Abstract
Korean red ginseng (KRG) has been traditionally used in Korea for health improvement. However, the clinical effect of KRG intake on the symptoms in patients with allergic rhinitis remains unknown. Our study was performed to identify the clinical effects of KRG on patients with allergic rhinitis and to examine the effect of KRG on allergic inflammatory reaction. We evaluated 60 patients with allergic rhinitis. All the patients were treated for 4 weeks. The patients were divided into 3 groups, according to the medication. Twenty patients were treated with KRG, 20 patients with the placebo, and 20 patients with the antihistamine. The patients recorded their symptoms in a daily symptom diary card. The patients checked the peak nasal inspiratory flow rate 2 times a day. Total serum immunoglobulin E (IgE) and serum-specific IgE were measured by ImmunoCap method before and after 4-week medication. The Th2 cytokines interleukin-4 (IL-4), IL-5, and IL-10 were checked in the serum before and after the 4-week treatment. The eosinophil counts in the nasal smears were checked. Korean red ginseng group has shown the significant improvement in rhinorrhea, nasal itching, and eye itching. Both the antihistamine and KRG groups showed a significant decrease in total IgE level at the end of treatment. The serum IL-4 level and eosinophil counts in the nasal smears were significantly decreased both in the antihistamine and in the KRG groups. In conclusion, KRG might be a useful treatment modality for patients with allergic rhinitis.
Introduction
Complementary and alternative medicine is commonly used to treat patients with allergic rhinitis. 1 Korean red ginseng (KRG), the steamed root of Panax ginseng Meyer, is traditionally used in Korea to improve health. The major components of ginseng are saponin and 37 different ginsenosides that exert different biological effects. Ginsenosides exhibit anti-inflammatory, anti-allergic, and antitumor activities. 2 -5 Ginsenosides Rb1, Rb2, Rc, Rd, Re, Rf, and Rg1 are the major ginsenosides. 6 Ginsenoside Rd elicits Th1 and Th2 immune responses by regulating the expression of Th1 and Th2 cytokines. 7 Ginsenosides Rh2 and Rg3 exert anti-inflammatory effects in vivo and were effective when used in an animal model of atopic dermatitis. 8,9 Ginsenoside Rh1 reduced the levels of interleukin-6 (IL-6) and total immunoglobulin E (IgE) in serum and reduced the degree of skin lesions similar to ear edema and atopic dermatitis in mice. 10
Korean red ginseng showed an anti-allergic effect on allergic mouse models and decreased Th2 cytokine activity. 11 The principal ginsenosides of KRG are Rg1 and Rb1; Rg1 inhibits the development of allergic symptoms reflected by rub scores and histamine, IgE, and IgG1 levels. Rg1 also reduced eosinophil and mast cell infiltration in a mouse model of allergic rhinitis. 12,13
However, no clinical effect of KRG on the symptoms of allergic rhinitis has yet been shown. Here, we identified such effects and explored whether KRG modulated allergic inflammatory reactions.
Patients and Methods
Patients
We identified 60 patients with allergic rhinitis who presented to the Gachon University Gil Medical Center. We recorded clinical histories, performed physical examinations, and ordered laboratory tests. Sensitization was considered present when the serum IgE levels against both Dermatophagoides pterynissinus and Dermatophagoides farine were ≥0.7 kU/L on ImmunoCap testing and the wheal diameters of D pterynissinus and D farine were equal to or greater than those of the histamine control on skin testing.
The severity of allergic rhinitis was assessed on a scale from I to IV, as recommended by the 2009 “Allergic Rhinitis and its Impact on Asthma” (ARIA) guidelines: I, mild intermittent; II, moderate–severe intermittent; III, mild persistent; IV, moderate–severe persistent. Before enrollment, we obtained written informed consent from all patients after a full explanation about this study. Our randomized, double-blind study was approved by the Gachon University Gil Medical Center Institutional Review Board Committee (GIRBA 2802-2012, approved on July 23, 2012). The exclusion criteria were prior immunotherapy and/or the use of oral medication (antihistamine or steroid) or nasal spray within 1 month prior to study commencement. Patients with sinusitis with anatomical abnormalities (septal deviation, turbinate hypertrophy, concha bullosa, etc) and/or with nasal polyps evident on X-rays were excluded.
Methods
Study design
All patients were treated for 4 weeks. The patients were randomly divided into 3 groups according to the medication. Twenty patients randomly received KRG, 20 received placebo, and 20 received an antihistamine (Allegra, fexofenadine). Korean red ginseng and placebo capsules were provided by the Korean Ginseng Corporation (Daejeon, Korea); the doses were 3 mg/kg/d (the adult dose recommended by the Korea Ginseng Corporation). Antihistamine was given as a single daily tablet (120 mg).
Symptom score diary
All patients recorded allergic rhinitis symptoms on a daily basis using diary cards. Rhinorrhea, the extent of nasal obstruction, sneezing, nasal itching, eye itching, smelling difficulties, and sleep disturbance level were scored using a 6-point Likert scale (0, no symptoms; 1, a few; 2, mild; 3, moderate; 4, severe; and 5, most severe discomfort). All patients were physically examined and invited to discuss their symptoms with physicians both prior to treatment and after the 4 weeks of treatment.
Peak nasal inspiratory flow rate
The peak nasal inspiratory flow (PNIF) meter is a portable instrument measuring the PNIF rate when a patient inspires as rapidly as possible. The flow rate reflects the extent of nasal obstruction. All patients recorded PNIF data twice daily (in the morning and evening) during the medication period; the data were recorded on the diary cards.
Laboratory tests
The levels of total serum IgE and D pterynissinus- and D farine-specific IgE were measured using the ImmunoCap method before and after 4 weeks of medication, as were the levels of IL-4, IL-5, and IL-10 (via enzyme-linked immunosorbent assay). The eosinophil counts in nasal smears were recorded before and after 4 weeks of treatment.
Statistical analyses
All data are presented as means ± standard errors. The t test and repeated-measures analysis of variance subroutines of SPSS version 11.0 were used in statistical analyses. The significance level was set at P < .05 for all comparisons.
Results
Patient Characteristics
The mean patient ages were 33.7 ± 9.6 years (KRG group), 35.6 ± 13.7 years (antihistamine group), and 35.5 ± 10.5 years (placebo group). The prevalence of atopic dermatitis was 5% (1/20) in the KRG group and 10% (2/20) in the antihistamine group. The prevalence of asthma was 5% (1/20) in the KRG group.
The 3 groups did not differ significantly with regard to mean age, gender, ARIA classification, prevalence of atopic dermatitis, or frequency of asthma. Allergic Rhinitis and its Impact on Asthma evaluation indicated that mild persistent patients comprised the majority of all groups (Table 1).
Demographic Characteristics of the Study Population.

Abbreviations: ARIA, Allergic Rhinitis and its Impact on Asthma; ARIA class I, mild–intermittent symptoms; II, moderate–severe intermittent symptoms; III, mild persistent symptoms; IV, moderate–severe persistent symptoms; KRG, Korean red ginseng; NS, not significant.
Safety Assessment
Of the 60 patients, 1 in the KRG group developed mild abdominal pain and 2 developed nausea. However, no change in pre- or posttreatment laboratory parameters was evident, and the symptoms disappeared without any need for medical intervention.
Symptom Scores
In the KRG group, sneezing and nasal and eye itching were significantly ameliorated, as evidenced by serial changes in the symptom scores (evaluated on a weekly basis). These symptoms did not improve in the other 2 groups (Figure 1).
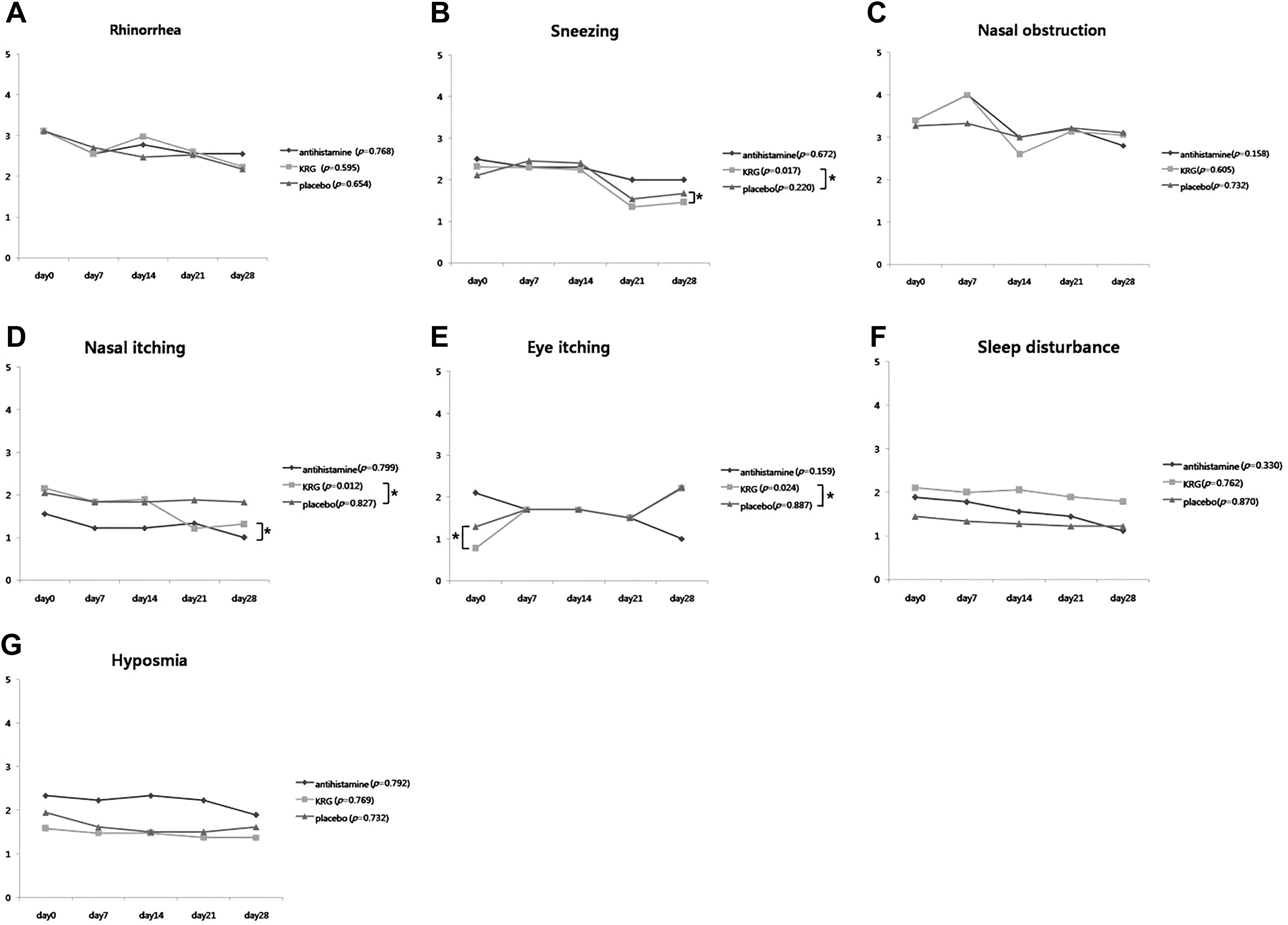
Serial symptom scores recorded in allergy diaries. Sneezing and nasal and eye itching were significantly ameliorated in the KRG group. These symptoms did not improve in the other 2 groups. (The repeated-measures ANOVA were used in statistical analyses.) A, Rhinorrhea, (B) sneezing, (C) nasal obstruction, (D) nasal itching, (E) eye itching, (F) sleep disturbance, and (G) hyposmia. ANOVA indicates analysis of variance; KRG, Korean red ginseng.
All allergic symptoms except nasal obstruction significantly improved in the antihistamine group. The KRG group exhibited significant improvements in rhinorrhea and nasal and eye itching. The pre- and posttreatment symptom scores did not differ significantly in the placebo group. Only eye itching differed significantly between the groups (Figure 2).
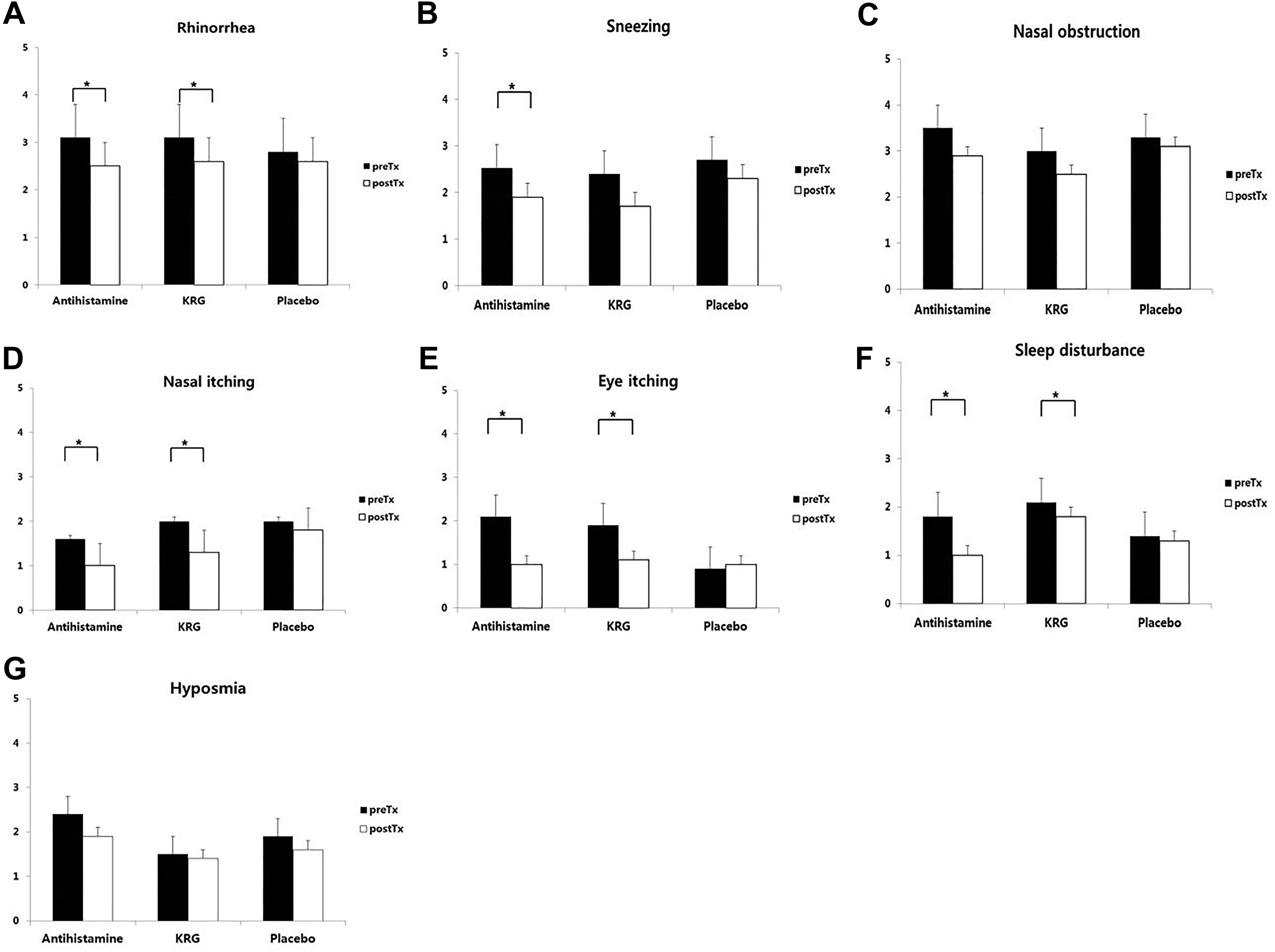
Symptom score improvements. All allergic symptoms except for nasal obstruction significantly improved in the antihistamine group. For KRG group, stress that all allergic symptoms were significantly improved except for nasal obstruction, sneezing, and hyposmia. The pre- and posttreatment symptom scores of the placebo group did not significantly differ. (The t test was used in statistical analyses.) A, Rhinorrhea, (B) sneezing, (C) nasal obstruction, (D) nasal itching, (E) eye itching, (F) sleep disturbance, and (G) hyposmia. KRG indicates Korean red ginseng.
Peak Nasal Inspiratory Flow Rate
Changes in PNIF rates between pre- and posttreatment were analyzed. The PNIF did not differ significantly before and after treatment in either the KRG or the antihistamine groups. Thus, KRG did not improve an objective measure of nasal obstruction (Table 2).
Changes in Peak Nasal Inspiratory Flow Rates (L/min).
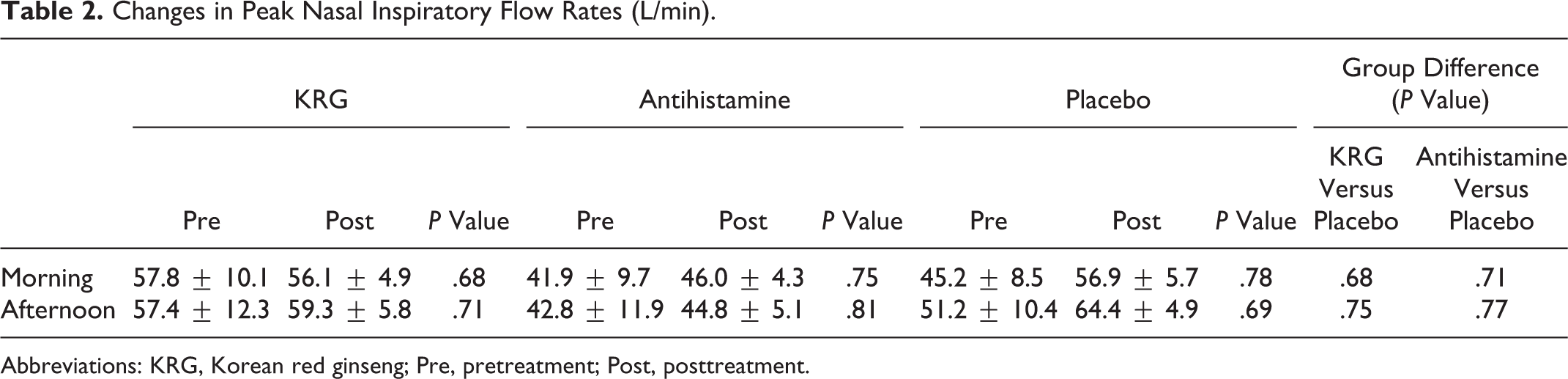
Abbreviations: KRG, Korean red ginseng; Pre, pretreatment; Post, posttreatment.
Laboratory Tests
Total serum IgE and specific IgE levels
Both the antihistamine and the KRG groups experienced significant reductions in total IgE levels after treatment, but the placebo group did not. According to the between-group analysis, the reductions were significant in both groups compared with the placebo group. The levels of IgE specific for D pterynissinus and D farine remained at baseline levels after treatment in all 3 groups (Figure 3).
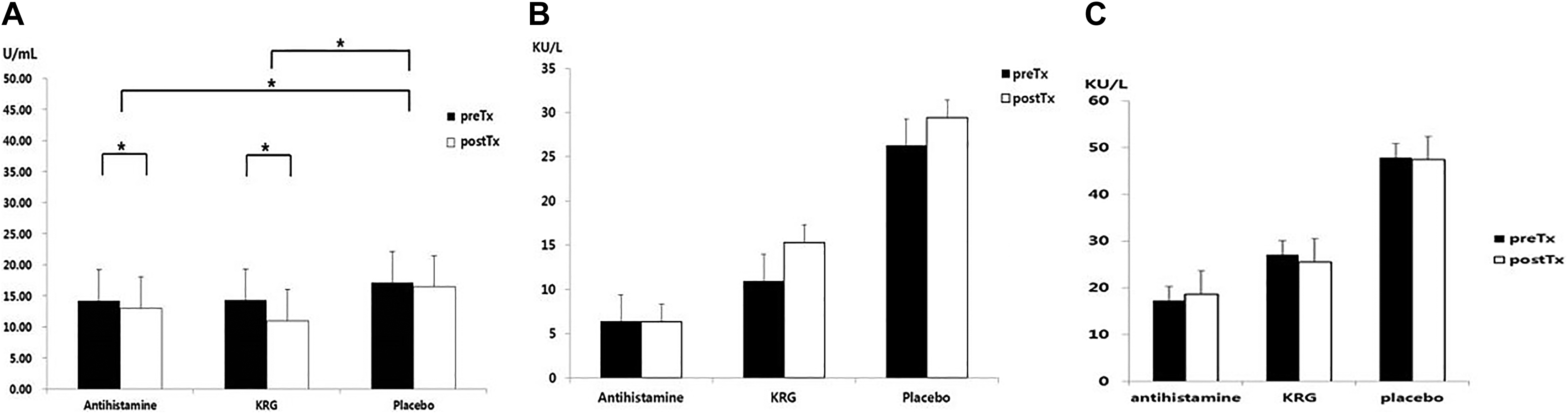
Changes in total IgE levels and the levels of specific IgEs recognizing Dermatophagoides pterynissinus and Dermatophagoides farine. A, Total IgE levels. B, Dermatophagoides pterynissinus–specific IgE levels. C, Dermatophagoides farine–specific IgE levels. Total IgE levels decreased significantly in both the antihistamine and KRG groups. However, no significant change in the levels of IgE specific for D pterynissinus or D farine was evident in either group. (The t test was used in statistical analyses.) IgE indicates immunoglobulin E; KRG, Korean red ginseng.
Cytokines in serum
The serum IL-4 level decreased significantly in both the antihistamine and the KRG groups but not in the placebo group. The IL-5 level did not change in any group. Interleukin-10 levels increased significantly in both the antihistamine and the KRG groups but not in the placebo group (Figure 4).
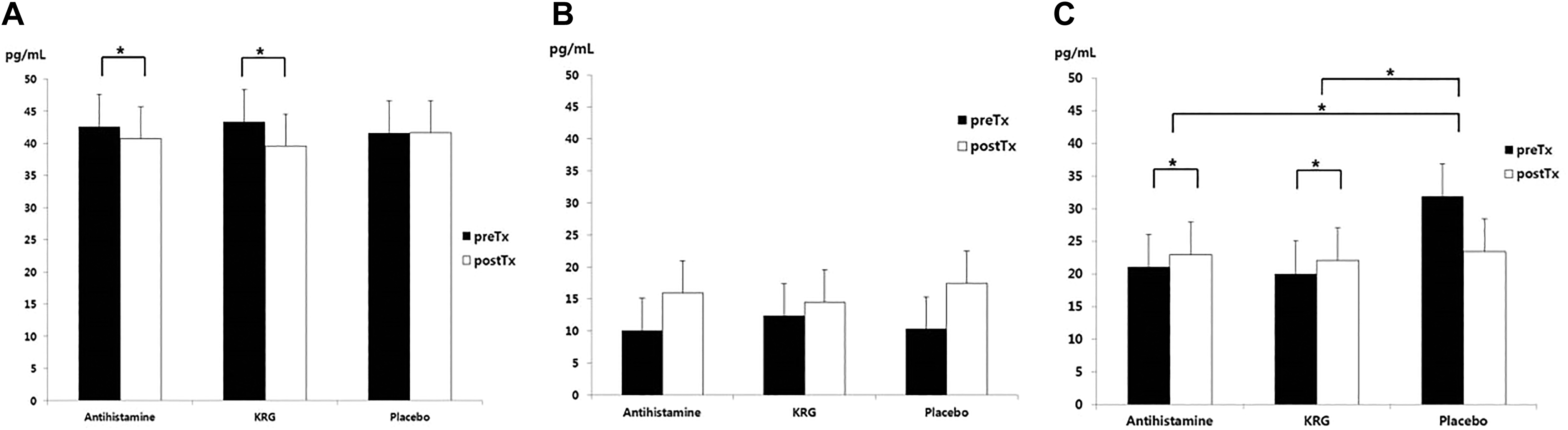
Changes in serum cytokine levels. A, IL-4 levels decreased significantly in both the antihistamine and the KRG groups but not in the placebo group. B, IL-5 levels did not change in any group. C, IL-10 levels increased significantly in both the antihistamine and the KRG groups. (The t test was used in statistical analyses.) IL indicates interleukin; KRG, Korean red ginseng.
Eosinophil counts in nasal smears
Eosinophil counts in nasal smears decreased significantly after treatment with the antihistamine or the KRG but not with the placebo (Figure 5).
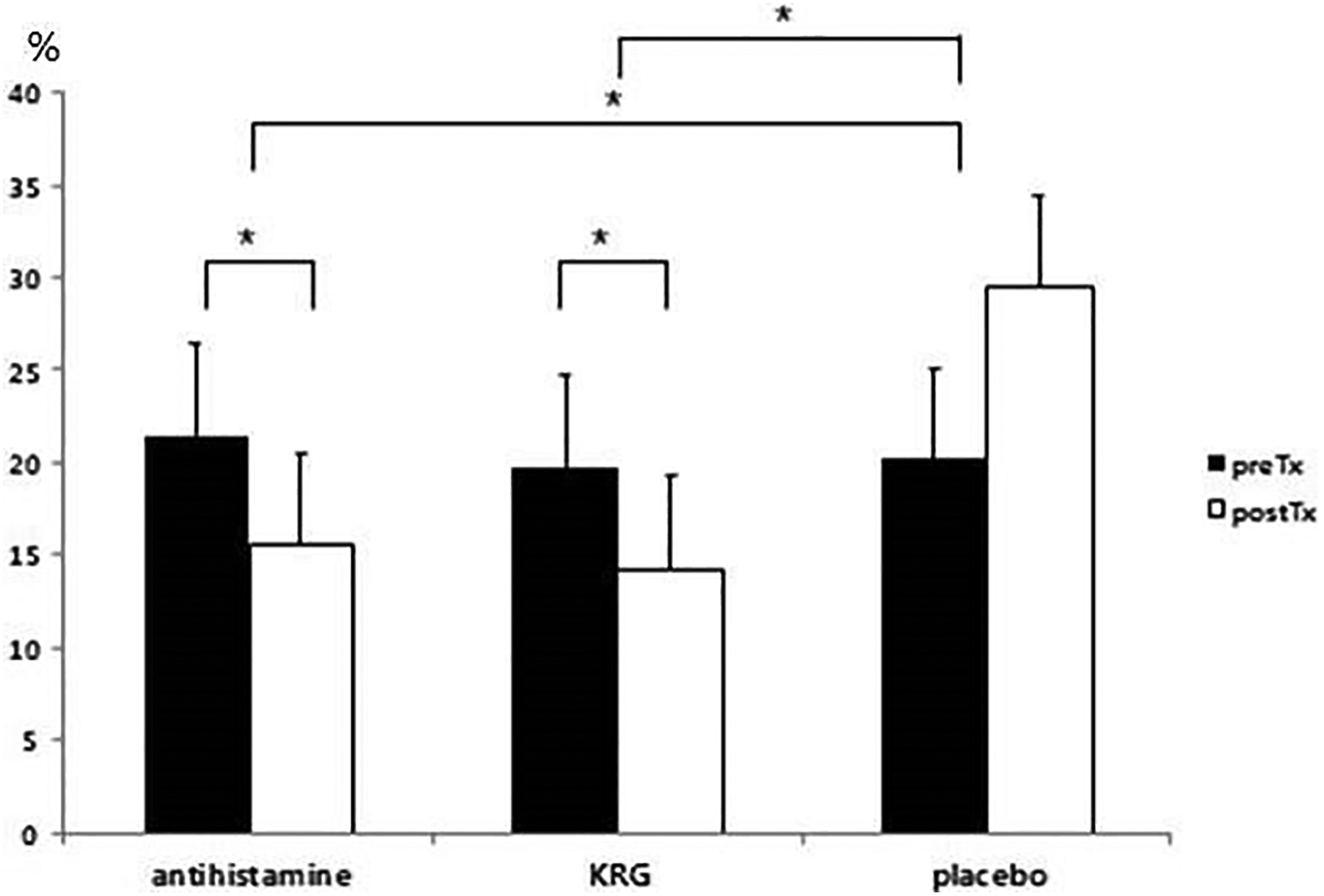
Eosinophil counts in nasal smears. The counts decreased significantly in the antihistamine and the KRG groups. (The t test was used in statistical analyses.) KRG indicates Korean red ginseng.
Discussion
Korean red ginseng is a traditional Korean medicine that appears to have several effects in addition to its anti-inflammatory, anti-allergic, and antitumor properties. Some research indicates that KRG improves cognitive function in healthy people and patients with Alzheimer disease. 14
It also reportedly improves sexual desire, arousal, orgasm, and satisfaction among postmenopausal women. 15 Moreover, patients with erectile dysfunction who were treated with KRG for 12 or 16 weeks scored higher on the 5-item version of the International Index of Erectile Function. 16,17 Ginsenosides reportedly inhibit chronic obstructive pulmonary disease pathogenesis via anti-inflammatory and antioxidative effects. 18 When KRG was administered in an ovalbumin (OVA)-induced mouse model of asthma, airway hyperresponsiveness, immune cell infiltration of bronchoalveolar regions, and inflammatory cytokine production were all reduced. Based on these findings, we decided to evaluate the effects of KRG in patients with allergic rhinitis. 19
We found that KRG was similar to an antihistamine in terms of improving rhinorrhea, nasal and eye itching, and sleep disturbance in patients with allergic rhinitis. Nasal obstruction levels improved slightly in both the KRG and the antihistamine groups, but the changes did not attain statistical significance.
Ginseng treatment reduced eosinophil major basic proteins, IL-5, germ cell number, and mitogen-activated protein kinase levels in bronchoalveolar lavage fluid, cells, and lung tissue of mice with OVA-induced allergic asthma. 20 Previously, we explored the effect of KRG on inflammation of the nasal cavity in a mouse model of allergic rhinitis. The IL-4 and IL-5 levels in nasal lavage fluid and those of eosinophils, IL-4, IL-5, and MUC5AC in nasal mucosa decreased with KRG treatment, indicating that KRG inhibited allergic inflammatory reactions by inactivating Th2 cytokines. However, the effect of KRG on allergic rhinitis has not yet been fully investigated. 11
We found that KRG reduced the serum IL-4 level and increased that of IL-10 in patients with allergic rhinitis. This suggests that KRG modulates allergic inflammation by inhibiting Th2 and activating Th1 cytokines. Fermented red ginseng ingestion for 4 weeks improved the nasal congestion symptoms and rhinitis quality of life scores of patients with perennial allergic rhinitis. However, there was no significant difference in total nasal symptom score (TNSS) or TNSS duration between the experimental and placebo groups. 21 Here, we compared antihistamine and placebo groups with a KRG group. Korean red ginseng was similar to an antihistamine in terms of improving the symptoms of rhinorrhea and nasal and eye itching, suggesting that a 4-week course of KRG was beneficial.
One limitation of our study may be the small size of patients. Additionally, we did not measure the long-term effects of KRG; it is necessary to explore how long the useful effects persist when KRG treatment ceases.
The objective nasal obstruction level measured via peak nasal inspiratory flowmetry did not differ significantly before and after treatment in either the antihistamine or the KRG group. Thus, this flow rate does not reflect the subjective improvements noted in nasal symptoms or patient satisfaction.
In the present study, the total IgE levels decreased significantly in both the KRG and the antihistamine groups. However, the levels of D pterynissinus- and D farine-specific IgEs did not change. The levels of these specific IgEs did not decrease over 1 year of treatment in an immunotherapy group. It would thus not be expected that a 4-week treatment would cause any meaningful change in specific serum IgE levels. Fermented red ginseng exerts antiallergic effects by reducing the serum levels of IgE, OVA-IgE, pro-inflammatory cytokines, β-lactoglobulin, and intestinal barrier–related molecules in OVA-sensitized mice. 22 Ginsenoside Rh1 effectively inhibits histamine release from mouse peritoneal mast cells and IgE-induced passive skin anaphylaxis. 23 Orally administered ginseng extract significantly reduced the increase in OVA-specific IgG levels but did not affect OVA-specific IgE levels in OVA-sensitized mice. 24
Tissue eosinophilia mediated by TH2 cytokine IL-5 is a typical symptom of allergic rhinitis; eosinophilic product releases allergy symptoms. 25,26 The TH2 cytokines IL-4 and IL-5 promote eosinophil recruitment and prolong tissue survival. In our present study, eosinophil counts in nasal smears were significantly reduced in the KRG group. In an allergic mouse model, eosinophil levels decreased in a KRG compared with a control group. Korean red ginseng reduced eosinophil counts in nasal epithelia, suggesting that KRG might prevent the allergy-induced increase in eosinophil recruitment. The principal ginsenosides of KRG are Rg1 and Rb1 (both at 4 mg/g). Rg1 is a ligand of the glucocorticoid receptor, downregulating that receptor, and stimulates the immune response to OVA in BALB/c mice. 27,28 Korean red ginseng can inhibit eosinophil recruitment by the nasal mucosa and can exhibit an antiallergic effect. The contained ginsenosides can exert a controlled glucocorticoid-like effect. 29,30
Safety issues associated with long-term administration of KRG should be considered. A multicenter, double-blind, randomized, placebo-controlled trial, involving 100 patients and conducted over a 24-week period, revealed no significant differences in adverse events between KRG and placebo groups. No serious side effects were observed, and 2 mg/d of KRG for 24 weeks was shown to be safe and tolerable for healthy adults. 31 In previous study, the interaction of KRG with major cytochrome P450 (CYP) enzymes and drug transporter P-glycoprotein (P-gp) was observed after administration of CYP substrate drugs (caffeine, losartan, dextromethorphan, omeprazole, midazolam, and fexofenadine) in healthy adult. Korean red ginseng did not interact with CYP enzymes or P-gp. 32,33
In cases of immune disease, another study found that patients with autoimmune arthritis treated with KRG extract improved in terms of clinical arthritis scores. 34 A previous study reported that administration of KRG in an acute multiple sclerosis model improved spinal demyelination, suggesting that this may be an effective novel treatment for autoimmune disorders including multiple sclerosis. Safety precautions for the administration of KRG to patients with other autoimmune diseases have not yet been determined. 35 A study of 25 patients receiving warfarin after cardiac valve replacement revealed no significant differences in international normalized ratios after treatment with KRG. Administration of KRG to patients receiving this anticoagulant appears to be safe under conditions of close monitoring and appropriate training. 36
We found that KRG exhibited anti-allergic actions similar to those of an antihistamine. Korean red ginseng reduced allergic symptoms and total IgE levels in patients with allergic rhinitis compared with individuals receiving a placebo. Korean red ginseng inhibited Th2 cytokine and eosinophil recruitment, suggesting that KRG might be useful in the treatment of allergic rhinitis. However, our sample was small, and additional, larger studies on the long-term effects of KRG are needed.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a 2012 grant from the Korean Society of Ginseng.
