Abstract
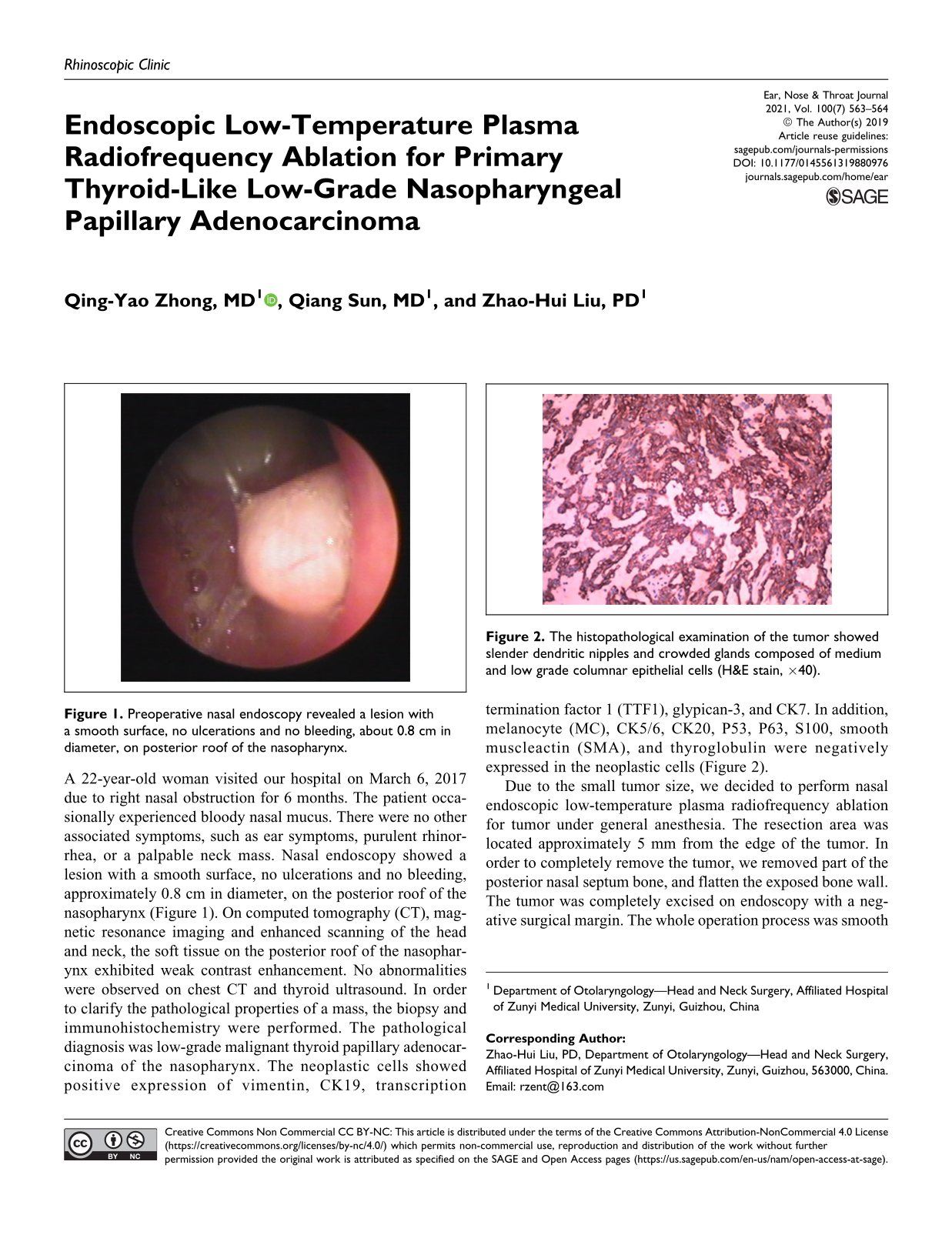
A 22-year-old woman visited our hospital on March 6, 2017 due to right nasal obstruction for 6 months. The patient occasionally experienced bloody nasal mucus. There were no other associated symptoms, such as ear symptoms, purulent rhinorrhea, or a palpable neck mass. Nasal endoscopy showed a lesion with a smooth surface, no ulcerations and no bleeding, approximately 0.8 cm in diameter, on the posterior roof of the nasopharynx (Figure 1). On computed tomography (CT), magnetic resonance imaging and enhanced scanning of the head and neck, the soft tissue on the posterior roof of the nasopharynx exhibited weak contrast enhancement. No abnormalities were observed on chest CT and thyroid ultrasound. In order to clarify the pathological properties of a mass, the biopsy and immunohistochemistry were performed. The pathological diagnosis was low-grade malignant thyroid papillary adenocarcinoma of the nasopharynx. The neoplastic cells showed positive expression of vimentin, CK19, transcription termination factor 1 (TTF1), glypican-3, and CK7. In addition, melanocyte (MC), CK5/6, CK20, P53, P63, S100, smooth muscleactin (SMA), and thyroglobulin were negatively expressed in the neoplastic cells (Figure 2).
Preoperative nasal endoscopy revealed a lesion with a smooth surface, no ulcerations and no bleeding, about 0.8 cm in diameter, on posterior roof of the nasopharynx.
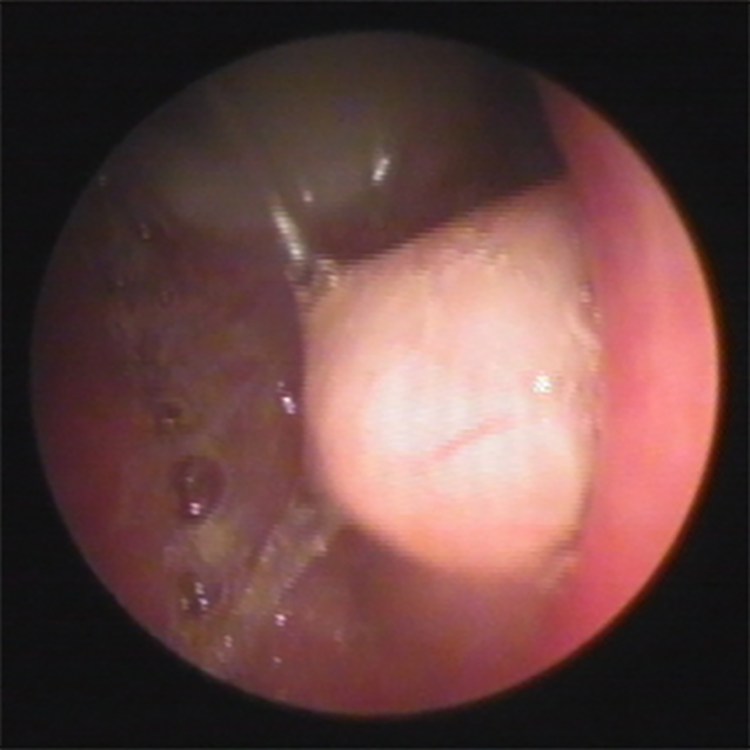
The histopathological examination of the tumor showed slender dendritic nipples and crowded glands composed of medium and low grade columnar epithelial cells (H&E stain, ×40).
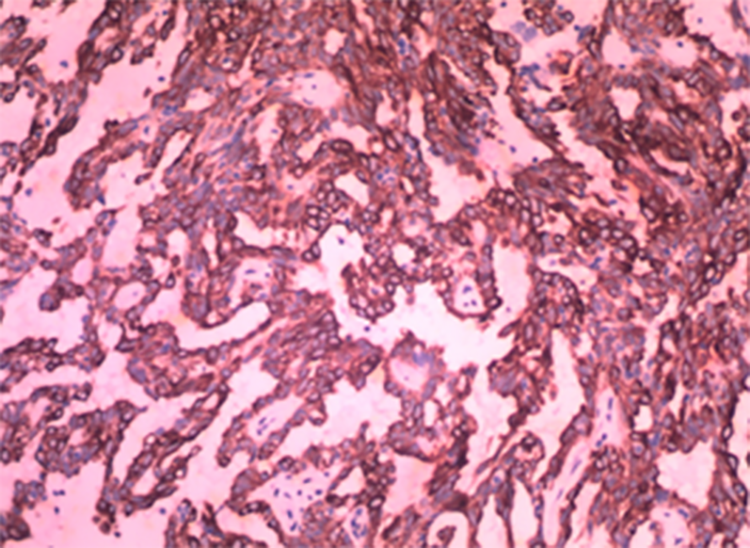
Due to the small tumor size, we decided to perform nasal endoscopic low-temperature plasma radiofrequency ablation for tumor under general anesthesia. The resection area was located approximately 5 mm from the edge of the tumor. In order to completely remove the tumor, we removed part of the posterior nasal septum bone, and flatten the exposed bone wall. The tumor was completely excised on endoscopy with a negative surgical margin. The whole operation process was smooth and the intraoperative bleeding volume was about 10 mL. Besides, no sign of recurrence was observed during the 1-year follow-up period (Figure 3).
Nasal endoscopy 1 year after the surgery revealed the nasopharyngeal mucosa was smooth and there were no new tumors.
Primary thyroid-like low-grade nasopharyngeal papillary adenocarcinoma (TL-LGNPPA) is an extremely rare malignant nasopharyngeal carcinoma originating from the surface mucosal epithelium. 1 As the pathological manifestations of this disease are easily confused with papillary thyroid carcinoma and other nasopharynx cancers, it is currently diagnosed by clinical history, nasal endoscopy, imaging examination, and pathological examination. 2 Nasal congestion, nasal discharge with blood and eustachian tube dysfunction are typical symptoms of TL-LGNPPA, which mainly occur at the top of the nasopharynx or the posterior margin of the nasal septum. In addition, slender dendritic nipples and crowded glands composed of medium- and low-grade columnar epithelial cells are typical pathologic characteristics of TL-LGNPPA. Immunohistochemically, it is well known that the positive expression of TTF-1, CK7, and CKl9 are important diagnostic features for TL-LGNPPA. 3
According to the review literature, patients with TL-LGNPPA have an excellent prognosis with almost no recurrence; thus, surgical resection is currently the better treatment for TL-LGNPPA than radiation and chemotherapy. There is no doubt that endoscopic surgery has great advantages for patients with small nasopharyngeal tumors. 4 In the present case, low-temperature plasma radiofrequency ablation is reported for the first time in the treatment of TL-LGNPPA. As a new technology, the principle of coblation is to use radiofrequency energy to stimulate charged plasma in a conductive medium, so as to generate enough chemically active free radicals. This results in dissolution of tissues at low temperature (40°C-70°C) and avoids damage to normal tissues; therefore, this technique has the advantages of reducing hemorrhage during surgery, relieving patients’ pain, and accelerating postoperative recovery. 5 The total intraoperative blood loss was only 10 mL in our patient.
Footnotes
Authors’ Note
Liu designed the study. Liu, Zhong, and Sun contributed to the surgical treatment and follow-up data collection. Zhong wrote the manuscript. Liu ZH revised the manuscript. All authors read and approved the final manuscript. The patient gave informed consent. Consent was obtained from the patient for publication of this report and any accompanying images.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
