Abstract
Introduction:
Sleep-disordered breathing (SDB) is associated with atherosclerosis. Peripheral arterial disease (PAD) is a manifestation of atherosclerosis in lower extremity arteries. No systematic review addressing the relationship between PAD and SDB was found. We performed this study aimed to summarize the relationship between SDB and PAD described in current clinical studies.
Material and Methods:
PubMed and Embase electronic databases were searched for clinical articles (published before 3 April, 2019) describing studies that evaluated the association between SDB and PAD. We showed the results involved in the association in clinical studies.
Results:
In total, 8 clinical studies have been included, and most of them were cross-sectional studies. Six articles demonstrated the coexistence of SDB and PAD, evidenced by high prevalence of SDB in patients with PAD and vice versa. Meanwhile, the included studies exhibited independent positive associations between SDB or sleep parameters and PAD after adjusting for multiple confounders.
Conclusion:
From present clinical prospective, positive association between SDB and PAD was shown. More prospective, randomized controlled studies are needed to establish the cause–effect relationships involved.
Introduction
Sleep-disordered breathing (SDB) is one of the most common sleep disorders, which is associated with cardiovascular morbidity and mortality, and is an independent risk factor for coronary artery disease (CAD), stroke, and atrial fibrillation. 1 –3 Atherosclerosis has been proposed as an intermediate between SDB and incident cardiovascular disease. 4 The pathophysiological effects of SDB-related atherosclerosis include large intrathoracic pressure swings, repetitive intermittent hypoxia (IH) and hypercapnia, reoxygenation, periodic arousals, and sleep fragmentation (SF). 5
Atherosclerosis often manifests as peripheral arterial disease (PAD), which is the atherosclerotic process of the lower extremity arteries leading to peripheral arterial obstruction and causing disabling symptoms as a result of reduced arterial blood flow and consequent arterial insufficiency. 6 Peripheral arterial disease affects about 20% of individuals over 60 years of age. 7 It is a marker of underlying atherosclerosis severity and prognosis that shares common risk factors with SDB, including hypertension, obesity, and diabetes mellitus (DM). 8 –10 Although several clinical studies have explored the relationship between SDB and PAD, the results were recapitulative.
The relationship between SDB and PAD is intricate and needs to be explored. In this review, we summarize the relationship between SDB and PAD in clinical studies. Issues related to confounding factors, epidemiology, and future research directions are also discussed.
Material and Methods
Search Strategy
A systematic search of the PubMed and Embase electronic databases was performed using the search terms: (obstructive sleep apnea) OR (sleep apnea syndromes) OR (OSA) OR (sleep apnea) OR (sleep disordered breathing) combined with (peripheral arterial disease) OR (peripheral occlusive arterial disease) OR (PAD). The final search was finished on 3 April, 2019. Additional reports were added when discovered by citation tracking. The searches were performed by Drs Xia and You separately.
Inclusion and Exclusion Criteria
We included studies that satisfied the following criteria: (1) SDB was confirmed by standard polysomnography (PSG), (2) PAD was confirmed by a history of walking distance limitation for angiological reasons or angiological diagnostics, and (3) the paper was in English. We excluded studies not referring to SDB or PAD.
Data Extraction
All of the included studies were retrieved, and the required information was extracted by 2 reviewers (Drs Xia and You). The characteristics extracted from each study included: the first author, publication year, location, study design, number of participants enrolled, percentage of males, age, SDB assessment, PAD assessment, details of the participants recruited in the article, number of cases of PAD, number of cases of SDB, and adjustment for covariates. Disagreements among the reviewers were resolved by consensus through a group discussion.
Results
Search Results
The initial search retrieved 151 references from PubMed and 428 references from Embase. Of these 579 citations, 38 papers were initially removed due to duplication. A further 525 records were removed after screening by titles and abstracts. We carefully read the full texts of the remaining 16 papers and then excluded 8 additional articles 11 –18 for the following reasons: (1) a review, (2) no extractable data, (3) overlapping data, (4) Burger disease, and (5) PAD not clearly identified as a cardiovascular event. Ultimately, the review included 8 articles. 19 –26 The flow diagram of the search procedure is shown in Figure 1.
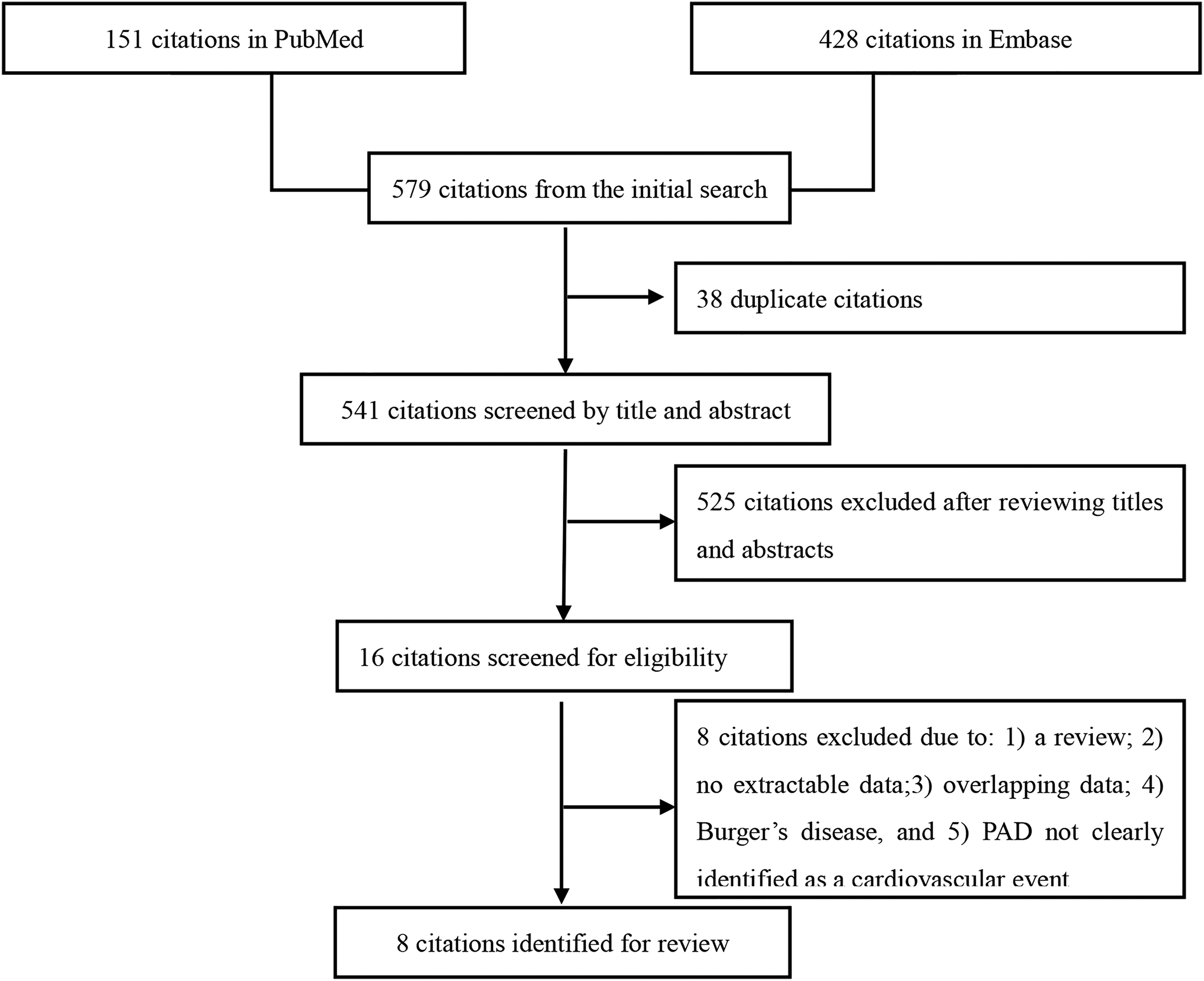
Flow diagram used to identify the qualifying articles.
Study Characteristics
The publication year of the included articles ranged from 2003 to 2017. Regarding the study design, 6 were cross-sectional studies, 19,21,23 –26 1 was a case–control study, 22 and 1 study included cross-sectional analyses and prospective longitudinal analyses. 20 The number of participants enrolled ranged from 59 to 47 268. All studies focused on middle-aged or older people. In these studies, the authors used ultrasound, angioplastic intervention, or a history of walking distance limitation for angiological reasons to assess PAD. To assess SDB, PSG or questionnaires were used for diagnosis. The details are summarized in Table 1.
Characteristics of Included Studies Exploring the Relationship Between SDB and PAD.
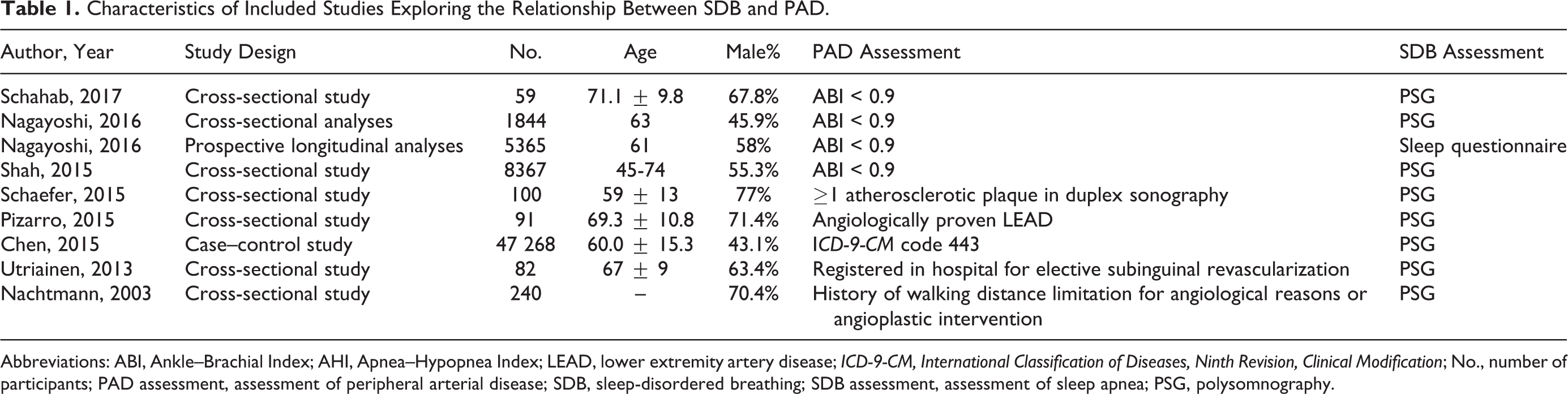
Abbreviations: ABI, Ankle–Brachial Index; AHI, Apnea–Hypopnea Index; LEAD, lower extremity artery disease; ICD-9-CM, International Classification of Diseases, Ninth Revision, Clinical Modification; No., number of participants; PAD assessment, assessment of peripheral arterial disease; SDB, sleep-disordered breathing; SDB assessment, assessment of sleep apnea; PSG, polysomnography.
Main Results
Coexistence of SDB and PAD
Like SDB and metabolic syndrome, SDB and PAD always coexist. High prevalence of SDB ranging from 70.3% to 85.4% in patients with PAD was demonstrated in 3 studies. 24 –26 Meanwhile, 2 studies exhibited higher prevalence of SDB in participants with PAD than those without. 19,22 In 240 patients with ischemic stroke, those with PAD had higher prevalence of SDB (75%) than those without PAD (23%), 19 and the same pattern went into the study performed by Chen et al in general population (0.8% vs 0.5%, P < .001 22 ; see Table 2).
Coexistence of SDB and PAD Showed in Studies.
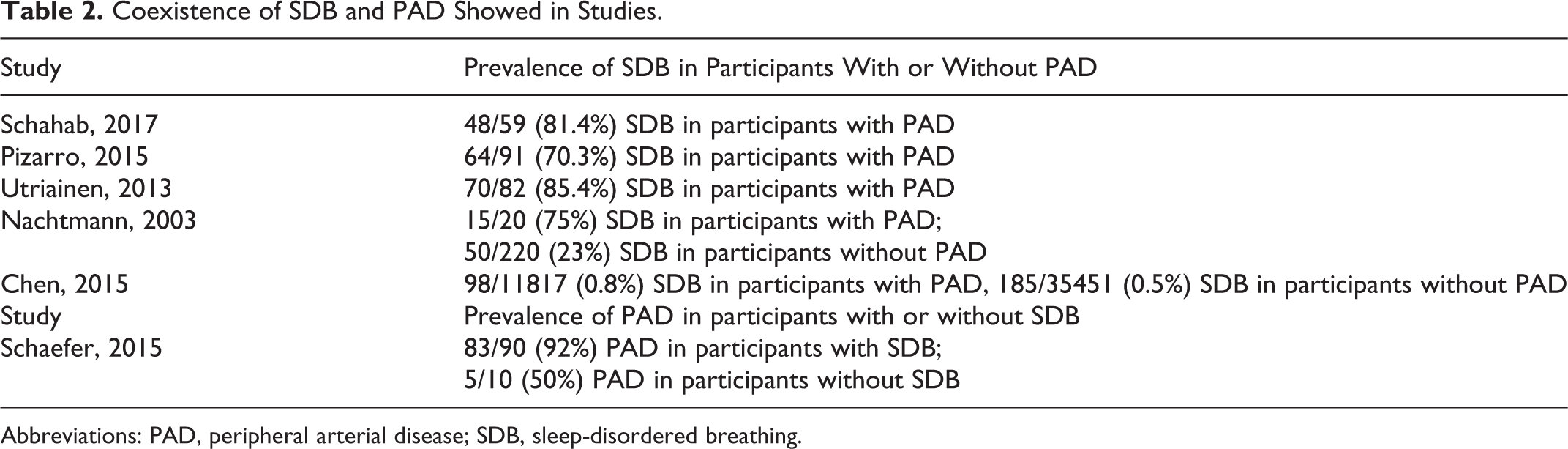
Abbreviations: PAD, peripheral arterial disease; SDB, sleep-disordered breathing.
Furthermore, high prevalence of PAD in patients with SDB was also seen. In one study of 100 patients with suspected SDB, 88% had PAD. Meanwhile, higher prevalence of PAD was seen (83/90, 92%) in participants with SDB than those without (5/10, 50% 21 ; see Table 2).
Associations between SDB and PAD
Apnea–Hypopnea Index (AHI) was found to be significantly correlated with advanced Fontaine stage (P = .042) in 59 patients with PAD 24 and increased with elevating PAD severity (P = .047) in 91 patients with PAD. 25
Five studies 19 –23 showed that the positive associations between SDB and PAD remained significant after adjusting for multiple confounders, and the details were demonstrated subsequently. Apnea–Hypopnea Index was shown to be positively associated with PAD (odd ratio, OR [95% confidence interval, CI]: 5.43 [1.07-27.57]) after adjusting for presence of claudication, oxygen desaturation index, minimal oxygen saturation, pulse-wave index, and central pulse wave velocity in 100 patients with suspected SDB. 21 Of 240 patients with ischemic stroke, after adjusting for age and sex, severe SDB remained significantly associated with PAD, OR (95% CI): 6.00 (1.63-15.97). 19 A large sample size study performed both cross-sectional analyses and prospective longitudinal analyses. 20 In their cross-sectional analyses including 1844 participants, they found that 101 (5.4%) participants had PAD and 1194 (64.8%) participants had SDB. After adjusting for age, sex, race/ethnicity, educational attainment, physical activity, smoking status, pack years, adiposity, PAD risk factors, severe SDB was associated with greater prevalent PAD among black participants, prevalence ratio (95% CI): 2.29 (1.07-4.89). In their prospective longitudinal analyses including 5365 participants, over a median of 9.2 years of follow-up, 229 incident PAD cases occurred. After adjusting for multivariate factors, participants with self-reported sleep apnea were at higher risk of incident PAD, hazard ratio (95% CI): 1.93 (1.05-3.53). Another study including 8367 participants demonstrated that participants with PAD had higher AHI than participants without PAD. 23 After adjusting for age, sex, body mass index, waist to hip ratio, hypertension, coronary heart disease, DM, dyslipidemia, C-reactive protein levels, smoking, alcohol use, physical activity, study site and Hispanic/Latino background, participants with moderate-to-severe SDB were 1.67 times more likely to have PAD, OR (95% CI): 1.67(1.10-2.51). Furthermore, one population-based case–control study with 47 268 participants found after adjusting for CAD or myocardial infarction, chronic kidney disease, hyperuricemia, and obesity, PAD was significantly associated with SDB (OR [95% CI]: 1.37 [1.07-1.76]). 22 However, the association was attenuated with further adjusting for hypertension, hyperlipidemia, and DM 22 (see Table 3).
Associations Between SDB and PAD Demonstrated in Clinical Studies.
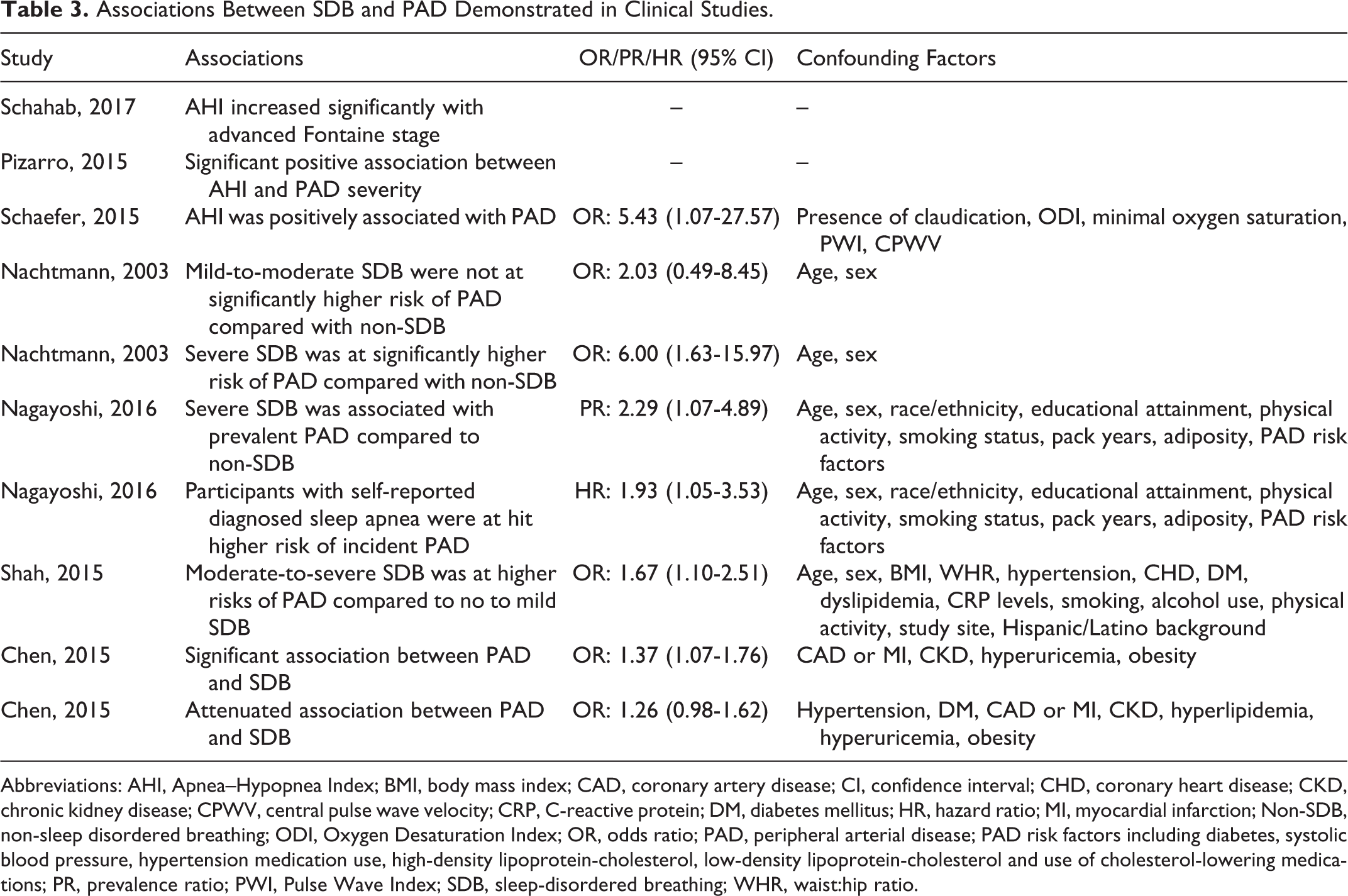
Abbreviations: AHI, Apnea–Hypopnea Index; BMI, body mass index; CAD, coronary artery disease; CI, confidence interval; CHD, coronary heart disease; CKD, chronic kidney disease; CPWV, central pulse wave velocity; CRP, C-reactive protein; DM, diabetes mellitus; HR, hazard ratio; MI, myocardial infarction; Non-SDB, non-sleep disordered breathing; ODI, Oxygen Desaturation Index; OR, odds ratio; PAD, peripheral arterial disease; PAD risk factors including diabetes, systolic blood pressure, hypertension medication use, high-density lipoprotein-cholesterol, low-density lipoprotein-cholesterol and use of cholesterol-lowering medications; PR, prevalence ratio; PWI, Pulse Wave Index; SDB, sleep-disordered breathing; WHR, waist:hip ratio.
Discussion
We can see that only a few studies have focused on the relationship between PAD and SDB. Six articles found that SDB and PAD always coexist, evidenced by high prevalence of SDB in patients with PAD and high prevalence of PAD in patients with SDB. Meanwhile, the included studies indicated independent positive associations between SDB or sleep parameters and PAD after adjusting for multiple confounding factors.
Actually, SDB is characterized with 2 typical pathophysiological processes, including IH and SF. Both IH and SF trigger a series of effects on arteries and ultimately promote atherosclerosis. Intermittent hypoxia causes repeated episodes of hypoxia interspersed with normoxia, potentially similar to the ischemia/reperfusion injury that results in the burst in the production of reactive oxygen species, 27 consequently leading to oxidative stress. Intermittent hypoxia and the ensuing oxidative stress activate pro-inflammatory signaling pathways involving transcription factors. 28 Meanwhile, IH and SF are associated with increased hypothalamic–pituitary–adrenal (HPA) axis activity. 29 –31 Oxidative stress, inflammation, and enhanced-HPA axis increase leptin expression, reduce growth factor, upregulate orexin neurons, and alter cerebral metabolism, which lead to insulin resistance and glucose intolerance, 32 obesity, 33 dyslipidemia, 34 hypertension, 35 type 2 DM, 36 and metabolic syndrome. 37 Furthermore, sympathetic nervous system activation and oxidative stress decrease nitric oxide synthesis 38,39 and promote endothelial dysfunction, which together with inflammation 40 contributes to hypercoagulability. 41 –44 Endothelial dysfunction and abnormal metabolism in SDB render the vascular endothelium vulnerable to damage. Hypercoagulability and inflammation promote plaque formation in arteries. Therefore, IH and SF prompt atherosclerotic processes in arteries. Peripheral arterial disease is the manifestation of atherosclerosis in the lower extremity arteries. Thus, we infer that SDB promotes PAD via the potential underlying mechanism described above. Prospective cohort and randomized controlled studies are warranted to confirm these findings.
Although the included studies demonstrated an independent relationship between SDB and PAD, there were a lot heterogeneities existing among the studies. First, the study designs differed among the studies, including 6 cross-sectional studies, 1 case–control study, and 1 study included cross-sectional analyses and prospective longitudinal analyses. Second, the characteristics of included participants varied a lot, as some studies included community population and some analyzed participants with specific diseases like ischemic stroke. Third, when exploring the independent association between SDB and PAD, different confounding factors were adjusted. For example, obesity that is known to be associated with both SDB and atherosclerosis is not included in several studies. Because of these heterogeneities, we cannot perform in-depth statistical analyses to draw conclusions on this clinically important issue.
There are some limitations in this study. First, this is a review of current evidence about the relationship between SDB and PAD, which lacks originality. Second, we only included papers in English; evidence in other language were dismissed. Third, due to the small number of studies focusing on the relationships between SDB and PAD, only 8 articles were included, resulting in low power. Finally, as described above, we did not synthesized results of included studies because of heterogeneities.
Thus, only a few studies focused on the associations between SDB and PAD at present. And most clinical studies indicated positive associations between PAD and SDB. We inferred atherosclerosis may be the underlying mechanism linking them together. But until now, most clinical studies exploring the relationship between SDB and PAD were cross-sectional and case–control studies, and the main shortcoming of observational studies is that they allow no inference regarding causal relationships. This prompts 2 questions: “Does the treatment of PAD alleviate SDB severity?” and “Does continuous positive airway pressure slow the progression of PAD?” Prospective, randomized controlled studies are needed to answer these questions.
Conclusion
In total, 8 clinical studies have been included, and most of them were cross-sectional studies. Six articles demonstrated the coexistence of SDB and PAD. Meanwhile, the studies including 3 large sample size studies exhibited independent positive associations between SDB or sleep parameters and PAD after adjusting for multiple confounders. From present clinical prospective, we infer that there is a positive association between SDB and PAD. More prospective, randomized controlled studies are needed to establish the cause–effect relationships involved.
Footnotes
Authors’ Note
Yunyan Xia and Kai You contributed equally to this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (NSFC) (Grant No. 81160126).
