Abstract
Introduction:
Image-guided surgery (IGS) has gained widespread acceptance in otorhinolaryngology for its applications in sinus and skull base surgery. Although the core concepts of IGS have not changed, advances in image guidance technology, including the incorporation of intraoperative imaging, have the potential to enhance surgical education, allow for more rigorous preoperative planning, and aid in more complete surgery with improved outcomes.
Objectives:
Provide a clinical update regarding the use of image guidance and intraoperative imaging in the field of rhinology and endoscopic skull base surgery with a focus on current state of the art technologies.
Methods:
English-language studies published in PubMed, Cochrane, and Embase were searched for articles relating to image-guided sinus surgery, skull base surgery, and intraoperative imaging. Relevant studies were reviewed and critical appraisals were included in this clinical update, highlighting current state of the art advances.
Conclusions:
As image guidance and intraoperative imaging systems have advanced, their applications in sinus and skull base surgery have expanded. Both technologies offer invaluable real-time feedback on the status and progress of surgery, and thus may help to improve the completeness of surgery and overall outcomes. Recent advances such as augmented and virtual reality offer a window into the future of IGS. Future advancements should aim to enhance the surgeon’s operative experience by improving user satisfaction and ultimately lead to better surgical results.
Keywords
Background
Since the introduction of image-guided surgery (IGS), it has gained widespread acceptance for its applications in endoscopic sinus surgery (ESS) due to IGS’s potential to improve surgical outcomes.1-9 Although no randomized controlled trials of IGS in ESS or skull base surgery have been conducted, multiple publications suggest that the number of sinuses entered,10,11 quality of life outcomes, 12 revision surgery/recurrence rates,13,14 major complication, and total complication rate may be improved with IGS utilization.4,6
Moreover, as computed tomography (CT) and magnetic resonance imagining (MRI) technologies have become physically smaller and more widely available, intraoperative imaging has been utilized in combination with IGS during functional ESS and anterior skull base surgery.15-22 The use of intraoperative imaging has the potential to improve the surgeon’s ability to achieve gross total resection in tumor cases and also the potential to confirm completeness of dissection during ESS and/or alter intraoperative decision-making.
Although the IGS core concepts have not changed since its introduction in the late 1980s and early 1990s, many improvements in the user interface have enhanced usability. Recent advancements include CT, MRI fusion capability, 23 three-dimensional (3-D) CT angiography, 24 preoperative planning software, virtual reality (VR) tools, 25 and augmented reality (AR) capabilities.26,27 The ensuing sections will further delve into the recent advances and potential of IGS during ESS and skull base surgery.
System Components and Registration
All systems used for IGS share similar components: a computer workstation, video monitor, tracking system, surgical instrumentation, and data transfer hardware. The computer workstation is central to all IGS systems as it supports the software governing the technology. The original operating systems ran on a UNIX platform; however, newer platforms utilize Windows operating systems (Microsoft) or LINUX. The monitor allows for the output of visual information from the system, and as systems have become more advanced, high resolution and even ultra-high definition (4K monitors) have become common place; as expected, these monitors offer improved visualization and contrast in the projected images.
The tracking system, which allows for the real-time determination of instrument position relative to surgical landmarks, is based on either electromagnetic (EM) or optical tracking (OT) technology. Regardless of the tracking technology, all systems require the placement of an intraoperative localization device (ILD) on the patient’s head in fixed position; this allows the system to compensate for the inevitable movement of the operative field (ie, the sinus cavities) during surgery. Electromagnetic tracking systems include an EM field generator and sensors, which together track instruments (and the patient’s head) in a 3D space. For OT systems, an overhead camera array tracks the position of light-emitting diodes or reflective spheres that serve to mark ILDs attached to the patient’s head and instruments.
The critical step for reliable and accurate IGS use is the registration process. In general, there are 3 paradigms for creating a one-to-one mapping relationship between corresponding points in the operating field volume and imaging data set volume: paired-point registration, contour-based registration, and now rarely used automatic registration.28-30 Today, the most commonly employed registration paradigm is contour-based followed by paired point. Paired-point registration is performed by first identifying fiducial points via attached external markers, chosen by the operating surgeon, or automatically by the IGS software. Next, the surgeon manually localizes each fiducial point with the registration probe. Finally, the IGS software aligns the corresponding points in the preoperative imaging data set and the operative field volume to complete the registration process. 28
For contour-based registration, the computer builds a 3-D model from the preoperative imaging data. The surgeon then identifies corresponding contours on the surface of the surgical volume. Commonly, this step requires an initial identification of 3 anatomic fiducial points; followed by the surgeon running the surgical navigation/registration probe across contours on the surface of the surgical volume to identify discrete points along a changing topography. The software then aligns the contours mapped in the surgical volume with the contours of the preoperative imaging volume to create the registration.
After the registration process is complete, one must assess the target registration error (TRE), which is the distance between the measured position of a tracked instrument tip (seen on the IGS computer monitor) and the position of the instrument tip in the real world. 29 It is important to assess TRE throughout the case as hardware issues (eg, slippage of the patient tracking sensor, deflection of precalibrated rigid suctions, etc) can lead to increased TRE, which places the surgeon at increased risk for an operative complication. The general consensus is that TRE should be 2 mm or less for surgical navigation to be useful. 31 Reported mean TRE values in published reports range from 1.5 to 2.3 mm. 29
Target registration error of any specific IGS platform depends on 3 separate factors: (1) ability of the system to sense instrument position, which will have some degree of error (less than 1.0 mm with current IGS systems), (2) quality of the imaging data used for registration (0.5-0.625 mm thickness for conventional high resolution CT) which results in a lower limit for TRE of at least 0.5 mm, and (3) the registration process of mapping points from the patient operative field volume to the imaging data introduces error. 32 The third aspect is dependent on user variability, and therefore it is important to emphasize the importance of registering with minimal disruption of the skin surface to improve TRE for contour-based registration. Generally, a TRE of 2.0 mm or less is considered clinically acceptable and current IGS platforms produce mean TREs in the 1.5 to 2.0 mm range in real-world scenarios. 32 Furthermore, some next generation systems have features to recalibrate and refine the original registration by adding more surface matching or fiducial points in an effort to decrease TRE.
Indications for IGS
The current recommendations for indications and use of IGS in rhinology, proposed and approved by the American Academy of Otolaryngology-Head and Neck Surgery (AAO-HNS), are also endorsed by the American Rhinologic Society and are found in Figure 1. 33

American Academy of Otolaryngology-Head and Neck Surgery (AAO-HNS) policy statement on indications for image-guided surgery in rhinology. 33
Image-Guided Primary and Revision Functional ESS
As the use of IGS in both primary and revision ESS has become widely accepted, several principles remain central to IGS-ESS
34
: Restore normal mucociliary clearance and allow for improved topical delivery of medications Careful anatomic dissection with mucosal preservation is an overriding principle in restoring sinus function IGS systems should not be used merely in a point-and-hunt manner, but rather for thorough preoperative planning, better understanding of complex anatomic relationships in 3 dimensions, and as a complement to standard ESS techniques
Although the rigid nasal endoscopes provide high-resolution image quality, these images are 2-D depictions of a complex 3-D space. Additionally, due to the wide-angle view, endoscopes create a variable amount of fisheye distortion to the images. Furthermore, the use of angled endoscopes can introduce perceptual error. When IGS is utilized as intended, the provided localization information facilitates the surgeon’s understanding of anatomic relationships and thereby provides a sense of depth to standard 2-D endoscopic views leading to reduced perceptual distortion.
Several results of IGS-ESS have been published since the technology became widely adopted. Metson, in 2003, reported on 42 surgeons and their initial use of IGS in 1000 patients. 2 These surgeons noted that IGS allowed for enhanced anatomic localization during sinonasal procedures and offered the potential for improved safety and efficacy during ESS. Tabaee et al 3 reported on 120 patients who underwent sinus procedures with IGS utilization for various sinonasal disorders, and no major complications were reported.
The surgical outcomes after primary and revision IGS-ESS have been debated over the years. The data to date remains sparse given that ESS has a low major complication rate (orbital injury, intracranial penetration, cerebrospinal fluid [CSF] leak, hemorrhage). The major complication rate of primary and revision ESS has been less than 1%, independent of the use of image guidance.35,36 Smith et al 37 published a systematic review on the indications for IGS-ESS in 2007. In their analysis, they noted that over 35 000 patients would be required in order to adequately power a study to show a 50% reduction in major complications during sinus surgery. In addition, randomized controlled trials are mostly impractical given that studies to address this issue may be unethical or would inadequately describe the situations in which IGS is most beneficial. 37
Even with these limitations, it is valuable to discuss the current literature that explores surgical outcomes with IGS use during ESS. In a randomized trial in which trainee sinus surgeons performed IGS-ESS and non-IGS ESS on 32 patients, the incidence of total complications was not different between groups, and no major complications were reported. 38 Interestingly, participants rated significantly increased willingness to take risks during ESS when utilizing IGS. In a subsequent study, Sunkaraneni et al 14 retrospectively analyzed outcomes after ESS with and without use of IGS in 355 total cases in addition to patients pooled from 7 other studies. Overall, no significant difference in complication rates were found. Only 1 of the 8 included studies showed an improvement in complication rates. 1 However, in a subsequent meta-analysis of 8 studies including over 800 total procedures by Vreugdenburg et al, 6 IGS use was associated with decreased risk of major (odds ratio [OR] = 0.36; 95% CI 0.18-0.75), orbital (OR = 0.38; 95% CI 0.17-0.83), and total complications (OR = 0.58; 95% CI 0.37-0.92). Similarly, Dalgorf et al investigated IGS use and complications rates in a later meta-analysis, and the overall (relative risk [RR] = 0.66; 95% CI 0.47-0.94) and major (RR = 0.48; 95% CI 0.28-0.82) complication rates during ESS were significantly reduced with IGS use compared to non-IGS cases. Authors noted that all 14 included studies were conducted at tertiary care hospitals with trainees, and thus results may not necessarily be generalized to the majority of nationwide sinus procedures. 4
One also needs to consider the utility of IGS to aid in more “complete” surgery and subsequently lower surgical revision rates. Completeness of surgery comparing IGS-ESS and non-IGS-ESS has been investigated in few studies to date. In 2002, Reardon 11 showed that with IGS, the mean number of sinuses entered increased from 4.7 (non-IGS mean) to 5.1 (P < .0001). Additionally, this study demonstrated that IGS significantly increased the rate at which the frontal sinus was addressed. In 2019, Giotakis et al 10 published the results of 169 IGS-ESS patients and compared them to historical controls. Overall, there was a significant decrease in the number of paranasal sinus compartments missed/not addressed during IGS-ESS compared to non-IGS-ESS. Similarly, Vincaut et al 39 performed a multicenter prospective study, including 311 patients undergoing ESS procedures at 16 hospitals in 2019. In this study, surgeons were able to perform a more complete sinus surgery in 81% of cases that utilized IGS and were able to identify and open previously unidentified sinus cells in 57% of cases.
One can speculate that if a more complete surgery is performed, where all diseased sinus compartments are addressed, then revision rates may be reduced. Although, this topic has been sparsely reported, in the previously mentioned study by Sunkaraneni et al, the authors noted non-IGS-ESS were 2 times or more likely to require revision ESS compared to the IGS-ESS group. It is important to note that all non-IGS cases were performed before IGS was readily available at the authors’ institution, and non-IGS cases only made up 22 of the 355 total cases. Furthermore, in a retrospective 2019 study by Galletti et al, 13 46 IGS-ESS cases were compared to 46 non-IGS-ESS cases. In this cohort, IGS use conferred a statistically significant advantage in terms of recurrent sinus disease rate, quality of life measures (SNOT-22 and RhinoQoL), and frontal sinus restenosis rates. Additionally, a different retrospective review found that IGS allowed for fewer complications and reduced recurrence rates in endoscopic resection of inverted papilloma. 40
Concerns have been raised about surgeon fatigue and potential for added stress with IGS utilization during ESS. However, multiple studies show that workload, operating surgeon stress, and overall perception of benefit improved with the use of IGS in ESS. Furthermore, in the only randomized trial to investigate training surgeon stress during ESS with and without IGS, there was no increase in measured markers for stress with IGS. 41 Perhaps more importantly, use of IGS has been shown to alter surgical decision-making. 42 Most recently, in the previously mentioned article by Vicaut et al, 39 surgical strategy was changed in over 75% of cases even in an experienced surgeon’s hands. Moreover, high surgeon satisfaction with IGS-ESS was noted (mean rating of 8.6 of 10), and IGS use was associated with decreased perceived stress in 95% of respondents. In a similar attempt to gauge satisfaction with IGS-ESS, Manzey et al 7 surveyed 213 surgeons in Germany. Overall, surgeons subjectively reported improved performance and patient safety with IGS-ESS. However, concerns over increased time pressure and mental demands were reported by less-experienced surgeons. Additionally, a few survey respondents cited a theoretical safety risk of overreliance on IGS. Contrary to the concerns for increased time associated with IGS-ESS, Reardon 11 showed that IGS did not add significantly to total operative time.
Image-Guided Skull Base Surgery
As image guidance gained widespread availability, its use during anterior skull base surgery has become increasingly common. In a 2018 survey of the North American Skull Base Society, 83.6% of respondents confirmed IGS use in all skull base cases regardless of the level of complexity. 43 In a 2007 cadaver model study, IGS use resulted in shorter time to sphenoid ostium identification and sellar window creation in cases with complex or aberrant sphenoid anatomy. 44 In a more recent study retrospectively analyzing over 600 transsphenoidal hypophysectomy cases, the non-IGS group tumor recurrence rate was statistically greater (7.9% vs 2.9%) as compared to IGS cases. 45 Also reported in this study were 2 major complications in the non-IGS group, including carotid injury and violation of the cribriform plate due to misidentification of critical structures compared to no major complications in the IGS cohort. Parameters such as operating time, blood loss, and gross total resection rate were similar among IGS and non-IGS cases. When compared to intraoperative fluoroscopy, Patel et al 46 showed that the use of IGS during endonasal pituitary surgery decreased total operative time from 131 to 107.9 minutes (P < .05). Furthermore, in a review of over 45 000 transsphenoidal pituitary resections performed in the United States, Chung et al 47 showed the use of IGS was associated with decreased rate of CSF leak, shorter hospital stay, and lower overall patient costs. Similarly, Jagannathan et al reported that IGS use in management of 176 sellar lesions resulted in increased accuracy of the surgical approach and a decrease in operative time. Additionally, in cases of revision skull base surgery, IGS has been demonstrated to allow for safe reduction of tumor burden. 48
Overall, it appears that use of IGS during endoscopic skull base surgery, most commonly pituitary surgery, can be used safely and efficiently with the potential for improved outcomes without a negative impact on surgical operative time. The use of IGS for endoscopic skull base surgery is in line with current widespread practice patterns and the AAO-HNS guidelines.
Use of Intraoperative Imaging in Sinus and Skull Base Surgery
A common criticism of surgical navigation is the system’s reliance on preoperative imaging and as such IGS will not reflect anatomic changes made during surgery. The incorporation of intraoperative imaging can alleviate this limitation. In the late 1990s, intraoperative MRI (iMRI) suites were introduced and used in sinus surgery. 15 However, due to concerns of increased cost and the need for MRI compatible operating room suites and MRI safe instruments, the technology was not widely adopted. Fluoroscopy provided a less expensive and more widely available intraoperative imaging technology, although its use in ESS is limited by poor image quality and single section imaging. 16 Nevertheless, improvements to both size and cost of CT scanners and diagnostic quality MRIs have made intraoperative use of both imaging modalities increasingly possible (Figure 2).
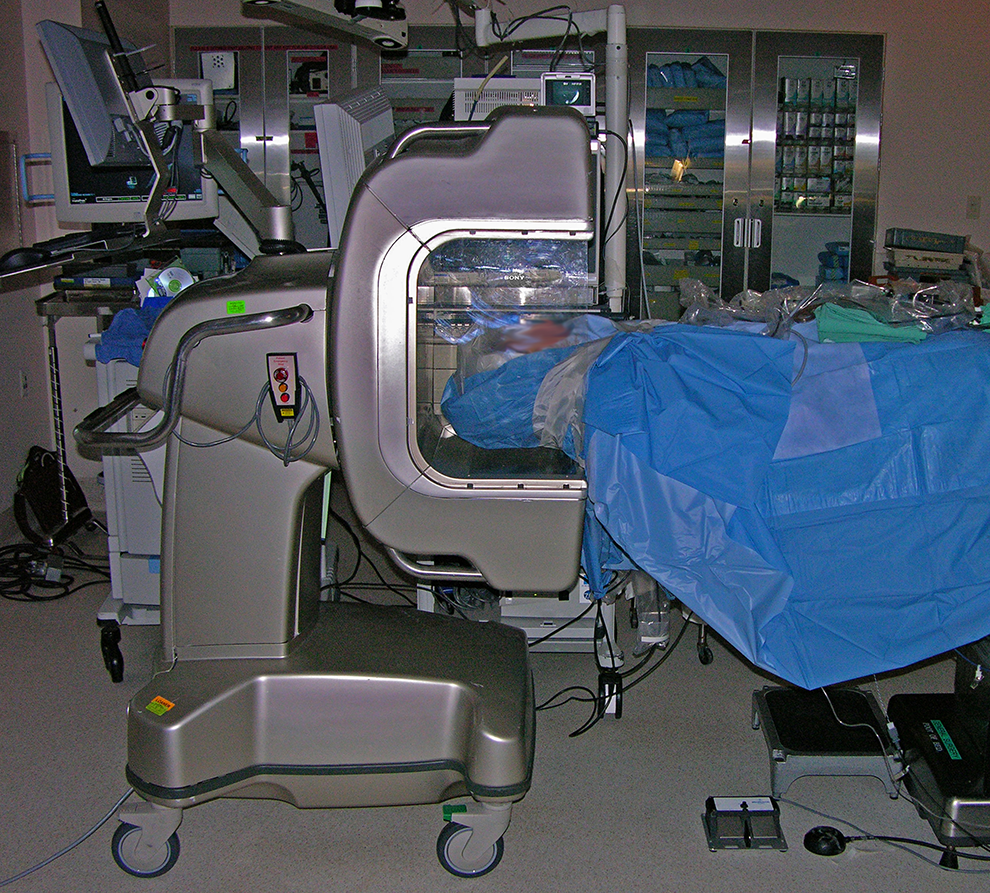
xCAT (Xoran Technologies) used during routine ESS. ESS indicates endoscopic sinus surgery.
In 2008, Wise et al 17 investigated the utility of intraoperative CT (iCT) as a teaching tool. The residents’ ability to identify structures in partially dissected sinuses with or without access to real-time iCT information in combination with standard IGS was measured. In this study, iCT allowed for improved ability to identify anatomic structures and all participants graded increased comfort level with iCT. 17 Similarly, Prisman et al 18 looked at iCT as a teaching tool for Otorhinolaryngology residents in a cadaver model. The use of iCT was deemed very useful in training residents and was specifically rated as moderately to very useful in completing ethmoidectomy, pterygopalatine fossa decompression, sphenoidotomy, and pituitary/clival resections.
In 2008, Batra et al 21 investigated the use of iCT in 25 patients undergoing a variety of image-guided endoscopic sinus and skull base procedures. In that study, iCT led to additional interventions (additional sinus dissection, tumor resection, frontal bone drilling, and repositioning of a frontal sinus stent) in 24% of patients. Jackman et al 22 also investigated the frequency in which iCT could change decision-making during IGS-ESS. In their study, 6 of 20 patients undergoing IGS-ESS had residual bony partitions confirmed on iCT that required further dissection. The most common location requiring further intervention was the frontal sinus. In a subsequent study, Conley and colleagues 19 investigated iCT using 3 commercially available CT systems. Authors reported that iCT was helpful in confirming complete resection of disease in 9 of 12 cases. Reasons for iCT “failure” were patient size preventing proper positioning within iCT scanner in one case and similarly, severe kyphosis, in another case. In a third patient, a petrous apex lesion required minimal dissection, and iCT did not change operative decision-making.
Since its introduction, the use of iMRI has gained increasing popularity, especially for use during anterior skull base/pituitary surgery. To date, multiple studies have examined the utility of iMRI at identifying residual pituitary tumor burden compared to a postoperative MRI. Studies evaluating extent of tumor resection utilizing greater than 0.5T iMRI devices demonstrated a sensitivity and specificity for residual tumor of over 90%.49,50 However, among studies using 0.5T or lower iMRI, the sensitivity and specificity ranges were more varied, but overall still reported favorable results.51-54 Several studies have examined the extent of tumor resection and progression-free survival with the addition of iMRI in endoscopic transsphenoidal cases. In 2016, Zaidi and colleagues 55 demonstrated that the percentage of cases achieving gross total resection increased from 60% to 80% with iMRI. In another study, Sylvester et al 56 compared results of pituitary adenoma resection utilizing either endoscopic or microscopic techniques with/without iMRI. The iMRI identified residual tumor in 35.9% of cases and increased extent of resection in approximately 10% of cases. Intraoperative MRI has also been utilized to aid in resection of functional pituitary tumors. In one study by Tanei et al, iMRI confirmed gross total resection in 50% of patients, and of those patients, 100% had endocrine remission of their secreting adenoma. 57
Although intraoperative image acquisition time can vary widely, most studies cite between 5 and 30 min to obtain a CT or MRI, respectively, in the operating room. A potential concern regarding the safety of intraoperative imaging is that a finding of residual tumor may lead to overresection resulting in an increased incidence of complications such as hypopituitarism and CSF leak. Nevertheless, several studies have not shown an increased complication rate with use of iCT and iMRI.19,20,58
State of the Art
Image-to-Image Registration
Computed tomography and MRI provide complementary information and are essential for understanding complex sinus pathology and lesions of the skull base. Computed tomography visualizes intricate bony detail, while MRI demonstrates the intracranial and extracranial soft tissue anatomy best. To maximize the benefits from both the CT and MRI, IGS platforms have since incorporated image fusion software.
To create fused images, the IGS software performs an image-to-image registration creating hybrid images with characteristics of both CT and MRI (Figure 3). 59 In one study, use of hybrid CT-MRI images for IGS was deemed helpful for preoperative assessment/planning and intraoperative navigation for sinonasal neoplasms, mucoceles, encephaloceles, and lesions impinging on the internal carotid artery. 23 Similarly, Chiu et al 60 described hybridizing CT, MRI, and magnetic resonance angiography (MRA) images to safely resect vascular tumors and tumors with surrounding aberrant carotid anatomy. In a more recent study, Micko et al 61 investigated the use of fusing CT, MRI, and MRA for IGS during pituitary surgery. Among 200 cases studied, the advanced fusion imaging protocol allowed for improved preoperative localization of sinonasal anatomy, fine vasculature, identification of the normal pituitary gland, and decreased cumulative complications compared to the standard CT only IGS protocol.
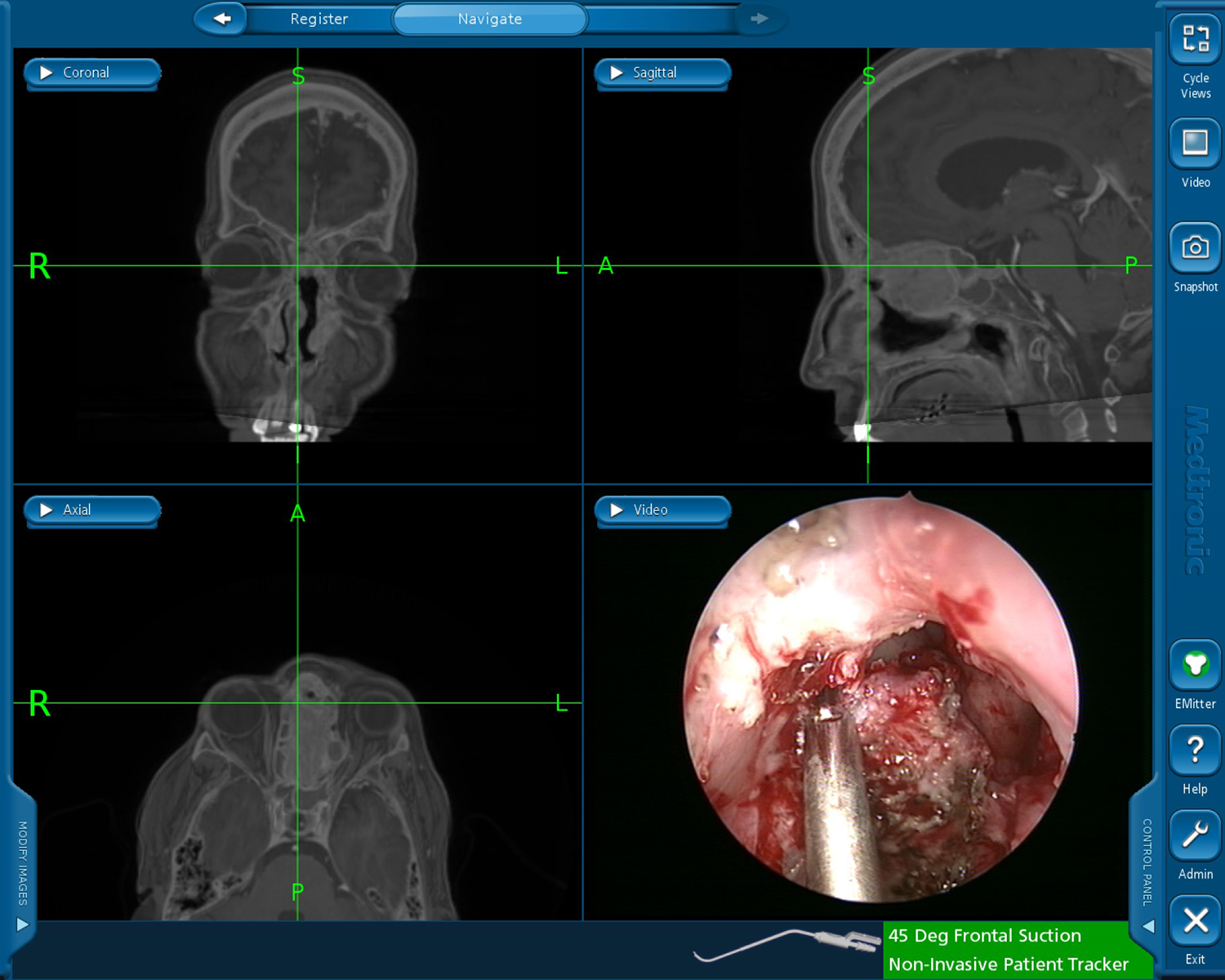
Computed tomography-magnetic resonance imaging (CT-MRI) fusion use during image-guided endoscopic tumor resection. The 3 radiographic panels show a fused image of the CT and MRI. The crosshair correlates to the tip of the instrument seen in the endoscopic view in lower right panel.
Given the wide range of tissue characteristics encountered in complex sinus and skull base procedures, the combination of CT and MRI modalities can help optimize both fine bony anatomy while preserving the ability to visualize soft tissues. This technology is an increasingly available feature on modern IGS platforms and appears to offer improved preoperative planning capabilities, ability to navigate abnormal/distorted anatomy, and potential for decreased complications.
Improving Tracking Technology
One of the basic limitations of IGS platforms is the requirement of space in the operating room. Most optical and EM tracking systems require either an optical system with direct line of sight to the operative field or an EM emitter that is usually attached to the operating table. In both circumstances, surgeons may be limited in where they can stand relative to the patient and IGS equipment in order to allow for continued navigation during the surgery and not to disrupt previously performed registration. Recently, TruDi and StealthStation IGS platforms, developed by Acclarent and Medtronic ENT, respectively, have introduced EM field generators that are flat and rest under a patient’s head (Figure 4). This seemingly small change to equipment location/space requirements can help improve ergonomics and room setup, while also saving space in small operating rooms and office settings.
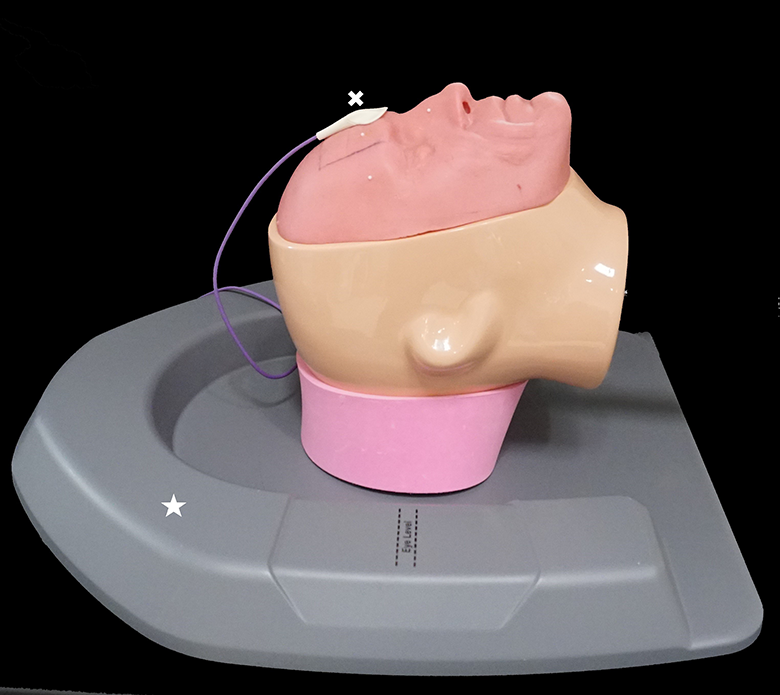
The TruDi electromagnetic (EM) field generator platform (Acclarent) incorporates 5 independent field-generating coils in a case placed under the patient’s head. The EM field generator denoted with (*). White (x) adjacent to the intraoperative localization device placed on the forehead in a fixed position.
An additional advancement in navigation technology are EM microsensors, which can be used for distal chip tracking. Image-guided surgery systems including Fiagon ENT Navigation (Fiagon AG Medical Technologies), Stryker TGS Navigation System (Stryker), and TruDi Navigation currently offer microsensors incorporated into navigation suction instruments (Figure 5). Distal microsensors afford more accurate and reliable tracking because factors such as inadvertent bending or flexing of precalibrated rigid instruments does not impact navigation accuracy. Moreover, this technology allows an instrument to be flexible throughout its length and has been utilized in prior studies to allow for IGS-guided balloon sinus dilation (Figure 6). 62

The TruDi instrument set (Acclarent) has a microsensor embedded in the wall of the distal instrument tip. Therefore, the accuracy of the surgical navigation will not be affected even if the shaft of the instrument is deformed.
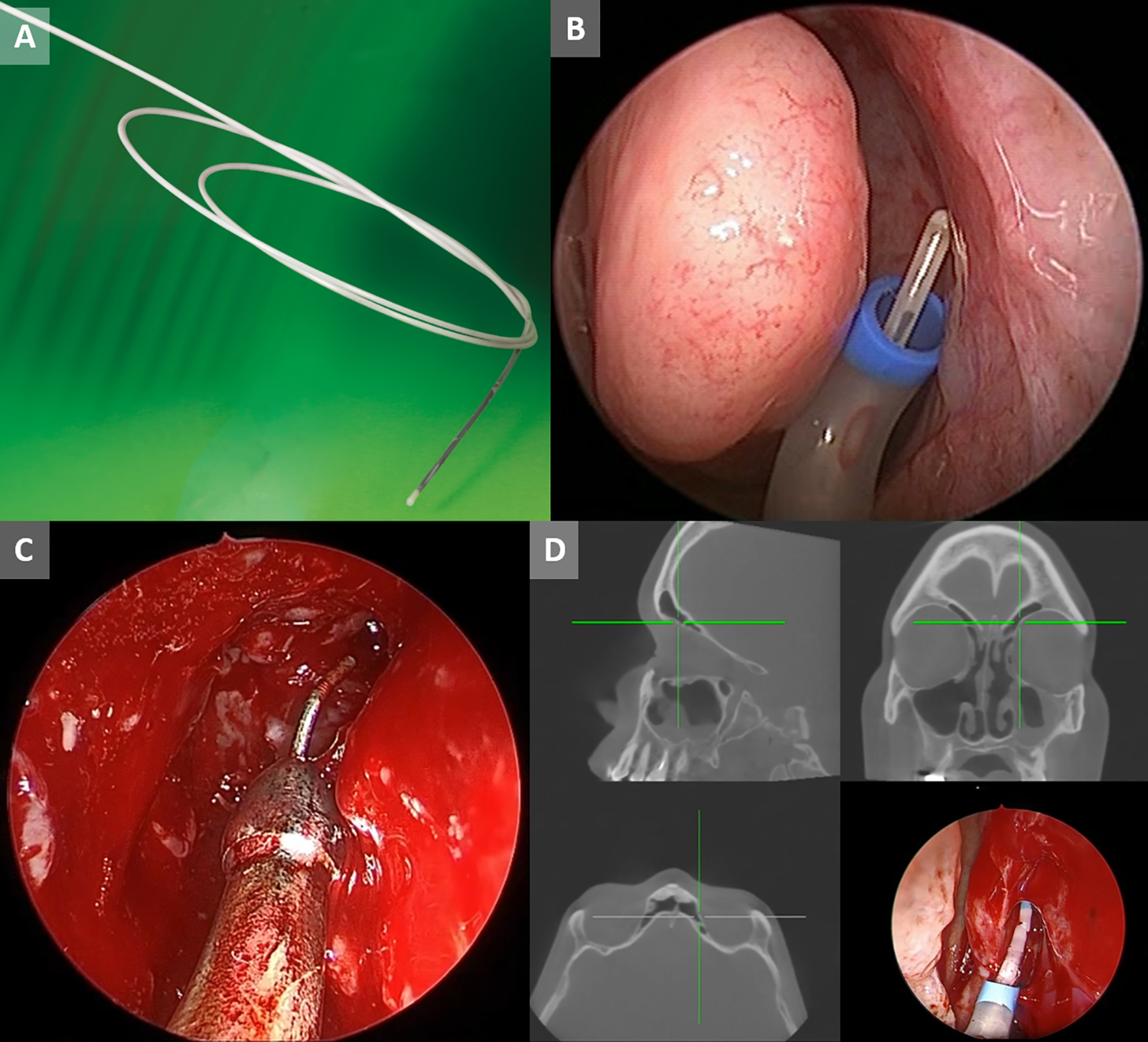
A, Fiagon GuideWire (Fiagon) features a microsensor with dimensions of 8 mm × 0.5 mm. B, The location of GuideWire (Fiagon) is shown at the tip of balloon sinus ostial dilation device. C, A microsensor on a wire can be passed through a conventional nonmagnetic suction, as shown in this image of the Acclarent Trudi wire (Acclarent). D, Microsensors can also be used to guide balloon deployment, as show in this instance of balloon-assisted left endoscopic frontal sinusotomy using the SpinPlusNav device (Acclarent). This image was obtained via an external dual channel recorder that simultaneously recorded both the endoscopic video output and the navigation video output.
Relatedly, proximal EM sensors have been incorporated into the handles of rigid sinus balloon devices. The NuVent EM Balloon Sinus Dilation System produced by Medtronic ENT employs several precalibrated rigid balloon sinus devices for use with the Fusion ENT Navigation System.
Augmented Reality and Virtual Reality
In recent years, the ability to overlay digital elements to the live endoscopic view (AR) or to add virtual information to computerized models of the sinuses during surgery (VR) has been introduced. In one of the earlier cadaveric trials evaluating VR uses in IGS-ESS, Dixon et al 63 showed experienced fellowship trained sinus surgeons graded their mental demand, effort, and frustration level as significantly lower utilizing 3-D virtual navigation images with proximity alerts compared to conventional IGS. Additionally, all participants in this trial stated that surgery was safer with use of the VR navigation due to increased confidence with dissection near critical structures. In a subsequent study, the same group studied the use of real-time virtual endoscopy during 16 skull base cases. 64 In this study, task workload was similar between conventional IGS to IGS aided by VR with proximity alerts. Overall, the participating surgeons subjectively noted the use of VR led to increased efficiency to ablate tumor and increased confidence while dissecting around critical neurovascular structures. Most recently, Zeiger et al 25 published a retrospective analysis of a mixed virtual 3-D IGS system used during a varied skull base surgery population. During 134 cases, surgeons graded their experience, and subjectively noted improved extent of bony dissection and ability to identify critical structures. However, standardized measures were not used making comparison to other published data difficult.
A new VR function, fast anatomic mapping (FAM), has also become available utilizing the Trudi platform. This technology has been extensively used in cardiac catheters designed for minimally invasive heart procedures.65-67 In IGS-ESS, FAM allows for a continuous 3-D reconstruction of a surgical space, which is then visualized on the registered patient CT images. Thus, FAM can provide a real-time virtual representation of the surgical progress made during sinus surgery by outlining which sinuses have been dissected/entered while excluding compartments that remain out of the surgical navigation sensors reach (Figure 7). Although there is a paucity of published data, this technology can aid in surgical education by helping novice sinus surgeons understand real-time changes in sinus anatomy during surgery and potentially highlighting areas that still need to be addressed. Additionally, in complex cases with altered anatomy, real-time updates of surgical progress could be valuable to sinus surgeons of all skill levels.
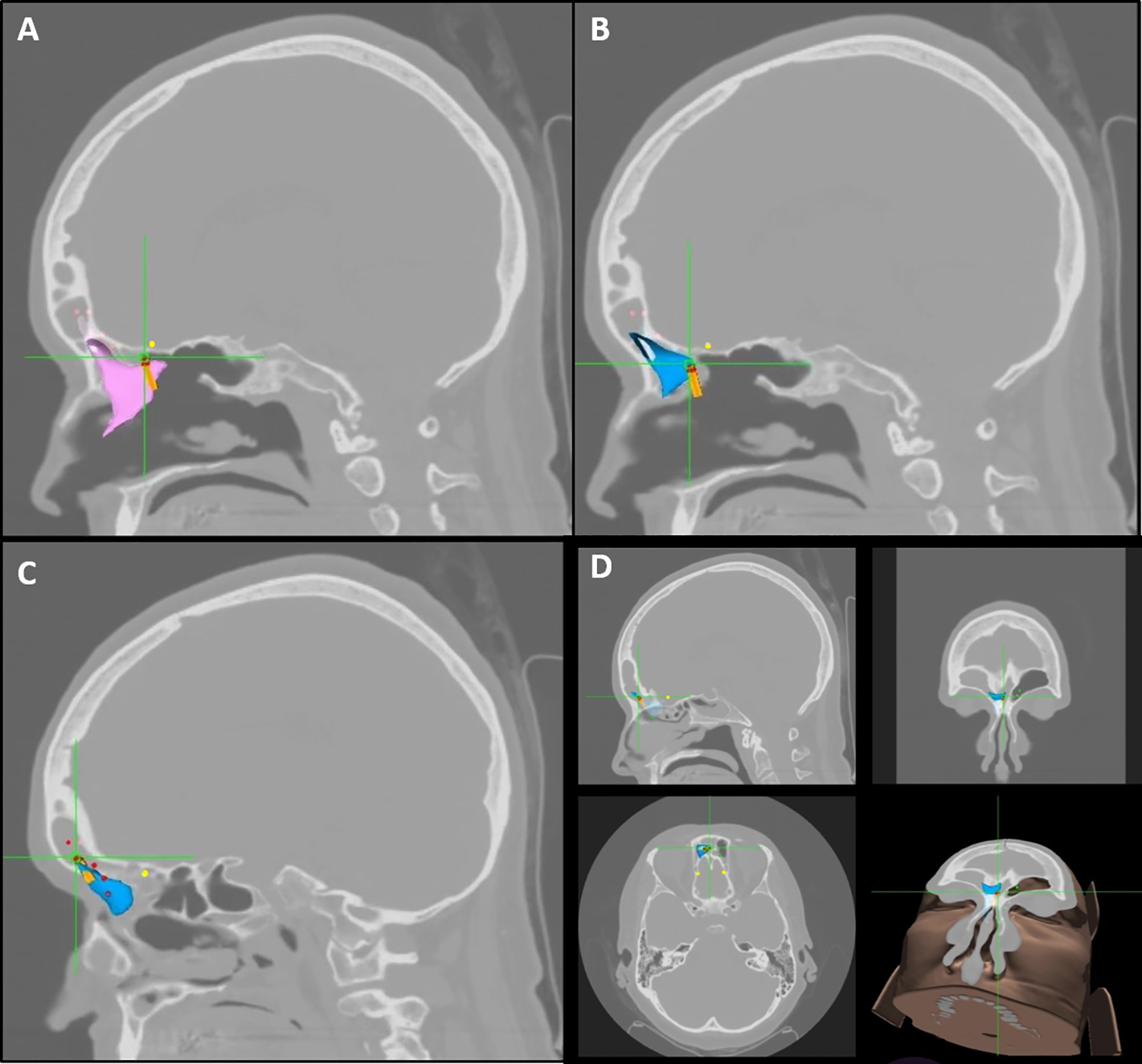
Fast anatomic mapping (TruDi, Acclarent) may be used to highlight residual cells and extent of surgical progress. A, Example of a residual supra bullar cell, whose bony margin is visually enhanced by the fast anatomic mapping (FAM) point cloud (purple). B, Examples of FAM highlighting the extent of surgical progress with the blue point clouds showing access to the frontal sinus. C, Further progress into frontal sinus with blue point cloud. Anterior ethmoid artery marked by yellow dots. Presurgical plan with proposed path into frontal sinus marked with red dots. D, Fast anatomic mapping map, triplanar and 3-D views, confirming access to frontal sinus medial compartment on the right side has been achieved (blue point cloud).
Similar to VR, AR aims to improve the operative experience. Augmented reality provides added virtual information that is integrated with a live surgical view. Once images are annotated during preoperative planning, AR software will allow the surgeon to visualize that information on the video monitor as an overlay of the anatomy in surgeon’s real-time endoscopic view (Figure 8). Currently, the Stryker TGS Navigation System incorporates a tool set known as Target Guided Surgery. This IGS platform and software allows for annotation and outlining structures of interest (targets) or structures to avoid (antitargets), as well as path planning to access a particular sinus. These preplanning tasks are performed directly on the IGS workstation. The value of the preplanning software for surgical education of trainees was investigated in a 2017 study by Agbetoba et al. Among 65 participants, nearly 90% stated they preferred learning frontal recess anatomy with the aid of 3-D planning models. 68 In a related study, Citardi et al 26 showed that this technology could be easily integrated into IGS-ESS in a cadaver model and provided useful orientation to surgeons attempting to cannulate an undissected frontal sinus and to highlight targets before they were uncovered surgically.
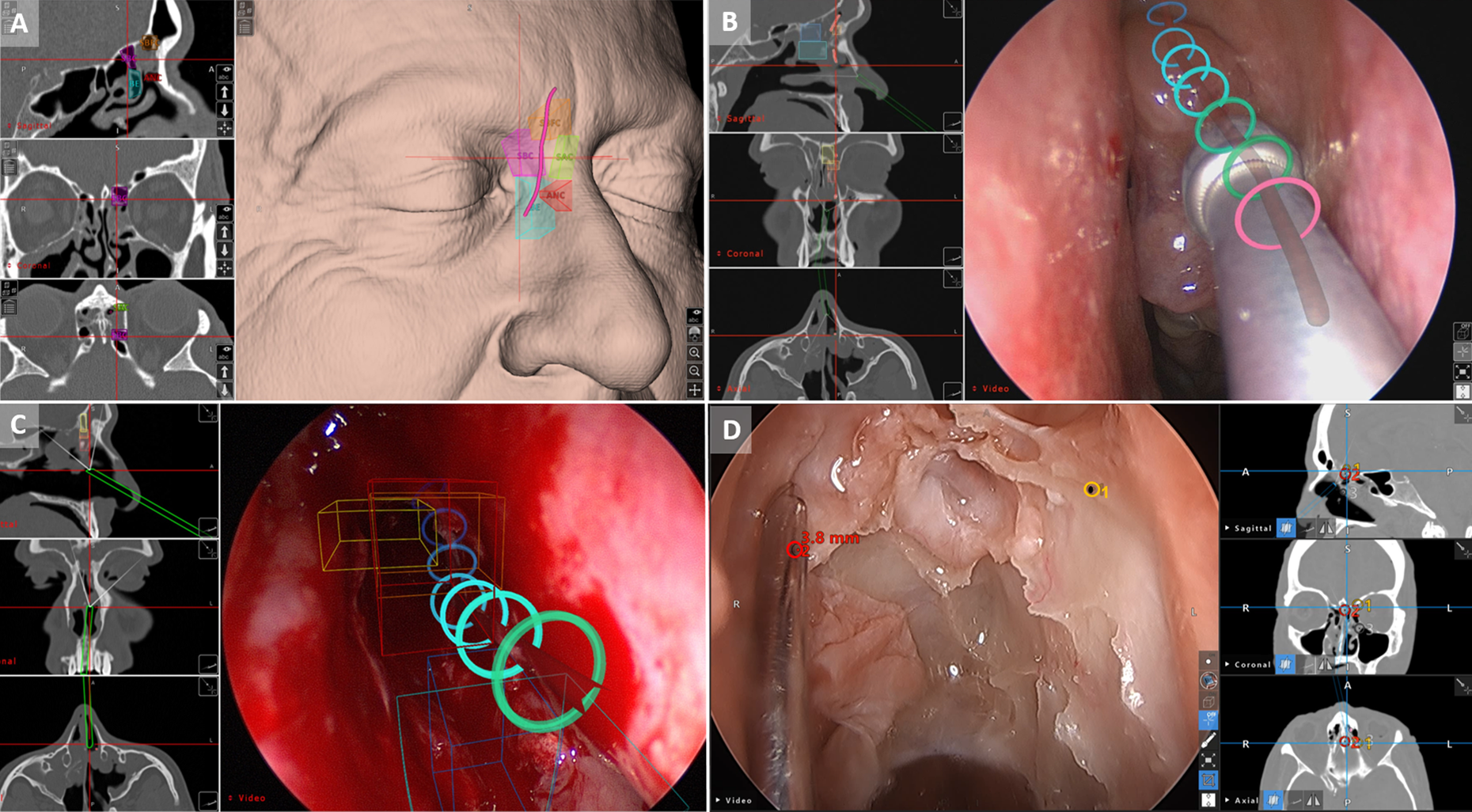
A, In Stryker TGS (Stryker), preoperative planning used to mark relevant anatomy as shown in this instance. Various frontal recess cells and proposed natural frontal sinus drainage pathway are clearly labeled. ANC, agger nasi cell; BE, bulla ethmoidalis; SBC, supra bulla cell; SBFC, supra bulla frontal cell and frontal sinus drainage pathway depicted as a pink line. B, Stryker TGS (Stryker) used to depict the path to target as an overlay on the endoscopic image. The path is animated to give surgeon a real-time feedback of instrument position relative to the preoperative plan. The rings represent the planned pathway with a color gradient from pink to blue depicting proximal to distal. The pink ring represent the portion of the pathway where the tracked instrument has already progressed. C, Multiple preoperative annotations, including frontal recess cells as well as the path to target may be shown as an overlay, as in this Stryker TGS (Stryker) screen capture. D, Screen capture during ESS, again demonstrating augmented reality features of Stryker TGS. The anterior ethmoid artery was marked preoperatively and a distance alarm can be seen as a superimposed annotation on the endoscopic image. ESS indicates endoscopic sinus surgery.
Overall, the field of AR and VR are being increasingly studied and developed given the possible benefits in IGS-ESS and skull base surgery. Some examples are the potential for increased confidence of dissection around critical structures and moreover, real-time intraoperative updates to sinus and skull base anatomy could be extremely valuable in difficult or revision cases. However, there are drawbacks to the technology as well. Depending on the system requirements, space taken up in the operating room can be an issue. Furthermore, the issue of “inattentional blindness” has been reported in previous studies, in which participants utilizing augmented IGS failed to recognize unusual findings of a skull base violation or presence of a foreign body. 69 While considering the limitations, the opportunity for augmented and VR to improve the IGS experience is great, and further studies exploring the indications for use and optimization of the technology are warranted.
Conclusion
As image guidance and intraoperative imaging systems have advanced, their applications in sinus and skull base surgery have expanded. Both technologies offer invaluable real-time localization feedback, and thus may improve the completeness of surgery, surgical safety and efficiency, as well as overall outcomes. Recent advances such as augmented and VR offer a window into the future of IGS by adding useful data to aid surgeons in perioperative and intraoperative surgical decision-making. Future advancements should aim to continue enhancing the surgeon’s experience during IGS, which could ultimately lead to further improvements in surgical results and reduce the need for revision surgery.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. A.U.L. serves as a consultant for Aerin Medical (Sunnyvale, CA), Lyra Therapeutics (Watertown, MA), and Stryker (Kalamazoo, MI). She is on the advisory board for ENTvantage (Austin, TX). She has served on advisory boards for Sanofi (Paris, FR) and Novartis (Basel, CH). M.J.C. serves as a consultant for Acclarent (Irvine, CA), BioMed ENT (San Antonio, TX), Medical Metrics (Houston, TX), and Stryker(Kalamazoo, MI). W.C.Y. serves as a consultant for Stryker (Kalamazoo, MI) and on the Speaker’s Bureau for Optinose (Yardley, PA).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
