Abstract
Painful neuromas are a devastating condition that is notoriously difficult to treat. The large number of techniques that have been attempted suggest that no one technique is superior. Neuromas often occur in the extremities, but iatrogenically caused pain in the head and neck area has also been described. This article describes 3 consecutive patients diagnosed with traumatic neuroma who underwent transection of the causative nerve, followed by capping of the nerve stump with a Neurocap. With a follow-up of 7 to 24 months, our results show a marked reduction in the pain scores of all 3 patients. The preliminary results indicate that this technique might be a viable treatment option for patients with a suspected neuroma in the head and neck area.
Introduction
Painful neuromas are considered a devastating condition for which many treatment options have been tried in the past. Unfortunately, they are still notoriously difficult to treat. 1 -5 Neuromas often occur in the extremities following trauma, but iatrogenic pain in the head and neck area has also been described. Trauma to the occipital nerves and the greater auricular nerve during excision of scalp masses, neurotological procedures, and parotid gland surgery can lead to sensory complications. 6,7 Iatrogenic pain in the eyebrow was a rather common consequence of an external approach to the frontal sinus. 8
To prevent an injured nerve from sprouting, various therapies were described in the past. Microbipolar cautery of the distal end of the nerve was tried, and nerves were buried into muscle, bone, or vein grafts. 9 -11 Moreover, a variety of materials were developed for capping purposes. 12 -14 However, a paucity of evidence exists to support one method over the other. This is partly due to the fact that the reporting of outcomes varied widely across the studies. Furthermore, previous studies have focused on the extremities, and therefore data cannot be extrapolated to our region of interest: the head and neck.
The Neurocap (Polyganics BV, Groningen, the Netherlands) is a transparent tubular device intended to protect a peripheral nerve stump and to separate the nerve from the surrounding environment, in order to prevent the development of a neuroma. It is made of a synthetic copolymer of lactide and caprolactone and it is supposed to be biocompatible and resorbable. It is the same material as used in the Neurolac which is a Food and Drug Administration/CE-approved nerve conduit for clinical repair of peripheral and cranial nerves. According to the manufacturer, its efficacy is being tested in 2 trials (the Surgical Treatment Of symPtomatic Neuroma and PROTECT studies), but the results have not yet been published.
In the last decade, our patients with neuroma are mainly treated with drug therapy for which the severe cases are referred to the Pain Clinic of the Department of Anaesthesiology. However, because neuropathic pain is often resistant to analgesics, better treatment options are demanded in clinical practice.
In this report, we describe the results of excision of iatrogenic neuromas with subsequent capping of the nerve stump with the Neurocap in 3 patients who had undergone ear, frontal sinus, and parotid gland surgery in the past. Our patients experienced reduced quality of life due to pain in the previously operated area. Many different colleagues had treated all 3 unsuccessfully. All 3 were desperate for a therapy with sustainable effect.
Case Series
The first patient was a 24-year-old female with severe otalgia. She scored a numerical rating scale (NRS) score (NRS 0-10) of 9. She had frequent grommet insertion for otitis media in childhood; at age of 22, she underwent myringoplasty in the left ear for a persistent perforation; retroauricular pain had developed since then. Scar revision surgery 1 year later resulted in temporary (8 weeks) relief of complaints. At clinical examination, she had no signs of ongoing infection, no retroauricular swelling or scar hypertrophy was noted, and otoscopy showed a normal aspect of the tympanic membrane. The pain could be relieved by peripheral nerve block (PNB) of the posterior branch of the greater auricular nerve, just proximal to the scar of the retroauricular incision. Peroperatively, a dissection of the greater auricular nerve was performed; the posterior branch could be followed into the scar tissue. This branch was transected and a 1.5-mm Neurocap was placed over the proximal stump. With the aid of a suture, the nerve was pulled into the cap without using its entire length—as in indicated in the manufacturer’s instruction—leaving a couple of millimeters of space in the front of the cap. Microscopically, a biopsy of the transected distal end showed sprouting into scar tissue as a consequence of previous surgery. Pathologic examination (see Figure 1) revealed a neuroma (positive S100 staining). At 24 months follow-up, the NRS score is 0.
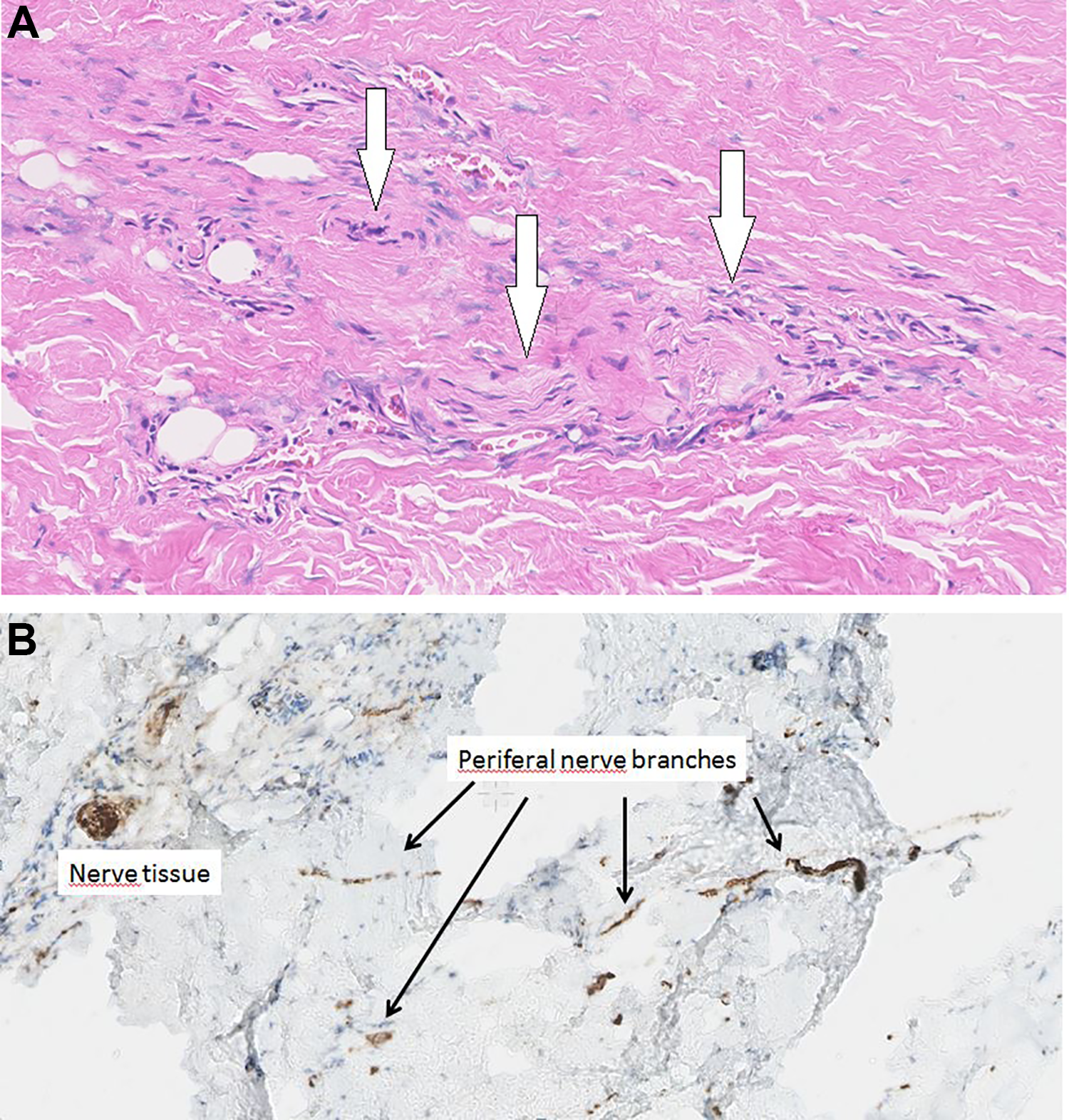
Histology imaging of nerve sprouting after iatrogenic damage. A, Hematoxylin and eosin (H&E) staining showing small nerves (arrows) in the tissue section. B, S100 immunohistochemistry marker showing small nerve offset in the nerve tissue (arrows).
Our second patient was a 55-year-old man with a history of multiple procedures of frontal sinus surgery via eyebrow incisions. The most recent of his eyebrow incisions was 10 years ago. He had a painful area in the region of the supraorbital nerve. He reported a NRS of 7. He could consistently locate the point of irritation deep in the soft tissue underneath the right orbital roof. Local anesthetic nerve blocks at that point gave only short-term relief of his complaints. Computed tomography scan confirmed the status following surgery, but no remaining pathology was found in the frontal sinus and supraorbital region. The patient was operated, and a neuroma of the supraorbital nerve was removed. A 2.0-mm Neurocap was placed over the proximal stump. The pathology report confirmed neuroma formation. At 7 months postoperatively, the NRS score was significantly reduced to 3. The remaining discomfort, that had been present before the capping as well, was located in the midfacial segment, a location outside our surgical area, unlikely caused by the neuroma and not expected to be resolved by the neurocapping. Because, our patient’s military career ended last year, he was lost to follow-up.
The third patient was a woman of 63 who underwent parotidectomy and postoperative irradiation because of a nonradically excised pleomorphic adenoma elsewhere. She was referred a year later to our hospital because of severe pain (NRS of 9) in the preauricular region. Physical examination and magnetic resonance imaging did not show recurrence of the pleomorphic adenoma or other pathology. Peripheral nerve block of the greater auricular nerve proximal to the parotidectomy scar gave complete relief of the pain for a couple of hours.
We performed an exploration and transection of this nerve. An incision was made a few centimeters caudally of the previous parotidectomy incision. From here, we could explore the greater auricular nerve and follow a couple of sprouts from the nerve into the dense scar tissue over the remaining parotid tissue. This tissue was excised; histology showed a traumatic neuroma. After transection of the nerve, the proximal end was capped with a 3.0-mm Neurocap (see Figure 2). At 19 months follow-up, the NRS in the preauricular area is 2. She reported a significant improvement in her quality of life.
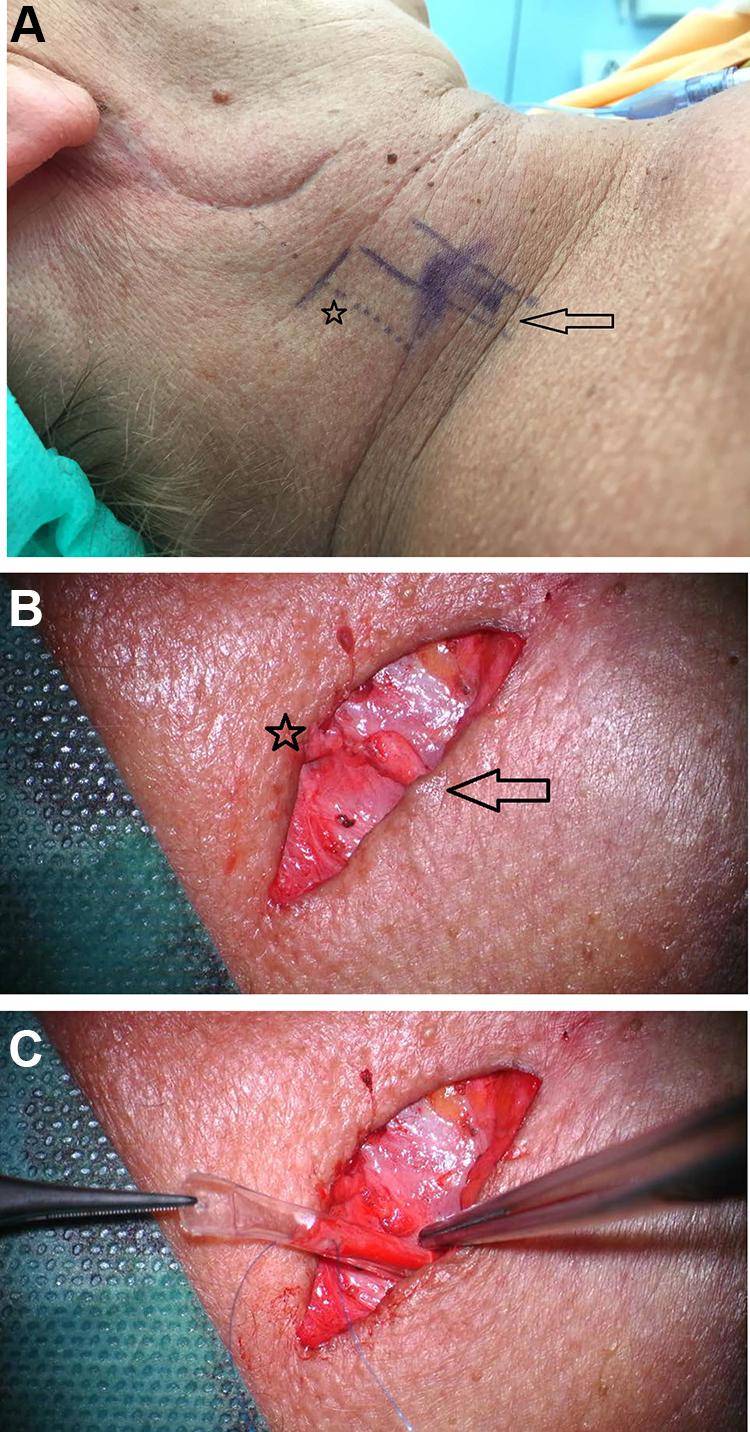
A, Right side of neck from patient 3 with the scar of the previous surgery clearly visible. Probable location of the greater auricular nerve is marked, and the skin crease for the more proximal incision is indicated. The nerve is usually located 1-cm posterior from the external jugular vein (marked by the arrow). B, The transected greater auricular nerve is shown, distal part (to the earlobe) is marked by an asterisk, and proximal part (from the punctum nervosum) is marked by an arrow. C, The Neurocap is sutured at the proximal nerve end.
Discussion
We report the short-term results, ranging 7 to 24 months, of 3 patients with a diagnosis of iatrogenic neuroma in the head and neck area. They were operated for neuroma removal, followed by placement of a Neurocap. To the best of our knowledge, this is the first report on resection of head and neck neuromas combined with capping the proximal stump with a Neurocap.
Neuromas usually develop following trauma or surgery and effect 2% to 60% of patients with nerve injury. 15 Why some patients develop painful neuromas and others do not is incompletely understood. 15 Often, patients will present with a history of pain for which they had surgery, and that pain resolved but was replaced with much worse pain after surgery. 15 This should raise suspicion for iatrogenic nerve injury and subsequent neuroma formation.
A systematic review on surgical treatment options for neuromas showed an overall success rate of 77% regardless of surgical technique used. 3 Twenty to thirty percent of neuromas seem refractory to surgical treatment. 3 Sustained input from the peripheral nervous system could even lead to central sensitization.
The large number of techniques that have been attempted suggests that not 1 technique is completely effective. 16 -18 Some authors describe good results with nerve capping with a nerve conduit for the clinical treatment of painful neuromas in the extremities, but most of the conduits were used as interposition grafting between the proximal and the distal ends of the nerves following removal of neuromas. 18
The importance of careful and correct diagnosis when considering surgical intervention for pain cannot be overemphasized. Allodynia, dysesthesia, or hyperpathia should raise suspicion of a neuropathic source of pain. 16 Our patients presented with life-altering chronic pain lasting for greater than 6 months. In all patients, imaging did not show any other explanation. No other treatment was successful and/or without side effects. Due to the prior surgery, the most probable single nerve to cause the deficit could be easily suspected. In addition, we made a temporary response to a PNB, a prerequisite for surgical exploration. For the size of the Neurocap, like Gould et al did, we chose a diameter that would slip easily over the nerve stump without being too tight or redundant. 19 In case conduits are used, Onode et al recently advised to use a conduit of 0.5-mm larger than the nerve to prevent constriction. 20 They found the appropriate nerve conduit length to be more than 4 times the diameter of the original nerve. 20 Yan et al, in 2 studies, had good outcomes by suturing the sciatic nerve 4 mm into a 1.5-cm long nanofiber conduit. 21,22
We think that there may be differences in the indications for when to use a conduit (in case outgrow of the nerve in an appropriate manner is needed) or to use a cap (to prevent outgrow—like in our cases).
Poppler et al defined a meaningful reduction in pain—among others—as a pain score reduction of 3 or more or final visual analogue pain scale score less than 4. 3 At the moment, the reported follow-up results of this approach seem satisfactory and fulfill the abovementioned criteria. Therefore, these results indicate that capping of iatrogenic traumatized nerves in the head and neck area could be a viable option for patients with severe neuropathic pain.
The good effect of the Neurocap might be due to its properties in that it takes 2 years to dissolve and therefore prevent nerve regrowth both by mechanical obstruction and that nerve sprouting stimulating factors from surrounding tissues do not reach the stump. Moreover, blocking the infiltration of the nerve stump with inflammatory cells was thought to be the primary mechanism of autotomy inhibition in a rat model. 23 Last but not least, just the insertion of the nerve stump in a nerve cap, for which no contraindications exist, seems to be a much simpler technique than the use of vein grafts or other conduits.
Like others, we think it is important during surgery to identify the nerve within the normal anatomy before entering the distorted anatomy of the previously dissected field; exposure of the nerve proximal is much easier and safer. With our patients, due to extensive scarring in the tissue innervated by the injured nerves, we did not try to restore integrity or search for intact distal nerve stumps; this was deemed impossible and/or superfluous. Not sensory loss but pain was the reason for surgery.
Reinnervating grafts, or bridging with a distal nerve end, within the head and neck region is typically used for the facial, recurrent laryngeal, and hypoglossal nerves that serve motor functions. While recovery of 2-point discrimination function after digital nerve repair in the hand has been described in the last decennium, sensory nerve repair has not been mentioned in the head and neck area. 13,24
Perhaps, if a larger sensory nerve is cut accidentally, leaving a distal end that is not located in scarred tissue, sensory nerve reconstruction—with or without the use of conduits—might be tried in the future. For good reviews on possible nerve conduits, we like to refer to Quigley et al and Kehoe et al. 25,26
Our series has some obvious drawbacks. First, our series only describes 3 patients. However, because neuroma constitutes a rarely reported pathology in the head and neck area, we still consider these cases a valuable contribution to the literature. Second, no control group is introduced, for example, capping with veins or burial in the muscle. Third, the follow-up, although significant, should preferably be longer.
Burchiel et al stated that the basic properties of axons are altered by injury such that, rather than high fidelity conductors, they become generators of abnormal activity. 2 This would lead to the conclusion that removal of the generator of abnormal neuronal activity should be beneficial. 2
Indeed, we believe that these interim results could help colleagues in making difficult treatment decisions involving patients with devastating pain in the head and neck region. Based on our results so far, we agree with Poppler et al that in appropriately selected cases, the dogma that “operating for pain will only result in more pain” may not apply. 3
Conclusion
Neuroma excision combined with capping of the proximal stump with a Neurocap might be a viable treatment option for iatrogenic neuropathic pain in the head and neck region.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
