Abstract

A 56-year-old male, former smoker, presented with right mandibular pain for several months, leading to a computed tomography (CT) scan showing a 2 to 3 cm mass. Subsequent fine-needle aspiration biopsy results were inconclusive. A nononcologic right submandibular resection performed at an outside institution revealed a pT3N0 poorly differentiated squamous cell carcinoma with positive margins and perineural invasion (PNI). He subsequently received 66 Gy of postoperative radiotherapy to the operative bed and 59.4 Gy to the regional lymphatics. He presented to our institution for follow-up care approximately 7 months after the completion of radiotherapy, complaining of persistent facial swelling and 2 months of diffuse right-sided facial numbness. Physical examination revealed numbness involving his right cranial nerves (CNs) V1-V3, as well as weakness of the right facial nerve. Additionally, soft tissue thickening was palpable in the operative bed. A computed tomography scan and subsequent F18 fluorodeoxyglucose positron emission tomography (PET) scan showed radiotracer uptake within the surgical bed, extending through the right foramen ovale, invading into the right parapharyngeal fat and right pterygoid plexus, and tracking up to the inferior medial right temporal lobe (Figure 1). A subsequent magnetic resonance imaging (MRI) study with intravenous gadolinium contrast confirmed these findings. He initiated reirradiation treatment to a planned dose of 60 Gy and platinum-based chemotherapy. Shortly after beginning therapy, he began experiencing right ocular blurring, diplopia, redness, and pain and was sent to an ophthalmology clinic. At that time, he was found to have lagophthalmos as well as a CN V-VIII palsy. The patient subsequently opted for comfort-based care and discontinued chemoradiotherapy.
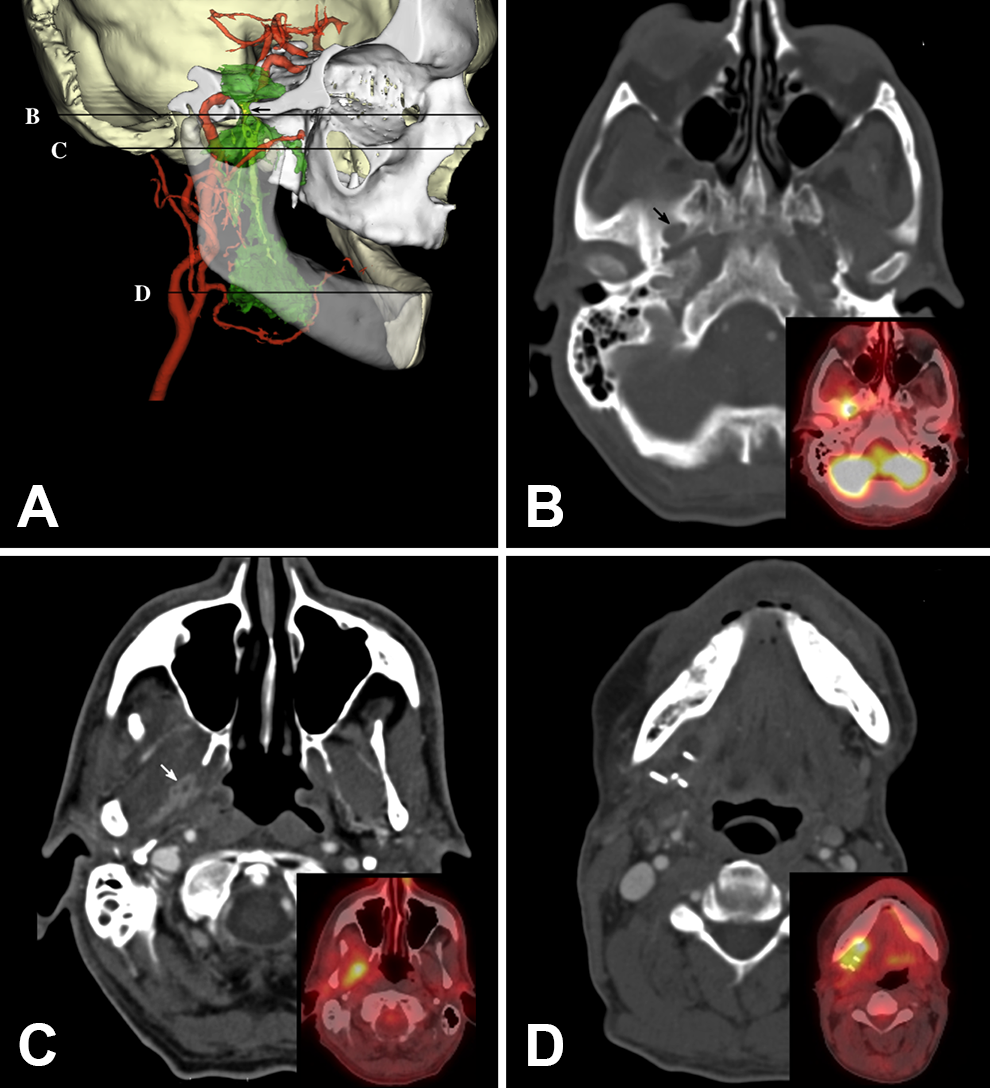
Computed tomography (CT) and F18 fluorodeoxyglucose (FDG) positron emission tomography (PET) scan. A, Computed tomography–based 3D reconstruction of skull (white, beige), tumor (green), cranial nerve V3 (yellow), and major arteries (red). Right side of skull (white) cut along sagittal plane at lateral aspect of foramen ovale. Horizontal black lines correspond to axial CT with inset PET/CT images (B-D). Arrows mark foreman ovale (black) and tumor encasement of cranial nerve V3 (white).
Perineural invasion is a phenomenon defined by the infiltration of neoplastic cells either within a peripheral nerve sheath or nearby the nerve, involving at least one-third of its circumference. 1 Perineural invasion is a histological finding, distinct from perineural tumor spread (PNTS). PNTS is a macroscopic finding either clinically or radiographically present, extending from the primary tumor. It commonly signifies more advanced disease. 2 The facial and trigeminal nerves are most commonly affected by PNI and PNTS given their expansive innervation of the head and neck. Although PNI is correlated with an aggressive phenotype among salivary gland malignancies, its independent prognostic significance is more nuanced. 3 As a result, the modalities and extent of treatment for salivary gland malignancies are still under debate. This case underscores the importance of PNI as a prognostic marker for recurrent disease.
The molecular correlates of PNI remain unclear. Previously, it was believed that PNI spread in the form of skip lesions. However, the continuous theory of spread is gaining more support. 4 Increasing evidence supports the role of brain-derived neurotrophic factor, nerve growth factor (NGF), and neutrophin-4 in tumor spread. Most of these molecules bind to a tyrosine kinase, growth factor, or NGF receptor, but there is much left to learn about the nature of PNI. 5
Less than 40% of patients with perineural growth present with symptoms, though rates may be an underestimated given that it is possible that subtle dysesthesias and paresthesias may be overlooked. 5 Recognition of clinical PNI is important; in addition to its prognostic ability for local recurrence and worsened survival, it may lead to larger radiotherapy fields and/or elective neural coverage to the base of skull. Additionally, radiological detection of PNI cannot supplant clinical detection; there are currently no effective, noninvasive techniques for detecting PNI. However PNI may still be suspected on MRI (more specifically with fat-saturated, contrast-enhanced T1-weighted MRI), CT, or PET.
The optimal radiotherapy volume along a nerve pathway still remains unclear due to the primarily retrospective nature of the literature and low aggregate numbers of recurrences along the nerve tracts. 6 Radiation of CNs at risk up to the skull base would, in theory, reduce the risk of both intracranial and extracranial recurrence. A recent review by Bakst and colleagues 2 recommens electively covering nerve pathways to the base of skull in all cases of clinical or radiographic PNTS. They also recommend consideration of coverage for tumors with extensive (rather than focal) PNI and/or involvement of named or large-caliber nerves, as well as tumors of adenoid cystic or salivary ductal histology.
Footnotes
Authors’ Note
Permission was obtained from the patient whose images are presented in this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
