Abstract

Clinical Report
A 63-year-old female with a painless progressive enlarging mass on her right parotid region with 1 year of history admitted to our otorhinolaryngology clinic. Physical examination revealed solid, fixed mass approximately 6 cm in size located in the parotid gland. There was no sign of facial nerve involvement. All other examinations of the head and neck including the nasopharynx were normal. Blood tests revealed nothing remarkable. Parotid ultrasonography reported a 67 mm × 41 mm well-capsulated hypoechoic heterogeneous mass located at the deep portion of the parotid gland. Magnetic resonance imaging illustrated a mass that was isolated from adjacent structures with heterogeneous intensity and having no invasion to the peripheral tissues (Figure 1A and B). Fine needle aspiration biopsy revealed malignancy suspicion and total excision of the mass was advised for accurate diagnosis.
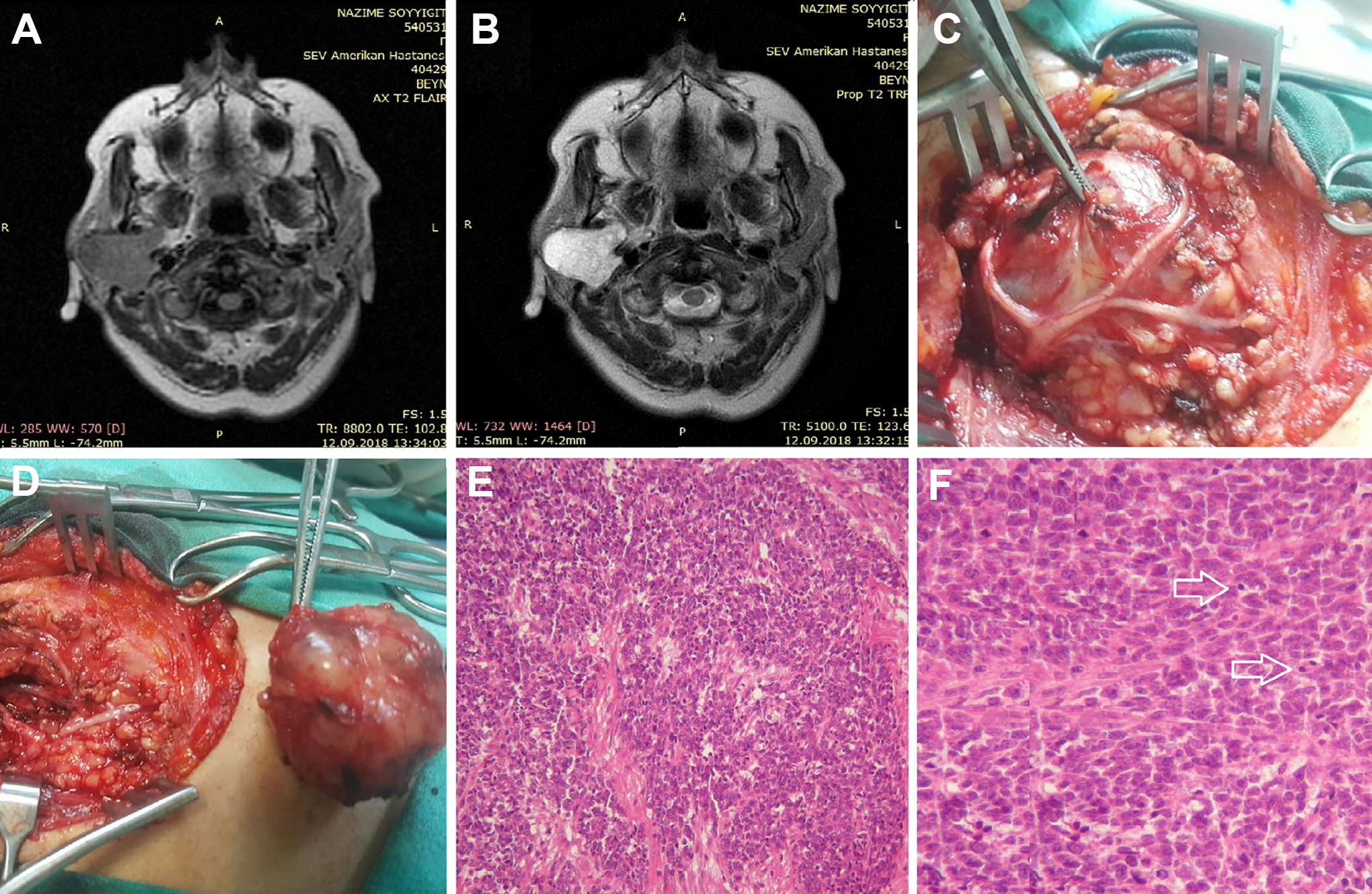
A-F, Magnetic resonance imaging, T2 axial section, shows a well-isolated mass without infiltration to peripheral tissues (A), MRI, contrast-enhanced T2 axial section, shows a parotid gland mass with heterogeneous density (B), a mass surrounded by branches of the facial nerve (C), an intraoperative view of totally excised mass (D), hematoxylin and eosin staining of the tumor under low-power (×100) microscopic field (E), hematoxylin and eosin staining of the tumor under high-power (×400) microscopic field. Arrows show tumor cells with round nuclei and deficient cytoplasm (F). MRI indicates magnetic resonance imaging.
The mass displaced the facial nerve laterally without invasion (Figure 1C). Following the superficial parotidectomy, the mass was completely removed with meticulous facial nerve dissection (Figure 1D). Hematoxylin and eosin staining of the tumor indicated that tumor composed of cells with round nuclei and deficient cytoplasm that lack differentiation significantly (Figure 1E and F). Mitotic figures and areas of necrosis were also observed in the histopathological examination. The preliminary examination was reported to be malignant, and further immunohistochemical evaluation was required for an accurate diagnosis. Immunohistochemical markers including synaptophysin, chromogranin, CD99, CD20, CD3, CAM52, S100, myoD1, myogenin, pan-cytokeratin, ttf-1, CK20, Desmin, Vimentin, FLI1, Ki-67, and leukocyte common antigenm (LCA) were applied. The specimen was negative with LCA, pan-cytokeratin, myogenin, CD3, CD20, myoD1, and desmin. The immunohistochemical staining revealed that the sample was strongly positive for CD99 (cluster of differentiation 99) antigen also known as MIC2 protein and Vimentin (Figure 2). MIC2 is a protein encoded by the human CD99 gene which found on the cell surface of Ewing sarcoma (ES)/primitive neuroectodermal tumor (PNET) family of tumors. 1 In some areas, the Ki-67 proliferative index was observed between 30% and 50%. To detect ESWR1-FLI1 rearrangement, the sample was sent to molecular pathology and fluorescence in situ hybridization (FISH) revealed ESWR1-FLI1 rearrangement. Afterward, the whole-body positron emission tomography (PET) scan was performed to confirm whether the tumor is primary or metastatic, and there was no evidence of tumor at other sites. Finally, the exact diagnosis was reported as primary extraskeletal ES of the parotid gland by evaluating histopathological–immunohistochemical staining pattern in conjunction with FISH test.

Immunohistochemical staining shows CD99 positivity in tumor cells.
The patient was referred to the oncology department, and the patient was treated by adjuvant chemotherapy and radiotherapy. After chemotherapy and radiotherapy were completed, the local recurrence and distant metastasis were investigated by PET scan. The whole-body PET scan revealed no evidence of local recurrence or any distant metastases. At the end of the 18-month follow-up period, no recurrence of disease observed.
Ewing sarcoma/PNET is difficult to discriminate from other small round blue cell tumors that exhibit similar histologic findings including neuroblastoma, rhabdomyosarcoma, mesenchymal chondrosarcoma, and poorly differentiated synovial sarcoma. Immunohistochemical staining of the MIC2 transmembrane protein with CD 99 antigen is helpful in differentiating this tumor from other small round-cell tumor members. 1 Nevertheless, CD 99 antigen expression is not exclusive for ES/PNET, as it might be expressed by other types of sarcomas, such as mesenchymal chondrosarcoma and poorly differentiated synovial sarcoma and lymphoblastic lymphoma. We excluded the other possible diagnoses by performing FISH for ESWR1-FLI1 rearrangement. The EWSR1/FLI1 fusion gene (t(11;22)(q24;q12)) is observed in approximately 85% of ES/PNET family of tumors. 2 Rare other translocations (t(21;22)(q22;q12), t(7;22)(p22;q12), t(17;22)(q12;q12), and t(2;22)(q33;q12)) fusing the EWSR1 gene with other genes that are related to the FLI1 gene may also cause ES/PNET. 2
Ewing sarcoma/PNET are aggressive malignant tumors with a high potential of distant metastases and local recurrence. On the other hand, the prognosis of ES/PNET appears to be favorable. An overall 5-year survival rate of ES/PNET is reported to be 71%. 3 Despite our follow-up duration (18 months) is relatively short as compared to those reported in the literature, we did not observe any local recurrence or distant metastasis during the follow-up period. Although the ES/PNET family of tumors are radiosensitive malignancies, radiotherapy alone does not appear to be a curative treatment option. Tumor resection with safe margins in combination with chemotherapy and radiotherapy is essential for a good prognosis. 3
To the best of our knowledge, the authors reported the first case of an extremely atypical presentation of primary extraskeletal ES originating from the parotid gland in the English literature. In conclusion, although primary extraskeletal ES of the parotid gland is an extremely rare clinical entity, it should be taken into consideration in the differential diagnosis of a progressive enlarging painless masses located at the parotid gland in adults.
Footnotes
Acknowledgments
The authors would like to acknowledge Dr Sevgul Kara, MD, for her invaluable contributions as editing intellectual content of this paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
