Abstract

Eosinophilic granulomatosis with polyangiitis (EGPA) is a rare form of systemic necrotizing vasculitis that affects small- and medium-caliber vessels, which can involve the lungs, nose and paranasal sinuses, intestinal tract, kidneys, heart, and skin. Eosinophilic granulomatosis with polyangiitis was formerly called Churg-Strauss Syndrome, from the names of the authors who first described it as an “allergic granulomatosis and allergic angiitis.” 1 According to the American College of Rheumatology (ACR), 2 the diagnosis of EGPA is mainly based on the finding of 6 clinical features: asthma, eosinophilia (>10% in peripheral blood), paranasal sinusitis, nonfixed pulmonary infiltrates, histological evidence of vasculitis with extravascular eosinophils, and multiple mononeuritis or polyneuropathy. The presence of at least 4 of these clinical parameters is necessary to formulate the diagnosis with a sensitivity of 85% and a specificity of 99.7%. A female patient aged 43 years, with a previous diagnosis of Loeffler Syndrome, came to our observation in December 2016, for the appearance of bilateral nasal obstruction and anosmia. Under rhinofibrolaringoscopic examination with flexible optical fibers, the presence of polypoid formations involving almost completely both nasal cavities was demonstrated (Figure 1). The patient underwent blood tests (erythrocyte sedimentation rate, C-reactive protein, creatinine, and blood count with formula), hematological tests (perinuclear antineutrophil cytoplasm antibodies [p-ANCA] and anti-myeloperoxidase [MPO] antibodies), and imaging examinations (computed tomography [CT] of paranasal sinuses and chest). A pneumological examination with spirometry and a rheumatological examination were also carried out. The results of the tests documented the presence of peripheral hypereosinophilia (>10%), absence of allergies, and anti-p-ANCA and anti-MPO positivity. Her pneumological examination and spirometric examination suggested mild obstructive dysventilation syndrome. Her CT scan of the sinuses showed nasal and sinus polyposis (Figure 2), and a CT scan of the chest showed “3-mm micro-nodular formation in the lower lobe of the right lung.” This diagnosis was further investigated with high-resolution CT, which revealed “multiple areas of ground-glass thickening and thickening of the right pulmonary apex” (Figure 3). In view of the fact that our patient presented 4 of the 6 clinical parameters, namely, peripheral blood hypereosinophilia, asthma, rhinosinusal polyposis, and nonfixed pulmonary infiltrates, required for the diagnosis of EGPA based on the clinical criteria of ACR, 2 her diagnosis of EGPA was reformulated. At the end of the diagnostic procedure, the patient was informed of the formulated diagnosis and on the different clinical course with respect to the previous diagnosis. She underwent a functional endoscopy sinus surgery, and an adequate periodic follow-up (every 6 months) was recommended. Currently, the patient is treated with methotrexate therapy with good control of her systemic symptoms and topical therapy with corticosteroids to control the symptoms of chronic rhinosinusitis. In EGPA, nasal sinus symptoms are one of the most common features. Ear, nose, and throat manifestations of the condition include allergic rhinitis, nasal obstruction, recurrent rhinosinusitis, nasal polyposis, otitis media with effusion, progressive sensorineural hearing loss, and facial nerve palsy. 3 The involvement of the nasal sinus area is so frequent that it represents 1 of the 6 diagnostic criteria. 2 The EGPA follows a chronological evolution in which 3 phases can be distinguished: the first is asthma with or without allergic rhinitis; the second is eosinophilia in the peripheral blood and eosinophilic infiltration in tissues (with an appearance similar to Loeffler syndrome), and the third is vasculitic infiltration with involvement of the lung, heart, peripheral nervous system, liver, lymph nodes, muscles, and skin. 4 However, the sequence of the clinical phases and the antibody profile are not always well defined. Different phases may overlap, or some may not appear at all as in our case. In effect, in our case, the appearance of the symptoms dates back to several years before the disease presented itself with a clinical picture corresponding to phase II of the EGPA, in which the symptomatology resembles that of the Loeffler syndrome. In Loeffler syndrome, unlike the EGPA, resolution of the symptoms is generally rapid, and the prognosis is favorable; therapy is based on corticosteroids and control of parasitic infestation. 4 In the reported case, the patient after her initial diagnosis of Loeffler syndrome received systemic corticosteroid therapy with only temporary resolution of her symptoms. In fact, asthma and hypereosinophilia reappeared when her therapy was interrupted. The correct and early classification of the nasal pathology in the context of a systemic disease such as the EGPA allowed us to provide the patient with important information on the course and prognosis of the disease and to make her aware of the need to perform targeted therapy and have periodic follow-up.
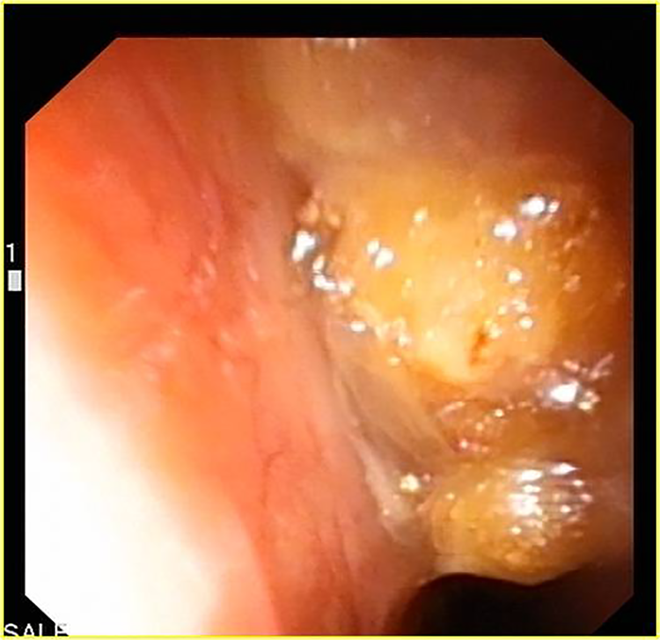
Endoscopic view of the polypoid formation involving almost completely the nasal cavity.
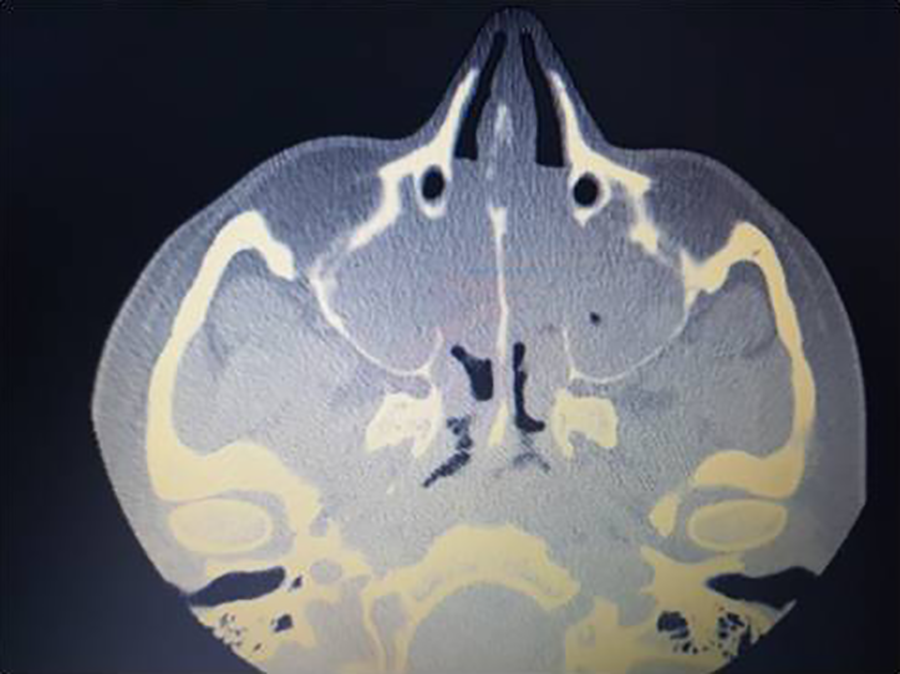
Computed tomography (CT) scan of the sinuses that showed nasal and sinusoidal polyposis.
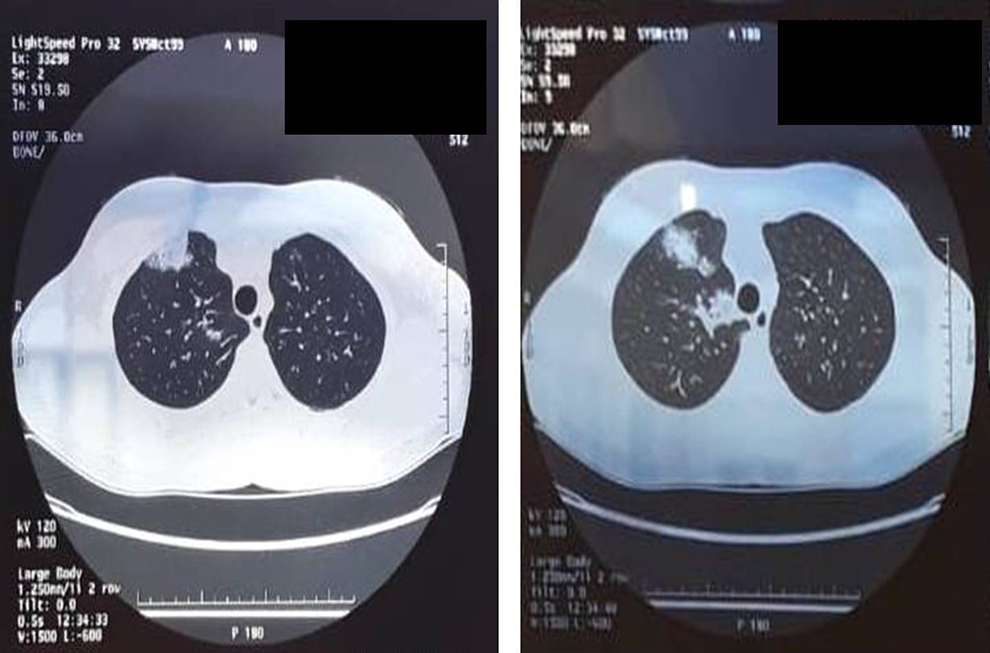
High-resolution computed tomography view of multiple areas of ground-glass thickening and thickening of the right pulmonary apex.
Footnotes
Authors’ Note
This article was presented at “XLII AOOI 2018,” Barletta, October 12-13, 2018, Italy.
Declaration of Conflicting Interests
The author(s) have declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
