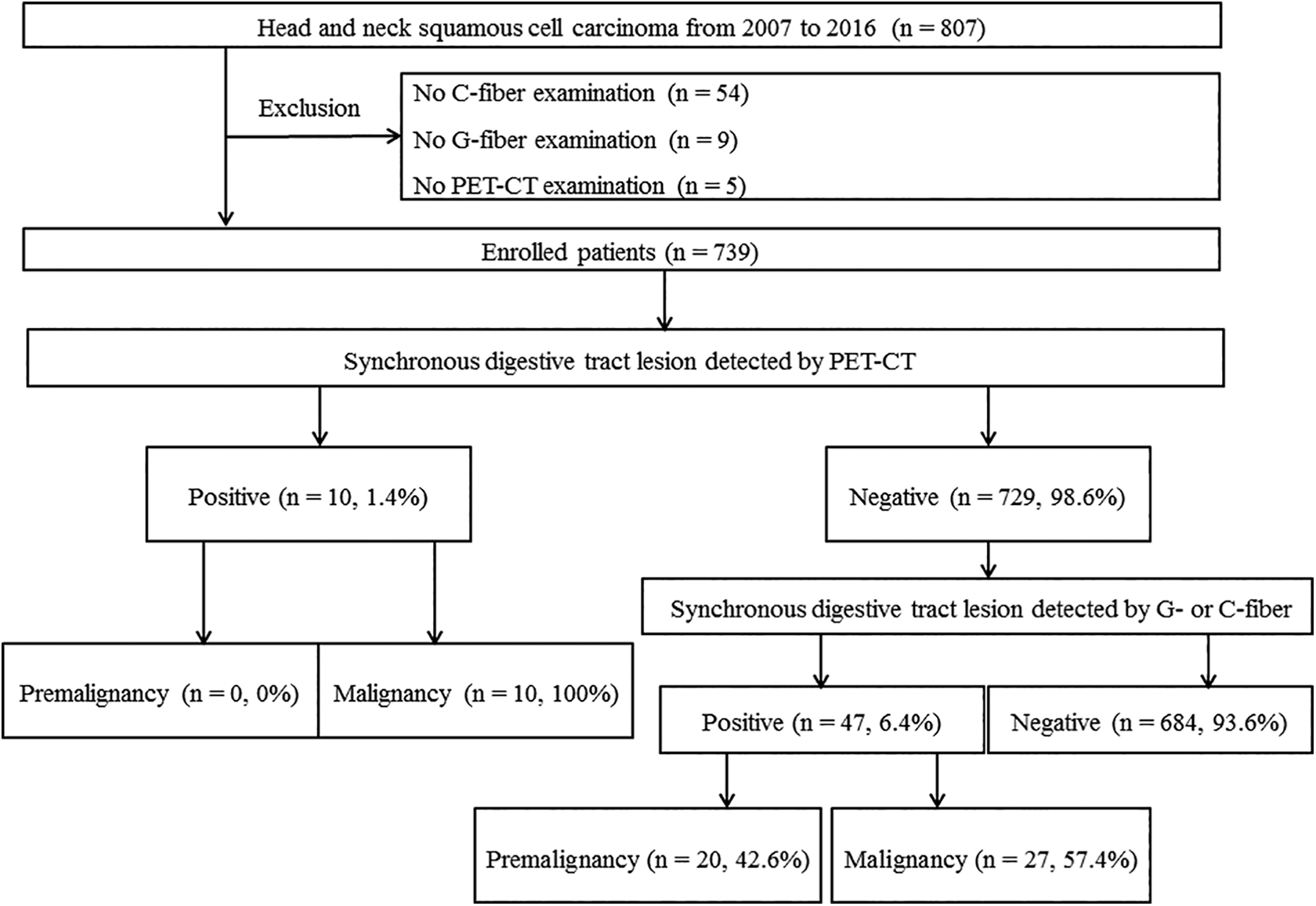
Flowchart of the study patients, summarizing the detection rates of synchronous second primary lesions by positron emission tomography–computed tomography and endoscopic examination. C-fiber indicates colonoscopy; G-fiber, esophagogastroduodenoscopy; PET-CT, positron emission tomography–computed tomography.
Protocol of the Initial HNSCC Staging Workup
When a patient with a lesion suspected of HNSCC visited hospital, we performed biopsy in an outpatient clinic or operating room. In addition, we performed general laboratory tests such as complete blood count, kidney function test, liver function test, electrolytes, coagulation test, chest radiography, and electrocardiography. If malignancy was confirmed on the basis of the biopsy result, the patient was registered in the Korean cancer registration system, whereby patients pay only 5% of the cost of their treatment during the next 5 years. Afterward, the patient underwent neck magnetic resonance imaging for T stage, neck CT for N stage, and whole-body FDG PET-CT for M stage. Thereafter, if G- or C-fiber were not checked within 6 months, they were performed in patients with HNSCC to explore for occult SSPLs. If a lesion was found in the whole digestive tract, the gastroenterologist performed biopsy during G- or C-fiber. If additional abnormalities were found in the abovementioned examinations, we conducted additional tests. Otherwise, we explained the results of the HNSCC workup and suggested the treatment plan to the patients in the multidisciplinary outpatient clinic.
Statistical Analyses
The Student t test, Pearson χ2 test, Fisher exact test, and linear-by-linear association test were used to identify significant associated factors with premalignant, malignant, and total SSPLs. The following clinical factors were adopted in the analysis of independent risk factors for the incidence of premalignant, malignant, and total SSPLs: age, sex, index tumor site, smoking history, alcohol consumption history, and TNM classification. A logistic regression analysis was used to estimate the odds ratios (ORs) and 95% CIs for analysis of significant prognostic factors for premalignant, malignant, and total SSPLs. A P value of <.05 was considered statistically significant. Statistical analyses were performed using the SPSS version 22.0 software (SPSS).
Results
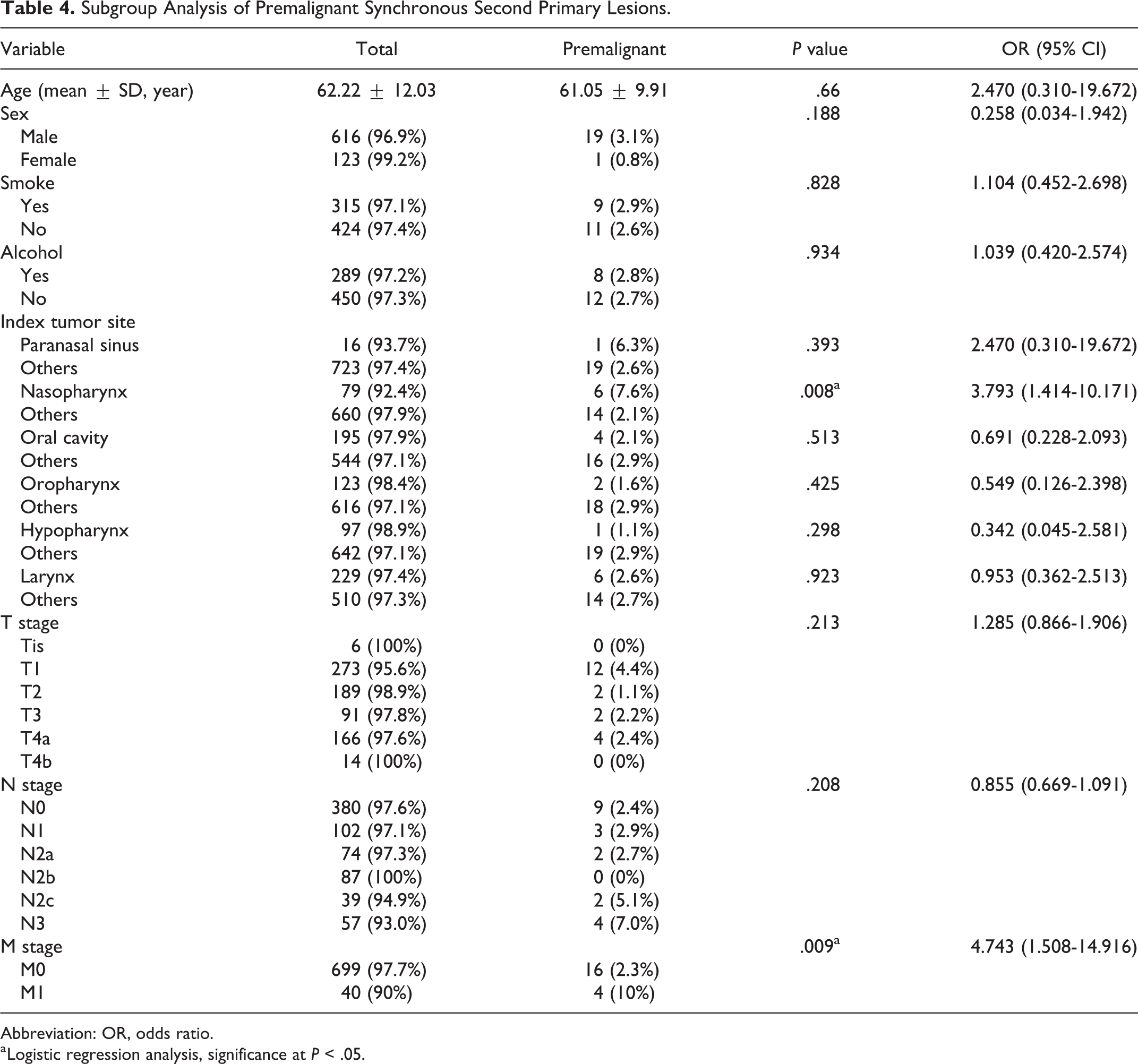
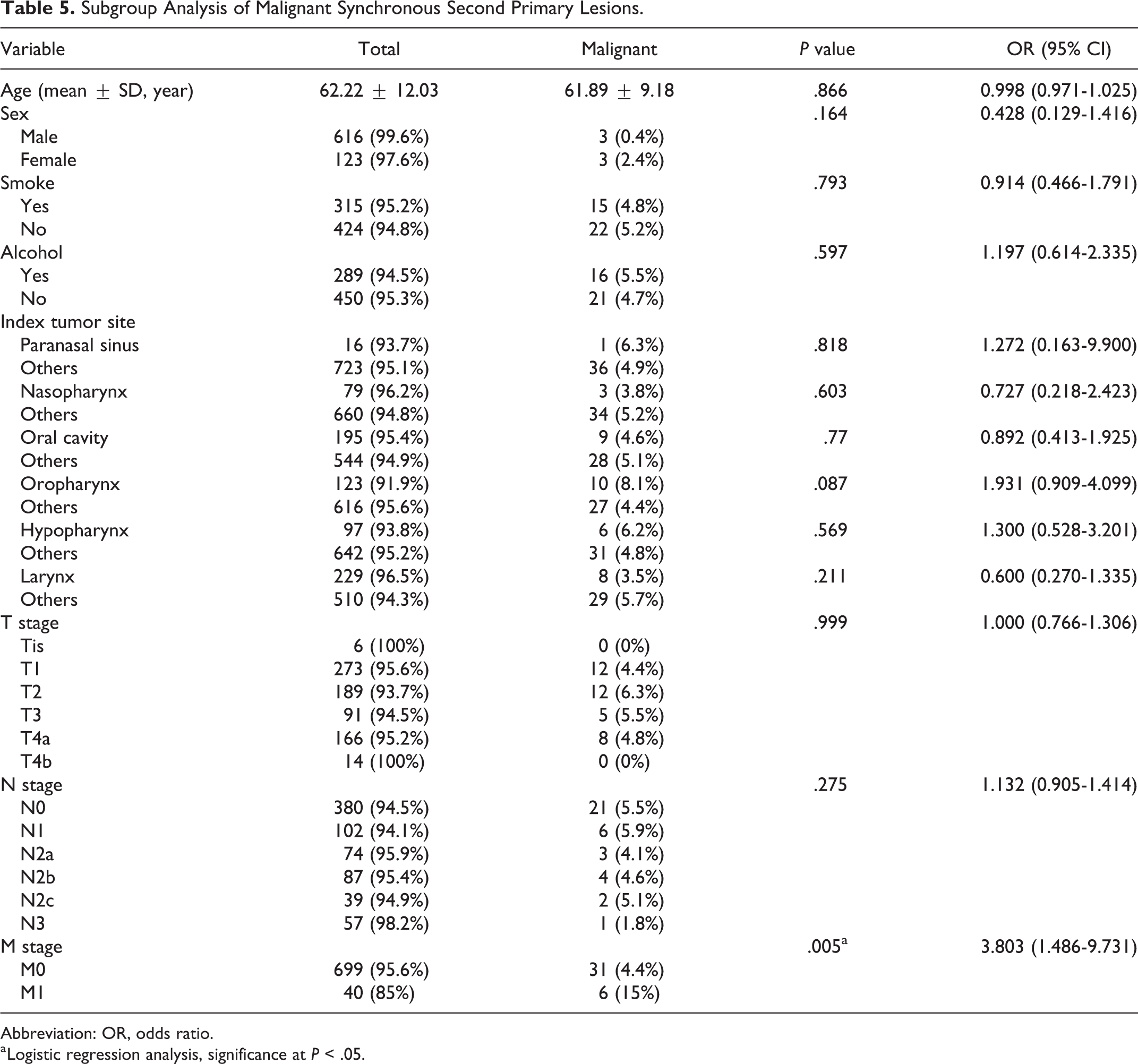
The patients’ demographic characteristics are shown in Table 1. Among the 739 patients, 57 had premalignant and malignant SSPLs, which consisted of 20 premalignant SSPLs and 37 malignant SSPLs. No premalignant SSPLs were detected by PET-CT (0 [0%] of 20). Some malignant SSPLs were detected by PET-CT (10 [27.02%] of 37; Table 2). In total patients, while PET-CT detected no premalignant SSPLs (0 [0%] of 739) and 10 malignant SSPLs (10 [1.35%] of 739), G- or C-fiber detected all 20 premalignant SSPLs (20 [2.71%] of 739) and all 37 malignant SSPLs (37 [5.00%] of 739; Figure 1). Distant metastasis (eg, lung, liver, or bone metastasis) was an independent risk factor for total SSPL (OR: 2.753; 95% CI: 1.159-6.538; Table 3), premalignant SSPL (OR: 4.743; 95% CI: 1.508-14.916; Table 4), and malignant SSPL (OR: 3.803; 95% CI: 1.486-9.731; Table 5). Nasopharynx cancer was an independent risk factor for premalignant SSPL (OR: 3.793; 95% CI: 1.414-10.171; Table 4).
General Characteristics of the Participants.a
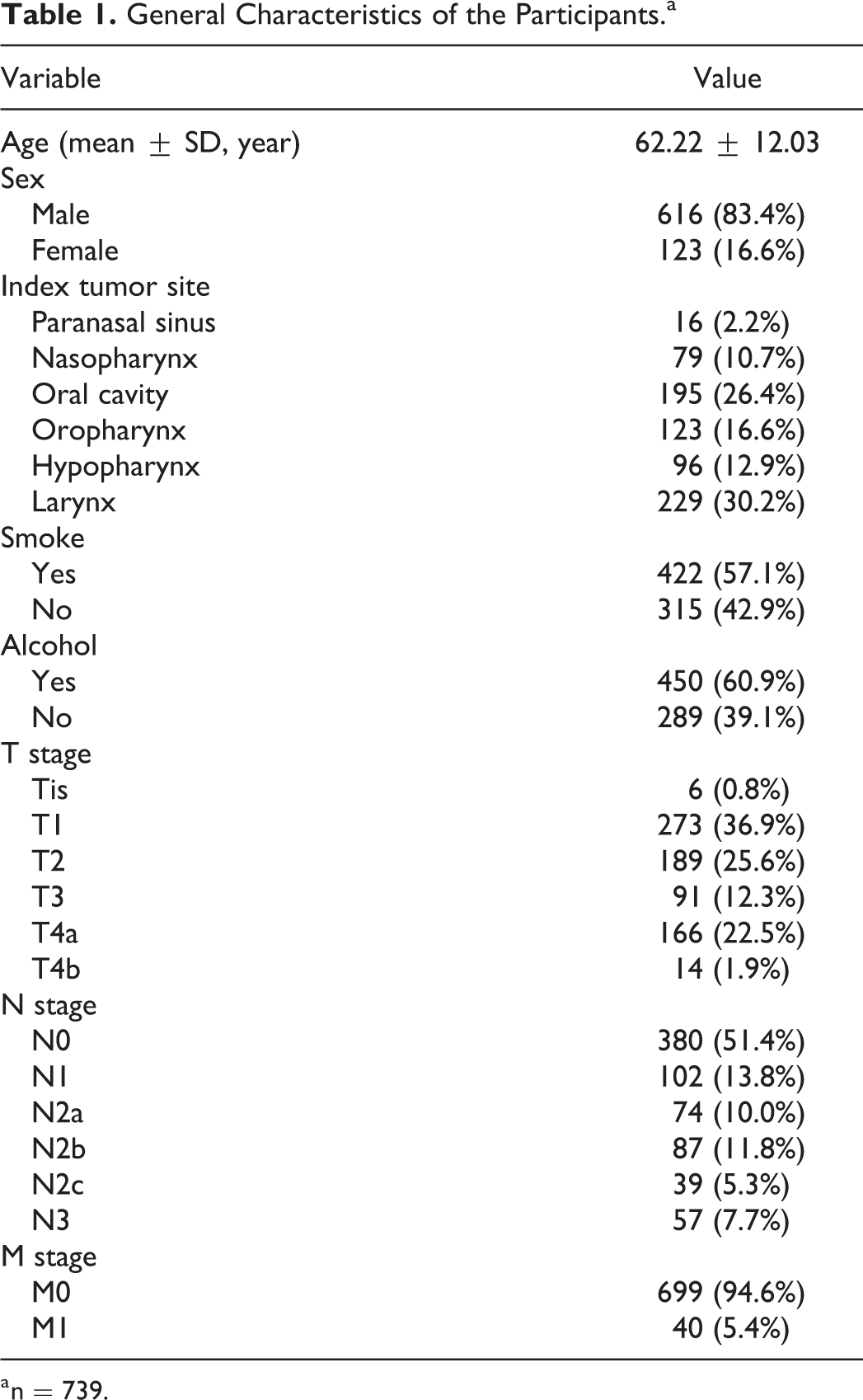
a n = 739.
Detection Rates of Premalignant and Malignant Synchronous Second Primary Lesions by Esophagogastroduodenoscopy or Colonoscopy, and Positron Emission Tomography–Computed Tomography.
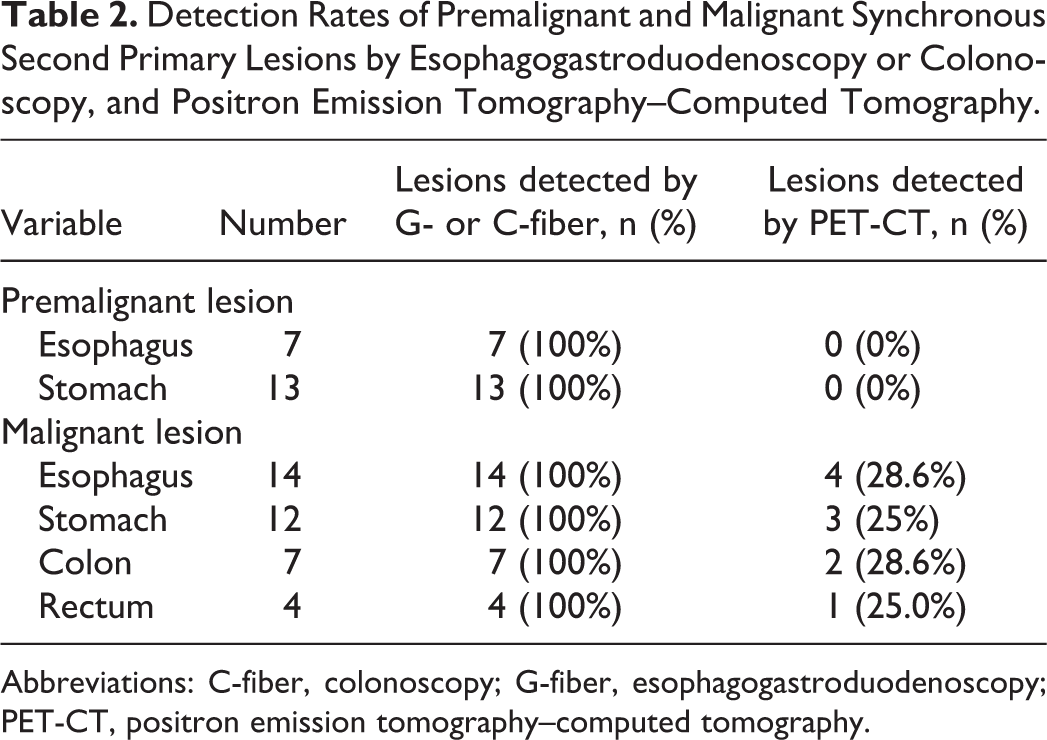
Abbreviations: C-fiber, colonoscopy; G-fiber, esophagogastroduodenoscopy; PET-CT, positron emission tomography–computed tomography.
Analysis of Cases of Total Synchronous Second Primary Lesions, Including Premalignant and Malignant Lesions.
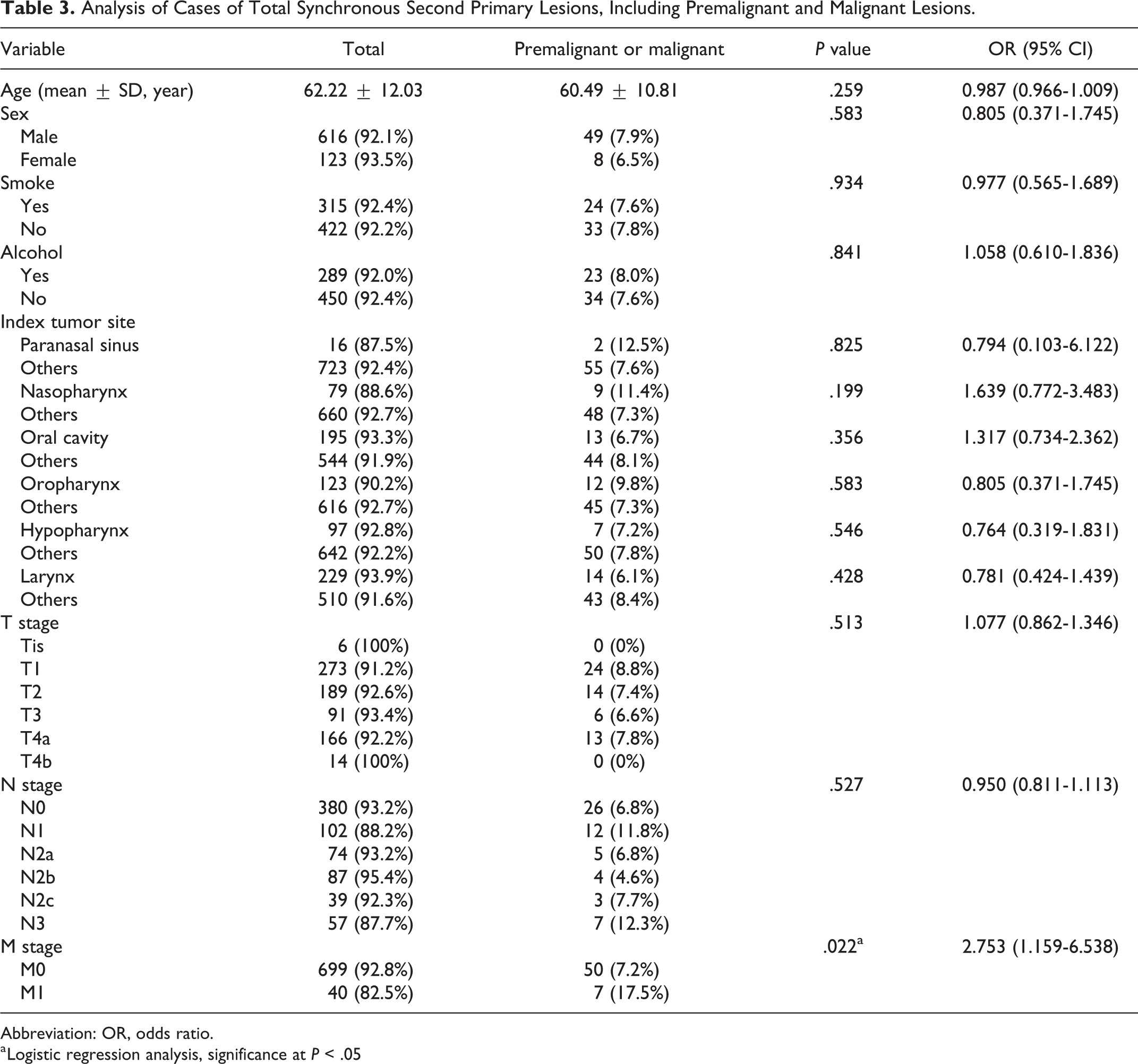
Abbreviation: OR, odds ratio.
a Logistic regression analysis, significance at P < .05
Subgroup Analysis of Premalignant Synchronous Second Primary Lesions.
Abbreviation: OR, odds ratio.
a Logistic regression analysis, significance at P < .05.
Subgroup Analysis of Malignant Synchronous Second Primary Lesions.
Abbreviation: OR, odds ratio.
a Logistic regression analysis, significance at P < .05.
Discussion
This study showed that many premalignant and malignant SSPLs (47 [82.4%] of 57) were not definitely detected only by PET-CT. In patients with advanced cancer who have distant metastases, the possibility of premalignant or malignant SSPL is relatively high. In addition, it is interesting that accompanying premalignant SSPL is relatively highly possible in patients with nasopharynx cancer in our results.
Our results suggested that the detection rate for SSPL by PET-CT was lower than generally expected. In fact, many Korean hospitals do not perform G- or C-fiber routinely in the initial staging of HNSCC, unlike PET-CT. Moreover, the recent national comprehensive cancer network (NCCN) guideline for head and neck cancer does not recommend routine endoscopic examination under anesthesia (EEUA) for patients with paranasal sinus, nasopharynx, oral cavity, and oropharynx cancers. The NCCN guideline recommends routine EEUA only for larynx and hypopharynx cancers. 11 However, like our study, a Japanese study also showed that the sensitivity of PET for detecting synchronous esophageal (1 [7.6%] of 13), gastric (2 [25.0%] of 8), and colon cancers (0 [0%] of 1) in patients with HNSCC was low. They suggested that because most of synchronous esophageal and gastric cancers were Tis or T1 stage, G-fiber should be performed to detect synchronous esophageal and gastric cancers in patients with head and neck cancer. 12
The role of endoscopic examination has been compared with that of imaging techniques, including the CT or PET in patients with HNSCC. Di Martino E et al showed that both PET and panendoscopy had a sensitivity of 95% and 100% for detection of second primary tumor. 13 However, the diagnostic value of endoscopic examination remains controversial in the literature. 14 Some studies showed that endoscopic examination was still considered as the most effective strategy to detect second primary tumors. They suggested that endoscopic examination cannot be replaced by any imaging procedure because early mucosal lesions are often undetected or overlooked by imaging procedures. Wang et al described that among 815 patients with HNSCC, 124 (15.2%) patients were diagnosed as having esophageal SSPL (66 low-grade dysplasia, 29 high-grade dysplasia, and 29 esophageal squamous-cell carcinoma). 15 Sharma et al demonstrated that SSPL prevalence was 5.56% (13 SSPL/234 HNSCC) and only 3 (23.13%) of 13 of SSPLs were exclusively found by triple endoscopy (direct laryngoscopy, bronchoscopy, and esophagoscopy). 16 However, other authors believed that endoscopic screening has a low yield and do not recommend it for concurrent use with other imaging modalities.10,17 Additionally, some studies suggested that the incidences of SSPL in HNSCC were low.17-19
This study showed that nasopharynx cancer accompanied by significantly premalignant SSPLs. In detail, 6 premalignant SSPLs, including 2 esophageal dysplasia and 4 stomach dysplasia, were detected in 79 patients with nasopharynx cancer patients (6 [7.6%] 79). On the other hand, 14 premalignant SSPLs were found in 660 patients with HNSCC (14 [2.1%] 660), except nasopharynx cancer. It is interesting that nasopharynx cancer was linked to stomach dysplasia. We hypothesized that the important reason for this finding can be Epstein-Barr virus (EBV) infection, which is known to be associated with the development of nasopharynx and stomach cancers. 20 Zhao et al reported that EBV is related with approximately 99% of nasopharyngeal cancers and 10% of gastric carcinomas. 21 Epstein-Barr virus–associated gastric cancer is 1 of the 4 subtypes of gastric carcinoma, as defined by the novel classification recently proposed by the Cancer Genome Atlas. 22 From these points, we can infer that nasopharyngeal cancer was significantly linked to premalignant SSPLs, especially in the stomach. It can be criticized that this is a phenomenon limited to East Asia. The incidence of gastric cancer in Korea is the highest worldwide. 23 Salted diet and gastric cancer are highly related. Korean diet contains high nitrites and nitrosamines in smoked fish and pickled foods like Kimchi. However, among Caucasian countries, the Swedish diet too contains many pickled foods and salted fish. As a result, they too have a higher incidence of gastric cancer.
A novelty of this study was the investigation of colorectal SSPLs in HNSCCs. Few studies have focused on C-fiber screening in patients with HNSCC. Asian countries are undergoing a rapid increase in colorectal cancer incidence due to economic development and Westernization of diet. 24 Our study showed that a few colorectal SSPLs were not clearly detected by PET-CT in the initial HNSCC staging. A previous Japanese study reported 1 case of synchronous colon cancer among 230 patients with head and neck cancer. 12 An additional novelty of this study was the investigation of premalignant SSPLs in HNSCCs. Only few articles reported about premalignant SSPLs in HNSCC. It may be important that premalignant SSPLs were not found at all on PET-CT, which is common sense. However, we think that undiscovered premalignant SSPLs may develop to cancer later on, which can worsen patient prognosis. However, the present study has some limitations. First, it was conducted in the one of the countries with the high incidence of gastric and colorectal cancer. Second, this study did not analyze the cost-effectiveness of G- and C-fiber screening for HNSCC. Esophagogastroduodenoscopy and C-fiber are cheaper in Korea (approximately US$100) than in other countries. Thus, future investigations would be required to analyze the cost-effectiveness of G- and C-fiber screening for HNSCC.
Synchronous SPL is a well-known independent poor prognostic factor in patients with HNSCC. Patients with HNSCC who have SSPL have a lower survival rate than those without SSPL.2,14,25 Furthermore, high-grade dysplasia in the esophagus should be removed as a malignant lesion. 26 Therefore, initial G- and C-fiber screening could help improve survival outcome and prognosis in patients with HNSCC. In this study, most patients with distant metastasis did not undergo curative treatment. However, in some cases, we performed treatment for both lesions and showed good results. For examples, one patient with oral and esophageal cancers underwent simultaneously surgical treatment on both. Another patient with nasopharyngeal and premalignant gastric lesion underwent CCRT after endoscopic mucosal resection. However, the yield of C-fiber was 1.4% with only 11 patients. Routine C-fiber screening will be discussed further because the yield is low in consideration of cost–benefit rate and increased risk of complications during endoscopy.
Conclusion
Esophagogastroduodenoscopy may be considered with PET-CT in the initial staging workup of advanced HNSCC for detecting occult premalignant or malignant SSPLs. However, C-fiber is not routinely recommended with HNSCC screening.
Footnotes
Authors’ Note
The manuscript was edited for proper English language, grammar, punctuation, spelling, and overall style by highly qualified native English-speaking editors at Editage.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (grant No. NRF-2017R1C1B1008842).
