Abstract

A 53-year-old male patient presented to our department with 1-year history of a slowly progressive left side nasal obstruction and mucopurulent rhinorrhea. He had been treated for primary atrophic rhinitis for 30 years. He was without sense of smell and taste for about 20 years, which was evaluated by an olfactometry test. Nasal endoscopy revealed a markedly large and wide nasal cavity, visibly dry, and thin nasal mucosa and a pedunculated lobular polypoid lesion arising from the medial portion of the left middle turbinate. A computed tomography scan revealed atrophic changes in the nasal and paranasal sinuses mucosa and a 2-cm mass attached to the left middle turbinate (Figure 1). Magnetic resonance imaging showed that the volume of both olfactory bulbs is 35 mm3, so it seems plausible to argue that it is unlikely that the patient will recover olfactory function. The polypoid lesion was removed endoscopically under general anesthesia. The specimen consisted of a 2-cm tan-pink, multinodular mass (Figure 2). Histopathological analysis revealed a lesion covered by respiratory epithelium and composed of lobular proliferations of seromucinous glands and ducts embedded within an inflamed and edematous fibrous stroma (Figure 3A). These glands and ducts are lined by a single layer of cuboidal or flattened epithelial cells (Figure 3B). These features suggested the diagnosis of a seromucinous hamartoma (SH). We followed up with the patient regularly over the course of 3 years. Nasal endoscopy showed atrophic rhinitis, but there was no evidence of hamartoma recurrence 3 years after surgery.
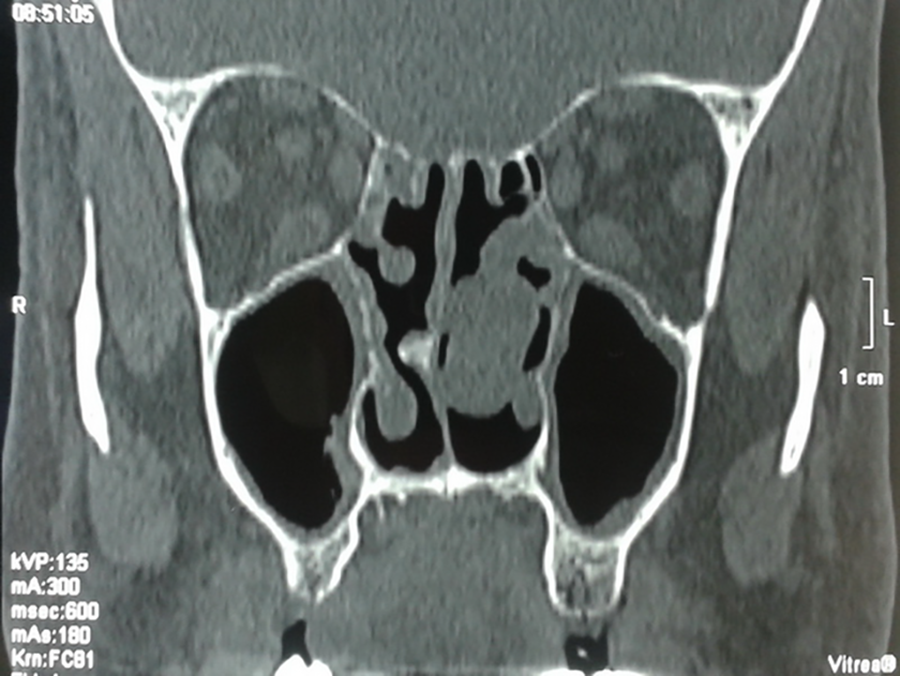
Computed tomography scan of the paranasal sinuses shows atrophic nasal and paranasal sinuses mucosa, especially in the region of the nasal turbinates and a 2-cm polypoid mass attached to the left middle turbinate.
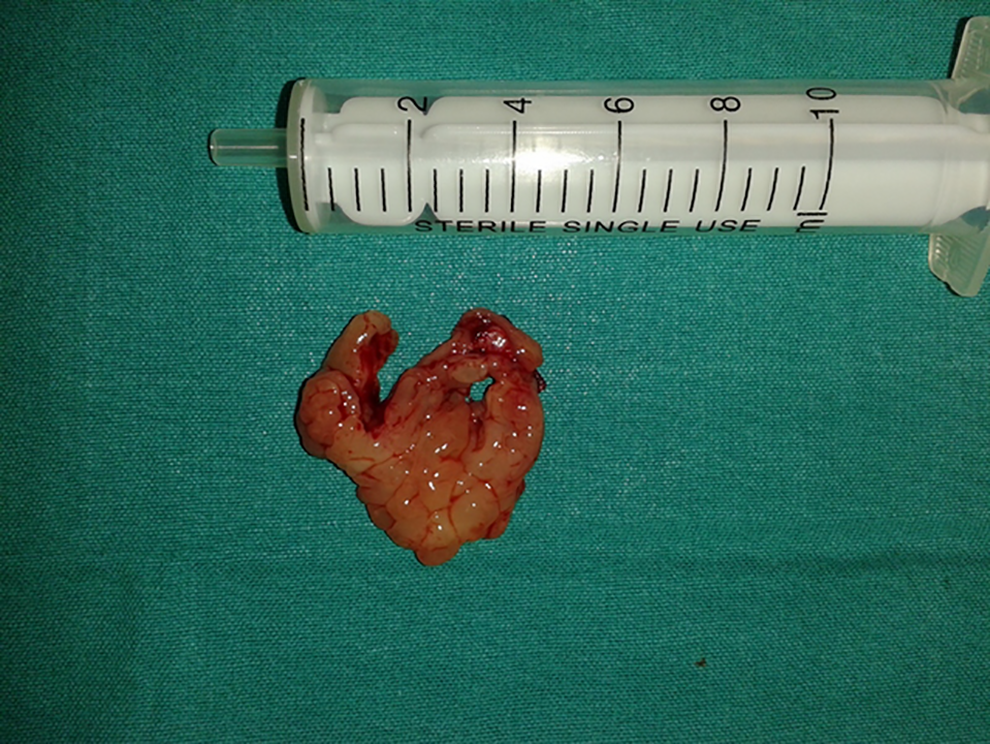
Macroscopic appearance of removed polypoid lesion consisted of a 2-cm tan-pink, multinodular mass.
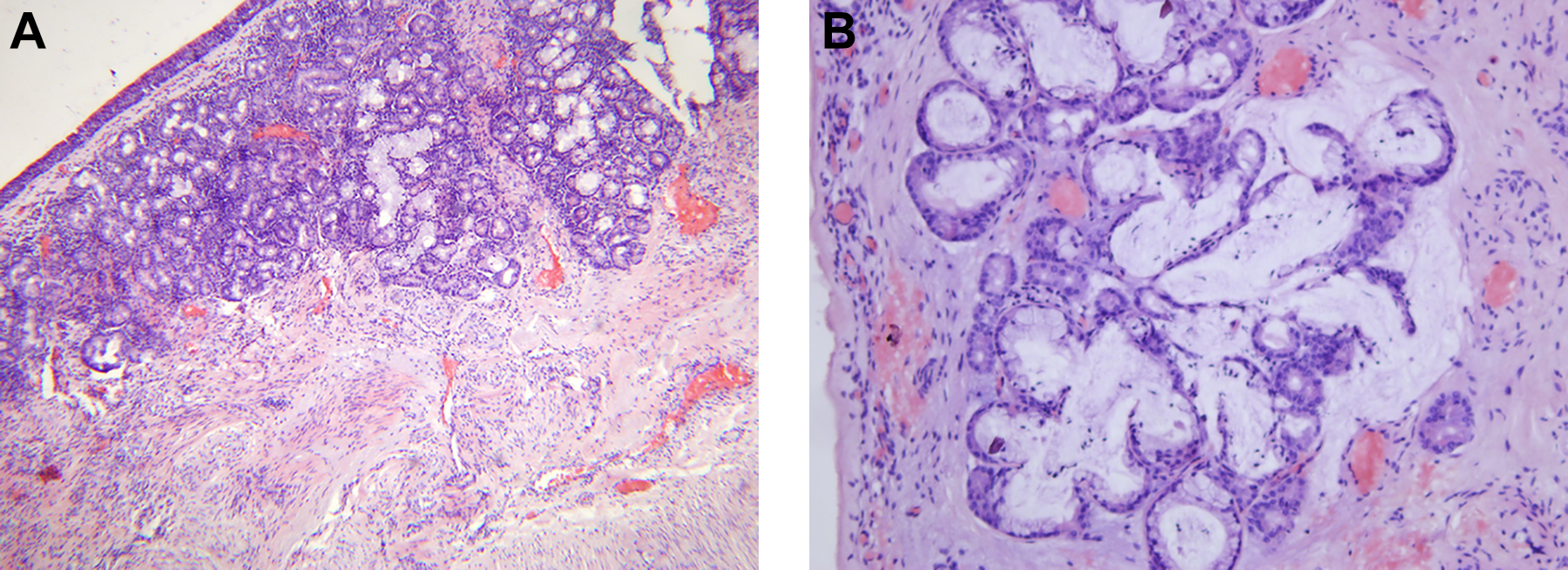
Microphotography of seromucinous hamartoma: (A) Histopathological analysis showing a lesion covered by respiratory epithelium and composed of lobular proliferations or seromucinous glands within an inflamed and edematous fibrous stroma (hematoxylin and eosin staining, magnification ×50); (B) Seromucinous glands and ducts are lined by a single layer of cuboidal or flattened epithelial cells (hematoxylin and eosin staining, magnification ×200).
Seromucinous hamartoma is an extremely rare benign glandular proliferation of the sinonasal tract and the nasopharynx. It consists of a respiratory epithelium-lined polypoid mass with subepithelial seromucinous glands in a lobular arrangement with a fibrous background. 1 This lesion typically arises on the posterior nasal septum and in the nasopharynx, although SH has also been observed on the lateral nasal wall.1,2 Only 39 cases of SH have been previously described in the literature.1-5 The male to female ratio is 3:2. 1 The most common symptoms are nasal obstruction, mucopurulent secretion, and epistaxis. It is unclear whether these lesions are true benign neoplasms or developmental tumor-like lesions.1-5 According to the 4th edition of the World Health Organization Classification of Head and Neck Tumors, SH and respiratory epithelial adenomatoid hamartoma (REAH) are classified as “respiratory epithelial lesions.” 6 The differential diagnosis for SH includes inflammatory nasal polyps, REAH, and low-grade sinonasal adenocarcinoma (LGSNA). Seromucinous hamartoma is characterized by numerous seromucinous glands, which are not present in inflammatory polyps.1-5 The stroma of inflammatory polyps is more edematous and the surface is smooth. 1 REAH is the most common hamartoma in the sinonasal tract, characterized by proliferation of pseudostratified respiratory epithelium-lined pseudoglands.1,5 On the other hand, SH is characterized by the presence of true, single-layered seromucinous glands.1-5 Low-grade sinonasal adenocarcinoma shows the presence of back-to-back glands with pleomorphisms, mitoses, anisocytosis, and cell hyperchromasia, and without a rich stroma. It usually grows in a papillary or cribriform pattern and may invade nearby normal structures.1,5 The results of a recent immunohistochemical study point toward the possibility that SH and some glandular components of REAH might be the precursory lesions for LGSNA. 5 Transnasal endoscopic resection is currently the standard management for all sinonasal hamartomas.1-5 The role of chronic inflammation in the growth of SH has also been considered. In addition to being associated with REAH, SH can also be associated with chronic inflammatory diseases of the sinonasal mucosa, especially with chronic rhinosinusitis with and without nasal polyps.1,3,5 In the case of our patient, the growth of SH could be supported by low-grade chronic inflammatory process associated with atrophic rhinitis. Primary atrophic rhinitis or ozena is a gradually progressive chronic degenerative condition of the nasal mucosa of unknown etiology, but genetic and infectious factors may be implicated. 7 Microbiological studies usually detect Klebsiella ozaenae and Staphylococcus aureus in ozena cases. The condition is more commonly associated with low socioeconomic status. Women are more frequently affected suggesting a possible endocrine factor in etiology. 7 Metaplastic changes in the nasal mucosa include atrophy of the serous and mucinous glands, loss of the cilia, atrophy of the olfactory neuroepithelium, and scar and crust formation. The main symptoms include paradoxaly nasal obstruction, sense of dryness, fetor, and progressively impaired sense of smell leading to functional anosmia. 7 The use of nasal irrigation solutions and antibiotics is the standard conservative therapy for ozena. 7 To our knowledge, this is an unique case of SH arising from the background of chronically changed mucosa in primary atrophic rhinitis.
Footnotes
Author’s Note
The patient’s permission was obtained. Presented as a poster at the 26th Congress of the European Rhinologic Society in conjunction with 35th International Symposium of Infection & Allergy of the Nose; July 3-7, 2016; Stockholm, Sweden.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
