Abstract

A 22-year-old male patient was referred to the Ear Nose and Throat department by his general practitioner with a 2-month history of a fluctuating left parotid swelling following an upper respiratory tract infection. He was otherwise fit and well, did not take any regular medication, and expressed no history of recurrent salivary gland infections or sialoliths. Physical examination revealed a 3 cm, hard mass within the left parotid gland. There was associated left level II tender lymphadenopathy.
Ultrasound examination revealed a 3 × 2 cm hyperechoic solid mass. A core biopsy was performed and was inconclusive. Magnetic resonance imaging (MRI) confirm a lesion involving the deep and superficial lobes of the gland measuring 3.5 cm (Figure 1). The changes suggest reaction in the parotid parenchyma consistent with a chronic inflammatory process, likely to be an IgG4 sclerosing disease.
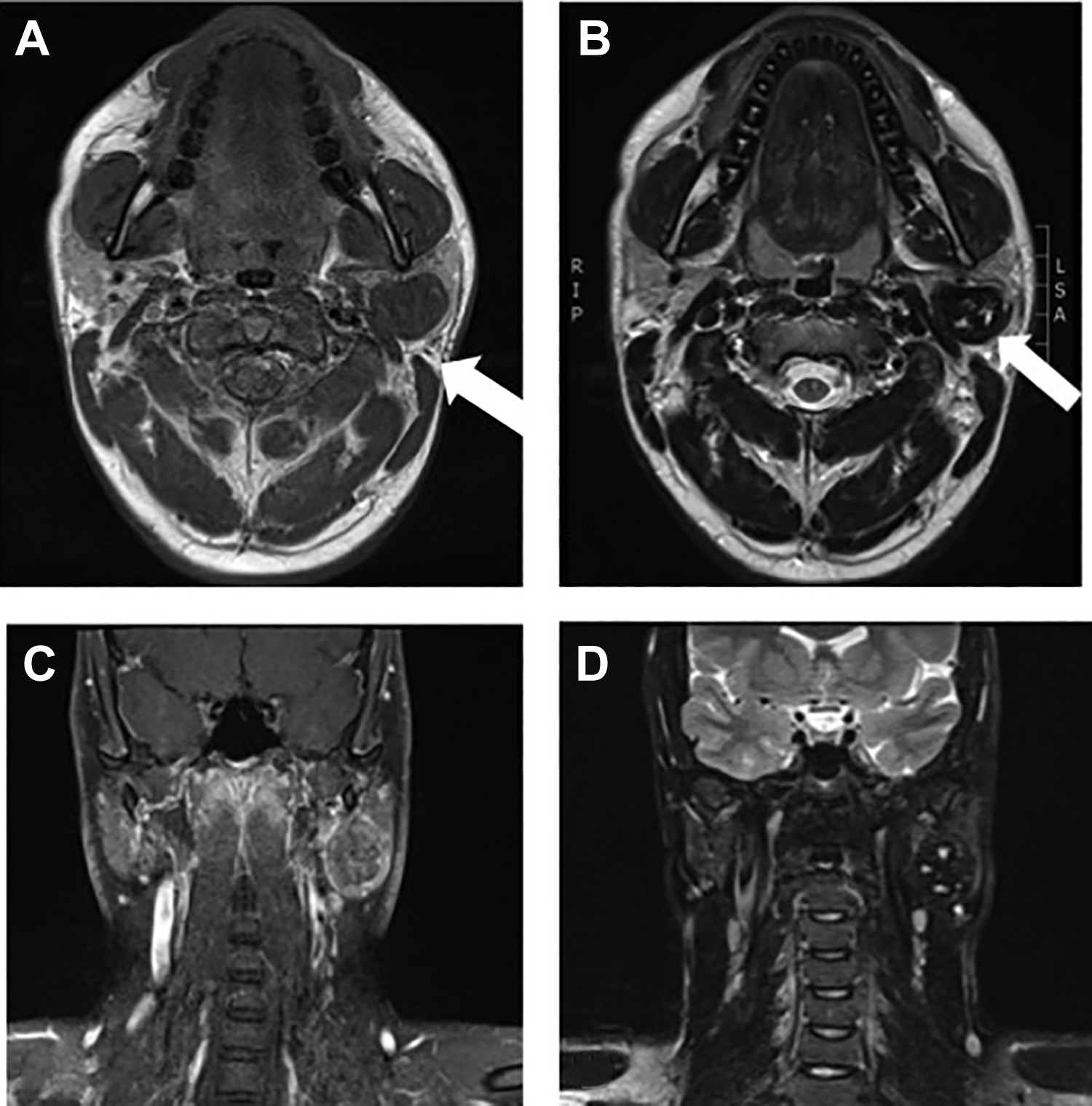
Magnetic resonance imaging. A, Axial T1W image revealed a hypointense mass in the left parotid gland involving the deep and superficial lobes (arrow). B and C, Axial and coronal T2W images, respectively, revealed hyperintensities suggesting ductal dilations with apparent hypointensities best seen on CT (not shown). D, Postcontrast coronal T1W image revealed minimally heterogenous enhancement of the parotid mass more pronounced in the peripherally consistent with chronic inflammatory reaction. CT indicates computed tomography.
The patient underwent a subtotal parotidectomy with facial nerve preservation to excise the lump and obtain definitive histology. Intraoperatively, the tumor presented as a hard, white mass, resembling fibrotic scar tissue. It was found at the junction between the superficial and deep lobe of the parotid gland, surrounding the main trunk of the facial nerve, with multiple adhesions and no plane of dissection between the nerve and the tumor. A decision was made to leave a thin cuff of tumor around the nerve, preserving the nerve entirely. The neural function was tested with a nerve stimulator (Neurosign) at the end of the procedure and it was intact. The postoperative period was uneventful, with no neuropraxia, and the patient was discharged after 2 days after removal of the drain.
Histology confirmed features consistent with an end-stage sclerosing inflammatory process or Küttner tumor with no features to suggest malignancy. The specimen was however negative for IgG4 immunohistochemistry staining (Figures 2 and 3).
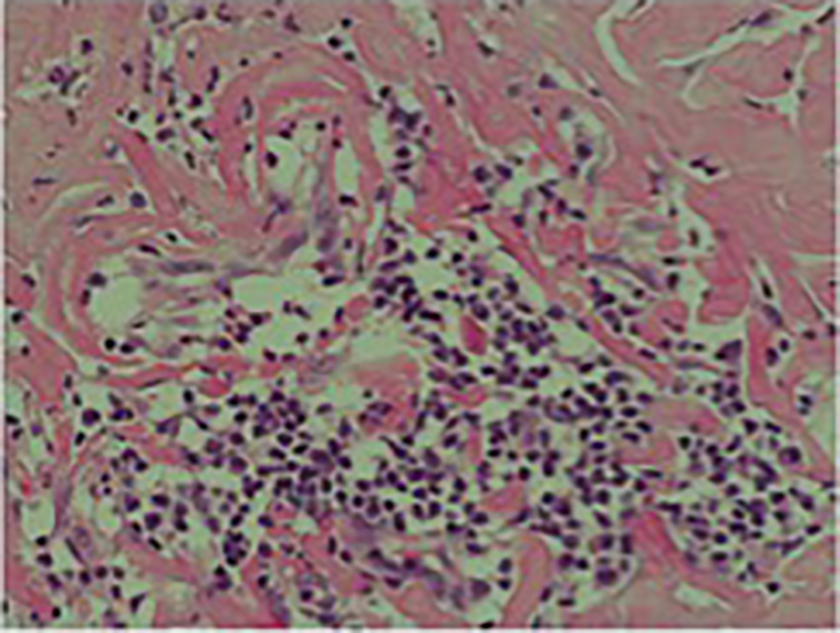
Dilated ducts, some of which were ruptured containing inspissated, inflamed intraluminal material.
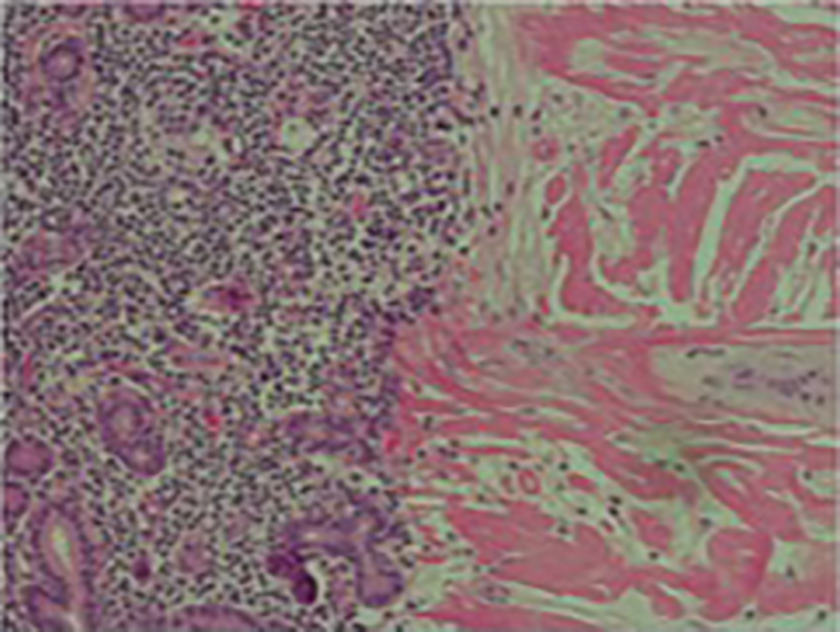
Lymphoid aggregates and numerous plasma cells.
Chronic sclerosing sialadenitis was first described by Küttner in 1896 as a benign inflammatory condition, usually affecting the submandibular glands. Clinically, it can present in a similar manner to neoplastic lesions of the salivary glands and therefore is classified as a tumor-like lesion of the salivary gland by the World Health Organization. 1,2 Chronic sclerosing sialadenitis or Küttner’s tumor usually present in individuals with a history of chronic inflammation such as recurrent sialoliths or salivary gland infections or in individuals with autoimmune inflammatory disorders such as IgG4-positive disease. 3,4
Parotid gland involvement, as in the case we present, is incredibly rare. We therefore conducted a literature review with Medline, Embase, and Cochrane library databases using the search terms “parotid,” “Küttner tumour,” and/or “chronic sclerosing sialadenitis.” Only full texts were considered for review. We identified 8 case reports of Küttner tumor affecting the parotid gland 2,5 –9 and 1 case series of 7 patients involving 1 patient with bilateral parotid involvement 9 and 1 case series of 4, one of which had parotid gland involvement. 10
One theory of Küttner tumor formation may be in the presence of sialoliths. 4,6 The rarity of Küttner tumors occurring in the parotid glands may therefore be directly proportional to the frequency of obstructive processes, such as calculi occurrence in parotids being much lower compared to submandibular glands. This is believed to be due to the secretions within the submandibular gland being more mucoid and less serous than that of the parotid glands, making stone formation more likely. 5,8
A second group of patients are those affected by systemic fibrosis where it may be that multiple glands may be susceptible. This entity of chronic sclerosing sialadenitis and associated IgG4-positive plasma cell infiltration can be found in patients with Sjögren syndrome, chronic sclerosing inflammatory fibrosis, and sialadenitis. 11,12
A third theory proposed by Seifert and Donath may explain the spontaneous occurrence of Küttner tumor in what appears to be a fit and well individual with no history of chronic salivary gland inflammation or salivary gland stones, as in our case presentation. 4 Siefert proposed that intracellular electrolyte disturbances within the salivary ducts leads to mucus plugging. These mucus plugs in turn obstruct the smaller ducts, thus triggering a local inflammatory reaction and the formation of Küttner tumor in such rare instances.
Both fine needle aspiration biopsy and core biopsy histology are unreliable in the diagnosis. Imaging may show heterogenous texture on MRI due to scarring with hypointense areas due to duct dilatation with areas of hyperintensity due to calcification. 13 These features are however not exclusive to a chronic sclerosing inflammatory process and may be confused with malignancy. 14 Therefore, surgery in form of a partial parotidectomy with preservation of the facial nerve should be considered the only definitive diagnostic method. 10
Footnotes
Authors’ Note
Material was presented at National Congress Italian Society of Otorhinolaryngology Head and Neck Surgery, Rimini, May 31, 2019.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
