Abstract
Skull base osteomyelitis (SBO) is an invasive infection refractory to therapy, closely linked with malignant otitis externa (MOE). It is characterized by a mild clinical presentation that can delay cross-sectional imaging considered as the key to revealing it. Skull base osteomyelitis typically affects elderly diabetics and immunocompromised patients (>70 years). It most commonly has an otogenic origin due to an extension of MOE. The prognosis can be very poor without the administration of adequate and timely therapy at an early disease stage. Nowadays, Pseudomonas aeruginosa remains the most common pathogen associated with SBO. Fungi are a rare cause of MOE. This report documents a rare case of otogenic SBO caused by Candida parapsilosis in a diabetic patient, with persistent otologic symptoms as clinical onset and resistance to medical treatment. Fungal MOE has more subtle symptoms and is more aggressive than its bacterial counterpart. When MOE is resistant to antibacterial drugs, this should raise the suspicion of a fungal etiology of MOE. The current guidelines do not exhaustively describe the diagnosis, antifungal drugs of choice, and optimum duration of treatment. The description of these rare clinical cases should help with the multidisciplinary management of this disease in order to optimize the diagnosis and therapeutic protocol.
Keywords
Introduction
Skull base osteomyelitis (SBO) is a serious infection refractory to therapy, closely linked with malignant otitis externa (MOE). The clinical concepts of SBO and MOE were formalized in the 1950s and 1960s. 1 Skull base osteomyelitis has an indolent, complicated course due to the risk of skull base neurovascular involvement and intracranial extension. 2 Skull base osteomyelitis is characterized by a mild clinical presentation that can delay cross-sectional imaging considered as the key to revealing skull base disease; it can be confused with malignancy on imaging, thereby delaying the correct diagnosis of SBO. 1
Skull base osteomyelitis typically affects elderly diabetics and immunocompromised patients (>70 years). 3 It commonly has an otogenic origin due to an extension of MOE, characterized by otological signs and symptoms such as ear pain. However, SBO can also be of sinogenic, rhinogenic, odontogenic, or pharyngogenic origin. Nonotogenic SBO is defined “central/atypical SBO.” It can present as a headache, facial pain, and cranial neuropathy.1,2 The prognosis can be very poor without the administration of adequate and timely therapy at an early disease stage.
Nowadays, Pseudomonas aeruginosa remains the most common pathogen associated with SBO, but the incidence of other pathogens has increased, such as Aspergillus spp, Staphylococcus aureus, Klebsiella oxytoca, Stenotrophomonas maltophilia, and Raoultella ornithinolytica.1,2,4 Fungi are a rare cause of MOE. 5
This report documents a rare case of otogenic SBO caused by Candida parapsilosis in a diabetic patient, with persistent otologic symptoms as clinical onset and a prompt response to medical treatment. We also provide a review of the literature findings concerning the management of SBO in order to optimize the therapeutic protocol.
Case Report
An 87-year-old Caucasian male patient was admitted to the Ear Nose Throat Unit at the University of Federico II in Naples with a 2-month history of recurrent right external otitis resistant to antibiotic therapy with worsening pain in the right temporal region, occipital area, and neck. Past medical history was positive for hypertension, uncontrolled diabetes mellitus type II complicated by peripheral vascular disease, and ischemic stroke without neurological sequelae. Notably, the patient had an ear swab negative for bacteria and fungi.
Physical examination showed right purulent otorrhea associated with edema of the external auditory canal, which was full of epithelial debris and granulation tissue. The skin on the right mastoid appeared hyperemic and sore at palpation. The tympanic membrane on the left ear was normal. For the preceding 2 weeks, the patient showed a complete right facial palsy of degree VI on the House-Brackmann scale (Figure 1). Tonal audiometric examination revealed high-grade mixed deficiency (85 dB) on the right ear and medium-grade sensorineural hearing loss on the left one (Figure 2). Inflammatory markers, for example, white count cells, erythrocyte sedimentation rate, and C-reactive protein, were higher than normal.
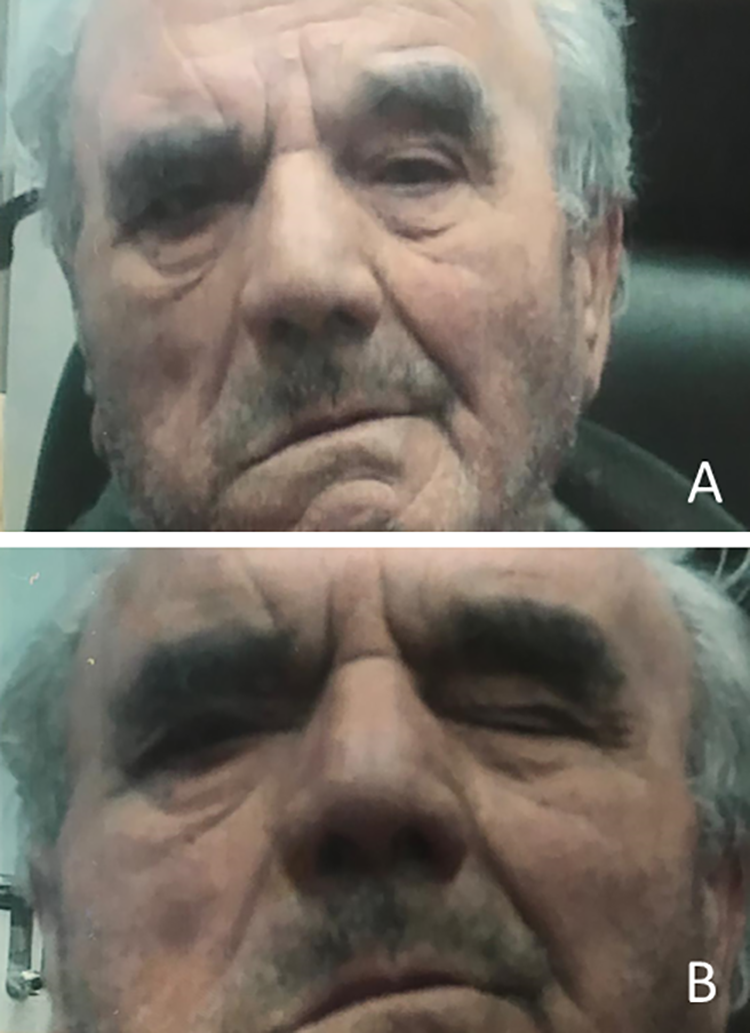
A, The patient face appeared asymmetrical, fixed in a static and dynamic position on the right side compatible with complete right facial paralysis—VI degree of House-Brackmann Scale. B, Bell’s phenomenon with attempted eye closure.
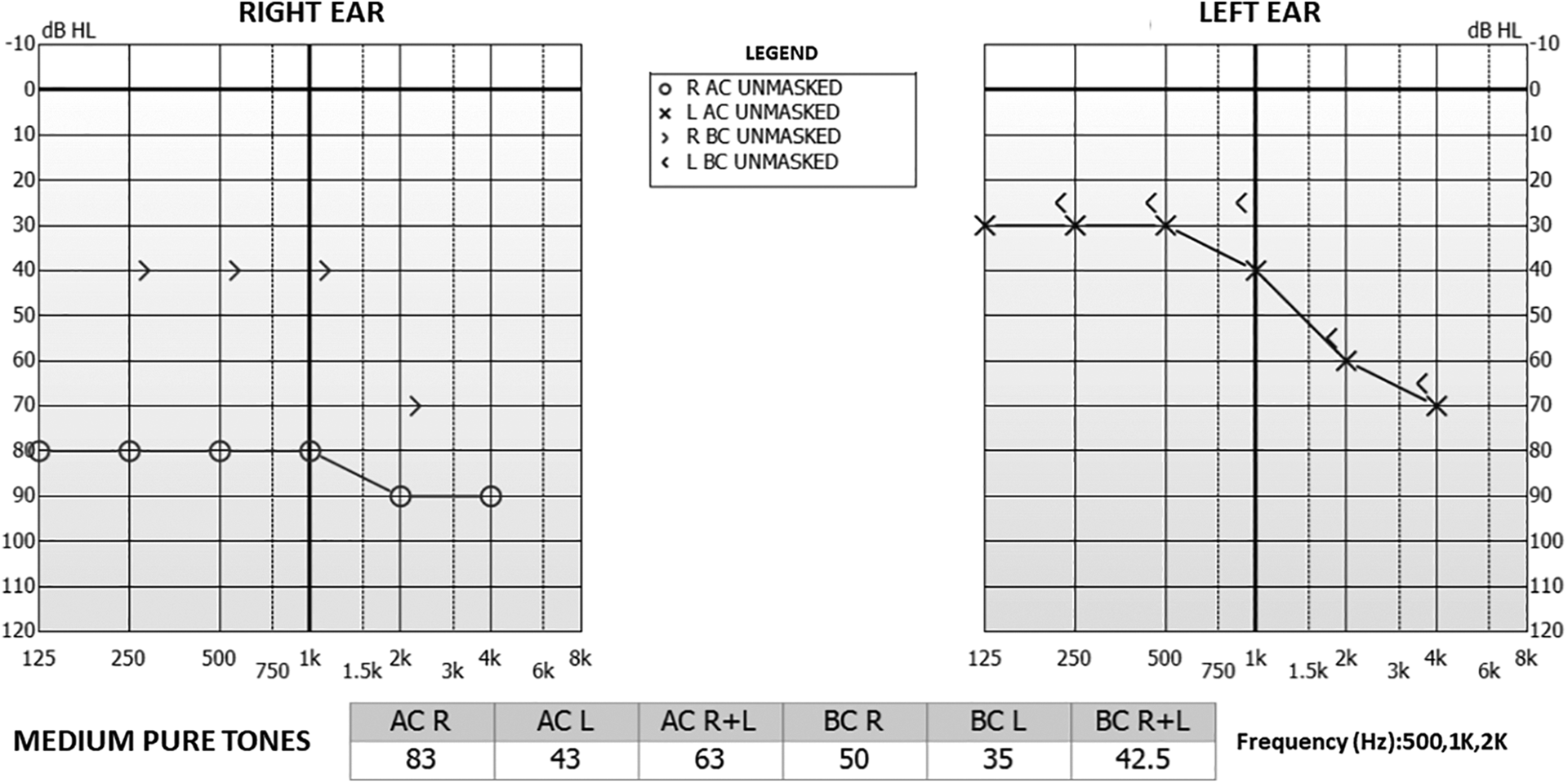
Tonal audiometric examination showed high-grade mixed deficiency (85 dB) on the right ear and medium-grade sensorineural hearing loss on the left one. O indicates right air conduction unmasked; X, left air conduction unmasked; >, right bone conduction unmasked;
The suspicion of right malignant external otitis was therefore raised, so empiric therapy was immediately started based on carbapenems and fluoroquinolones (meropenem associated with oral ciprofloxacin for 2 weeks) in combination with intravenous corticosteroids despite known diabetes to ensure early good control of edema and pain. In the meantime, the patient underwent both high-resolution computed tomography (CT) scan and contrast-enhanced magnetic resonance imaging (MRI) of the skull base. The CT scan (Figure 3) showed a thickened right external auditory canal, middle ear cavity opacification with slight widening of the antrum and tegmen tympani, and involvement of the right nasopharynx and parapharyngeal space through the Eustachian tube. The masticator space and retrocondylar soft tissues were also thickened, with mild and inhomogeneous postcontrast enhancement. The apex of the petrous bone was remodeled and partly eroded, with a reduction in the diameter of the right internal carotid artery (ICA) compared to the contralateral side, especially in the horizontal intrapetrous segment. The jugular vein and intracranial sigmoid sinus opacification were normal. Magnetic resonance imaging examination (Figure 4) confirmed an asymmetric nasopharynx and parapharyngeal space due to right-sided thickening with soft tissue swelling and opacification of the mastoid air cells. Postcontrast acquisition showed increased enhancement with stenosing soft tissue thickening extending from the right external auditory canal through the Eustachian tube up to parapharyngeal space, temporomandibular joint, and medial pterygoid muscle, with inner nonenhancing necrotic areas. Petrous bone apex involvement with reduced right intrapetrous ICA diameter and sigmoid sinus patency were confirmed, while no sign of intracranial invasion or dural enhancement was observed.
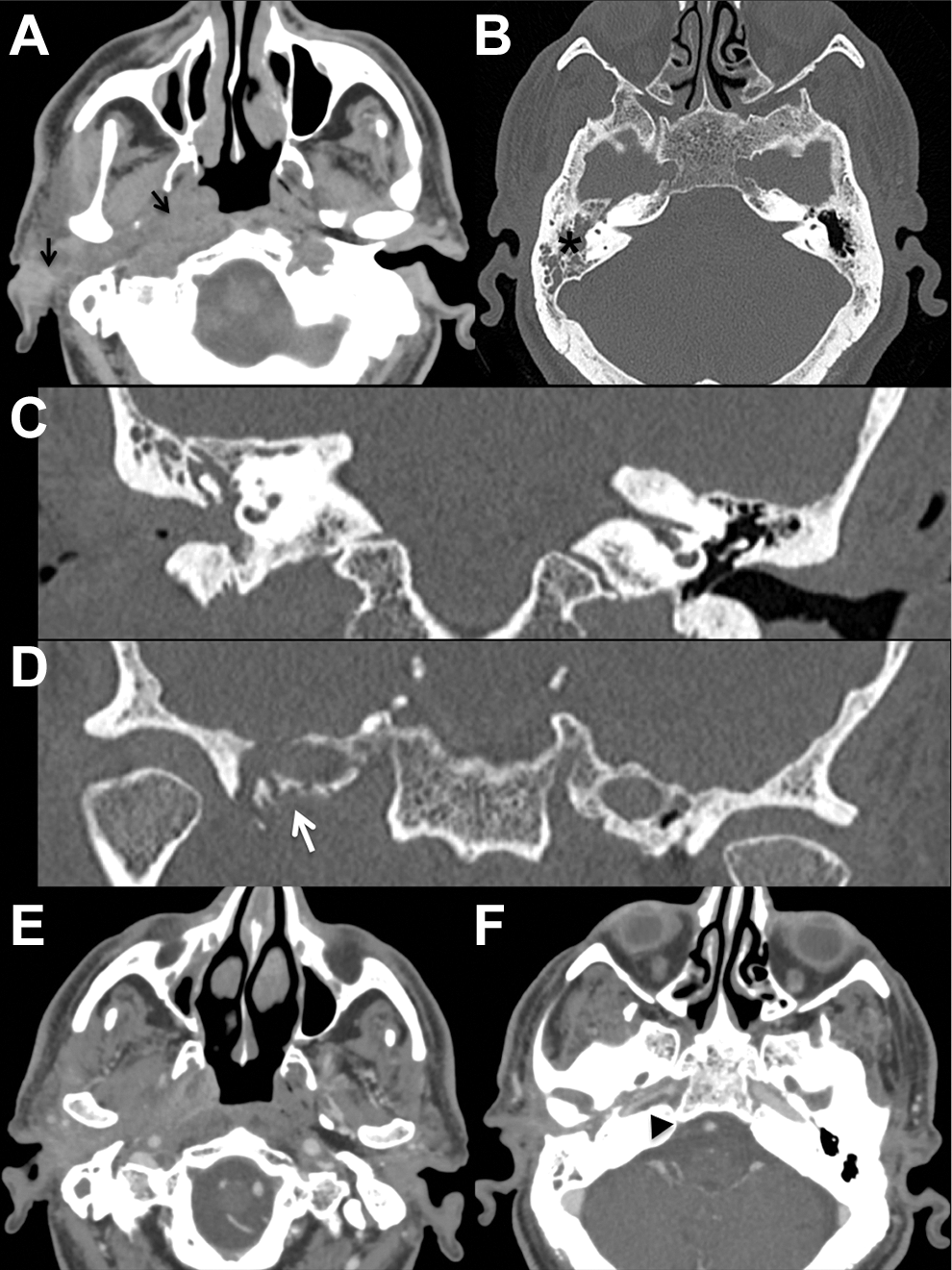
Axial computed tomography (CT) scan (A) showing thickened right external auditory canal extending to ipsilateral nasopharynx and parapharyngeal space through the Eustachian tube (black arrows); retrocondylar tissue, masticator space, and retrocondylar soft tissues were also involved. High-resolution CT scan of the petrous bone with bone algorithm and coronal reconstruction (B-D) showed middle ear cavity and mastoid opacification (black asterisk) and remodeled and partially eroded petrous bone apex (white arrow). Postcontrast axial images (E-F) revealed diffuse and inhomogeneous soft tissues enhancement, coupled to reduction in right intrapetrous internal carotid artery diameter (black arrowhead) close to bone remodeling areas; jugular vein and intracranial sigmoid sinus opacification was normal.
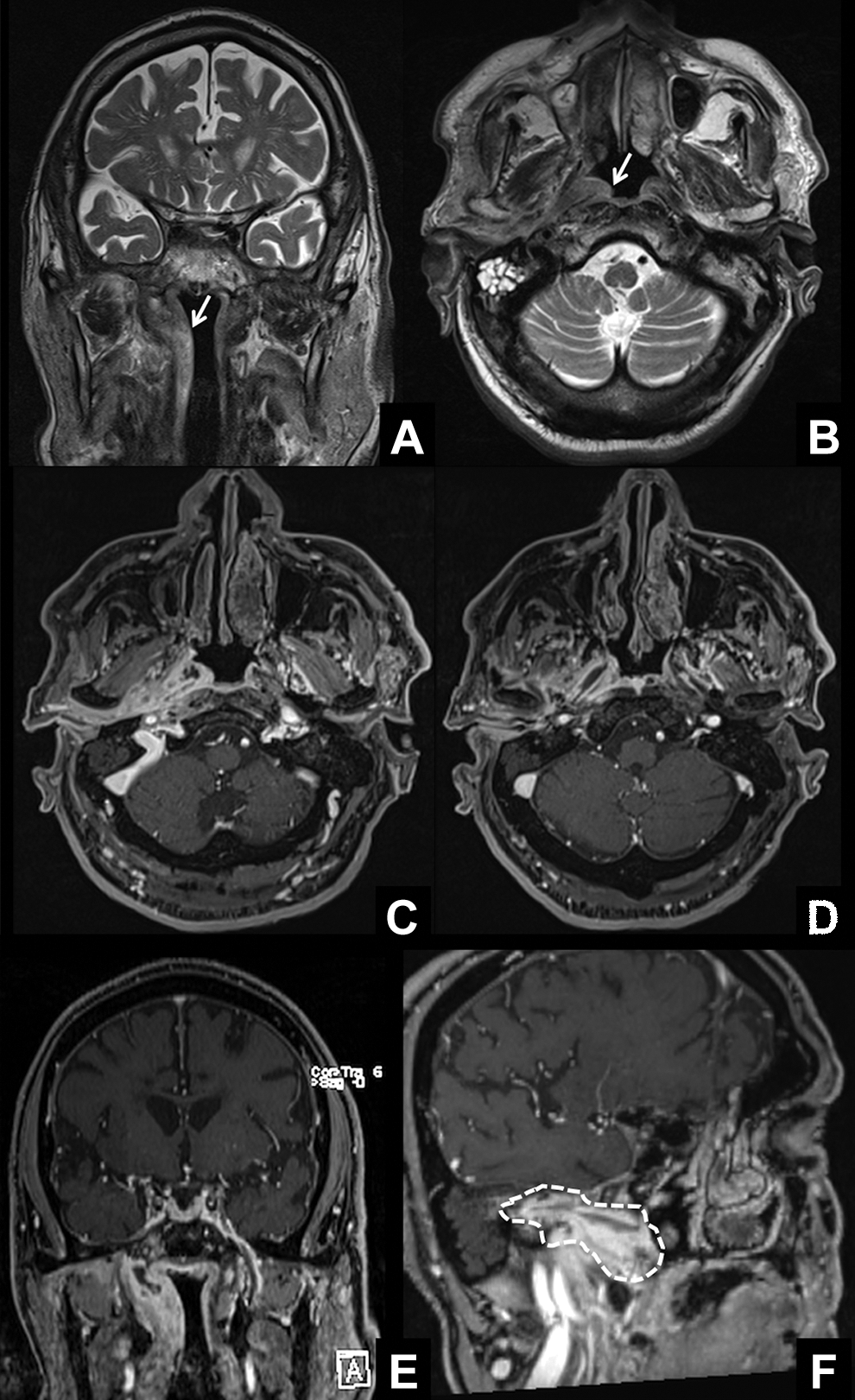
Coronal and axial Turbo Spin Echo (TSE) T2-weighted magnetic resonance imaging (A and B) showed asymmetric nasopharynx and parapharyngeal space with soft tissue swelling on the right side (white arrow) and opacification of the mastoid air cells (B). Fat-saturated volumetric T1-weighted images after contrast media administration (B-F) showed increased enhancement with stenosing soft tissue thickening extending from the right external auditory canal through the Eustachian tube up to parapharyngeal space (dotted line), temporomandibular joint, and medial pterygoid muscle; inner nonenhancing necrotic areas are also visible. Petrous bone apex involvement with reduced right intrapetrous internal carotid artery diameter was also present; no sign of intracranial invasion or dural enhancement was visible, and right sigmoid sinus opacification was normal.
As this case was refractory to therapy, a culture examination was carried out on the right ear swab, demonstrating the presence of C. parapsilosis sensitive to fluconazole (Diflucan). Simultaneously, soft tissue biopsy under endoscopic view (2.7 mm 0° rigid endoscope; Storz) on the right external auditory canal and on deep nasopharyngeal soft tissue excluded malignancy and confirmed chronic inflammatory changes. Therefore, oral and topical antifungal treatment (fluconazole) was added to the previous antibiotic therapy. Moreover, the patient received a combination of paracetamol and opioids for pain control.
After 20 days, the clinical picture improved with regression of the right facial nerve palsy and a reduction in facial asymmetry. The patient remains under outpatient follow-up. The patient provided signed informed consent.
Discussion
Malignant otitis externa is an invasive infection of the external ear and temporal bone that can typically affect elderly diabetic patients such as in our case.5-8 The term “malignant” highlights the aggressive behavior of this pathology spreading outside the external auditory canal. 6 In fact, the infection begins as external otitis with ear pain, otorrhea, and aural fullness up to involving the middle ear and the skull base with high mortality and morbidity due to the proximity of these structures. This clinical behavior can lead to fatal complications such as mastoiditis, SBO, cranial nerve involvement, cerebral venous sinus thrombosis, arteritis and/or pseudoaneurysms of intracranial arteries, and brain abscess.5,6
Skull base osteomyelitis is an inflammatory process of bony structures of the cranial base.
8
In the literature, Mortazavi et al
9
and Das et al
10
proposed an SBO classification based on 2 general categories: “sinorhino-otogenic” or “otogenic” and “non-sinorhino-otogenic” or “nonotogenic” groups due to different clinical features.9,10 As in our case, otogenic SBO represents one of the higher risk complications due to the spread of the external ear canal infection to the temporal bone and subsequent skull base involvement through the fissures of Santorini.
6
We distinguish 3 types of otogenic SBO: Malignant otitis externa with extension to the central skull base, central SBO following resolved MOE, and central SBO as a primary or isolated presentation.
1
According to the literature, our patient developed a type 1 SBO. Main clinical features of otogenic SBO are extremely painful otitis externa and facial palsy such as in our case along with headaches and cranial nerve neuropathies.8,11 Considering the clinical complexity of SBO, multidisciplinary management is necessary, involving an otolaryngologist, neurosurgeon, radiologist, and infectiologist. 8
Pseudomonas aeruginosa is a Gram-negative aerobe that colonizes the external auditory canal with a high moisture content, especially in immunocompromised subjects due to the presence of a mucoid layer (hindering phagocytosis) and lytic enzymes (promoting necrotizing vasculitis and arteritis). Pseudomonas aeruginosa is considered the etiological agent in 50% to 90% of MOE patients; although different microorganisms can also be isolated, especially in nondiabetic patients (5% to 20% of cases) such as Staphylococci (S. aureus, especially Methicillin-resistant S. Aureus - MRSA , Staphylococcus epidermidis), Streptococci, and Gram-negative bacteria (Klebsiella spp, Proteus mirabilis, or Raoultella ornithinolytica).4,6
Conversely, fungi rarely cause MOE (incidence of 10%),6,12,13 with Aspergillus spp and Candida albicans as the most implicated fungi (Candida glabrata, C. parapsilosis, and Candida ciferrii are only occasionally reported).5,14 The pathophysiology of fungal MOE is not well understood, and local complications are more frequent compared to bacterial MOE,5,15 as confirmed by our report where C. parapsilosis produced an invasive clinical condition with rapid clinical evolution and considerable local aggressiveness.
The diagnosis of fungal MOE is challenging and requires microbiological confirmation. 16 Diagnosis of MOE is based on a multimodal approach based on history, physical examination, and laboratory and radiological findings. 7 Samplings of ear secretions and granulation tissue biopsies from the auditory canal or the skull base (as performed in our patient) are fundamental for pathogen identification. However, high rates of culture-negative specimens in MOE are not uncommon and have been reported in the literature, such as in our case initially. In the case of initially negative microbiologic studies, further examination including fungi or slow-growing pathogens should always be performed, ideally using new molecular techniques available in other fields to optimize diagnosis and treatment with new drugs.5,17,18 Moreover, in fungal MOE, positive bacterial cultures can occur due to previous therapy causing an alteration in the existing flora and delaying the diagnosis of a fungal etiology. 16 Moreover, fungal MOE usually occurs in high-risk patients affected by diabetes, congenital or acquired immune-deficiencies, malignancies, chronic renal failure, and users of prolonged steroid or antibiotic therapy.5,19
Additionally, neuroimaging through CT and MRI scanning is crucial to determine disease extension for a prognostic evaluation and in posttreatment monitoring.3,5,20-24 Computed tomography describes bone details, whereas MRI enriched with diffusion-weighted intensity sequences identifies soft tissue abnormalities.22-24 Recently, some authors have shown that 18F-FDG (18F-fluorodeoxyglucose) positron emission tomography can be considered a good diagnostic and therapy monitoring tool for SBO. 25
The therapy of MOE has evolved over the last 2 decades. Early therapy for MOE prevents cranial nerve involvement and improves the prognosis. Currently, the standard antimicrobial treatment is long-term antibiotic or fungal cultured-directed therapy, generally intravenously for 4 to 6-8 weeks, followed by 3 to 12 months of oral drug administration.7,26 Generally, the 6-week duration of the therapy is based on the 3 or 4 weeks that bone takes to revascularize. Based on antibiogram findings, the most common antibiotics are systemic quinolones (ciprofloxacin), ceftriaxone, piperacillin-tazobactam, meropenem, or clindamycin. They have a 90% to 100% success rate. 26 The use of newer, more effective and safer antibiotics has changed the indication for surgical debridement from a former first-line treatment to very limited cases with bony sequestra or abscesses, which our patient lacked. 5 Our case had a negative initial culture on an ear swab, as documented in the literature case. 5 So, considering the clinical history of our patient, he began empiric antibiotic treatment (meropenem and oral ciprofloxacin) for 2 weeks.
In our patient, resistance to empiric antibiotic therapy led us to rapidly reconsider a bacterial MOE etiology. A further ear swab culture was positive for C. parapsilosis, confirming the suspected fungal etiology. Based on the swab result, the patient was treated with 3-month oral and local fluconazole administration. In recent times, some authors have proposed to start empiric antifungal drug treatment for patients unresponsive to antibacterial therapy or relapsing after treatment, with negative cultures from ear swabs or granulation tissue biopsies. Fluconazole is the first drug option in the case of fungal infection by Candida spp. 26 Amphotericin B, itraconazole, and voriconazole are considered the gold standard antifungal drugs for Aspergillus MOE treatment. 13 In addition to empirical antimicrobial therapy, supportive treatment should also include strict glycemic control, correction of electrolyte imbalance, and immunocompetency recovery when possible, along with analgesics for pain control.6,18 Some authors have also investigated hyperbaric oxygen as a promising adjunct therapy, demonstrating a higher rate and speed of recovery than those treated with antimicrobials alone.5,27
A severe and life-threatening complication of MOE, especially fungal MOE, is infection of the intracranial arteries as this can lead to the formation of pseudoaneurysms; these represent 0.7% to 4% of all cerebral aneurysms. In our case, imaging revealed early signs of arteritis of the right ICA, a rare MOE complication followed by almost complete recovery after targeted therapy. According to the literature, the mechanism by which the infection reaches the arterial lumen can involve hematogenous seeding or direct invasion of the arterial adventitia from a surrounding infection.5,28 Nontreatment or delayed treatment of arteritis can lead to the formation of infected aneurysms. The latter can cause fulminant sepsis, spontaneous arterial rupture, and death. So, earlier detection of infected aneurysms or previous arteritis, such as in our case, is critical to ensure the appropriate therapy and to optimize patient outcome. 28 In this light, CT and MRI scan performed early allow for the timely detection of complications in MOE as well as in many different head/neck, skull base, and otogenic disorders.29-31 According to the literature, the mortality rate of MOE and SBO ranges between 7% and 10%, so prompt multidisciplinary evaluation is mandatory for this pathology.26,32
Conclusion
Malignant otitis externa is a serious condition that can lead to grave complications, such as SBO. Fungal MOE can exhibit more subtle symptoms and be more aggressive than its bacterial counterpart. Arteritis of the intracranial vessels is a life-threatening complication that is seldom reported in the literature. Furthermore, a poor response of MOE to antibacterial drugs should raise the suspicion of a fungal etiology of MOE. The current guidelines do not exhaustively describe the diagnosis or therapy, particularly regarding the antifungal drugs of choice and the optimum duration of treatment and follow-up. The description of these rare clinical cases should help clinicians to diagnose this disease and to optimize the therapeutic protocol with multidisciplinary cooperation.
Footnotes
Authors’ Note
Informed consent was obtained from the patient.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
