Abstract
The use of alternative medicine in chronic rhinosinus-itis (CRS) continues to increase in popularity, for the most part without meeting the burden of being based on sound clinical evidence. New and emerging treat-ments, both natural and developed, are numerous, and it remains a challenge for otolaryngologists as well as general practitioners to keep up to date with these therapies and their efficacy. In this systematic review, we discuss a number of alternative therapies for CRS, their proposed physiologic mechanisms, and evidence supporting their use. This analysis is based on our re-view of the English-language literature on alternative therapies for CRS (we did not include any therapies that are already recommended by accepted profession-al bodies). Data collection was performed using the PubMed database (not restricted to MEDLINE due to the nature of the subject matter), the Cochrane data-bases, and bibliography searches. We found that while many of the alternative therapies we reviewed might have a firm basis in science, they lack any clinical ev-idence to support their use specifically for CRS. Some emerging therapies, such as therapeutic ultrasonog-raphy and phonophoresis, show some promise, based on a growing body of positive evidence. In addition, the use of baby shampoo, thyme honey, and bromelain additives to saline lavage in CRS are all supported by clinical evidence, as is Sinupret, an oral preparation that contains echinacea. However, higher levels of ev-idence gleaned from large, well-designed, prospective, randomized, controlled trials are needed before any of these therapies can be recommended.
Introduction
The standard ENT consultation is changing in some ways within both specialist and general practices. Pa-tients’ own Internet research and the widespread use of social media platforms by commercial entities to promote their products 1 have allowed some patients to form very mature opinions of their disease and how it might be treated with alternative therapies. Physi-cians have a duty to know about these therapies, es-pecially with respect to their risks of potential harm to patients.
In a systematic review published by the World Health Organization in 2000, Ernst reported a wide discrepancy in the proportion of patients who reported using com-plementary and alternative medicine (CAM), ranging from 9 to 65%.2 In Germany and the United States, there is evidence to suggest that the use of CAM has become increasingly prevalent since 1945. 2
In otolaryngology, perhaps the condition most often treated with CAM therapies is chronic rhinosinusitis (CRS). According to a 2010 report by Rotenberg and Bertens, 15.6% of CRS patients in one Canadian co-hort admitted to using alternative therapies to treat their CRS. 3 A study published in 1999 by Krouse and Krouse included 120 patients with sinusitis; 35% of them reported using chiropractic therapy, 29% herbal therapy, and 19% acupuncture. 4
Increasing healthcare costs and poor access to healthcare have been reported as possible reasons for the growing use of CAM in the United States. 5 Social networking sites are also used to promote the use of CAM by healthcare professionals and amateurs alike. 1
In a 2012 Dutch study, Kooreman and Baars obtained economic data from general practices indicating a re-duction of 0 to 30% in cost when the use of CAM was combined with or used instead of allopathic medicine. 6 However, their study did not compare disease entities within rhinosinusitis, and population bias 2 might have existed because the cohorts using CAM were primarily made up of younger women. For these reasons, their claims were disputed by Sampson et al, who contended that the analysis was based on an incorrect interpre-tation of the data. 7
In the state of South Australia, a survey of 3,015 residents found that 52.2% were CAM users. 8 Some 49.7% used CAM concurrently with allopathic medi-cines, although fewer than half of them admitted this use to their physician. In an alarming finding, half of the survey respondents incorrectly believed that these medicines had been independently tested for quality, safety, and efficacy by the Australian government’s Therapeutic Goods Administration.
As for the economic benefits of CAM, the data are conflicting. 9 The disparities likely represent the varied nature of the conditions treated and the efficacy of the CAM agents available.
In this article, we present our systematic review of all the alternative therapies currently reported in the literature and the corresponding clinical evidence where applicable.
Materials and methods
Our study looked at all published therapeutic options for CRS that we classified as CAM. Our research team limited our search to English-language journals, but we specified no timeline.
We searched the PubMed database using the search terms “chronic rhinosinusitis,” “CRS,” “chronic rhini-tis,” “chronic sinusitis,” “natural,” “alternative,” “com-plementary,” “Chinese,” “traditional,” and “herbal.” Because of the scarcity and poor quality of the research in some cases, it was not possible to confine ourselves to using only MEDLINE indexes, and this was taken into account when we formulated our clinical recom-mendations. Despite the lack of well-founded data, we thought it was important to include these therapies to create an up-to-date resource on this topic. We excluded studies that were specifically performed on acute rhinosinusitis. However, many studies did not specify a specific subtype of disease, and we included these in our discussion for the sake of completeness. We also included articles in which multiple subtypes of sinusitis, including CRS, were mentioned (e.g., CRS with allergic rhinitis).
Our exclusion criteria were designed to focus our review outside the realm of mainstream therapies, and therefore we decided to exclude therapies that have received recommendations (including negative recommendations) for use in CRS by a professional body as outlined in the European Position Paper on Rhinosinusitis and Nasal Polyps 2012. 10
For CRS without nasal polyps, these therapies included topical and systemic steroids, topical and systemic antibiotics/antimycotics, saline irrigations, bacterial lysates, mucolytics, proton-pump inhibitors, decongestants, systemic antihistamines, probiotics, and immunotherapies. In the postoperative setting,
we included xylitol and sodium hypochorite additives to saline lavage.
For CRS with nasal polyps, we excluded furosemide, immunosuppressants other than steroids, and biologic drugs (e.g., anti-IL-5 antibodies). However, we did not exclude phytotherapy from our review because we aim to include any CAM treatments within the rapidly ex-panding natural medicine field that may have evidence for use in CRS.
Results
Topical antibiotics might play an adjunctive role in the treatment of refractory CRS without nasal polyps; they are not indicated as a first-line therapy, and their use is not supported in CRS with nasal polyps. Other topical mainstream therapies such as antihistamines and decongestants do not affect quality of life in the long term, and they are not further discussed here.
Baby shampoo. Baby shampoo (Johnson’s baby shampoo unless otherwise stated) consists of three surfactants: PEG-80 sorbitan laurate, cocamidopropyl betaine, and sodium trideceth sulfate, and it belongs to a family of surfactants that can be used to treat CRS. (Another member of this family is SinuSurf, a previously available surfactant that was removed from commercial production because it was implicated in causing a loss of smell and possibly other side effects in some users. 13 ) For treating CRS, nonperfumed baby shampoo is added to isotonic saline lavage fluid at a 1% dilution. Lavage is performed as usual. The action of baby shampoo is twofold; it acts as a mucolytic and a bactericidal detergent.
It is believed that surfactants, including baby sham-poo, may also exert antibiofilm activity due to their detergent action against Staphylococcus aureus and Pseudomonas aeruginosa, two major CRS pathogens. 14 An in vitro study by Chiu et al published in 2008
demonstrated the antibiofilm-forming activity of baby shampoo against planktonic P aeruginosa and biofilm formation at standard dosing concentrations of 1%.15 The authors did not observe any action against estab-lished biofilms. 15
Study results have been conflicting with respect to ciliary dysfunction after the use of topical surfactants. A murine explant model of mucociliary function demonstrated increased ciliary beating when a modified bicarbonate -buffered surfactant solution was applied to the tissue. 16 That study found no ciliary toxicity in the murine model. An ex vivo study using human tissue from diseased sinuses to determine the potential toxicity of SinuSurf indicated that no toxicity occurred at the recommended dose, although a dose-related toxicity was observed at higher doses. 17
Clinical studies of surfactants for use in CRS are of varied strengths. In a comprehensive review published in 2013, Rosen et al discussed three human clinical studies of irrigated surfactants (baby shampoo). 14 But only one of these studies was a randomized, controlled trial. In a randomized controlled study of 40 patients, Farag et al found no statistically significant differences in subjec-tive postoperative symptoms when baby shampoo (1% in isotonic saline) was compared with saline alone. 18
The in vitro study by Chiu et al in 2008 found sig-nificant improvement in recalcitrant postoperative symptoms, although the findings in the treatment group might have been confounded by a small cohort within that received concomitant antibiotics. 15
The paucity of adequately powered studies of baby shampoo makes it difficult to draw conclusions as to its efficacy in nasal lavage, although the available data do indicate that the potential for toxicity is likely to be low when it is used in the recommended amount. The most commonly reported side effects were burning of the mucosa and nasal irritation.
Honey. Investigators have reported that manuka honey, manuka-type honey, and Sidr honey exert an-tibiofilm activity against S aureus and P aeruginosa. 19 -21 An in vivo study of the anti-S aureus biofilm activity of manuka-type honey and its toxicity in sheep found a significant reduction of biofilm in treated subjects and no reported toxicity when it was used in a topical concentration of 1.8 mg/ml in normal saline. 21
A human clinical trial conducted by Thamboo et al found no significant differences in subjective sino-nasal outcome test (SNOT-22) scores and endoscopic grading scores between nostrils treated with manuka honey and untreated control nostrils. 22 In another clinical trial, Hashemian et al used thyme honey (honey made from thyme pollen) as a postoperative adjunct along with saline lavage and an intranasal steroid. 23 During a 2-month follow-up period, they
found no significant differences in endoscopic grad-ing scores and SNOT-22 scores between the thyme honey users and placebo controls, although the thyme honey group did show a greater reduction in the endoscopic scores.
Capsaicin. Capsaicin is an active component of chili peppers. It is a known stimulator of sensory nerve C fibers, and it is known to stimulate the release of calci-tonin gene-related peptide (CGRP). 24 For the treatment of CRS, a concentration of sensory neuropeptides such as CGRP has been found to positively correlate with inflammatory cell concentrations and worsening symptoms. In pig and guinea pig models, it has been demonstrated that capsaicin application leads to a decrease in CGRP levels and destruction of sensory C fibers in the nasal mucosa, suggesting a mechanism of its use in CRS. 25
In a study published in 1991, Lacroix and colleagues followed 16 patients with CRS and rhinitis medica-mentosa over 5 weeks as they applied capsaicin nasal spray weekly. 26 The authors noted reduced levels of CGRP in mucosal biopsies after application and a significant reduction in symptoms over a 6-month follow-up. Since capsaicin is a direct stimulator of C fibers, it is necessary to apply it with the use of local anesthesia, which calls into question its feasibility as a treatment for CRS.
Méndez-Sanchez et al described a nonrandomized, noncontrolled case series on manual therapy for CRS in 11 women with CRS and midfacial pain. 27 It is unclear if the diagnosis of CRS was made by a physician or by a physical therapist using criteria from the American Academy of Otolaryngology–Head and Neck Surgery. During the 7-week study, patients underwent three sessions of physical therapy. Patients who were tak-ing prescription medications or who had undergone head and neck surgery, including facial surgery, were not included in the analysis. The authors found that manual therapy conclusively reduced symptoms of CRS and midface pain. To the best of our knowledge, this is the only published study to date that has examined the link between manual therapy and CRS symptoms. However, in view of the small number of patients, the exclusion criteria, treatment bias, and the lack of a control group, reaching conclusions about this therapy is impractical.
While ultrasonography had already been advocated as a treatment for CRS, it was Bartley and Young in 2009 who suggested that the mechanism behind its effect was its inhibition and destruction of biofilms and its bactericidal properties, which had been established in vitro. 29
The therapeutic ultrasonography procedure in-volves the placement of 1- to 15-mHz ultrasound probes just inside the nasal vestibule and on the skin
overlying the maxillary and frontal sinuses. Ultra-sound waves are believed to exert three effects within the diseased tissue—thermal energy, cavitation, and acoustic streaming:
• Thermal energy is an unwanted effect of therapeu-tic ultrasonography. It can be significantly reduced by pulsing the ultrasound probe on and off. • Cavitation occurs when bubbles of gas form from dissolved gases during the administration of ultra-sound wave cycles as energy is released. • Acoustic streaming refers to the shear stress that oc-curs in the cell membranes as ultrasound waves are deliv-ered. This stress interferes with cell membrane signaling.
Therapeutic ultrasonography is believed to play a role in reducing bacterial biofilms and exerting anti-in-flammatory action through the interference created by cavitation and the disruption of physiologic processes by acoustic streaming. 30
Karosi et al conducted an in vitro study of patients who had received a diagnosis of CRS with nasal pol-yps. 31 They examined the surgically resected polyps and treated them with 0.4 mHz of ultrasound for 5 minutes. Treatment resulted in a significant reduction in biofilm thickness and inflammatory cell count in the stromal layer of the polyps.
In a recent review, Bartley et al looked at five published studies in which therapeutic ultrasonography was used to treat CRS in a total of 169 patients. 30 One of these stud-ies was a single-blind, randomized, controlled trial by Ansari et al, who found a significantly greater reduction of CRS symptoms in the ultrasonography group than in the sham ultrasonography group. 32 Another study by Ansari et al comparing pulsed and nonpulsed thera-peutic ultrasonography found no significant difference in outcomes between the two groups. 33 The results of the review by Bartley et al were supportive of the use of therapeutic ultrasonography in CRS. 30
In a randomized, double-blind study of 60 patients with CRS, Ansari et al compared ultrasonography with erythromycin phonophoresis or ultrasonography alone. 34 The authors found a statistically significant improvement in total symptom scores in both groups; the degree of improvement was significantly greater in the erythromycin phonophoresis group. This emerg-ing treatment shows promise for patients with CRS, as the supporting positive evidence base appears to be growing.
In a study by Wang et al, phototherapy performed on rats with asthma was shown to considerably reduce levels of interleukin 4 (IL-4) in the bronchial lavage fluid and increase the levels of interferon gamma (IFNγ). 36 In addition, the concentration of eosinophils was decreased in the lung parenchyma and serum. Phototherapy resulted in a statistically significant improvement in the laser-treated rats, but the differ-ence between them and a control group treated with the inhaled steroid budesonide was not significant. These results in a disease process with pathophysi-ologic processes similar to those of CRS with nasal polyps suggest that phototherapy might play a role in treating the latter.
In a small clinical pilot study, Naghdi et al treated 15 CRS patients with low-level laser therapy over the frontal and maxillary sinuses for a total of 198 seconds per sinus. 37 The estimated amount of energy delivered to the treated area was 6 J. The entire regimen was delivered three times a week for 3 weeks plus once more for a total of 10 sessions. The authors reported
a significant reduction in symptoms compared with preintervention questionnaire scores. A similar study by Krespi and Kizhner demonstrated the clinical effi-cacy of near-infrared illumination with and without a topical photosensitizer, although these patients were not compared with those of a control group. 38
Phototherapy for CRS is in its relative infancy, and better-designed studies are needed to properly assess its clinical effect.
The results of a human trial of acupuncture conducted by Kim et al showed promise in the treatment of allergic rhinosinusitis. 40 Sertel et al published a double-blind, randomized, placebo-controlled trial of acupuncture that looked specifically at nasal blockage due to either CRS without nasal polyps or chronically hypertrophic inferior turbinates. 41 They found significant improve-ment in nasal airflow on anterior rhinomanometry in the treatment arm.
In a three-armed study by Stavem et al, one group re-ceived mainstream CRS treatment (antibiotics, steroids, saline lavage, and decongestants) for 3 months while the other two groups were administered acupuncture and sham acupuncture. 42 The differences in treatment results were not statistically significant, but they did trend toward favoring the mainstream treatment. No clinically significant difference was observed between acupuncture and sham acupuncture.
A similar study by Rössberg et al using the same three study arms for 2 to 4 weeks of treatment found no significant differences between acupuncture and mainstream CRS treatment based on patient question-naire scores and computed tomography findings over a 12-week follow-up. 43
Based on the results of these studies, we do not recommend the use of acupuncture for treating CRS.
Herbal supplements. In a North American study, Blanc et al surveyed the alternative medicine use of 175 individuals with rhinosinusitis, some of whom were not taking a prescription medication. 44 Their findings were surprising in that 26% of the respon-dents reported using at least one herbal remedy. Some 9% of respondents were taking a traditional Chinese herbal remedy.
Most herbal supplements are marketed in the form of an extract from one or more botanical sources. They
can be taken orally in capsule form or as an additive to tea. The most commonly used herbal supplements in CRS are echinacea, bromelain, and a multitude of Chinese herbal extracts and herbs. 44 Less common-ly used herbal supplements include ascorbic acid, N-acetylcysteine, quercetin, undecylenic acid, Urtica dioica, Esberitox, Myrtol, cineole, Bi Yuan Shu, and Ecballium elaterium.
Echinacea is a popular alternative medicine and is widely studied (Table 1). In CRS, the most common species used are Echinacea pallida, Echinacea purpurea, and Echinacea angustifolia. 45 Echinacea’s immuno-modulatory effect appears to be attributable to antiviral activation of the immune system, which is evident by the upregulation of the proinflammatory cytokines IL-1β, IL-6, IL -10, and tumor necrosis factor alpha (TNFα) in human macrophages. 46
Selected characteristics of some systemic herbal medicines proposed for the treatment of chronic rhinosinusitis.

Key: IL = interleukin; TNFα = tumor necrosis factor alpha; CRS = chronic rhinosinusitis; GI = gastrointestinal; NSAIDs = nonsteroidal anti-inflammatory drugs; IFNγ = interferon gamma; NF-κB = nuclear factor kappa-light-chain enhancer of activated B cells; FESS = functional endoscopic sinus surgery.
Bromelain is a pineapple extract consisting of prote-ase and nonprotease components (Table 1). Bromelain inhibits nuclear factor kappa-light-chain enhancer of activated B cells (NF-κB) and cyclooxygenase-2 (COX-2), and it stimulates the production of proinflammatory cytokines IL-1β, IL-6, TNFα, and IFNγ. 47 Three separate, randomized, controlled trials found that it reduced symptoms of CRS; however, all three trials are now more than 50 years old, and they involved patients with both chronic and acute rhinosinusitis. 48 -50
Butterbur (Petasites hybridus), a member of the sun-flower family, is purported to be effective in treating allergic rhinitis. 51,52 In CRS, evidence supporting its use is negligible.
Chinese traditional medicine. Ge Gen Tang is a traditional Chinese medicine that is made up of many herbs. It is prescribed by therapists for the treatment of gastrointestinal upset and upper respiratory tract infections. 53 A survey by Yen et al of CRS patients who took traditional Chinese medicines found that Ge Gen Tang use was common. 54 In another study, Chang et al used multiple in vitro cell lines to demon-strate that Ge Gen Tang exerts antiviral action against respiratory syncytial virus (RSV). 55 The mechanism of this action is still not understood, but the expo-sure of RSV to Ge Gen Tang resulted in a significant reduction in TNFα expression. There are no clinical data to suggest that Ge Gen Tang has any efficacy in the treatment of CRS.
Bi Yuan Shu is an herb mixture that contains Mag-nolia liliiflora, Xanthium strumarium, and Astragalus membranaceus (Table 1). It has been evaluated clinically in patients recovering from functional endoscopic sinus surgery (FESS) in combination with intranasal steroid and antibiotic therapy. In a study of 340 patients, a group randomized to Bi Yuan Shu combined with standard post-FESS therapy experienced significantly
less pain, discharge, and breathing difficulty than did those taking the standard treatment alone. 56 In patients with allergic rhinitis, Yang et al found that levels of IL-5, IFNγ, and COX-2 were significantly lowered when they were treated with a formula of similar Chinese herbs, suggesting a proposed mechanism for symptom reduction in CRS. 57
Homeopathy. Homeopathy in CRS involves treating both the underlying disease and its predominant symptoms with dilutions of precipitating, or “start-ing,” substances. There is much debate as to whether these dilutions actually contain any molecules of the original substance and hence whether any clinical effect is attributable to a placebo effect or study bias. 58 Starting substances may contain Sepia (cuttlefish ink), Pulsatilla (windflower plant), Lycopodium (club moss or wolf’s claw plant), or any number of other remedies.
Clinical studies that have evaluated homeopathic remedies in CRS are scarce. Nayak et al did conduct such an investigation—a prospective, observational study of 550 Asian Indian patients who had been di-agnosed with CRS and commenced on homeopathic treatment. 59 At 3 and 6 months of follow-up, significant improvements were observed in quality-of-life scores and radiologic evidence of CRS. 59 However, their study
did not include a control group, and the homeopathic treatments varied greatly among patients, which made the results difficult to interpret.
Sinfrontal is one homeopathic remedy that has been shown in a prospective, randomized, double-blind, pla-cebo-controlled trial to significantly reduce symptoms of acute maxillary sinusitis. 60 However, the exclusion criteria in that study included recurrent infections (three in the preceding 12 months) and CRS, so its efficacy in CRS was obviously not determined.
Discussion
Modern otolaryngologists and general practitioners must arm themselves with the knowledge of alterna-tive therapies that their patients may consider as part or all of their treatment for CRS. They must do so to educate their patients on the evidence surrounding their choices and the risks involved, to warn them of potential ineffective or even dangerous treatments that may lead to disastrous consequences.
It should be noted that significant bias (positive pub-lication bias) has been purported regarding studies of CAM therapies. This bias is likely reflected in the dis-cussion of the results in our review. Very few alternative therapies have been studied in true comparative clinical trials—or if they have, they remain unpublished. We have aimed to take this bias into our considerations and recommendations.
Most of the treatments discussed in this review are outside the realm of mainstream, and for the most part they lack sufficient evidence because of small study numbers, poor study design, or negative findings. However, some therapies, such as phonophoresis and therapeutic ultrasonography, do show some promise based on a growing body of positive evidence (Table 2). Similarly, positive clinical evidence has been found for the oral preparation Sinupret and the saline lavage additives baby shampoo, thyme honey, and bromelain.
Alternative therapies recommended for CRS.
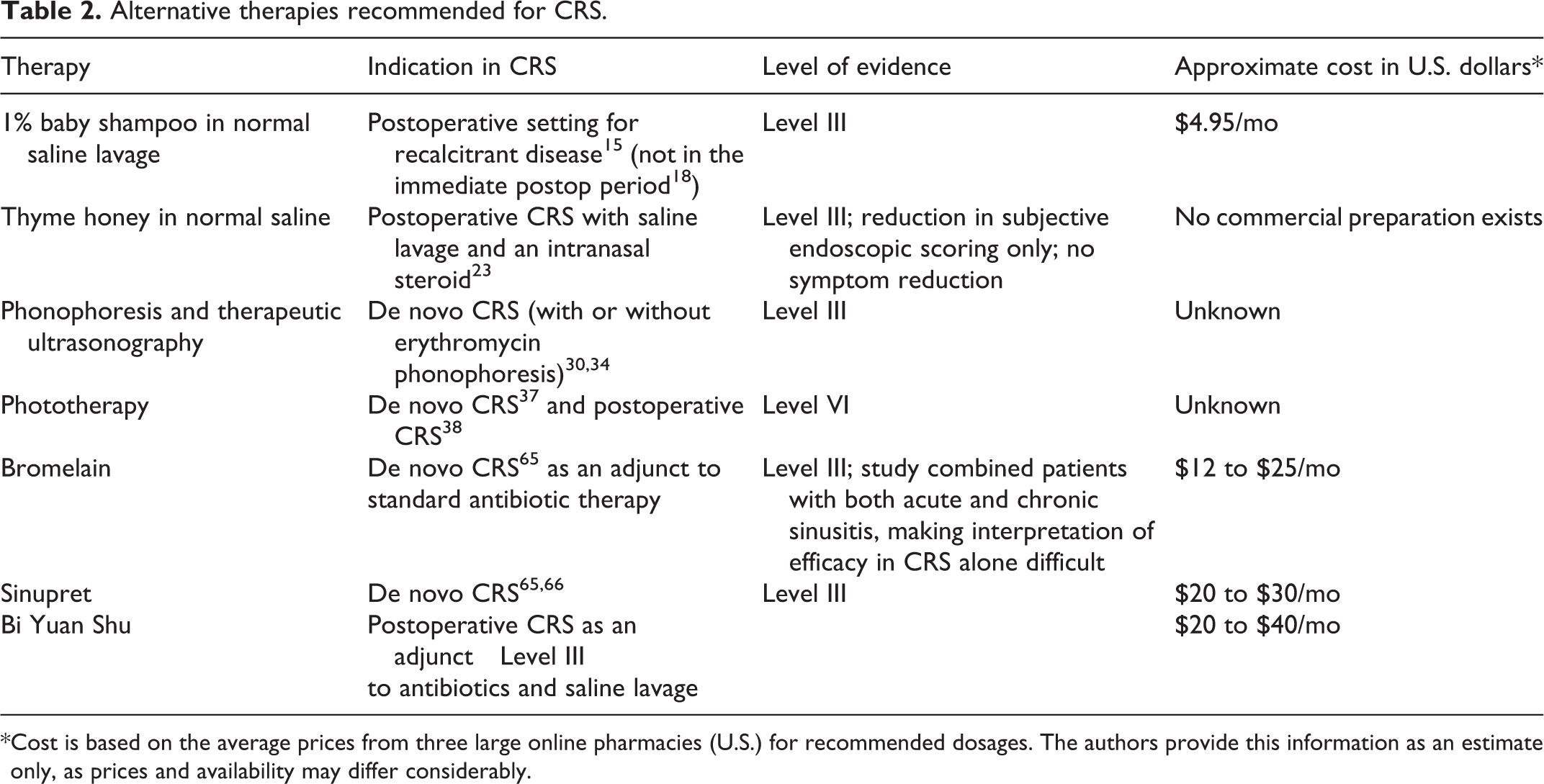
*Cost is based on the average prices from three large online pharmacies (U.S.) for recommended dosages. The authors provide this information as an estimate only, as prices and availability may differ considerably.
Overall, based on the low quality of evidence and poor study design associated with most clinical trials of alternative therapies, we cannot recommend any of these therapies for widespread use in CRS. The fact that most of these treatments are associated with a relatively low risk profile makes them an alluring alternative to mainstream treatment with steroids and antibiotics for some CRS patients. We urge readers to consider the nature of the therapy, the quality of the evidence, and individual patient factors before advising patients on their use.
Where possible, tertiary care referral centers should enroll patients in well-designed and well-powered clini-cal trials to expand the evidence base for these therapies.
