Abstract

Two patients presented to the otolaryngology department of our university with a history of left nasal obstruction. The first case, a 50-year-old man, presented with a 3-month progressively worsening history of left maxillary swelling with mild facial pain and left nasal obstruction. A previous diagnosis of AIDS was reported; tuberous sclerosis was excluded by clinical workup. No alcohol consumption or smoking was reported. Nasal endoscopy revealed the presence of a reddish mass in the middle meatus, arising from the left inferior turbinate (Figure 1A). Narrow band imaging evaluation showed a relatively regular arrangement of intraepithelial capillary; the microvascular network and submucosal veins are nearly invisible (Figure 1B). The patient underwent a maxillofacial computed tomography (CT) scan with contrast that confirmed the presence of a soft density tissue of 13 mm in diameter, occupying the posterior portion of the middle turbinate, without evidence of bony erosion in the medial wall (Figure 1C). The patient underwent an endoscopic resection of the mass. No signs of recurrence of the disease were observed 1 year after surgery.
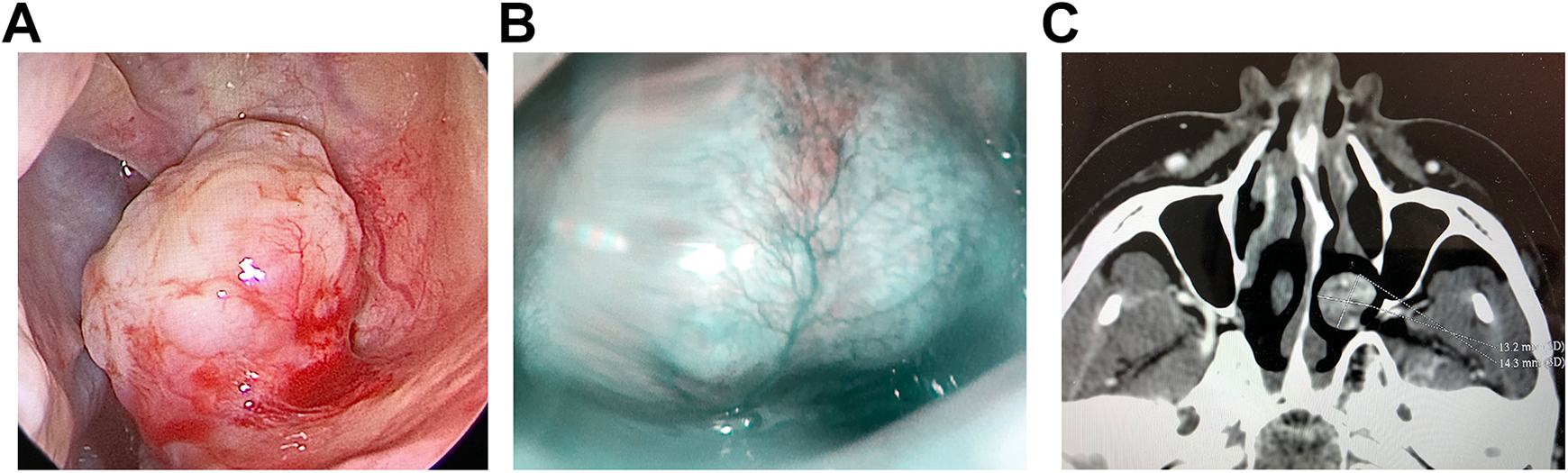
Case 1: Intraoperative nasal endoscopy (A) displaying an exophytic reddish mass in the middle meatus, arising from the left inferior turbinate covered by normal mucosa. Narrow-band imaging (NBI) evaluation (B) in the same patient shows a relatively regular arrangement of intraepithelial capillary. Computed tomography (CT) scan (C) reveals a soft density tissue of 13 mm in diameter, occupying the posterior portion of the middle turbinate, without evidence of bony erosion in the medial wall.
The second case, a 63-year-old male, presented with left-side nasal obstruction and rhinorrhea. Anterior rhinoscopy revealed in the left nasal fossa a smooth spherical nasal mass arising from the head of the left inferior turbinate, without bleeding on touch. Contrast-enhanced CT scan showed a soft density tissue, arising from the head of the left inferior turbinate, without evidence of bony erosion (Figure 2). These clinical features were suggestive of a benign tumor; thus, a complete excision with nasal endoscopy was performed. No signs of recurrence of the disease were observed 5 months after surgery.
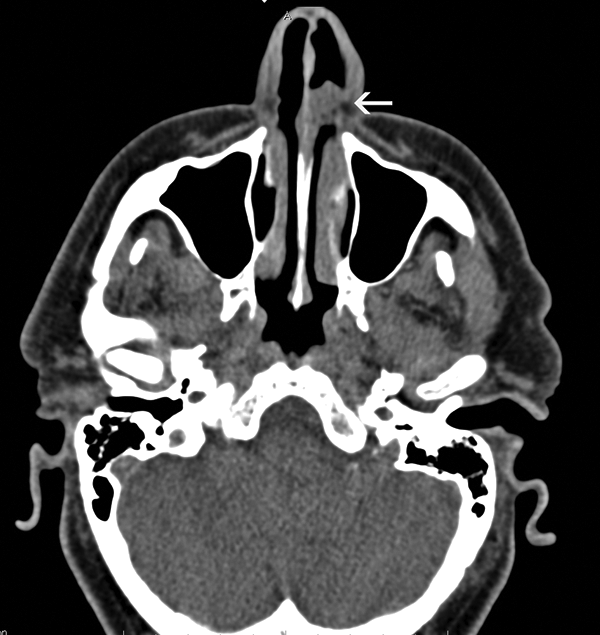
Case 2: Computed tomography (CT) scan shows a soft density (white arrow) tissue, arising from the head of the left inferior turbinate. No evidence of bony erosion is observed.
Both excised tumors consisted of vascular channels surrounded by thick muscular coats associated with intersecting fascicles of spindle cells. Cavernous-like vessels were also recognized in case 1 and aggregates of lymphocytes in case 2. Of note, mature adipocytes were identified in both tumors. While in case 1 they formed large aggregates, in case 2 they were distributed either singularly or in very small clusters. In both tumors, mitosis, pleomorphism, and necrosis were absent, and immunohistochemical analysis performed as described previously 1 demonstrated positivity of the spindle cells for smooth muscle actin and muscle-specific actin and negativity for S100, HMB45, MelanA, estrogen receptor (ER) and progesterone receptor (PR); the proliferative activity (Ki67) was less than 3%, and Epstein-Barr Encoding Region (EBER) in situ hybridization was negative (Figure 3). Based on these findings, the pathologic diagnosis of sinonasal-ALM with adipocytic differentiation 2 was made.
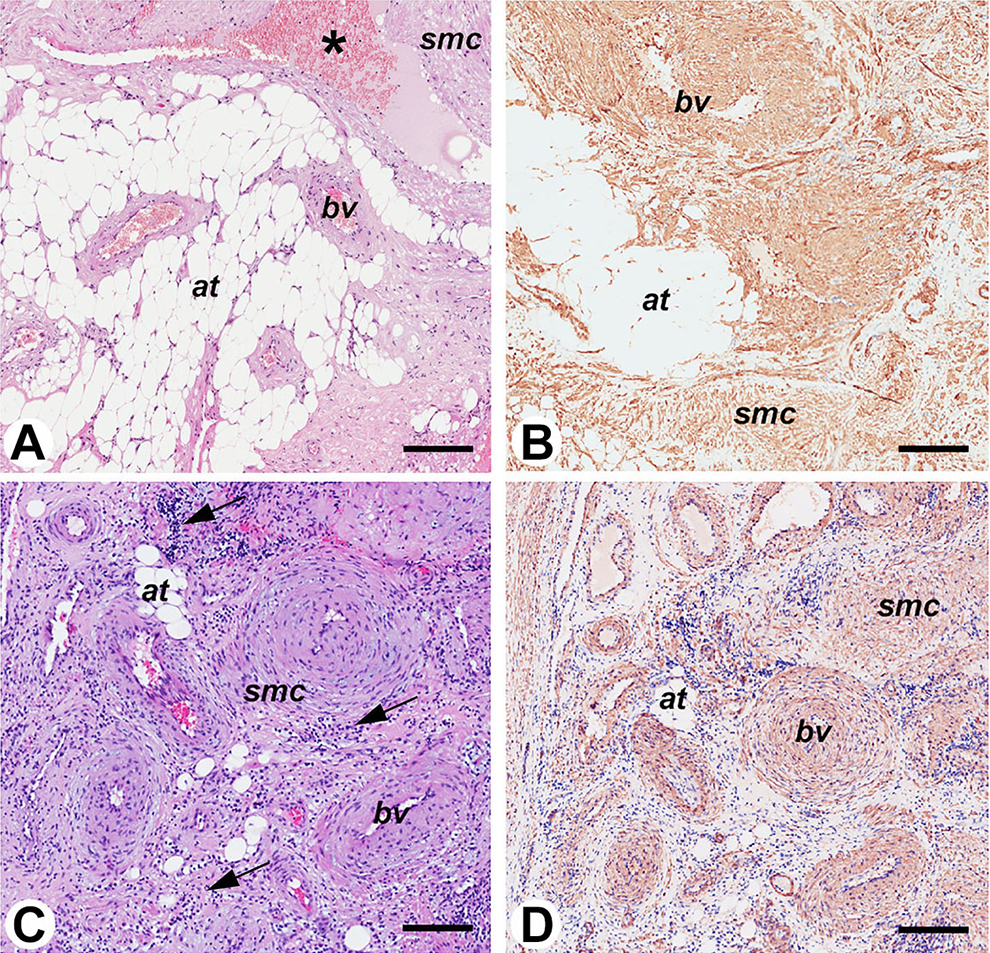
Pathologic features of the tumors. Case 1: A and B. Case 2: C and D. Both tumors (A and C) consist of blood vessels (bv) surrounded by thick muscular coats, smooth muscle cells (smc), and mature adipose tissue (at). In case 1, blood vessels are encircled by the aggregates of adipocytes and, focally, dilated (asterisk in A). In case 2, aggregates of lymphocytes are also present (arrows in C). The abundant amount of smooth muscle cells is highlighted by the immunohistochemical stain for smooth muscle actin (B and D). Bar: 200 μm; A and C: hematoxylin and eosin.
The occurrence of mature adipocytes in an otherwise typical sinonasal-ALM is not uncommon. Indeed, the term angiomyolipoma has been inappropriately used to describe typical sinonasal-ALM. However, angiomyolipoma is a completely different entity, since lesional cells co-express melanocytic (mainly HMB45) and myogenic markers. In analogy with the rare cutaneous and soft tissue counterpart, the previously suggested term of sinonasal-ALM with adipocytic differentiation (or alternatively the more descriptive term angiolipoleiomyoma) seems appropriate to identify the ALMs involving the sinonasal region that contains mature adipose tissue.
Since recent data indicate that ER and PR may be expressed by the smooth muscle cells in sinonasal-ALM, 3 immunohistochemical stains were performed for both receptors, but the result was negative. In addition, since in particular in the context of immunodeficiency smooth muscle tumor may be related to Epstein Barr Virus (EBV) infection, 4 EBER in situ hybridization was performed on both tumors, but the result was negative. Although our results overlap that obtained in other studies, 1,5 the role of ER and PR and EBV infection in sinonasal-ALM requires further studies on larger series of cases.
It has been estimated that sinonasal-ALMs represent approximately 10% of head and neck and no more than 1% of all ALMs. 2 Sinonasal-ALM arises most frequently from the turbinate and consists of a slow-growing sessile or polypoid well-circumscribed nonencapsulated mass. Nasal obstruction and epistaxis are the most common presenting symptoms. Complete surgical excision is the recommended treatment. 2 Size, location, and extension of the tumor, as also the experience of the surgeon, are the main factors that influence the surgical approach. In majority of cases, transnasal endoscopic excision is the treatment of choice, since the majority of the tumors are limited to the nasal cavity. Preoperative embolization has been considered for large tumors with abundant vascular supply or a prominent vascular pedicle at the base. While recurrence of sinonasal-ALM has been rarely described, likely as a result of an incomplete primary excision, malignant transformation has never been reported.
Footnotes
Authors’ Note
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
