Abstract
We present a case of primary systemic amyloidosis diagnosed by endoscopic sinus surgery. A 75-year-old woman had blurred vision in her left eye; computed tomography and magnetic resonance imaging showed shadows of the bilateral paranasal sinuses. Endoscopic sinus surgery was performed, and amyloidosis was diagnosed by histopathology. She had previously been diagnosed with amyloidosis of the stomach, and therefore, she was diagnosed with primary systemic amyloidosis. A systemic workup for additional amyloid deposits revealed no evidence of other diseases. The patient remained under follow-up without further treatment, as no further amyloid deposition or progression of the lesions was seen. Amyloidosis is a rare condition characterized by the deposition of abnormal protein filaments in the extracellular tissue. Generally, systemic amyloidosis does not involve the head and neck region, and the presence of amyloid in the nasal and paranasal sinus mucosa is more likely to be indicative of a localized process. However, in our patient, the lesions were located in both the sinonasal tract and the stomach, indicating systemic amyloidosis. To our knowledge, there have been no previous reports of systemic amyloidosis involving the sinonasal tract, and therefore, we consider this case to be extremely rare.
Introduction
Amyloidosis, a rare condition characterized by the deposition of abnormal protein fibrils in the extracellular tissue, is classified as systemic amyloidosis and localized amyloidosis. The most frequent types of systemic amyloidosis are immunoglobulin amyloid light-chain amyloidosis (AL; primary amyloidosis, as in the present case) and serum amyloid A protein–associated amyloidosis (AA; secondary amyloidosis). Amyloid light-chain amyloidosis usually occurs in the heart, kidney, or digestive tract, such as the stomach or intestines. Generally, systemic amyloidosis does not involve the head and neck region, and the presence of amyloid in the nasal and paranasal sinus mucosa is more likely to be indicative of a localized process. Localized amyloidosis of the nasal and paranasal sinuses is extremely rare, with only 17 reported cases. 1 However, there has been one report of systemic amyloidosis involving the nasopharynx. 2 Here, we report an extremely rare case of AL amyloidosis occurring in both the sinonasal tract and the stomach.
Case Report
A 75-year-old woman with blurred vision in her left eye for 2 months visited an ophthalmologic clinic. She had a history of cervical cancer, cataract, hives, hyperlipidemia, and hypertension, but she had no family history of serious disease. Although her ophthalmologic findings were normal, an abnormal shadow in the paranasal sinus was seen on magnetic resonance imaging (MRI) of the head. She was referred to our department for otorhinolaryngological examination. She had no nasal symptoms, and nasal endoscopy found no nasal polyps or masses. Coronal T2-weighted MRI revealed a hypointense lesion on the bilateral membranous portions of the maxillary sinuses and a hyperintense lesion in the right maxillary sinus, indicating entrapped secretions in the sinuses (Figure 1A). Computed tomography (CT) revealed a calcified lesion on the bilateral membranous portions of the maxillary sinuses and the right side of the nasal septum (Figure 1B). We performed endoscopic sinus surgery with the patient under general anesthesia. Surgery was performed for 2 reasons: to obtain an accurate diagnosis of the sinonasal lesions by biopsy and to treat the sinusitis by removing the lesions and opening the sinuses. During the operation, bone thickening–like lesions were found on the right side of the nasal septum (Figure 2A), the bilateral membranous portions of the maxillary sinuses (Figure 2B, C), and the bilateral anterior ethmoid sinuses (Figure 2D). The bone thickening–like lesion of the right membranous portions of the maxillary sinuses was very hard (Figure 2B), and therefore, it was difficult to open the right-sided maxillary sinuses. To avoid damaging the brain and eyes, we removed the bone thickening-like lesion using biting forceps rather than a powered instrument such as a bur. Purulent nasal discharge in the maxillary sinuses was removed by suction (Figure 2D). As much as possible of the affected tissues was removed, and all the sinuses were widely opened. The mucosa of the ethmoid and maxillary sinuses was greatly thickened by sinusitis, but the frontal and sphenoid sinuses had almost normal mucosa.
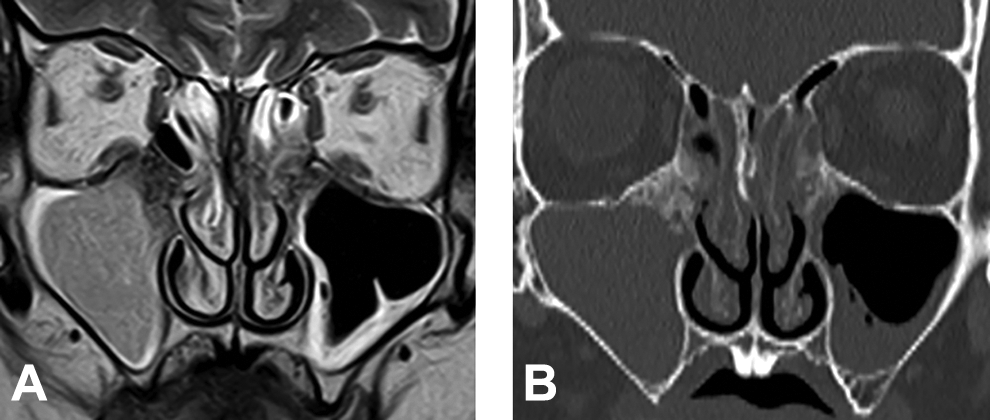
Coronal T2-weighted magnetic resonance image (MRI) showing a hypointense lesion in the bilateral membranous portions and a hyperintense lesion indicating entrapped secretions in the sinuses (A). Coronal computed tomography scan showing a calcified lesion in the bilateral membranous portions and sinusitis (B).
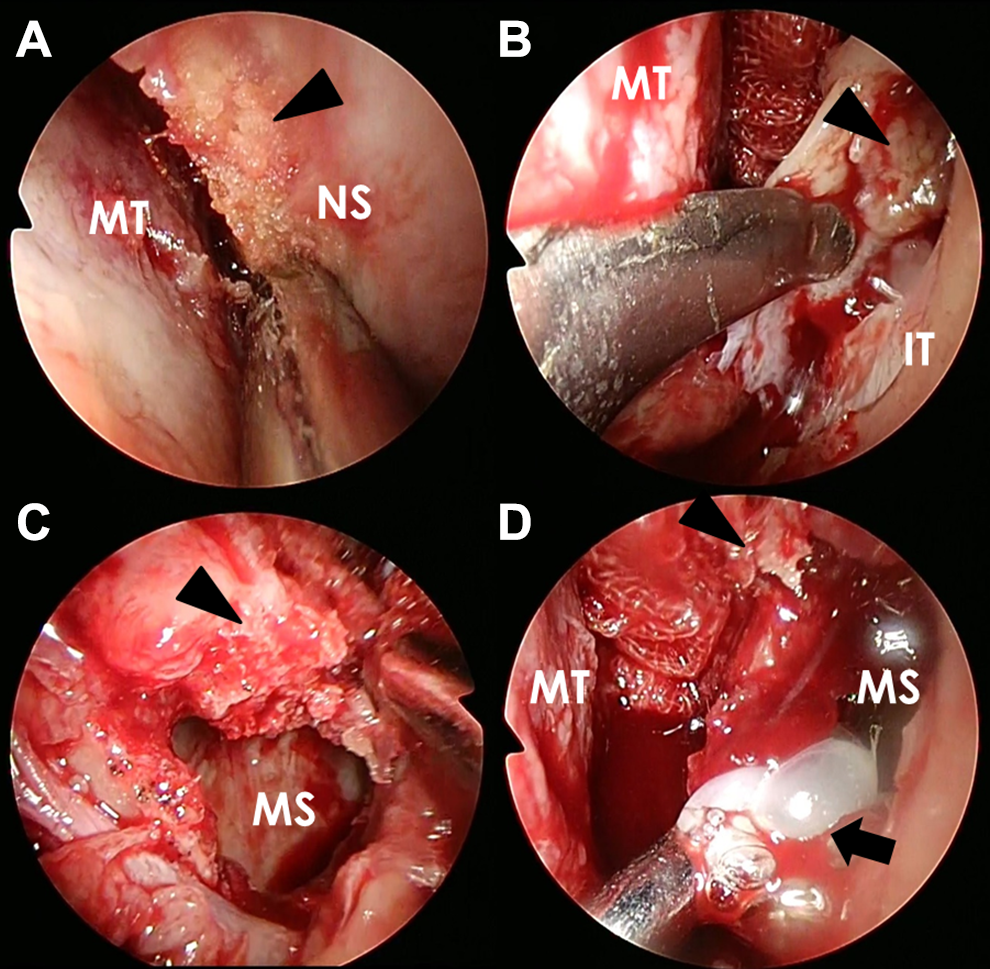
Intraoperative endoscopic view showing bone thickening-like lesions (arrowheads) on the right side of the nasal septum (A) and on the bilateral membranous portions, maxillary sinuses, and ethmoid sinuses (C, right; B, D, left) and purulent nasal discharge (arrow) in the maxillary sinuses (D). IT indicates inferior turbinate; MS, maxillary sinus; MT, middle turbinate; NS, nasal septum.
Hematoxylin–eosin staining of the resected tissues at the bone thickening–like lesions on the right side of the nasal septum (Figure 2A), the bilateral membranous portions of the maxillary sinuses (Figure 2B, C), and the bilateral anterior ethmoid sinuses (Figure 2D) revealed homogeneous nonstructural deposits of an acidophilic mass around the blood vessels that were positively stained with both Congo red and direct first scarlet (DFS) stain. The histopathological diagnosis of the lesion was amyloidosis (Figure 3A-C). When we informed the patient after surgery that the pathological diagnosis of the sinonasal lesion was amyloidosis, she told us that she had received a diagnosis of amyloidosis on the greater curvature side of the lower gastric body about 1 year previously. Therefore, we diagnosed her disease as a systemic amyloidosis involving the sinonasal tract and stomach.
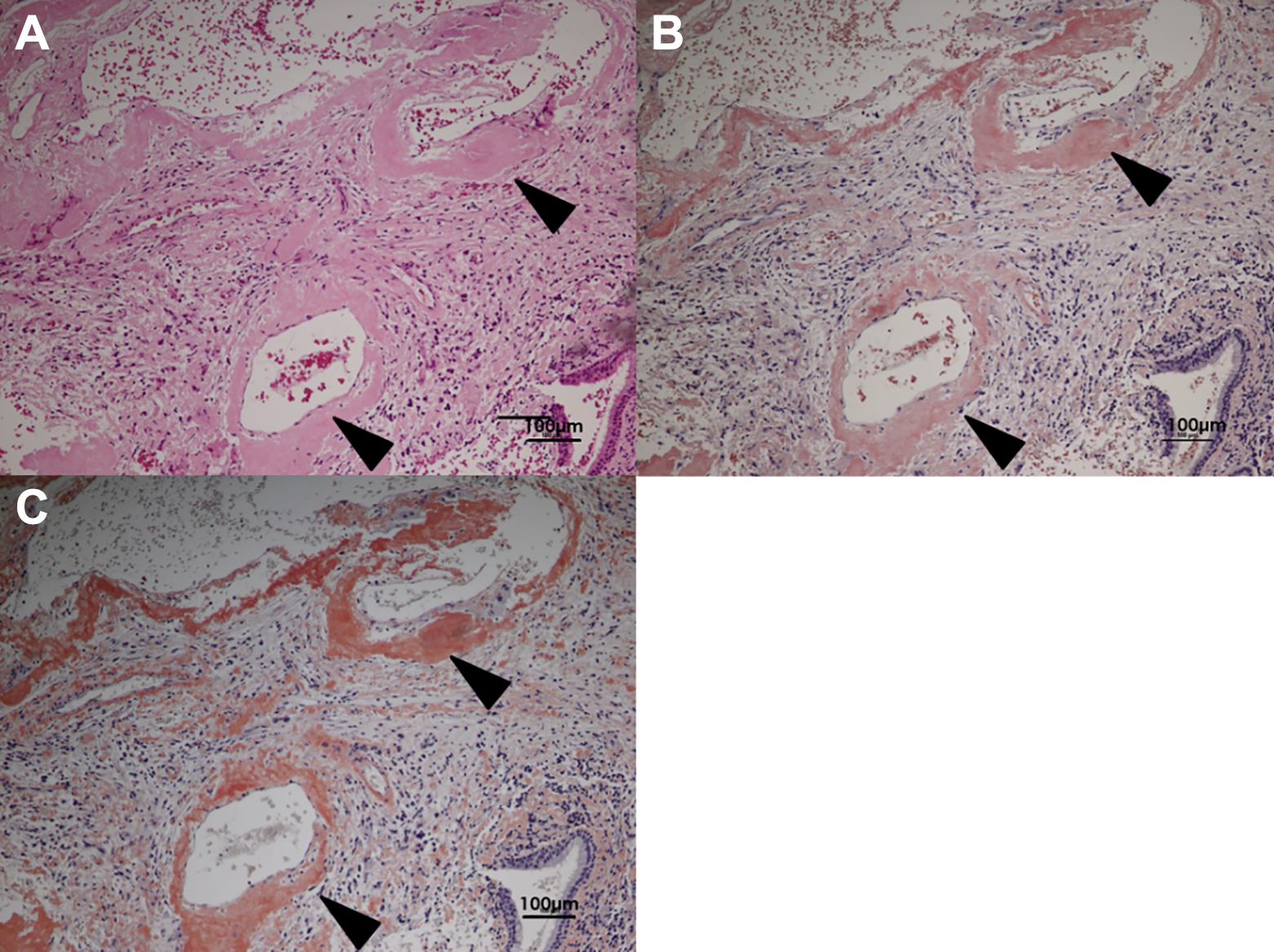
Hematoxylin–eosin stain of the resected tissues showing homogeneous nonstructural deposits (arrowheads) of an acidophilic mass around the blood vessels (A), which was stained orange with Congo red (B) and direct first scarlet (C). Magnification ×200.
To search for additional amyloid deposits, several examinations were performed, including cardiac sonography, bone marrow aspiration, and blood and urine examination. No amyloid involvement was found in other organs. Finally, immunohistochemical examination of the resected tissues established a diagnosis of AL amyloidosis.
One year after surgery, the nasal cavity mucosa was partially adhered (Figure 4A, B), and the gastric lesion was not changed. Although there were some residual calcified lesions on postoperative CT, nearly all of the sinuses remained widely open and had good aeration (Figure 4C, D). The patient remained under follow-up without further treatment for 1 year and 6 months, in cooperation with the internist.
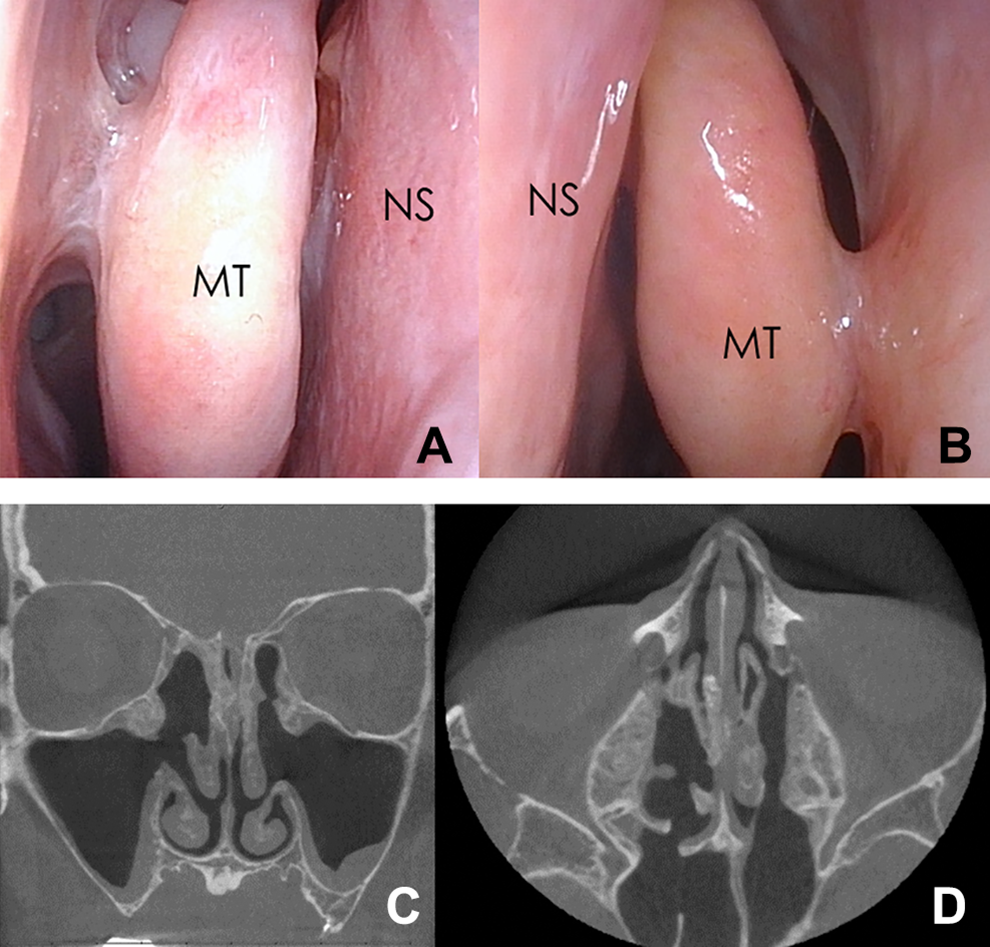
Endoscopic nasal view showing adherence between the middle turbinate and lateral nasal wall in both nasal cavities (A: right, B: left). Postoperative computed tomography scan after 1 year showing aeration of the sinuses and residual calcified lesions in coronal (C) and axial (D) views. MT indicates middle turbinate; NS, nasal septum.
Discussion
Amyloidosis can be classified as localized or systemic, depending on the distribution of the amyloid fibrils. The distinction between localized and systemic disease is important because localized amyloidosis can be managed conservatively with an excellent prognosis, whereas systemic amyloidosis is associated with significant morbidity and mortality. The presence of systemic amyloidosis may be detected by M protein in the serum, Bence Jones protein in the urine, or increase of free light chain in the blood. However, the diagnosis of amyloidosis is based on the findings of amyloid deposits in a biopsy specimen, such as the skin, kidney, or stomach. Congo red or DFS staining of the biopsy specimen produces apple green birefringence under polarized microscopy. 3 The most frequent type of systemic amyloidosis is AL amyloidosis, followed by AA amyloidosis. 4 Amyloid light-chain amyloidosis is caused by the deposition of protein derived from immunoglobulin-free light-chain fragments released by abnormal plasma cells. Amyloidosis associated with immune disorders, such as rheumatoid arthritis and inflammatory bowel disease, is AA amyloidosis, not AL amyloidosis. Primary AL amyloidosis is not associated with underlying diseases, such as multiple myeloma or primary macroglobulinemia. Our patient was considered to have primary AL amyloidosis because amyloid deposits were detected by biopsies of the stomach and nose, and she had no history of immune disorders such as rheumatoid arthritis or inflammatory bowel disease.
Amyloid is deposited in various organs, resulting in their dysfunction. Amyloid deposition usually occurs in the heart, kidney, digestive tract (stomach and intestine), and peripheral nerves; it rarely occurs in the head and neck region. The larynx is the most frequently involved site in the head and neck region (61%), followed by the oropharynx (23%), trachea (9%), orbit (4%), and sinonasal tract and nasopharynx (3%). 4 Only 17 cases of isolated amyloidosis of the nasal and paranasal sinuses have been reported. 1 Generally, systemic amyloidosis does not involve the head and neck region, 5 and the presence of amyloid in the nasal and paranasal sinus mucosa is more likely to be indicative of a localized process. However, our patient had lesions in both the paranasal sinuses and the stomach, indicating systemic amyloidosis. There have been 2 reports of systemic amyloidosis involving the pharynx2,6 and 8 reports of systemic amyloidosis involving the tongue. 5 However, to our knowledge, there has been no report of systemic amyloidosis involving the sinonasal tract, so our case is extremely rare. The reason why systemic amyloidosis occurred in the sinonasal tract in this case is unclear. In general, the clinical symptoms of sinonasal amyloidosis are frequent nasal symptoms, such as epistaxis and nasal obstruction. Our patient had no nasal symptoms, and nasal endoscopy did not find nasal polyps or masses. Therefore, sinonasal amyloidosis may be accidentally found, as in this case. Systemic primary AL amyloidosis in the sinonasal tract may be present in hidden cases.
Amyloid light-chain amyloidosis can be diagnosed by elevation of kappa or lambda light chains in the blood or urine, but immunohistochemical examination is needed to confirm the diagnosis. 7 The diagnosis in our patient was established not by biochemical examination but by tissue immunohistochemistry. Patients who progress to multiple myeloma or who have general organ dysfunction due to amyloid deposits have a poor prognosis. In these patients, systemic chemotherapy and peripheral blood stem cell transplantation are standard treatments; however, asymptomatic or elderly patients without progression of the lesion may be observed without further treatment, as in our patient.
The clinical symptoms of sinonasal amyloidosis are common nasal symptoms, such as epistaxis and nasal obstruction, depending on the site of the amyloid deposits. A previous study reported that the most common sites of origin were the conchae and the lateral nasal wall. 8 Episodes of epistaxis are associated with the loss of vascular integrity (as the amyloid tends to infiltrate the blood vessels) or with the appearance of fragile, irregular mucosa that covers the amyloid masses. 7 Amyloidosis of the sinonasal region may progress slowly for months or years before diagnosis. Fungal rhinosinusitis is well known as a disease showing a calcified lesion on CT. The CT appearance of amyloidosis of the nasal and paranasal sinuses is relatively nonspecific; however, Chin et al reported that it had a “fluffy” appearance, possibly caused by an osteoblastic reaction induced by the deposition of proteinaceous amyloid fibrils. 9 Another study reported that T2-weighted MRI showed distinctive hypointensity at the amyloid deposition sites, which excluded many of the other differential diagnoses. 10 Nakayama et al reported that T2-weighted MRI was useful to evaluate the extent of the amyloidosis. 7 In the present case, the CT and MRI findings were very similar to those described above.
Most cases of sinonasal amyloidosis are treated with surgery, but mucosal adhesion often occurs after surgery. Furthermore, complete removal of amyloid is very difficult because of the risk of damaging the brain and eyes, and residual lesions are inevitable. Debulking of the amyloid without impairing the function of the surrounding organs is recommended for the treatment of sinonasal amyloidosis. 2 Despite a high rate of recurrence (up to 50%), surgery provides at least symptomatic improvement in the majority of patients. 11 Recurrent amyloid deposits can be simply re-excised, and therefore, complete removal of amyloid deposits is not recommended if it carries a risk of significant morbidity.
Conclusion
This study is only a case report, and the follow-up period is still very short. We report a case of systemic primary AL amyloidosis involving the sinonasal tract and showing characteristic radiological findings on both CT and MRI. This was an extremely rare case of AL amyloidosis with amyloid deposits in both the sinonasal and the gastric tracts. The patient remains under follow-up without further treatment, as no further amyloid deposition or progression of lesions is seen. We should pay attention to amyloidosis when we find calcified lesions in the bilateral sinonasal tract on CT.
Footnotes
Acknowledgments
The authors thank Dr. Yuji Shimura, Department of Hematology and Oncology, Kyoto Prefectural University of Medicine, for his therapeutic advice and cooperation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
