Abstract
Bone conduction implants based on percutaneous abutment acoustic transmission have been implemented in patients with satisfactory outcomes. However, adverse soft tissue outcomes present a limitation. Transcutaneous bone conduction devices (t-BCDs) are an alternative that may mitigate these complications. A retrospective review was performed of patients who underwent implantation of a t-BCD from 2013 to 2017. Surgical outcomes were reviewed including wound complications, frequency of device use, patient concerns regarding the device, and reported patient satisfaction. A total of 37 patients were implanted with the bone-anchored hearing aids, BAHA Attract (Cochlear™ Baha® Implant System). Average follow-up time was 271.8 days. Postoperatively, 7 (18.9%) patients complained of soft tissue changes or issues with wound healing. Twelve (32.4%) patients requested adjustment of their devices. The t-BCD is an excellent option for hearing rehabilitation. Overall, the complication rate is low, patient satisfaction is high, and successful conversion from a percutaneous device is possible with minimal risk.
Introduction
Bone-anchored hearing aids (BAHAs) have become widely implemented for use in both unilateral and bilateral conductive or mixed hearing loss. They have been well established for use in patients with single-sided deafness and patients with congenital defects such as aural atresia, for who conventional hearing aids are either poorly tolerated or not indicated. The principle of bone conduction, whereby sound is transferred to the inner ear using osseous vibrations across the calvarium to bypass the external and middle ear, was documented in the second century
The BAHA was first reported by Branemark and Tjellstrom in 1977 as an evolution of dental implants. 4,5 Traditionally, the system is comprised of a titanium implant with a percutaneous abutment and a sound processor that is attached to the abutment. This apparatus allows direct coupling of sound transmission from processor to bone via the abutment and implant. Although these devices have proven useful, there remains resistance among potential candidates due to aesthetic concerns and issues with skin breakdown, overgrowth, or infection attributable to the skin-penetrating abutment. 5 -7 Some patients may have medical comorbidities that make it difficult to perform the daily cleaning required to maintain the skin penetration site. Infections surrounding the abutment site have been reported in 8% to 59% of patients, and revision surgery is required in 5% to 42% of cases. 8,9 These issues are often deterrents for patients seeking intervention. A nonpenetrating transcutaneous device may represent a more viable option for these patients. Magnetic bone conduction hearing implants use the same digital sound processing technology and osseointegrated implant devices that are employed in direct percutaneous bone conduction; however, the skin-penetrating abutment is replaced with an implanted magnet for the transmission of sound energy across the skin to the external component. The external magnetic device is fitted with a lining of soft material, thereby distributing the pressure across the skin surface allowing for stable single-point bone fixation and evenly distributed contact pressure, with efficient transmission and minimal skin complications.
These transcutaneous bone conduction hearing implants have been well-documented, with encouraging success rates. 10,11 The main obstacles to the successful implementation of these devices include energy dissipation and loss of amplification due to the layer of intervening skin, resulting in decreased transmission compared to direct percutaneous bone conduction. 12 -14 The need for adequate magnetic coupling in order to ensure device retention and effective sound transmission must also be balanced with the potential for soft tissue complications. Advances in sound-processing technology and modifications in implant devices have, however, permitted successful compensation for sound attenuation by increasing the amplification in the affected frequencies. 15 -17
The present study evaluates our institutional clinical experience and the performance of the transcutaneous magnetic bone conduction hearing implant system. Our investigation represents the largest single institutional study, within North America, to date evaluating hearing performance in comparison to unaided hearing, device retention, soft tissue complications, and overall patient benefit. Additionally, while the majority of reports thus far have documented only early outcomes, we report intermediate-term follow-up of patients.
Methods
An institutional review board-approved retrospective chart review was performed. All procedures for placement of bone-anchored hearing device surgery between December 2013 and January 2017 were identified. Those procedures in which patients received percutaneous devices were excluded; however, patients who were converted from percutaneous to transcutaneous were noted. All patients had pre- and postoperative audiologic evaluation. More specifically, they underwent pure tone average audiometry in addition to speech test battery, including AzBio testing in unaided and aided conditions.
The surgical procedures were performed according to the manufacturer’s recommended specifications. The implantation site is identified either using the provided template or by measuring 6.5 cm posterosuperior to the superior margin of the external auditory canal, in the plane of the superior margin of the auricle. The site is marked, and methylene blue dye is used percutaneously to stain the calvarium and periosteum below the planned site. A curved incision is made approximately 3 to 4 cm anterior to the planned implant site and carried down to the periosteum. A skin flap is raised, and the methylene blue dye identified. A cruciate incision is made in the periosteum, and this is elevated. A socket for implant placement is then created using a combination of 3- and 4-mm drill bits, depending on calvarial thickness. Bone at the base of the socket and/or dural integrity are confirmed. The 3-mm or 4-mm countersink is then used. The implant is then placed with the drill set to 45 N-m. A bone bed indicator is used to verify no contact of the magnet with underlying bone. If contact is anticipated, the bone is reduced using 5-mm diamond burr. Using the supplied torque wrench, the implantable magnet is then affixed to the implant to a torque of 25 N-m. The incision is closed, and a Glasscock dressing is placed for pressure on the implant site.
Postoperative follow-up was measured in days. The electronic medical record was reviewed for patient concerns regarding the device, issues with skin irritation or breakdown, and device satisfaction and use.
Results
A total of 37 patients were identified. These patients underwent a total of 38 surgeries with a total of 39 implants. The average age at device implantation was 36.6 years with a range of 5.98 to 83.59 years. This included 19 females and 18 males, 13 of who were pediatric patients (aged <18 years). A total of 38 surgeries were performed, including 4 patients who initially underwent implantation with the Connect abutment (Cochlear™ Baha® Implant System) but who subsequently underwent conversions to the Attract transcutaneous system. One patient had 2 implants placed during a single procedure, and another had 2 implants placed during separate procedures. Operative time average was 52 ± 0.015 minutes. The average follow-up time was 271.8 days (range: 71-824 days). With regard to indications for surgery, 14 patients had unilateral conductive hearing loss (CHL), 11 had unilateral sensorineural hearing loss (SNHL), 8 had bilateral CHL, 3 with unilateral mixed hearing loss, and 1 patient with bilateral SNHL (Table 1). The underlying etiologies of hearing loss varied. Eleven patients had previously undergone tympanomastoidectomy with CHL, one of which experienced bilateral CHL. One patient had previously experienced a bout of labyrinthitis with resultant unilateral SNHL. Another individual had unilateral SNHL following temporal bone fracture. One patient had enlarged vestibular aqueduct in the context of head trauma and unilateral SNHL. Several patients had congenital anomalies including aural atresia, microtia, or Goldenhaar syndrome.
Patient Data and Outcomes.
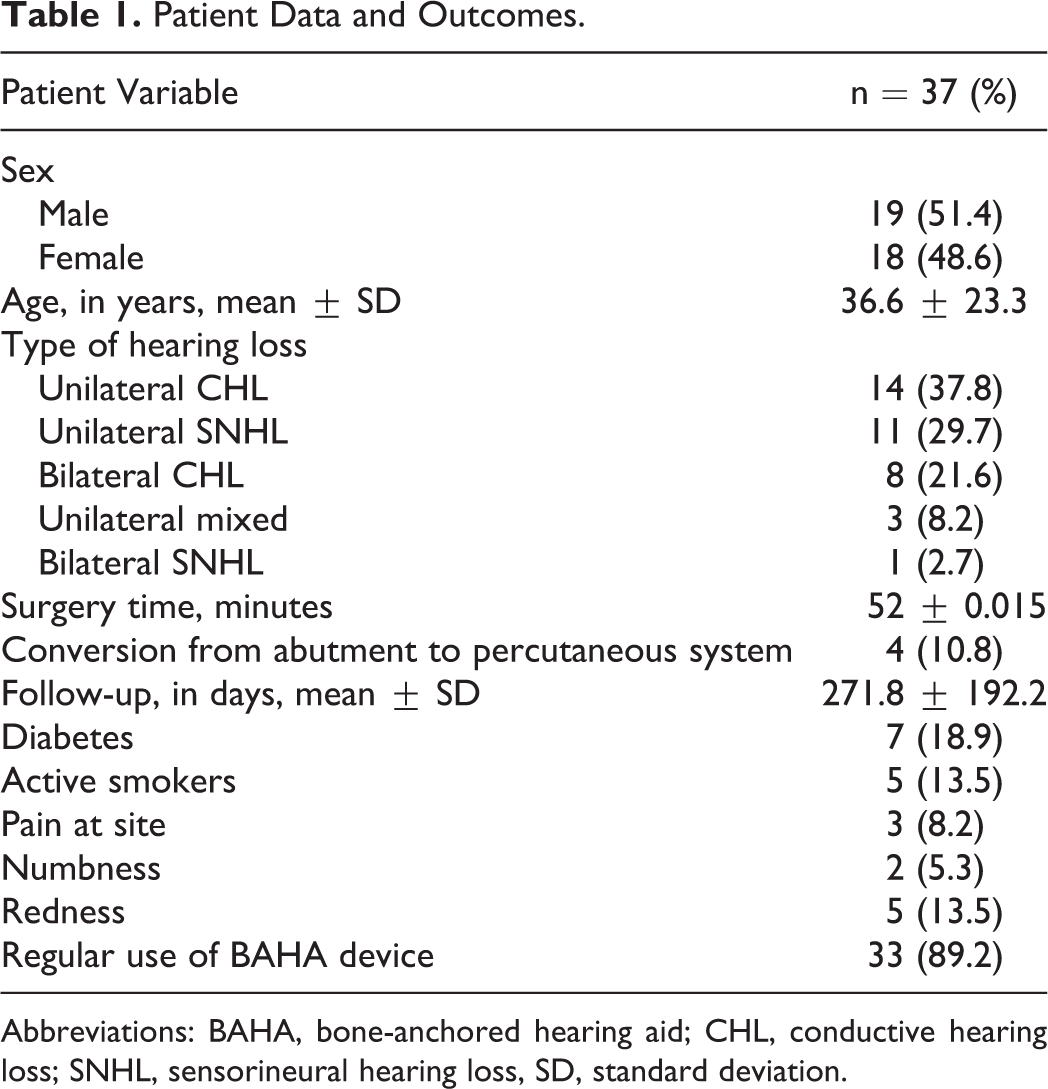
Abbreviations: BAHA, bone-anchored hearing aid; CHL, conductive hearing loss; SNHL, sensorineural hearing loss, SD, standard deviation.
Postoperatively, 7 (18.9%) patients complained of soft tissue changes or issues with wound healing. The majority of these patients experienced erythema surrounding the wound site that resolved without need for intervention. One patient experienced eczematous reaction necessitating use of topical steroid ointment with full resolution of his symptoms. A single patient experienced a small hematoma, and this individual was a former smoker with a medical history significant for peripheral artery disease and controlled diabetes. The hematoma did not require intervention and had resolved on follow-up examination. Another individual with a history of psoriasis experienced development of granulation tissue that persisted 6 months postoperatively. Interestingly, of those complaining of soft tissue changes, none were active smokers and only 1 had diabetes that was well controlled. Only 3 (8.1%) patients complained of postoperative pain which was short term and had resolved well before 6-month follow-up. Two (5.4%) patients complained of numbness or paresthesia at the implant site. Four (10.81%) patients reported that they were not using their implant, 1 of these individuals complained of irritation when using the device; yet another described issues with amplitude adjustment. The remainder of individuals reported regular daily, or at least weekly, use of the device. Twelve (32.4%) patients requested adjustment of their devices. Two (5.41%) individuals reported issues with feedback, 4 (10.81%) reported skin irritation, 3 (8.11%) complained of magnet connectivity issues, 2 (5.41%) experienced headaches that were self-limited and had resolved by their second postoperative visit, and 1 (2.7%) experienced discomfort with sound amplitude that resolved with adjustment of the device. The majority of patients reported no issues with their BAHA devices and endorse improvement in their hearing with regular use. All patients are scheduled to return in 6 months for post-BAHA activation testing. Our data collection is in progress; however, preliminary scores show a trend toward improvement in scores 6 months postactivation.
All 4 individuals who initially underwent Connect abutment system implantation, but later required conversion to the Attract transcutaneous magnetic implant, underwent conversion due to local wound healing complications with the percutaneous device. Three patients had significant skin overgrowth and local tissue reactions preventing the use of their abutment. Upon conversion to the magnet-based system, they had no further complications and on follow-up were using their devices without issue. The fourth individual initially experienced a nonhealing wound at the abutment site; however, on conversion to BAHA Attract (Cochlear ™Baha1Implant System), he experienced appropriate healing and was able to tolerate use of the device with minimal skin irritation.
Audiologic fitting concerns note the BAHA power devices are bulkier and have added weight which can be problematic, as the patient may require a stronger magnet for retention. The increased magnet strength provides improvement in sound quality and reduces audible feedback; however, close monitoring of the magnet site is advised. We have found that patients with very thick hair present significant problems with retention and feedback. Those patients are advised to cut the hair under the magnet site. Patients with stronger magnets should be periodically checked for skin degradation, and family members are counseled to check the site weekly.
Discussion
Percutaneous bone-anchored hearing devices have been utilized for many years, and their efficacy has been well-documented. 18,19 However, these systems are limited by their soft tissue complications that are a direct result of the percutaneous abutment. 20,21 Overall, implantation of BAHA is considered both safe and efficacious with limited serious complications. However, tissue overgrowth and abutment site infections continue to be frequent and can limit patient comfort and compliance. 21 Although modifications in implantation techniques and implant design have been made in order to decrease complications, continued difficulties with soft tissue healing have resulted in a shift toward magnet-based transcutaneous systems. These systems have their limitations as well with soft tissue attenuation, particularly at higher frequencies, and issues surrounding implant retention. The development of modern sound processor technology has, however, allowed compensation for skin attenuation by implementation of increased amplification at higher frequencies.
The majority of our patients used their devices on a daily basis, a subjective measure of benefit, as individuals with little or no improvement in hearing would likely discontinue use of the device. Prior studies have found usage of transcutaneous devices to range from 5 to 12 hours daily. 22,23 Several patients underwent conversion from percutaneous-based bone-anchored systems to the BAHA Attract transcutaneous system. These patients did well with continued regular use of their devices without further skin complications or issues with compliance. A recent systematic review of the BAHA devices evaluated data from 10 articles analyzing 89 cases; the overwhelming majority of the data was favorable with regard to both functional and audiologic outcomes with complication rates similar to those presented here. 24 There was variability between these studies in objective measures; therefore, comparisons are difficult. Additionally, no multicenter randomized controlled trials have been performed evaluating the magnet-based system. Comparisons between our study and similar studies of transcutaneous devices are difficult particularly due to varied study design and measured outcomes. However, the greater majority of studies have indicated an overall benefit to patients with negligible complications.
Our institutional experience indicates that magnetic bone conduction hearing implants are associated with minimal complications and are generally safe. There were no major complications necessitating implant removal or loss, indicating that these implants are a safe and reliable alternative to the percutaneous abutment system. Magnetic transcutaneous implants have improved aesthetics and eliminate the necessity of daily cleaning. Our research, although ongoing, indicates that patients are able to obtain appropriate sound transmission without significant soft tissue attenuation. This is consistent with prior studies indicating that patients show functional gains at all frequencies with use of the BAHA Attract although, as expected, soft tissue attenuation is experienced at higher frequencies. 25,26 Despite our positive results, we do recommend that preoperative testing with a soft band sound processor continued to be used, as it has previously been a reliable predictor of functional outcomes. 6,27 -29 Soft tissue complications were minimal, only 7 patients complained of issues with skin irritation, and these cases were self-limited or resolved with adjustment of magnet strength. Our results confirm prior studies, which have indicated that the Attract device is associated with significantly less adverse soft tissue reactions than those implementing skin-penetrating abutment. 30 Complications that were reported in less than 5% of patients included pain, paresthesia, headache, feedback, or connectivity issues. Additionally, the majority of our patients reported daily use, and those with part-time use continued to endorse benefit from use of the device. The high proportion of active users is likely attributable to the minimal discomfort and ease with which transcutaneous devices can be maintained. The rate of connectivity issues or difficulty with implant retention was low (3 patients; 8.1%). Retention difficulties may be further addressed by decreasing soft tissue flap thickness below 6 mm or use of magnets with increased strength, particularly in patients with postoperative swelling and edema. 22 Notably, all patients initially implanted with percutaneous abutment devices with local skin complications were reimplanted with transcutaneous Attract systems without further skin issues, consistent with prior studies. 31 Additionally, the pediatric patients in our study displayed excellent long-term wound healing without issues and were able to wear their devices on a consistent basis. These data indicate that utilization of the Attract system may be of particular use in the pediatric population during critical periods of language development and acquisition. 31
Conclusion
Our experience with the BAHA Attract transcutaneous magnet system demonstrates its safety, ease of use, and overall utility. No major surgical complications are associated with implantation of the device, and in the greater majority of patients, there are minimal skin complications with appropriate healing, functional, and aesthetic outcomes.
Footnotes
Authors’ Note
These results were presented at the American Academy of Otolaryngology–Head & Neck Surgery (AAO-HNSF) annual meeting in Atlanta, Georgia, October 7-10, 2018.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
