Abstract
Ototoxicity is the general name of cochlear and vestibular organ injury resulting from encountering various therapeutic agents and chemical substances. Cisplatin is commonly used in the treatment of many cancers. In this study, the efficacy of intratympanic steroids was compared for preventing cisplatin ototoxicity. In this study, 32 (64 ears) rats were used by separating into 4 groups. Cisplatin was administered intraperitoneally to the first group (n = 8). Methylprednisolone and then cisplatin were administered intratympanically to the second group (n = 8). On the third group (n = 8), dexamethasone and then cisplatin were administered intratympanically. To the fourth group (n = 8), 0.9% NaCl and then cisplatin were given intratympanically. Otoacoustic emission (OAE) measurements and auditory brainstem responses (ABRs) tests were performed on all groups before and 72 hours after the procedure. Pretreatment of ABR-IV values were 4.29 ± 0.19 milliseconds in group 2 and 4.27 ± 0.16 milliseconds in group 3, whereas posttreatment ABR-IV values were 4.95 ± 0.35 milliseconds in group 2 and 4.65 ± 0.26 milliseconds in group 3. The ABR-IV values were measured significantly shorter in the rats given dexamethasone and methylprednisolone, according to control and cisplatin groups (P < .001). Pretreatment of ABR I-IV interval values were 2.98 ± 0.34 milliseconds and 3.03 ± 0.42 milliseconds in group 1 and group 4, respectively, and ABR I-IV interval values in group 1 and group 4 posttreatment were 3.49 ± 0.39 milliseconds and 3.5 ± 0.39 milliseconds in group 1 and group 4, respectively. Auditory brainstem responses I-IV interval was significantly longer in the cisplatin and control group than in the rats given dexamethasone and methylprednisolone (P < .001). After cisplatin treatment, OAE amplitudes decreased significantly in group 1 and group 4 for all frequencies, while OAE values were protected in methylprednisolone and dexamethasone group (P < .001). In conclusion, it has been shown that both agents have protective effects on cisplatin ototoxicity, with dexamethasone slightly more than methylprednisolone.
Introduction
Ototoxicity is the cellular degeneration and dysfunction that may be even permanent, caused by various medications and chemicals in the cochlea and vestibular organ. 1,2 Cisplatin is a commonly used antineoplastic agent and peculiar with its highly toxic effects on inner ear. Cisplatin (cis-diamminedichloroplatinum) is mainly used in adults and children in the treatment of many malignant diseases, including especially head and neck tumors, urogenital system, central nervous system, respiratory system, and esophageal cancers. 3 Cisplatin-dependent ototoxic effect occurs mainly via DNA damage, reactive oxygen species, ion channel blockage, and lipid peroxidation. It causes hyperpolarization and auditory threshold elevation in hairy cells by blocking the ion transition channels on the membranes of outer hairy cells. 4 Another mechanism of cisplatin ototoxicity involves the formation of reactive oxygen species in the cochlea. Free radical formation occurs as a result of decreased levels of intracellular glutathione and thus changes in the antioxidant enzyme activity. The deterioration in the antioxidant defense system causes an increase in lipid peroxidation and thereby leads to apoptosis in hairy cells, support cells, stria vascularis, and nerves. 5 The clinical picture appears as irreversible, bilateral, sensorineural hearing loss, which initially involves high-frequency sounds and then low-frequency sounds including speech sounds. The audiological revealing of the ototoxicity could be performed by audiometric examinations, distortion product otoacoustic emission (DPOAE) and auditory brainstem response (ABR). There is no standard treatment for preventing cisplatin-related ototoxicity today.
Steroids are one of the medications that have been investigated for the prevention of cisplatin ototoxicity. 6 Steroids were shown to limit the effect of reactive oxygen species in the inner ear. 7 Intratympanic steroids are widely used to prevent the side effects related to the systemic steroids and to achieve a higher dose of perilymphatic medication concentration. Transtympanic medication applications are used in the treatment of sudden hearing loss, Meniere disease, sensorineural hearing loss, and autoimmune inner ear diseases. In this study, the efficacy of intratympanic steroids was compared to DPOAE and ABR in order to prevent cisplatin ototoxicity.
Materials and Methods
Animal Care and Treatment
This study was carried out with the ethics committee approval of Experimental Animal Research Center of Kırıkkale University. A total of 32 female, adult, healthy, 3-month-old Albino-Wistar rats (64 ears) were used in our study. The rats were rested for 48 hours in the appropriate temperature and humidity after they were moved, in order to adjust their new environment. Rats were kept in an environment in experimental Animal Research Center where the temperature was 22°C ± 2°C, humidity 65% to 70%, with 12-hour light/12-hour dark, and a free access to food and water, in addition to medication application times. External and middle ear examinations of the rats were performed under anesthesia with the otoscopic examinations. Ears with plugs were cleaned, and rats with infection in the external auditory canal, opacification, and perforation in the tympanic membrane and those with an infection in middle ear were excluded from the study.
Anesthesia and Drug Application
Animals were randomized into 4 groups by the simple randomization technique. Eight experimental animals (rats) were used in each group. Groups were formed as group 1 (cisplatin group, n = 8), group 2 (methylprednisolone group, n = 8), group 3 (dexamethasone group, n = 8), and group 4 (control group, n = 8). All rats underwent general anesthesia with 60 mg/kg intraperitoneal (ip) ketamine hydrochloride (Ketalar, Eczacibasi Parke-Davis, Istanbul, Turkey) and 10 mg/kg ip xylazine HCl (Alfazyn, Alfas International B.V., Woerden, The Netherlands) before the procedures. In group 1, 15 mg/kg ip cisplatin (Cisplatin DBL, Hospira Australia Pty Ltd, Victoria, Australia) was administered via slow infusion. In group 2, 62.5 mg/mL methylprednisolone ampoule was applied with a dose of 0.05 mL intratympanically to both tympanic membranes of the rats under the microscope. In group 3, 4 mg/mL dexamethasone ampoule was applied with a dose of 0.05 mL intratympanically to both tympanic membranes of the rats under the microscope. In group 4, 0.9% NaCl was administered with a dose of 0.05 mL intratympanically to both tympanic membranes of the rats under the microscope. In groups 2-4, 15 mg/kg ip cisplatin was given 30 minutes following the medication administration. The doses of medications were established based on previous studies. 8 -10
Auditory Brainstem Response Measurements
Auditory brainstem response recordings were taken with the Interacoustics Eclipse EP15 device (Interacoustics A/S, Middelfart, Denmark). Measurements were made before and 72 hours after the medication administration. Newborn ear probes were inserted into the ear from the external ear canal of the measured side. Subdermal stainless-steel monops needle electrodes were placed on vertex (positive), mastoid region (negative), and dorsum (earth). Impedances were checked to ensure that the impedance of the electrodes was less than 2 kΩ. Stimulations were produced in the first 10 milliseconds and all clicks were filtered (100-3 kHz). Stimulation level started at 11 pps from 100 dB hearing level and reduced by 10 dB every step. Hearing threshold was defined as the visible, reproducible ABR produced at the lowest stimulation intensity. An average of 1500 click/stimulus was applied for all levels. The ABR-I, ABR-IV, and ABR I-IV interval and threshold values were used in the measurements.
Distortion Product Otoacoustic Emission Measurements
Distortion product otoacoustic emission recordings were taken with the Otodynamics OAE System device (Otodynamics Ltd, Hatfield, United Kingdom). Measurements were made before and 72 hours after the medication administration. With the probe used for DPOAE, pure sound stimuli at 2 different frequencies (f1 and f2) were given simultaneously, and the strongest emission in the cochlea was found with the formula 2f1 − f2. These acoustic responses were obtained via the microphone inside the probe. The frequencies of 1416, 2002, 2832, 4004, and 5652 kHz were measured in DPOAE. The procedures were made in a quiet environment.
Results
Both ABR and DPOAE were performed to all groups before and after the procedure. The ABR-I, ABR-IV, and ABR I-IV interval and threshold values were analyzed in the ABR test. As for in DPOAE test, the measurements were made in the frequencies of 1416, 2002, 2832, 4004, and 5652 kHz. There was no significant difference between pretreatment ABR values, ABR I-IV interval, and ABR threshold values of the groups (Table 1, P > .05). Auditory brainstem response-I values did not differ between groups after cisplatin administration. Auditory brainstem response-IV values were significantly shorter in rats treated with dexamethasone and methylprednisolone. Auditory brainstem response I-IV interval was significantly longer in cisplatin and control groups. Even though the protective effect of dexamethasone on ABR-IV and ABR I-IV interval was slightly more notable than methylprednisolone, the difference was not statistically significant. Threshold values were 25 (25-30) dB in pretreatment methylprednisolone group and were 60 (55-70) dB after the treatment. Threshold values were 25 (20-30) dB in pretreatment dexamethasone group and were 60 (50-60) dB after the treatment. The threshold values were found to be protected with the methylprednisolone and dexamethasone treatment when compared to the control and cisplatin groups. Cisplatin treatment was determined to significantly prolong the ABR-I and ABR-IV values in all groups when ABR test changes were evaluated before and after treatment in each group (P < .001; Table 1). Threshold values were found to increase significantly in all groups under the cisplatin effect (P < .001).
Comparison of ABR and Threshold Values Before and After Treatment Within Groups.
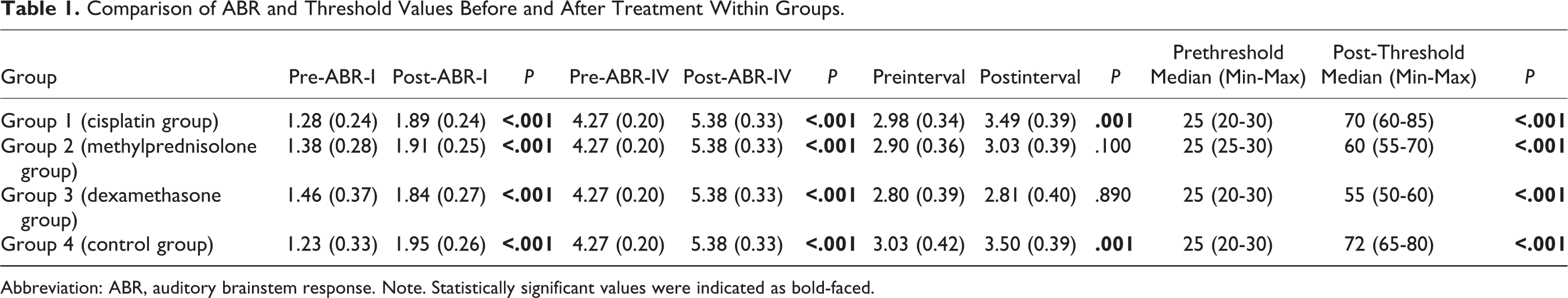
Abbreviation: ABR, auditory brainstem response. Note. Statistically significant values were indicated as bold-faced.
All groups were similar in terms of OAE values before the cisplatin treatment (P > .05). After cisplatin effect, OAE amplitudes decreased significantly in group 1 and group 4 for all frequencies, and OAE values were maintained in rats treated with methylprednisolone and dexamethasone (P < .001; Table 2). In the dexamethasone group, while pretreatment OAE values were 18.70 ± 1.59 dB at the frequency of 2832 kHz, 19.73 ± 1.93 dB at 4004 kHz, and 25.72 ± 2.86 dB at 5652 kHz, posttreatment values were 16.48 ± 2.03 dB at the frequency of 2832 kHz, 18.88 ± 2.96 dB at 4004 kHz, and 21.33 ± 2.40 dB at 5652 kHz. As in the methylprednisolone group, while pretreatment OAE values were 17.29 ± 2.03 dB at the frequency of 2832 kHz, 21.1 ± 1.42 dB at 4004 kHz, and 25.69 ± 2.81 dB at 5652 kHz, posttreatment values were 13.81 ± 1.09 dB at the frequency of 2832 kHz, 15.99 ± 1.82 dB at 4004 kHz, and 18.6 ± 1.398 dB at 5652 kHz. The protective effect of dexamethasone at 2832, 4004, and 5652 frequencies was significantly more notable than methylprednisolone. When the changes of OAE amplitudes in different frequencies were examined within the groups, it was found that cisplatin decreased OAE amplitudes in all frequencies and that this decrease was statistically significant in all frequencies, except 4004 Hz.
Comparison of DPOAE Values Before and After Treatment Within Groups.
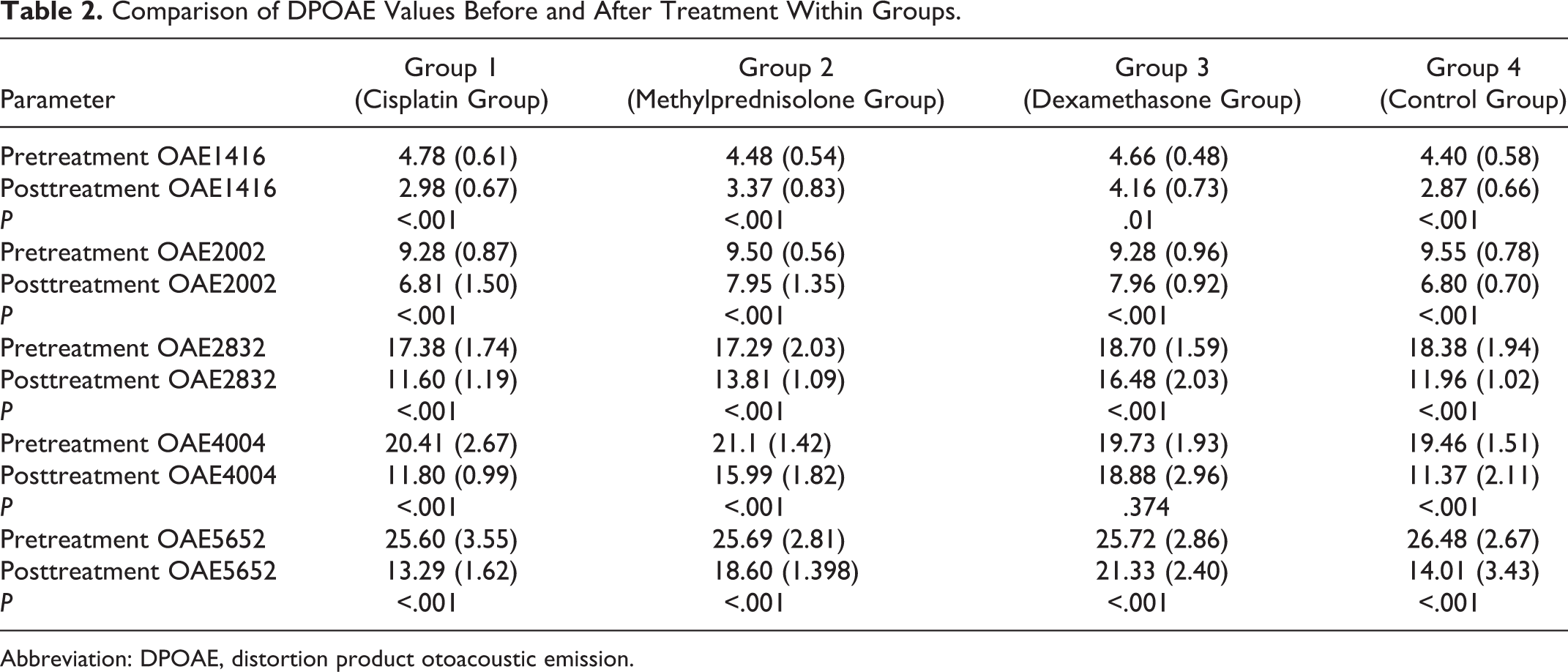
Abbreviation: DPOAE, distortion product otoacoustic emission.
Statistical Analysis
The data corresponding to the normally distribution were represented as mean (standard deviation), but the data corresponded to not normally distribution were represented as median (min-max). The Kolmogorov-Smirnov test demonstrated whether the distribution of continuous variables was normal. Morphological characteristics of the rats (body weight), ABR-I, ABR-IV, and ABR I-IV interval and DPOAE frequency parameters, were found to be normally distributed (P > .05). One-way analysis of variance test was used in the statistics of these values. Pre- and post-ABR threshold values were not normally distributed. Kruskal-Wallis H test was used in the statistics of these values. The value of P < .05 was considered statistically significant. Student t test was used to compare the study and control groups that were normally distributed and P < .05 was considered statistically significant. All data were analyzed using SPSS version 13.0 program (SPSS version 13.0, Chicago, Illinois).
Discussion
In the current study, we found that intratympanic steroids were found to significantly inhibit cisplatin ototoxicity. The knowledge on the comparison of 2 different intratympanic steroid treatment methods on cisplatin ototoxicity is quite limited. Özel et al have first published an experimental research investigating the protective role of intratympanic dexamethasone and intratympanic methylprednisolone on cisplatin-induced ototoxicity. 8 They have measured only DPAOE as an audiological test for the comparison. In the current study, however, we investigated the role of 2 different intratympanic steroid regimens by the measurement of both DPAOE and ABR in order to increase the reliability of the results regarding the audiological function.
Balance disorder and/or hearing loss due to the loss of function in the inner ear tissues as a result of the use of a substance or medication is called ototoxicity.
11,12
Cisplatin is an antineoplastic agent that is frequently used in the treatment of various malignant diseases. It is one of the most effective chemotherapeutic agents, especially in pediatric patients with a cure rate of approximately 85%.
3
There is no known treatment of ototoxic and neurotoxic effects of cisplatin. The ototoxicity-related hearing loss is a bilateral, irreversible, and progressive sensorineural hearing loss at high frequencies. All cochlea, especially outer hairy cells, are irreversibly affected in the ototoxicity of cisplatin. Audiometric studies showed an increase in hearing thresholds of 75% to 100% during cisplatin treatment.
3
It is possible to monitor cisplatin ototoxicity by audiometric examinations, OAE and ABR.
13,14
In addition, successful monitoring of ototoxicity and the association of frequencies with the clinical appearance could be ensured with DPOAE.
15
The agents that were investigated for their efficacy in preventing cisplatin ototoxicity are mainly steroids, resveratrol, melatonin, Na-thiosulfate, salicylate, Gingko biloba, vitamin E, vitamin C, N-acetylcysteine, and
Steroids are the agents that have been mainly investigated for the prevention of cisplatin ototoxicity. 6 Steroids prevent ototoxicity in the inner ear by preventing the effect of reactive oxygen species. 7 The demonstration of corticosteroid receptors in rat inner ears is an indicator of the corticosteroid efficacy on cochlea. 18
Steroids could be administered intratympanically to prevent their systemic side effects and to achieve a greater concentration in the inner ear. Intratympanic dexamethasone was reported to have no ototoxic effect. 19 Sun et al 20 showed that the protection against cisplatin ototoxicity could be achieved by nanoparticles loaded with dexamethasone. Han et al 21 reported that the combined steroid treatment was significantly better in meta-analyses comparing systemic steroid therapy with combined steroid therapy (intratympanic steroid and systemic steroid). The efficacy of methylprednisolone was shown in the study conducted with the methylprednisolone-loaded nanoparticles. 22 Rauch et al 23 compared oral prednisolone with intratympanic methylprednisolone in a multicentered, prospective, randomized study of 250 patients with the unilateral sensorineural hearing loss. Intratympanic methylprednisolone administration was shown not to be more ineffective than oral prednisolone therapy.
Based on previous publications, ABR and DPOAE were performed when there was no residual drug in the middle ear (after 72 hours). 9,24
The efficacy of intratympanic steroid treatment could also be demonstrated by ABR. Hill et al 24 demonstrated the protective effect of intratympanic dexamethasone in cisplatin ototoxicity by ABR in an animal experiment. In our study, ABR thresholds were found to be maintained with methylprednisolone treatment, although not as much as with dexamethasone treatment. Both ABR-IV and ABR I-IV interval values were found to be protected with the methylprednisolone and dexamethasone treatment.
Marshak et al 25 demonstrated the protective effect of intratympanic steroids in patients with cancer, especially at high frequencies with DPOAE. In their study which they compared intratympanic dexamethasone and methylprednisolone, Özel et al 8 reported that both agents were effective, but dexamethasone was more effective than methylprednisolone. In a meta-analysis which included 184 studies, intratympanic dexamethasone was reported to be more effective in improving hearing loss compared to intratympanic methylprednisolone. 26 In our study, the dexamethasone group was shown to be more effective than methylprednisolone at DPOAE of 2832, 4004, and 5652 frequencies. Although not as much as in the dexamethasone group, the threshold values were found to be maintained significantly in the methylprednisolone group compared to the control and cisplatin groups.
Conclusion
There is no curative treatment of cisplatin ototoxicity as there is no molecule to reach all the affected areas. Furthermore, it should be demonstrated by other studies whether the agents used to prevent ototoxicity would reduce the antineoplastic effect of cisplatin.
In future studies, the efficacy of intratympanic steroids can be demonstrated by repeated drug administration and long-term audiological and histological parameters.
Footnotes
Authors’ Note
This study was presented as an oral presentation at the 11. International Otorhinolaryngology & Head & Neck Surgery Congress, April 5-7, 2018, Ankara. B.M.T. and G.Ş. contributed to concept and wrote the manuscript. B.M.T. designed the article and contributed to literature search. G.Ş. and R.K. supervised the article, did analysis and/or interpretation, and critically reviewed the manuscript. B.M.T. and M.A. made funding and contributed to materials. M.A. done data collection and/or processing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
