Abstract
Objectives:
Free tissue reconstruction of the head and neck must be initially overcorrected due to expected postoperative free flap volume loss, which can be accelerated by adjuvant radiation therapy. In this study, we aim to identify patient and treatment-specific factors that may significantly contribute to this phenomenon and translate these characteristics into a predictive model for expected percent free flap volume loss in a given patient.
Methods:
Patients with a history of oral cavity and/or oropharyngeal cancer who underwent nonosseous free flap reconstruction were reviewed between January 2009 and November 2018 at a tertiary care center. Demographics/characteristics, total radiation dose, radiation fractionation (RF), and pre/postradiation free flap volume as evaluated by computed tomography imaging were collected. Free flap volume was measured by a fellowship-trained neuroradiologist in all cases. Only patients receiving adjuvant radiotherapy with available pre/postradiation imaging were included. Multivariable linear regression modeling for prediction of free flap volume loss was performed with optimization via stepwise elimination.
Results:
Thirty patients were included for analysis. Mean flap volume loss was 42.7% ± 17.4%. The model predicted flap volume loss in a significant fashion (P = .004, R2 = 0.49) with a mean magnitude of error of 9.8% ± 7.5%. Age (β = 0.01, P = .003) and RF (β = −0.01, P = .009) were individual predictors of flap volume loss.
Conclusions:
Our model predicts percent free flap volume loss in a significant fashion. Age and RF are individual predictors of free flap volume loss, the latter being a novel finding that is also modifiable through hyperfractionation radiotherapy schedules.
Keywords
Introduction
The use of free tissue transfer for reconstruction of oncologic defects of the head and neck has become the standard of care over the last 2 decades and is routinely performed at tertiary care medical centers. However, reconstructive surgeons continue to face the challenge of how much volume to add in complex oral cavity and oropharyngeal defects as the reconstruction must balance the added bulk thought to improve swallowing and speech function versus upper airway patency.1,2 Further complicating the reconstructive surgery algorithm is the impact of adjuvant radiation therapy (RT) thought to significantly reduce adipofascial and muscle volume beyond the natural contracture of the flap soft tissue. Similarly, other factors such as type of flap, loss of body weight, muscle atrophy, and host protoplasm may play a role in flap volume loss.2-7
As an adequate reconstructive volume is vital for good functional and aesthetic outcome, inadequate initial free tissue volume leads to suboptimal long-term results.8-10 In fact, the shape and volume of oral cavity reconstructions have been closely linked to functional swallowing postoperatively, which patients have identified as being the most important outcome.10-12 Accordingly, most reconstructive surgeons overcorrect the initial defect in regard to free tissue volume.6,13,14 However, the appropriate degree of overcorrection may vary among patients. Although prior literature has identified patient-specific attributes contributing to this variation, no studies have sought to translate these variables into a predictive model that approximates the adequate volume of initial reconstruction. Furthermore, no patient or treatment specific modifiable factors have been identified that can lead to maintenance of reconstructive volume postoperatively. In this study, we aimed to develop such a model to help optimize head and neck reconstruction based on patient-specific characteristics with the goal of enhancing long-term cosmesis and function.
Methods
All cases of head and neck oncologic reconstruction using nonosseous free tissue transfer at a tertiary care medical center between January 2009 and November 2018 were retrospectively reviewed. Institutional Review Board approval was obtained for this study. Cases with defects outside the oral cavity and oropharynx were excluded, as were oral cavity reconstructions that did not reconstruct the tongue. Similarly, cases that did not undergo adjuvant postoperative irradiation at our institution were excluded, as radiation simulation and surveillance imaging were used to quantify free flap volume. Finally, cases of innervated reconstruction, free flap failure, and/or partial flap necrosis were excluded to prevent confounding factors in the resultant final flap volumes. Demographics, outcomes, and complications were reviewed. Free flap volume was measured within the patients’ radiation simulation and disease surveillance follow-up imaging. Follow-up imaging was performed at least 6 months following completion of RT. In all cases, the imaging reviewed was computed tomography (CT) imaging with ≤ 5.0-mm slice thickness. Patients who did not have these imaging studies available for review due to inadequate follow-up, failure to complete RT, or completion of RT at an outside institution were also excluded.
Free flap volume was measured by a fellowship-trained neuroradiologist in all cases. Volumetric flap measurements were made using Carestream Vue PACS version 12.2.2.0105. The flap tissue was hand segmented using Livewire Mode segmentation on all axial slices to generate a calculated volume (Figure 1). Best estimate of flap margins was used in areas of severe imaging artifact. In areas of difficult to determine flap margins, a similar estimate was used on pre- and postradiation studies. Percent free flap volume loss was calculated by dividing the post-RT completion volume by the initial free flap volume and subtracting that value from 1.
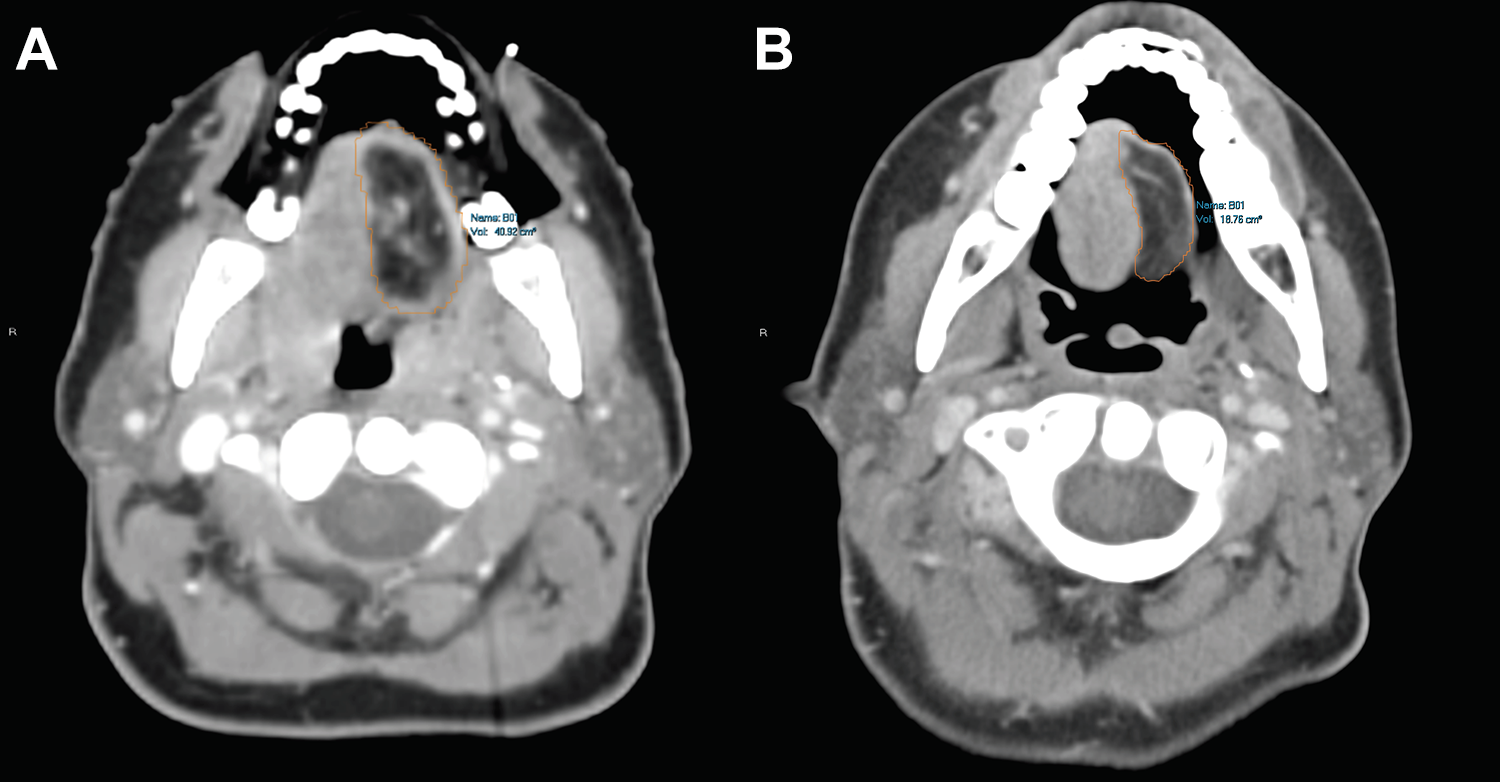
Representation of flap volume calculation in pre (A) and postradiation (B) imaging.
Multivariable linear regression modeling was performed evaluating the following independent variables in prediction of percent free flap volume loss: age, sex, body mass index, perioperative albumin, total radiation dose (RD), radiation fractionation (RF), smoking history, location of defect, and free flap type. The final model was created with inclusion of independent variables using a combination of forced entry and stepwise elimination, in that variables that had been previously associated with free flap volume loss2-7 were included in the base model, with subsequent elimination of variables not contributing to model fidelity as evaluated by the model’s adjusted R2. 15 The model was then internally validated with 5-fold cross-validation and root-mean-squared error (RMSE) and R2 values across the 5 model iterations were summarized with descriptive statistics. 16 Analysis was performed in Stata 15 (StataCorp LLC) with an alpha of .05 for statistical significance.
Results
In all, 939 cases of free tissue transfer were performed in the above time period; 291 of these cases were performed for nonosseous oral cavity or oropharyngeal oncologic reconstruction; 261 of these cases were excluded as they either did not have sufficient imaging available for review (ie had radiation at an outside institution etc), had evidence of recurrence on follow-up imaging, had nonglossectomy oral cavity defects, underwent innervated reconstruction, or had partial flap necrosis/flap failure. As such, 30 patients were included in the final analysis and model development. Fifteen patients (50%) had partial glossectomy defects (hemiglossectomy or subtotal glossectomy), 6 patients had total glossectomy defects (20%), 5 patients (17%) had isolated oropharyngeal defects, and 4 patients (13%) had defects spanning both subsites that were not total glossectomy defects. The average age of included patients was 59.7 ± 11.8 years, while men and women were approximately evenly distributed with females making up 47% (14/30) of the cohort. The majority of defects (21/30, 70%) were reconstructed with radial forearm free flaps (RFF), while the remainder were reconstructed with anterolateral thigh (ALT) free flaps. Mean total RD was 61.9 ± 0.9 Gy and mean percent free flap volume loss was 42.7% ± 17.4%. Post-RT imaging was performed at a mean of 10 ± 5 months following completion of RT (Table 1).
Cohort Demographics and Characteristics.

Abbreviations: BMI, body mass index; OC/OP, defect spans both subsites but is not a total glossectomy; OP, isolated oropharyngeal defect; PG, partial glossectomy; RFF, radial forearm free flap; SD, standard deviation; TG, total glossectomy.
The final predictive model for percent free flap volume loss included the following independent variables: age, sex, RF, type of free flap, and perioperative albumin. This model was found to be a significant predictor of percent free flap volume loss (P = .004) with an R2 of 0.49 and RMSE of 0.14. Among the aforementioned variables, age and RF were found to be independent predictors of percent free flap volume loss when controlling for the other variables in the model. Increasing age was found to be a significant positive predictor (β = 0.01, P = .003), meaning that each increase of patient age by 1 year correlated with a 1% decrease in flap volume. Conversely increasing RF through a hyperfractionation radiotherapy schedule was a significant negative predictor of free flap volume loss (β = −0.01, P = .009), with each additional fraction that the total RT dose was delivered over correlating to preservation of 1% flap volume. The complete predictive model can be found in Table 2. The mean magnitude of error (MOE) in prediction of percent free flap volume loss was 9.8 ± 7.5% across all cases. Five-fold cross-validation of the model demonstrated mean R2 and RMSE values of 0.52 ± 0.06 and 0.14 ± 0.004 respectively, indicating that the model was not overfit.
Multivariable Linear Regression Model for Prediction of Free Flap Volume Loss.a,b
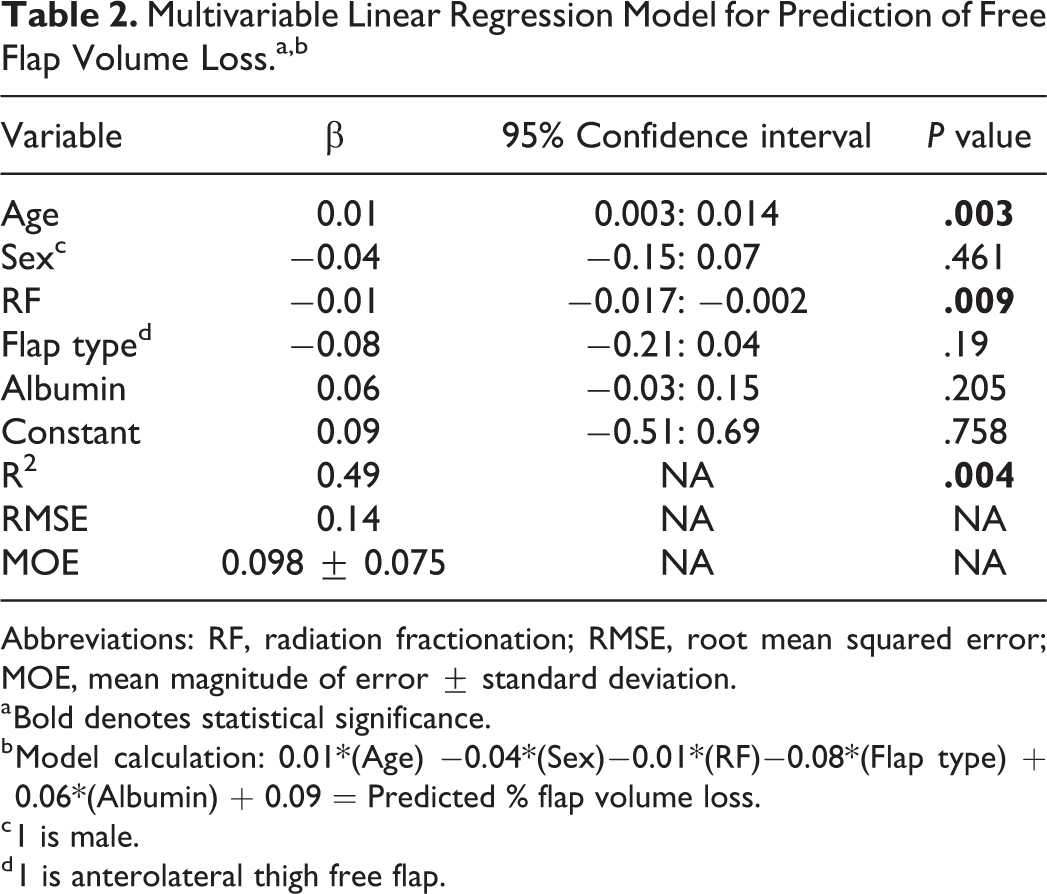
Abbreviations: RF, radiation fractionation; RMSE, root mean squared error; MOE, mean magnitude of error ± standard deviation.
a Bold denotes statistical significance.
b Model calculation: 0.01*(Age) −0.04*(Sex)−0.01*(RF)−0.08*(Flap type) + 0.06*(Albumin) + 0.09 = Predicted % flap volume loss.
c 1 is male.
d 1 is anterolateral thigh free flap.
Discussion
It is well recognized that the volume of free tissue transfer reduces over time, resulting in the need for initial overcorrection6,7 (Figure 2). However, overcorrection is currently an empirical endeavor, with the degree suggested ranging from 10% to 40% within the literature.3,5,7,11 This is despite the known importance of adequate reconstructive bulk, particularly within the oral cavity where it is a vital factor in obtaining favorable speech and swallow outcomes postoperatively.5,17 Moreover, although certain patient-specific factors have been linked to greater free flap volume loss, there has been no translation between the presence/absence of one or more of these characteristics and the ideal initial reconstructive volume in a given patient up to this point.2-7 Similarly, no modifiable factors have been identified that contribute to the loss of free flap volume following adjuvant RT. As such, this is the first study, to our knowledge, that develops a model that predicts the degree of flap volume loss based on patient-specific characteristics, while also identifying a modifiable treatment specific factor that may help preserve free flap volume.
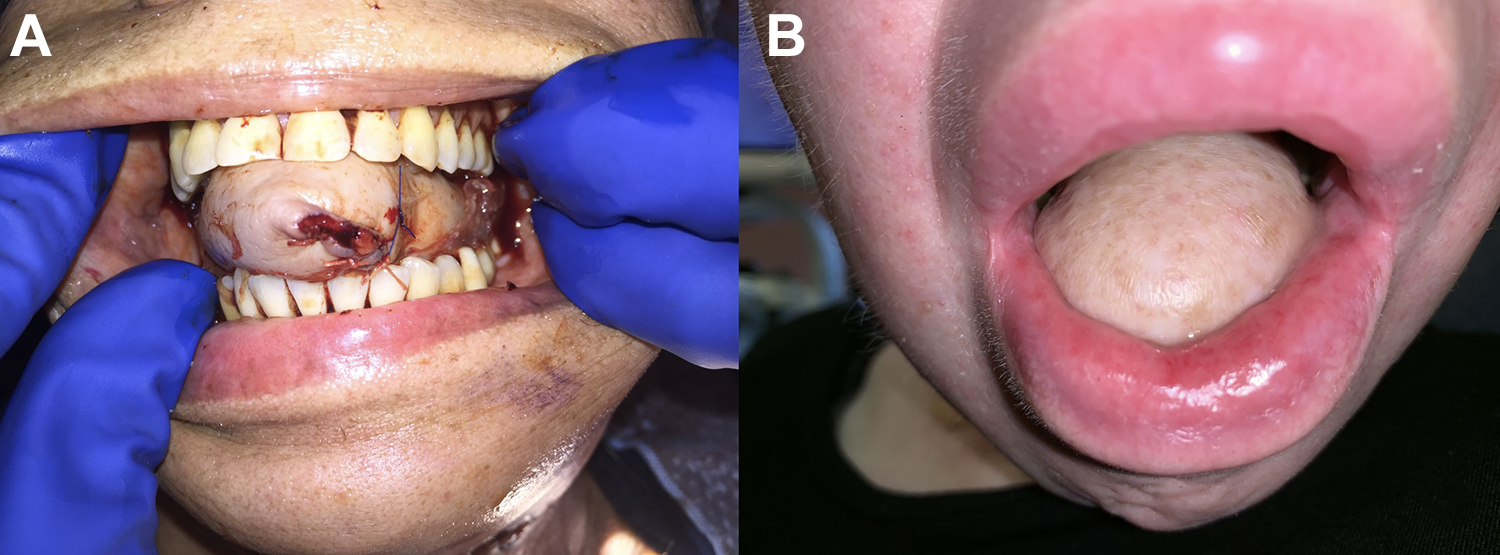
Pre (A) and 1 year postradiation photo (B) of a patient who underwent total glossectomy with anterolateral thigh free flap reconstruction. Note the soft tissue volume loss of almost 40% postradiation in this patient.
The model developed in this study significantly predicted percent of free flap volume loss (P = .004, R2 = 0.49), while 5-factor cross-validation demonstrated that the model was not overfit. The calculated R2 value signifies that the model was able to account for 49% of the variability seen in percent free flap volume loss across the 30 cases. Moreover, it was able to predict the percent free flap volume loss to within an average of 9.8% ± 7.5% in each case. In other words entering the patient-specific factors of age, sex, free flap type (RFFF vs ALT), planned RF, and perioperative albumin into the model provided in Table 2 predicted each patients postradiation percent flap volume loss to within an average of 9.8%. This can be done by multiplying a given patient’s demographic information by the respective β value and summing these values with the constant (Table 2).
When considering other potential sources of variability in free flap volume loss and error in the model, the most notable source is the radiographic delineation of the free flap margins. Although, a single fellowship-trained neuroradiologist hand-segmented all free flap volumes, the ease with which the flap margins could be identified varied in each case due to factors such as dental artifact. Moreover, magnetic resonance imaging (MRI) may be a better tool to accurately identify flap margins, regardless of the presence/absence of artifact. Given the retrospective nature of this study, this imaging modality was unavailable for review. That being said, the overall mean flap volume loss of 42.7% is consistent with prior literature examining flap volume loss following RT as evaluated by MRI, which reported a mean volume loss of 44.2%. 7
The model identified the influence of RF on percent free flap volume loss when controlling for the other variables investigated. Radiation fractionation was found to be an individual negative predictor of free flap volume loss, in that each additional fraction reduced the degree of free flap volume loss by 1% (β = −0.01, P = .009). In our study population, RF represented an increase in the number of radiotherapy fractions delivered using a hyperfractionation radiotherapy schedule, where the total number of fractions is increased but delivered twice daily at a lower than standard dose per fraction. The finding that RF is protective of free tissue reconstruction volume is novel, and to our knowledge, has not been previously described. This impact of RF supports our a priori hypothesis that based on radiobiologic principles, the risk of late-manifesting normal tissue complications is influenced not only by the total RD but by the dose per fraction. Our group postulated that the use of a hyperfractionated schedule would be beneficial when reirradiation was indicated. Moreover, reirradiation was commonly used in the postoperative setting where flap reconstruction with unirradiated tissue offered potentially more effective neovascularization of the surgical wound. Our findings are consistent with a hyperfractionated radiotherapy schedule associated with a trend toward less late normal tissue complications in RTOG 9003 with 10 years of clinical follow-up 18 and a lower risk of radiation-induced optic neuropathy (RION) and retinopathy.19.20 Whether the reduced flap volume loss was due to less direct normal cell injury or endothelial cell injury due to the reduced radiotherapy dose per fraction delivered is not clear. The importance of this finding is that RF may be a modifiable risk factor, in that utilization of hyperfractionated schedule for adjuvant radiotherapy may help preserve the volume of free tissue reconstruction. This is something that requires further prospective study.
Conversely, age was found to be a positive predictor of percent free flap volume loss (β = 0.01, P = .003). In the context of RF providing a protective effect on the free flap, we speculate that the impact of age may represent a reduced ability to repair the cellular sublethal damage caused by radiation. Alternatively, the impact of age may represent the vascularization health of the donor vessels and of the flap. That RION is felt to be related to vascular injury by radiation and similarly increased with age19.20 is consistent with our finding that age also influences the free flap volume.
Although our study provides valuable preliminary data, barriers remain to clinical implementation and use of this model to appropriately size the reconstructive volume. Given the retrospective study design, we were limited to inclusion of patients who had postoperative RT as radiation simulation and follow-up imaging was used for free flap volume determination. Moreover, these patients had to have undergone RT at our institution to have imaging available for review. This both severely limited our sample size for model development, while also failing to provide information regarding flap volume change in patients undergoing single modality surgical management. On the other hand, it is a rare clinical occurrence for a patient to require a free flap reconstruction, but not require radiation postoperatively as multimodality treatment is inherent in these advanced staged tumors requiring complex reconstruction. Furthermore, as there was very little variability in our cohort in total RD (mean dose of 61.9 ± 0.9 Gy) we were unable to adequately draw conclusions regarding a potential dose dependent relationship between irradiation and free flap volume loss. As previously discussed, we relied on CT imaging for free flap volume determination as opposed to MRI, which may be more accurate for flap margin delineation. Finally, radiation simulation imaging was used to determine baseline flap volumes. The timing of this imaging in relation to when the initial procedure was performed may vary among patients. This could potentially lead to errors in determining the true initial volume of the flap, as patients undergoing early postoperative simulation may have had overestimation of flap volume owing to postoperative edema, while those with delayed radiation may have underestimation due to flap contracture. To ultimately validate our findings and develop a model to be used clinically, a prospective study utilizing interval MRI’s and nonradiated control patients may be necessary. Nevertheless, our preliminary work provides a promising foundation for such a study to be designed from.
Conclusions
Free flap volume loss can be predicted in a significant fashion in oral cavity and oropharyngeal reconstruction in those receiving adjuvant RT based on patient-specific characteristics. Age and RF are individual predictors of free flap volume loss, with increased age being a propagating factor for volume loss and increased RF being a protective factor, which is a novel finding. Our findings will be valuable in the prospective development of similar models to be used clinically for accurate sizing of initial reconstructive volumes. Doing so may ultimately lead to enhanced functional speech and swallow outcomes in oral cavity and oropharyngeal reconstruction, and eliminate the need for secondary reconstructive surgeries.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded in part by an NIDCD Grant: NIDCD T32 DC000027.
