Abstract

A 49-year-old man presented to the otolaryngology department of our hospital and reported having intermittent right-side epistaxis and progressive nasal obstruction for 2 months. He reported no headache, facial paresthesia, facial pain, or visual changes. Anterior rhinoscopy revealed a fungating mass partially occupying the right nasal cavity. No enlarged cervical lymph nodes were noted, and other aspects of the physical examination were unremarkable. Nasal endoscopy revealed a well-defined, solitary, friable mass with an irregular surface that easily bled upon contact (Figure 1A). Computed tomography revealed an expansile, homogeneous soft-tissue mass 2 cm in diameter in the middle part of the right nasal cavity without destruction of the surrounding bone structure (Figure 2). Subsequently, the patient underwent transnasal endoscopic resection. Before excision, we confirmed that the mass was anchored to the nasal septum and exhibited no adhesion or invasion to the mucosa of the adjacent middle and inferior turbinate. A frozen section of the tumor was determined to be malignant. Using bipolar cauterization, we performed a wide excision of the stalk of the mass and the surrounding mucoperichondrium of the nasal septum (Figure 1B). Resection margins were negative for carcinoma. Histopathological evaluation revealed solid nests of basaloid cells that exhibited high mitotic rates and necrosis, with foci of cribriform growth (Figure 3A). Myoepithelial differentiation with extracellular matrix formation was also evident. The surface squamous epithelium exhibited severe dysplasia. Immunohistochemical study indicated strong and diffuse p16 immunoreactivity (Figure 3B). The specimen was subjected to a polymerase chain reaction for human papillomavirus (HPV) genotyping and was positive for HPV 33. On the basis of these findings, a diagnosis of HPV-related multiphenotypic sinonasal carcinoma (HMSC) was confirmed. The patient received no postoperative chemotherapy or radiotherapy. However, a small nasal septal perforation developed postoperatively (Figure 1C). Figure 1C to F presents the subsequent nasal endoscopic images captured during follow-up visits. To date, no signs of recurrence or distant metastasis were observed after 18 months of regular follow-up (Figure 1D–F). Written informed consent was obtained from the patient. The Ethics Committee of the Cardinal Tien Hospital approved the case report (CTH-108-3-7-001).
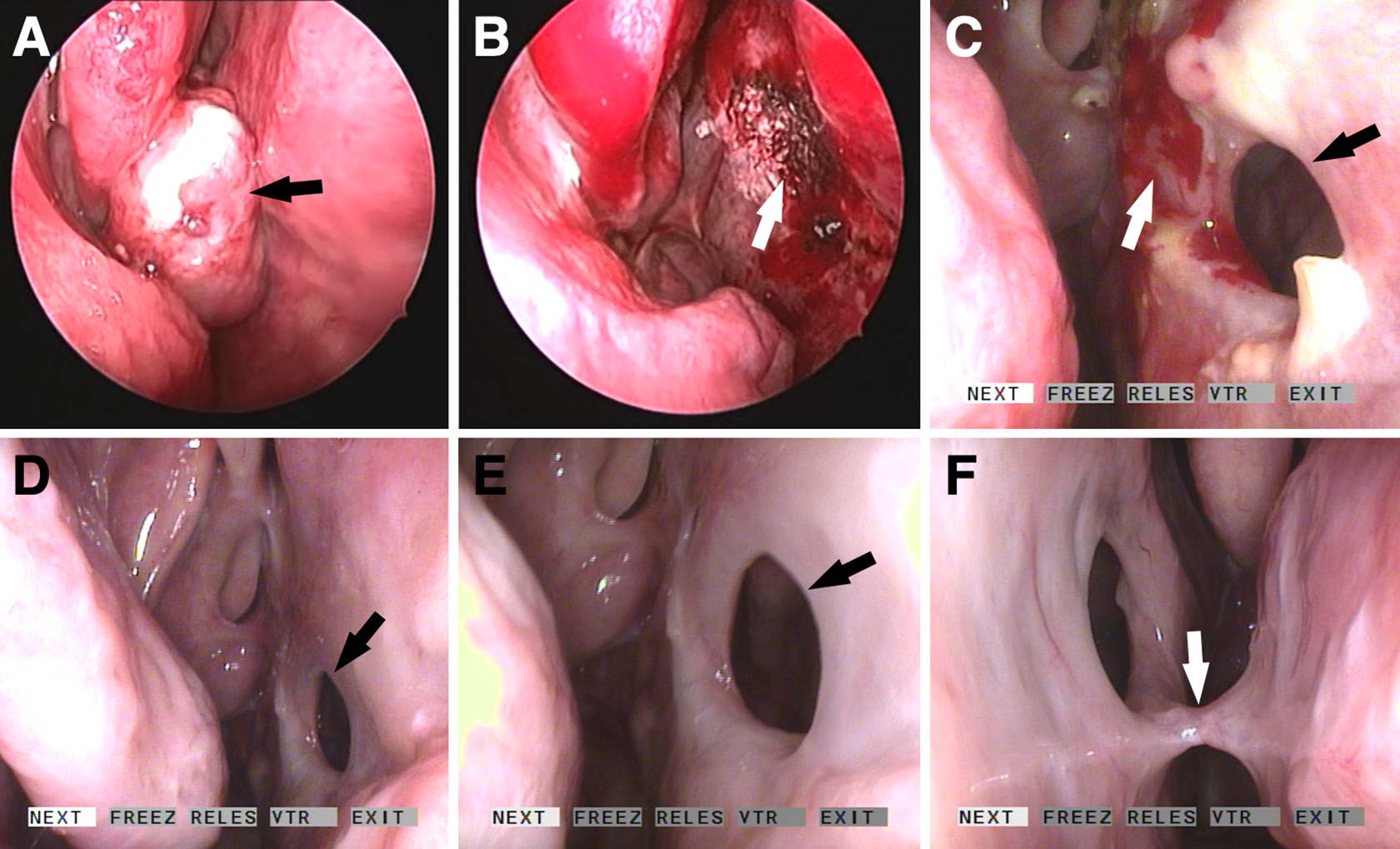
A, Preoperative endoscopic view of a fungated tumor in the right nasal cavity (black arrow). B, The postoperative image of a raw surface after tumor resection (white arrow). C, Image of a septal perforation (black arrow) and crusting (white arrow) 3 months after operation. D to F, Follow-up image revealing no evidence of local recurrence and a persistent septal perforation (black arrow) and contralateral synechia (white arrow) 18 months after operation.
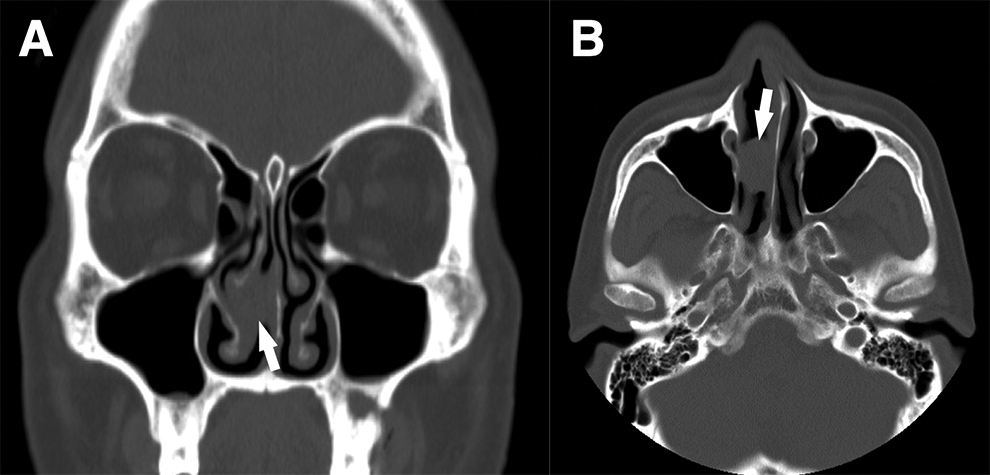
Coronal (A) and axial (B) computed tomography images revealing a homogeneous soft-tissue tumor in the middle part of the right nasal cavity without bone destruction.
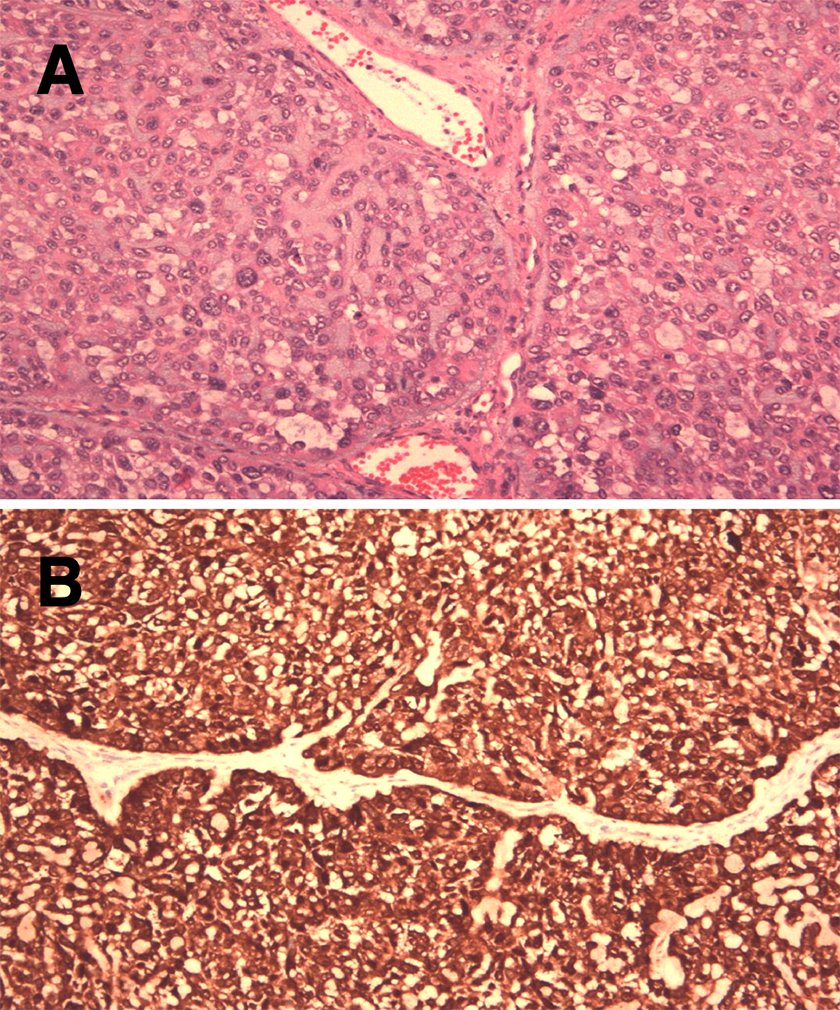
A Solid nests of basophilic tumor cells exhibiting high mitotic rates and necrosis. Myoepithelial differentiation with extracellular matrix formation is evident (×200). B, Highly positive in p16 immunostaining (×200).
Sinonasal cancers account for less than 1% of all cancers and approximately 3% to 5% of all head and neck malignancies. 1 Human papillomavirus is known to play a major role in the oncogenic process, 2,3 and the relationship between HPV and oropharyngeal cancer has recently been confirmed. Up to 80% of oropharyngeal cancer cases are related to HPV type 16, but comprehensive HPV testing of all head and neck cancers has suggested that HPV-related carcinoma might be more common. 4 In 2013, Bishop et al 5 first identified a new subset of sinonasal carcinoma; these carcinomas are HPV positive (mainly type 33) and histologically similar to adenoid cystic carcinoma. Thus, this type of carcinoma was initially named HPV-related carcinoma with adenoid cystic-like features. 6 In response to an increasing number of cases that exhibited considerable histological diversity, Bishop et al 7 suggested a name change to HMSC. To date, more than 60 cases of HMSC have been reported. 8 –12 However, most of the reports have focused on discussing pathological differential diagnosis. 9 –11 We have added to the literature a complete set of recordings of clinical endoscopic images taken before and after successful surgical treatment of this emerging disease entity. Clinically, all cases of HMSC originate from the sinonasal tract. All salivary gland tumors outside the sinonasal tract are HPV negative. 5,7 More women develop this disease than men (57% vs 43%). Presenting symptoms include nasal obstruction, epistaxis, nasal discharge, epiphora, and various ophthalmic symptoms. The age of patients with HMSC ranges from 28 to 90 years. Clinical staging at presentation is variable, but half of the reported cases have presented at an advanced stage (T3 or T4) with a high-grade histological appearance. 7 However, HMSC exhibits a paradoxically indolent prognosis, 13 with frequent and late local recurrence but no lymph node metastasis or distant metastasis. Only one case of very late local recurrence (after 30 years) 14 and one case of tumor-related death has been reported to date. 8 Overall, HMSC has better prognosis than other sinonasal cancers. 6 Surgery is the primary treatment, 15 but some patients undergo postoperative chemotherapy or chemoradiation therapy. 8 For this patient, identifying the attachment of the mass before endoscopic resection was critical. Using bipolar cauterization may have reduced the volume of blood loss. Operating in this manner helps to easily obtain negative margins while minimizing patient morbidity.
Footnotes
Acknowledgments
The authors thank Chiu-Ping Wang and Shu-Hwei Fan for their assistance in preparing this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Cardinal Tien Hospital under grant CTH-108A-2A05.
