Abstract

Malignant peripheral nerve sheath tumors (MPNSTs) arise most commonly in patients with neurofibromatosis type 1 (NF-1) but may occur after radiotherapy as also in the absence of any clinical predisposition. 1 A 2-tiered grading system with low- (neurofibroma-like) and high-grade MPNSTs has been proposed, 2 the latter typically characterized by limited, if any, evidence of Schwannian differentiation at immunohistochemical level.
In the parapharyngeal space, MPNST is rare. 3 It has been reported both in patients with and without NF-1 and exceptionally has been demonstrated to arise from the hypoglossal nerve. 4
We report a 50-year-old woman with a 6-month history of right neck painful swelling, right ear fullness and otalgia, and progressive dysphagia to solid foods. Prior radiotherapy was denied by the patient. Clinical examination revealed a tough and poorly moveable cervical mass and medialization of the right anterior tonsillar pillar and right lateral deviation of the tongue during the protrusion (Figure 1A). Laboratory investigations and radiography of the chest were normal. Contrast-enhanced computed tomography scan (Figure 1B) and contrast-enhanced magnetic resonance imaging (Figure 1C and D) reveled a nodular enhancing lesion of 43 mm × 40 mm × 30 mm with a necrotic core at the level of the carotid bifurcation. Enlarged lymph nodes were not detected. The mass was excised through a transcervical approach. At surgery, it was localized in the poststyloid compartment of the parapharyngeal space and appeared strictly adherent to the hypoglossal nerve. A careful sharp and blunt dissection was performed with sparing of the trunk of the hypoglossal nerve. Based on the clinical evidence of tongue paresis and on the strict association of the tumor with the hypoglossal nerve, the tumor was considered to be arisen from it.
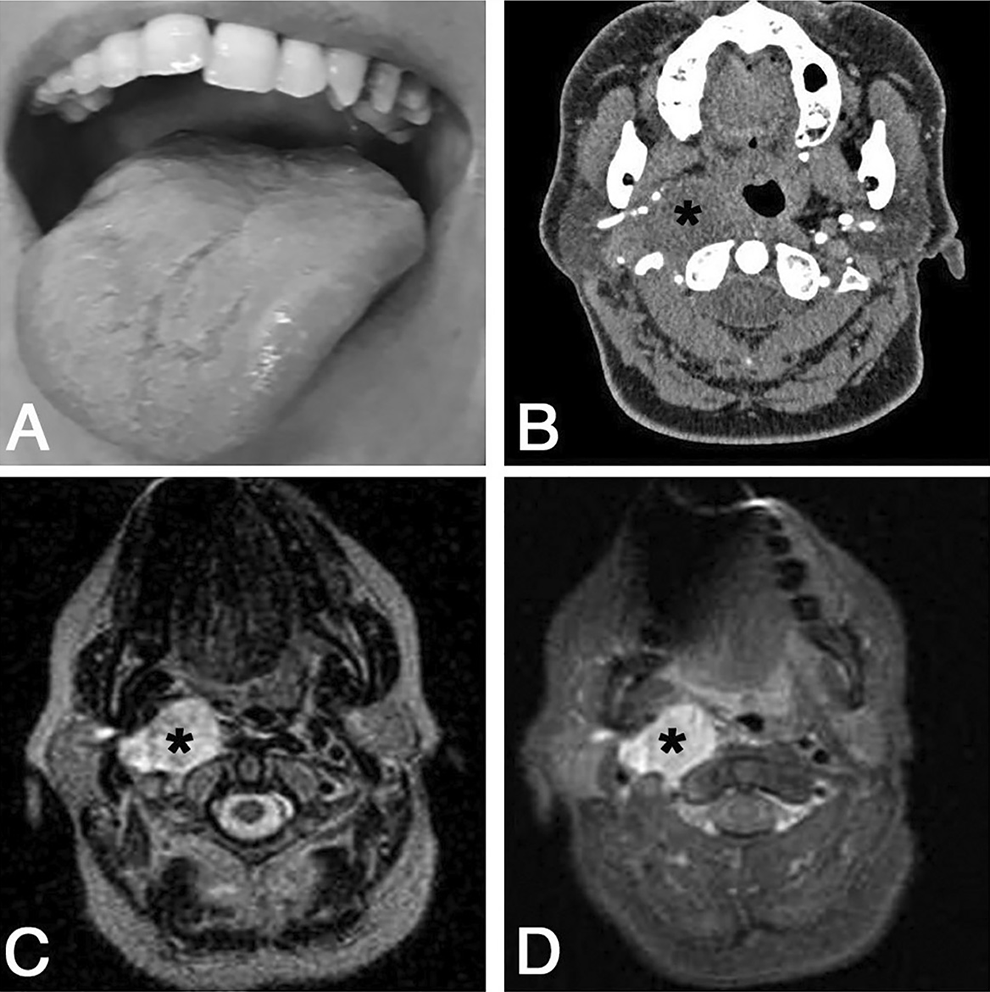
Right lateral deviation of the tongue during the protrusion is illustrated in (A). Contrast-enhanced computed tomography scan and axial T2-weighted and fat-sat magnetic resonance imaging are illustrated in (B), (C), and (D), respectively. The tumor involves the right parapharyngeal space, shows a necrotic central core, and determines medialization of the pharyngeal wall displacement of the right internal and external carotid arteries. The tumor is identified by the asterisks (B-D).
Pathologic examination of the mass revealed a neoplastic tissue composed of spindle cells showing a fascicular pattern of growth. Densely cellular areas (Figure 2A and B) alternated with hypocellular (Figure 2C and D) ones. Focally, neoplastic cells show marked pleomorphism (Figure 2E). Mitosis were abundant and necrosis extensive. Skeletal muscle elements, bone, cartilage, and glandular structures were not detected. Immunohistochemical analysis (Figure 3), performed as described previously, 5 revealed immunoreactivity of the neoplastic cells for vimentin and INI-1 and focal for CD34 but not for S100, GFAP, smooth muscle actin, muscle-specific actin, myogenin, desmin, CD117, DOG1, STAT6, p16, and CKAE1AE3. “Intact” H3K27me3 was revealed by immunohistochemical staining according to previously published criteria. 6 The proliferative activity (Ki67) was about 20%. The pathological findings were considered consistent with a high-grade MPNST.
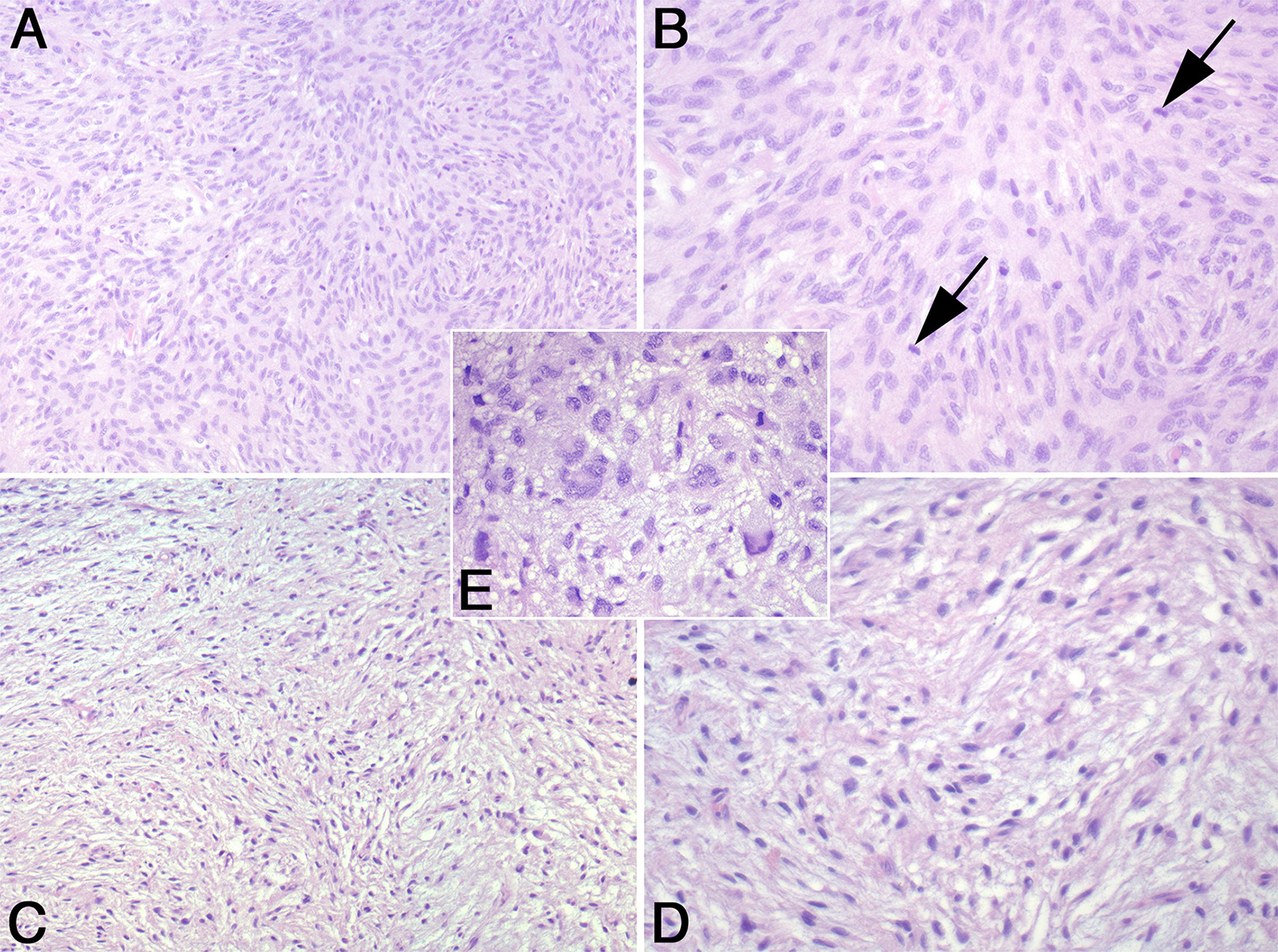
Low- (A and C) and high-power magnification (B and D) of the hypercellular (A and B) and hypocellular (C and D) areas of the tumor. The neoplastic spindle cells are arranged in fascicles. An area with pleomorphic cells is shown in (E). The arrows in (B) indicate mitotic figures. (A-E) Hematoxylin-eosin. Original magnification: ×40 for (A) and (C), ×80 for (B) and (D), and ×100 for (E).
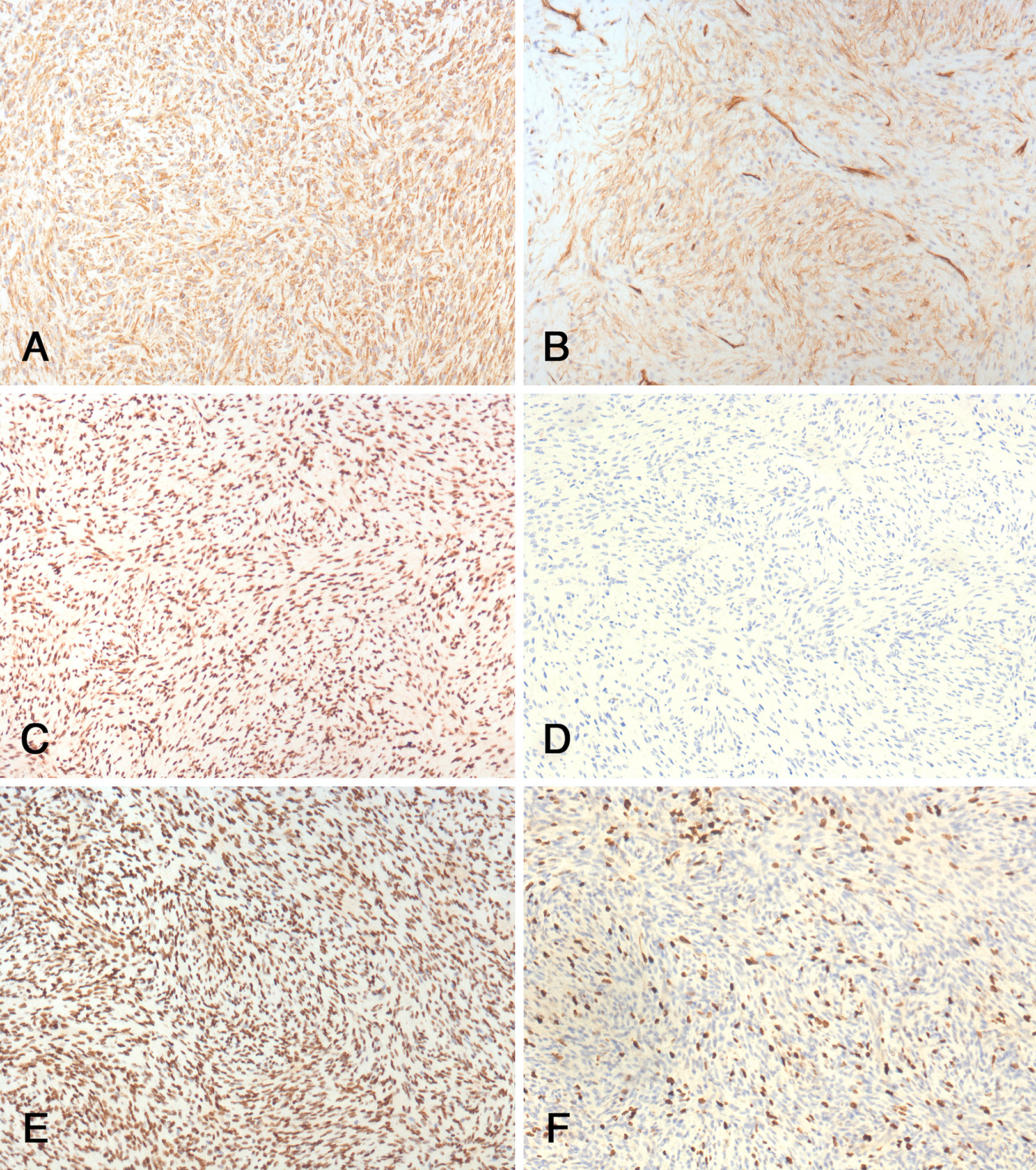
Immunohistochemical profile of the tumor. A, Vimentin. B, CD34. C, INI-1. D, S100. E, H3K27me3. F, Ki67. Original magnification ×40.
Based on this diagnosis, clinical workup for NF-1 was performed but it was negative. After a multidisciplinary discussion of the case, chemoradiotherapy was proposed. However, it was refused by the patient who decided to undergo treatment and follow-up in another institution.
Malignant peripheral nerve sheath tumors of the parapharyngeal space represents a relevant differential diagnostic challenge for clinicians, in particular for the huge variety of benign and malignant tumors that may arise in this region and for the lack of specific symptoms. 3 Similarly, the diagnosis of MPNST remains challenging for the pathologist because of its nonspecific morphologic features and lack of pathognomonic immunohistochemical markers. Indeed, MPNSTs may show significant morphological overlap with other soft tissue tumors including cellular or atypical neurofibroma or high-grade sarcomas such as undifferentiated pleomorphic or spindle cell sarcoma, and immunoreactivity for S100, the most specific marker of Schwannian differentiation, is positive (usually focally) in less than 50% of the cases. 1 As a consequence, a high interobserver variability may exist particularly when MPNST is encountered outside the well-established predisposing clinical conditions (NF1 and prior radiotherapy). In sporadic MPNST, the diagnosis is mainly made on the basis of matching morphologic features, patchy S100 protein immunostaining, involvement of a nerve, and exclusion of other possible pleomorphic and spindle cell neoplasms. A recently identified important diagnostic tool is the loss of H3K27me3 by immunohistochemistry. 6 In our case, H3K27me3 was “intact.” However, this does not contrast with the diagnosis. In one series, H3K27me3 was reported to be retained in 68% of sporadic MPNST. 6
Independent of the site of origin, MPNSTs are aggressive tumor, with a high rate of local recurrence and distant metastasis.1,7 In a recent study on head and neck MPNSTs, 62% of patients developed local recurrence and 46% distant metastasis. 7 Surgical resection with negative margins is the mainstay for the treatment of MPNST. The role of radiotherapy and adjuvant chemotherapy is limited and recommended in the setting of metastatic disease, large-size tumors, and late-stage tumors.
Footnotes
Authors’ Note
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request. All the clinicopathologic investigations detailed in the manuscript have been conducted in accordance with the Declaration of Helsinki and its later amendments or comparable ethical standards. Written informed consent for publication of data and images was obtained from the patient.
Acknowledgments
The authors gratefully acknowledge Prof Angelo P. Dei Tos (Department of Medicine, University of Padua, School of Medicine, Padua, Italy) who confirmed the pathologic diagnosis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
