Abstract

A 38-year-old female presented with a 12-month history of a palpable mass below her left ear that had been stable in size. She denied any fevers, chills, night sweats, cough, or weight loss. Her medical history was otherwise unremarkable. She was born in the Dominican Republic and moved to the United States 4 years prior to her initial evaluation. She denied any personal or family history of tuberculosis (TB). Physical examination revealed a 2-cm firm, tender posterior auricular mass at the angle of the left mandible with no overlying skin changes. Facial nerve function was intact.
Fine needle aspiration cytology (FNAC) was performed showing abundant lymphocytes and cysts containing foamy macrophages and scant oncocytes, consistent with Warthin’s tumor. No malignant cells were detected. Computed tomography (CT) imaging demonstrated a multilobulated, peripherally enhancing structure at the superolateral aspect of the left parotid, measuring 2.1 cm × 1.1 cm × 1.1 cm (Figure 1). Multiple enlarged cervical lymph nodes on the ipsilateral side were identified. In contrast to the cytology, the features on CT were more suggestive of an infectious or inflammatory process.
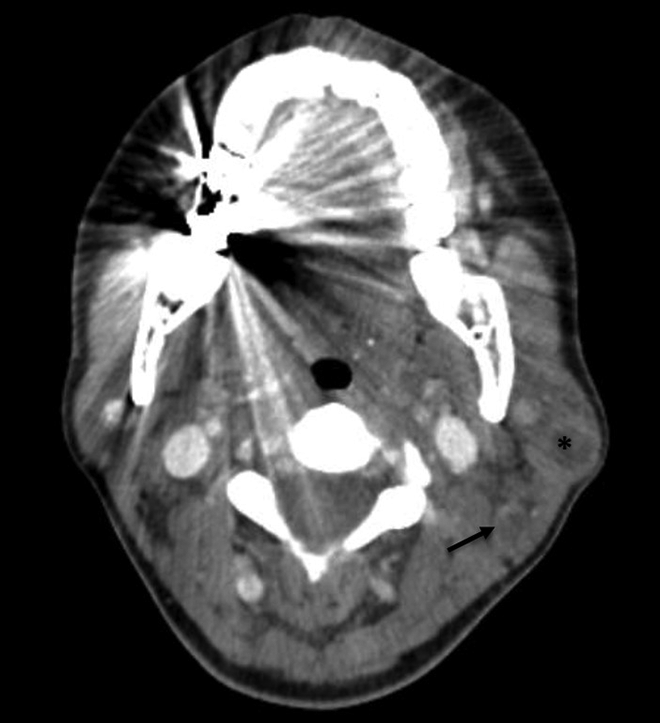
Computed tomography showing a superficial, multilobulated, peripherally enhancing, internally necrotic cystic mass in the left parotid gland (asterisk). An ipsilateral centrally low attenuation level IIB lymph node is also present (arrow).
Despite this imaging result, the patient had no active signs of infection and no recent travels outside of the United States. Therefore, considering the FNAC results, which were confirmed by a second cytopathologist, and the patient’s preference for surgical removal, a left superficial parotidectomy with facial nerve preservation was performed, and a tan-pink, rubbery mass was excised. Culture of the specimen was not obtained. Histopathological examination demonstrated a necrotizing granulomatous inflammation of an intraparotid lymph node (Figure 2). The surgical specimen was negative for fungal and acid-fast bacilli (AFB) stains. A serum interferon γ assay, performed using the QuantiFERON-TB Gold Plus (QIAGEN), was positive. Chest radiography was normal. Follow-up CT performed 2 months postoperatively showed resolution of the previous necrotic mass within the left parotid gland.
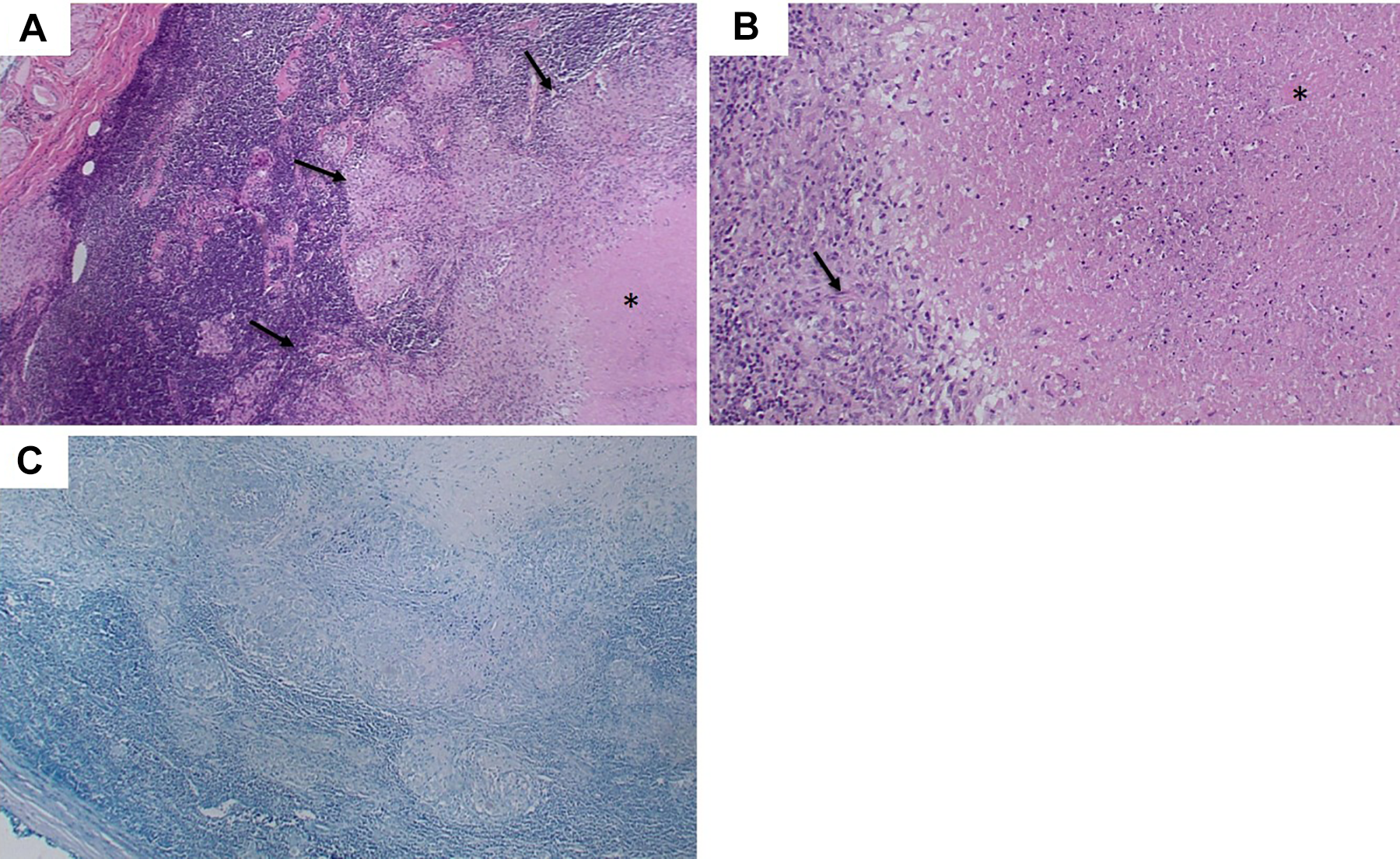
A, Hematoxylin–eosin-stained photomicrograph at ×40 shows necrotizing granulomatous inflammation replacing a lymph node within the parotid gland with central necrosis (*) and granuloma (outlined by arrows), B, Hematoxylin–eosin-stain at ×200 demonstrates the granuloma composed of epithelioid macrophage (arrow) and necrosis (*). C, Acid-fast-stained specimen at ×40 is negative for the presence of acid-fast bacilli.
Due to the presence of necrotizing granulomata on pathology, paraffin blocks were sent to the Centers for Disease Control and Prevention (CDC) in Atlanta, Georgia. Molecular testing using genetic material extracted from the specimen was performed, which was positive for the Mycobacterium tuberculosis complex species. The patient was diagnosed with active TB disease of the left parotid gland and started on 4-drug antituberculous therapy with isoniazid, rifampin, pyrazinamide, and ethambutol. Six weeks into treatment, results of additional molecular testing done at the CDC showed a katG gene mutation, which is associated with isoniazid resistance. Isoniazid was subsequently switched to moxifloxacin, with a plan to continue the patient on this regimen for at least 6 months. 1
Extrapulmonary manifestations of TB disease are not uncommon, representing approximately 20% of active TB cases in the United States. 2 Within the head and neck, cervical lymphadenopathy accounts for the majority of cases, followed by laryngeal TB and tuberculous otitis media.3,4 Tuberculosis of the parotid gland is rare even in endemic regions of the world. 3
The typical clinical presentation of parotid gland TB is insidious onset of a slow-growing, asymptomatic localized lesion within the gland. Constitutional symptoms are usually absent. 3 As such, the mass is often mistaken for a neoplasm.5,6 Tuberculosis should be suspected in individuals at high risk for the disease, including those born in or who have traveled to endemic countries or who reside in congregate settings. Compared to a typical patient with malignancy, these patients present at a younger age, usually 30s to 50s.3,4
The detection of mycobacterial species on culture remains the gold standard for diagnosing TB disease.3,4 However, a culture-confirmed diagnosis may be elusive, as demonstrated by our patient in which a culture was not initially performed due to lack of suspicion. Furthermore, the negative AFB stain on the surgical specimen suggested a paucity of organisms so it is unclear if a culture would have yielded growth. Based on our experience, we suggest that when TB is suspected, a core biopsy or repeat aspiration be attempted first with specimens sent for AFB staining and culture. Histology is typically characterized by granuloma formation with or without caseating necrosis with epithelioid cells or Langerhans giant cells.3,4,7 Additionally, polymerase chain reaction (PCR) can serve as an important diagnostic tool, especially when a culture cannot be obtained.3,7 For our patient, the positive PCR allowed for confirmation of TB disease following the initial suspicion based on the surgical pathology result.
The differential diagnosis of a parotid gland mass remains broad, including sarcoidosis, Sjögren syndrome, abscess, and benign versus malignant neoplasm. Nevertheless, a high degree of suspicion and timely recognition of parotid gland TB may spare patients unnecessary surgery and lead to prompt treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
