Abstract
Skull base osteomyelitis (SBO) is an invasive infection of the external auditory canal, with involvement of the skull base, typically in the elderly diabetic population. Diagnosis may be challenging, as it requires a combination of clinical, laboratory, and radiographical findings. The mainstay of treatment is long-term antibiotic therapy, but surgical debridement of the temporal bone may be necessary in refractory cases. Commonly reported complications include cranial neuropathies, meningitis, temporal lobe abscess, and dural venous sinus thrombosis. A rare and life-threatening complication of SBO is petrous internal carotid artery (ICA) blowout, which has been described as presenting with bleeding from the ear. Here, we describe the case of a 77-year-old woman with SBO complicated by a petrous ICA blowout, which presented with fulminant epistaxis. To our knowledge, this is the second reported case of a massive hemorrhage from a petrous ICA blowout secondary to SBO and the first presentation with massive epistaxis. We present this case to raise awareness of this potential impending complication in patients with SBO and recommend consideration of this etiology when assessing patients with massive epistaxis in the appropriate clinical setting.
Introduction
Skull base osteomyelitis (SBO), also known as malignant otitis externa, is an invasive infection of the external auditory canal (EAC) and skull base. This condition typically affects the elderly diabetic population, likely due to the increased pH of cerumen and microangiopathy of the EAC in these individuals. 1 Pathogenesis involves soft tissue infection of the EAC, by Pseudomonas aeruginosa in more than 90% of cases, which then progresses through the fissures of Santorini and tympanomastoid suture to the base of the skull. 1,2 Patients may present with severe otalgia, otorrhea, aural fullness, hearing loss, and given the proximity of the EAC to the stylomastoid foramen, unilateral facial nerve palsy. More advanced infection may lead to temporomandibular joint (TMJ) pain, ipsilateral facial pain, and headache. 3,4 Diagnosis involves a combination of history, physical examination, as well as laboratory and radiographical findings. The mainstay of treatment is long-term antibiotic therapy, typically intravenously for 6 to 8 weeks, followed by 3 to 12 months of oral antibiotics, with some patients requiring surgical debridement or hyperbaric oxygen. 2 Commonly described complications from SBO include cranial neuropathies, meningitis, intracranial abscess, and dural venous sinus thrombosis. 5
A rare and potentially fatal complication of SBO is internal carotid artery (ICA) blowout. Internal carotid artery blowout, typically associated with head and neck cancer, requires emergent endovascular or surgical treatment. Mortality is high, with reported rates between 40% and 63%. 4,5 Here, we present a case of a petrous ICA blowout in a patient with refractory SBO, which is particularly notable given its etiology, as well as its presentation as fulminant epistaxis. Thus, the aim of this project is to alert treating physicians of this catastrophic complication and postulate interventions in the hopes of avoiding this complication.
Case Report
A 77-year-old woman with a past medical history of type II diabetes mellitus, end-stage renal disease, and hypertension presented to the emergency department (ED) with a 2-month history of severe left ear and facial pain. Computed tomography (CT) demonstrated complete opacification of the left mastoid air cells and middle ear cavity with soft tissue thickening and opacification involving the EAC, as well as irregularity along the bony EAC. Additionally, MRI demonstrated soft tissue inflammatory changes along the left TMJ and infratemporal fossa with extension medially toward the nasopharynx (Figure 1), concerning for SBO. Culture of the EAC was notable for few cutaneous flora, and the patient was started on broad-spectrum intravenous (IV) antibiotics and ear drops consisting of ciprofloxacin and dexamethasone via a wick. After considerable improvement in her pain, she was discharged on a prolonged course of IV cefepime.
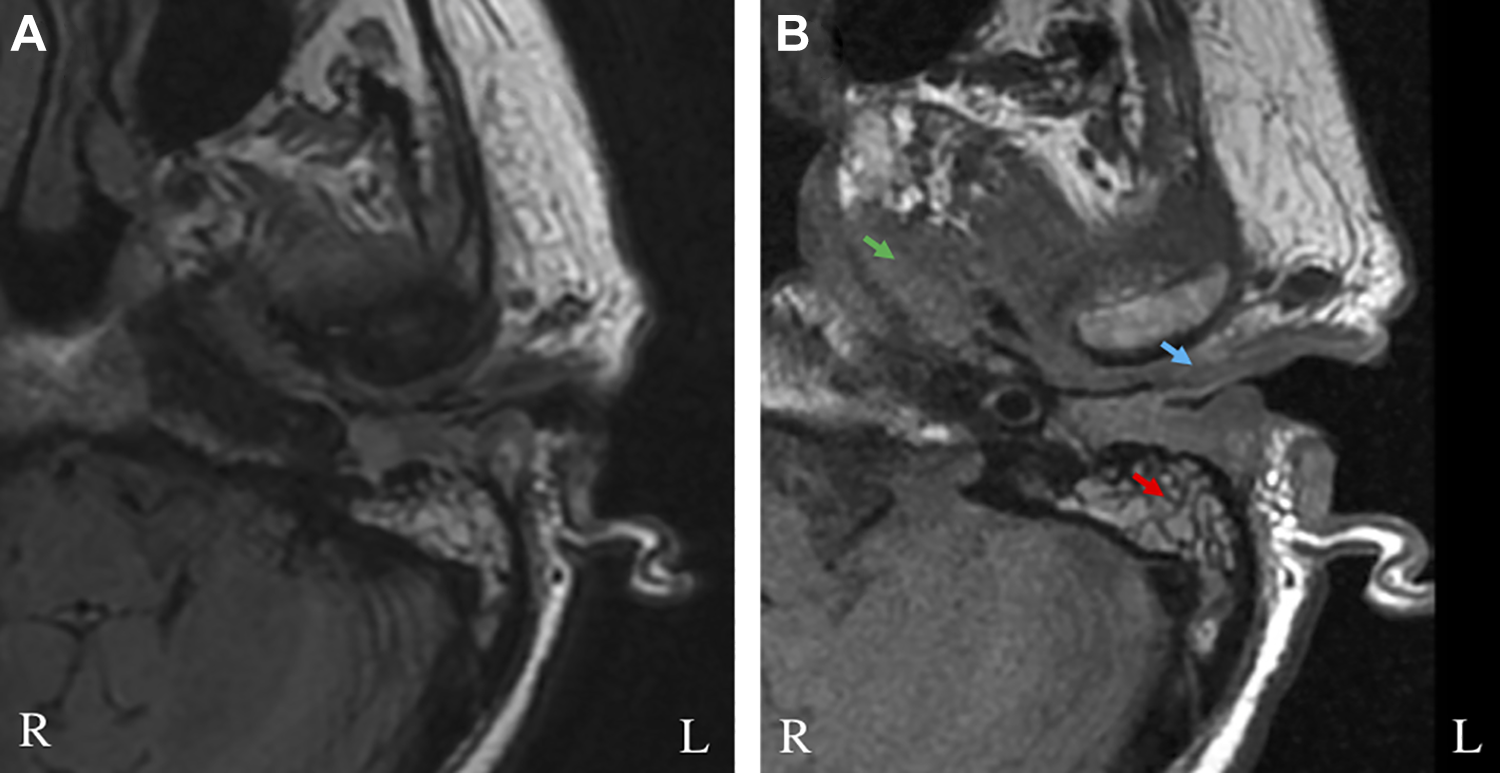
Magnetic resonance imaging without contrast.
Over the next 3 months, the patient presented twice for exacerbations of SBO, each treated with wick placement, ear drops consisting of ciprofloxacin and dexamethasone, and broad-spectrum IV antibiotics with antipseudomonal coverage. At her third presentation for worsening ear pain, a CT temporal bone showed continued opacification of the left EAC, middle ear cavity, and mastoid air cells, with new fullness of the nasopharynx and pterygoid muscles, as well as narrowing of the left ICA and left jugular vein, representing possible arteritis or vasospasm (Figure 2). Due to the need for better tissue diagnosis for culture-directed antibiotic treatment, canal wall up tympanomastoidectomy was performed. Intraoperatively, a severely edematous EAC was noted medially. A large cortical mastoid abscess was also found, which was drained. Intraoperative cultures grew aspergillus and nonanthrax bacillus, and the patient was started on amoxicillin clavulanate, vancomycin, and isavuconazole, with improvement in symptoms.
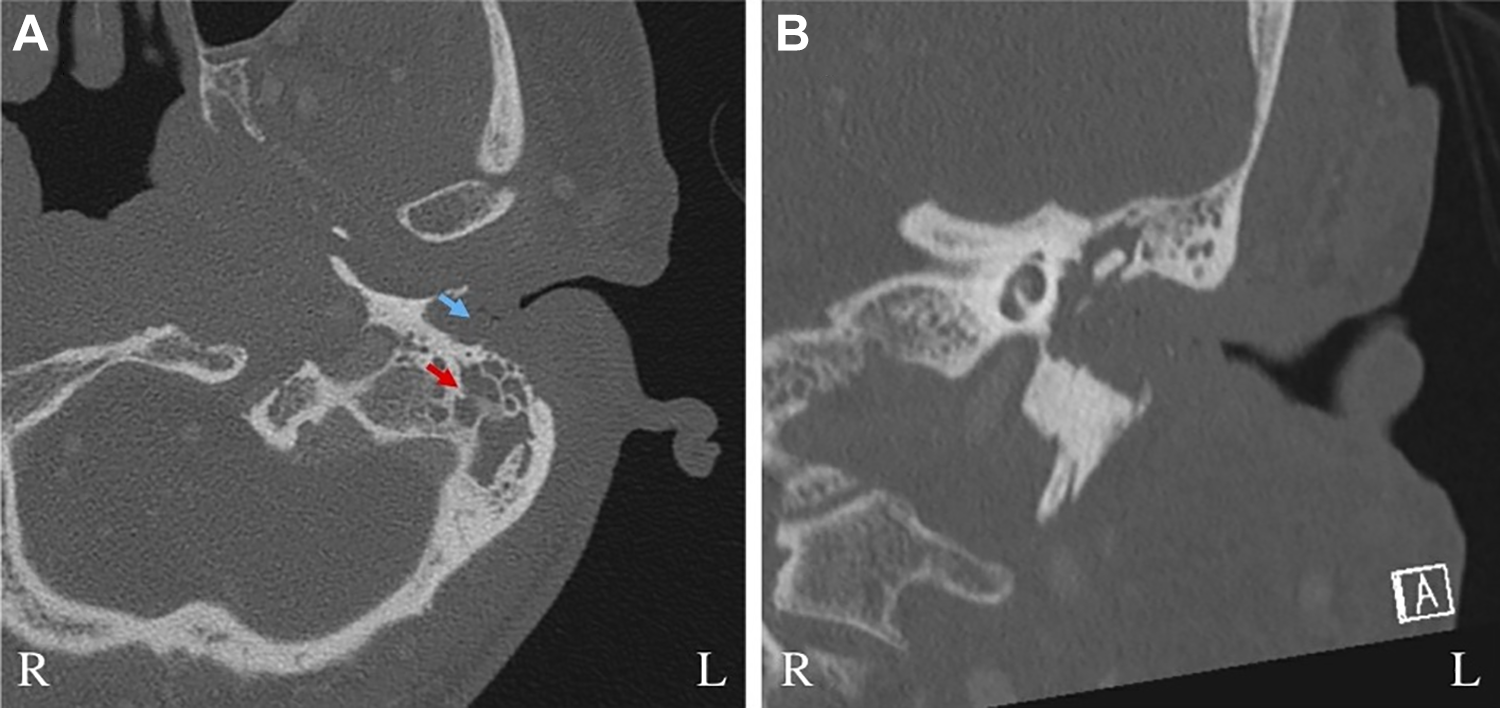
Computed tomography (CT) temporal bone.
Ten days after discharge, the patient presented again to the ED with bilateral epistaxis, which was initially managed conservatively with nasal packing. The following day, she was noted to be profoundly anemic, hypotensive, and lethargic, necessitating admission. She then developed fulminant bilateral epistaxis, requiring emergent intubation for airway protection. During this acute event, copious bright red blood was suctioned bilaterally from the nares, but a clear source was not identified. Bilateral anterior–posterior packing was performed, and the patient was taken emergently for angiography (Figure 3). The source of bleeding was found to be the petrous ICA. After attempted stent-coil placement, the vessel was ultimately glue embolized, with good hemostasis. The packing was subsequently removed, without further bleeding. However, follow-up imaging showed bilateral acute strokes, and with a poor neurologic prognosis, the patient was terminally extubated 2 weeks later.
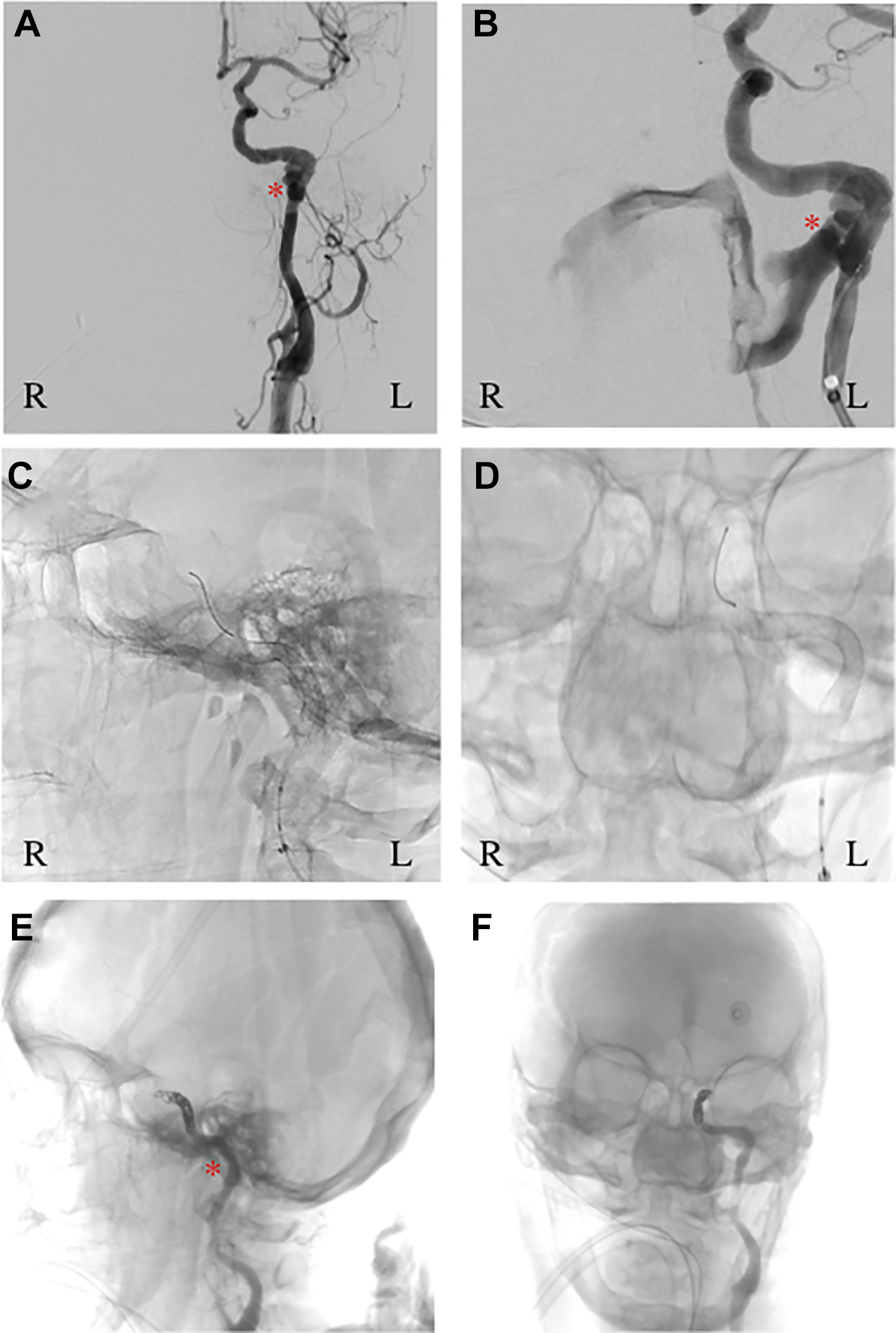
Cerebral angiogram.
Discussion
Here, we present a patient with SBO complicated by a petrous ICA blowout, presenting as fulminant epistaxis. To our knowledge, only 2 other cases of ICA blowout in association with SBO have been reported. The first case described a patient with known SBO who presented with ear and oral bleeding secondary to a large petrous ICA pseudoaneurysm. Despite attempted ICA ligation, the patient succumbed to the illness. 6 In the second case, a tympanomastoidectomy was performed in a patient with known SBO, during which significant pulsation was noted around the carotid canal and eustachian tube. Postoperative CT angiography was performed, which demonstrated a petrous ICA pseudoaneurysm. The patient then underwent endovascular stenting of the pseudoaneurysm and had no further vascular complications. 7
To our knowledge, there has been only one other case of ICA blowout described that presented with massive epistaxis. Specifically, Hsiao et al reported a 71-year-old man with nasopharyngeal carcinoma with a distant history of radiation therapy who presented to the hospital as a result of a minor ischemic stroke. During his hospital course, he developed fulminant epistaxis from a large pseudoaneurysm of the left ICA, which was ultimately underwent endovascular coiling. 8 Our case is the first to describe this presentation of ICA blowout in association with SBO, rather than malignancy.
Epistaxis is commonly seen by otolaryngologists and often treated in the acute inpatient setting without deep investigation into etiology. Moreover, complications of SBO, which is thought to arise from otitis externa, are typically attributed to the ear and thus, epistaxis in these patients may often be treated as an unrelated entity. As with ICA blowout in other clinical settings, our patient presented with what may have been a sentinel bleed, which was initially managed with nasal packing. In retrospect, if bleeding was from the petrous ICA, it is possible that vasospasm, rather than packing, may have controlled the initial bleed. With known SBO and epistaxis requiring packing, early CT angiography, as is often performed in patients with head and neck cancer, may have helped in diagnosis and should be considered in this patient population.
In addition, it is worth considering whether further intervention may have helped to prevent this complication. Reports of circumferential petrosectomy for progressive disease have been described with good results. 5 Our patient presented on multiple occasions with new symptoms and worsening imaging suggesting progressive disease and earlier removal of the infected temporal bone may have aided in recovery. However, most patients with SBO have significant comorbidities, thus making the decision to operate difficult. Moreover, given the diffuse nature of the infection, complete removal of infected bone is often impossible, making it unclear whether further surgery would have been of benefit.
Conclusion
We present this case of SBO resulting in petrous ICA blowout manifesting as fulminant epistaxis to raise awareness of the possibility of this etiology in patients presenting with epistaxis with known skull base disease. We encourage providers to consider a lower threshold for imaging studies, such as CT angiography, in the appropriate clinical setting, in hopes of preventing this life-threatening complication.
Footnotes
Authors’ Note
R.E.W, A.S.P., S.K.G., and C.E.C. contributed to analyzing the case, drafting the article, revising the article critically for important intellectual content, and gave final approval of the version to be submitted.
Acknowledgment
There were no nonauthor contributors to be included in the Acknowledgment section. The subject provided written informed consent to publish their case.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
