Abstract
Background:
Cisplatin-induced ototoxicity is related to oxidative stress. Astaxanthin is one of the most powerful antioxidants in nature.
Aims/objectives:
To investigate the protective effect of astaxanthin on cisplatin-induced ototoxicity.
Materials and Methods:
Thirty-five Sprague Dawley female rats were divided into 5 groups: control, cisplatin, and cisplatin with 10, 20, and 40 mg/kg astaxanthin groups. Cisplatin group received a single intraperitoneal injection of 14 mg/kg cisplatin. While saline was administered in the control group, in the other 3 groups, 10, 20, and 40 mg/kg daily doses of astaxanthin were administered through orogastric cannula before administration of cisplatin. Baseline and 10th day otoacoustic emission tests were administered. An intracardiac blood sample was taken to measure total antioxidant capacity (TAC), and the cochleas of the animals were investigated histopathologically.
Results:
Hearing level of astaxanthin 40 mg/kg + cisplatin group was higher at 24 kHz and 32 kHz frequencies compared to the cisplatin group. The TAC value of the cisplatin group was lower than both the control and astaxanthin + cisplatin groups (P < .05). On histopathological examination, the other groups were deformed compared to the control group, but no statistically significant difference was observed between the astaxanthin + cisplatin and cisplatin groups.
Conclusions and significance:
Astaxanthin showed protective effect at high frequencies when it was administered at high dose. Thus, astaxanthin may have protective effect against cisplatin-induced ototoxicity.
Introduction
Cisplatin is a widely used chemotherapeutic agent in the treatment of a number of human cancers including testicular, ovarian, lung, and urinary bladder carcinomas in addition to head and neck cancers. Ototoxicity, nephrotoxicity, and neurotoxicity are dose-limiting side effects of cisplatin. 1 While these side effects cause comorbidities affecting patient’s quality of life, they also lead to difficulty in achieving desired efficacy in cancer treatment due to dose limitation and treatment interruptions. There have been multiple publications related to cisplatin-induced ototoxicity, ototoxicity mechanisms, and the methods that can be used for the prevention of cisplatin-induced ototoxicity in the literature. However, a routine protocol could not be established yet for the prevention of cisplatin-induced ototoxicity. 2
It is known that cisplatin-induced ototoxicity occurs through the production of reactive oxygen species (ROS). When the excessive generation of ROS occurs, it activates proapoptotic pathways, and consequently, cell damage and cell death occur.
Astaxanthin is known as one of the most powerful antioxidants in nature, and it is a red carotenoid pigment belonging to the group of xanthophyll carotenoids. It is plentifully present in a wide variety of microorganisms such as Haematococcus pluvialis as well as in seafood, such as salmon, trout, and shrimps. 3
Antioxidant activities of astaxanthin are higher compared to the other known antioxidants. According to the study performed by Zhang et al, ROS scavenging capacity of astaxanthin is 6000 times higher than vitamin C, 800 times higher than coenzyme Q10, 550 times higher than vitamin E, 200 times higher than polyphenol, 150 times higher than anthocyanin, and 75 times higher than α-lipoic acid. 3 It has also antiapoptotic, anti-inflammatory, neuroprotective, anticarcinogenic, antidiabetic, and antiobesity activities. 4,5
In our study, we aimed to investigate the possible protective effect of astaxanthin against cisplatin-induced ototoxicity using otoacoustic emission measurements, plasma total antioxidant capacity (TAC) level, and histopathological examination results of the cochlea.
Methods
Animals
The protocol of this experimental study was approved by local animal care and ethics committee. Thirty-five 6-week-old Sprague Dawley healthy female rats weighting 220 to 240 g were used in this study. The animals were housed as groups including 4 rats in a room where the temperature was set to 21°C, with free access to pellet food and tap water (ad libitum) with 12-hour light–dark cycles and a background noise level less than 50 dB.
Materials
During the study, ketamine hydrochloride (Ketalar 500 mg/10 mL, İstanbul, Turkey; Pfizer) for anesthesia, cisplatin (Cisplatin Ebewe 100 mg/100 mL, İstanbul, Turkey; Liba) as ototoxic drug, and astaxanthin (Astaxanthin 2% powder; Nootrabiolabs, Jupiter, Florida) as an antioxidant were used. A solution was prepared by diluting 10 g of astaxanthin 2% powder with 100 mL physiological saline. Solutions prepared were kept in a lightproof container. A solution containing astaxanthin 10 g 2% in 100 mL, that is, 200 mg in 100 mL, was administered to the animal groups in an appropriate amount through orogastric gavage at a dose equal to 10, 20, or 40 mg/kg according to the body weight of the animal. OtoPhyLab ABR DP-gram (RT Conception, Ferrand, France) device was used for otoacoustic emission measurements.
Study Design
Thirty-five Sprague Dawley rats were divided into 5 groups as follows: Control group: Only physiological saline was administered through orogastric gavage from the first day to the seventh day of the experiment. The distortion product otoacoustic emission (DPOAE) measurements were performed on the 1st and 10th days of the study. Cisplatin group: A single intraperitoneal injection of 14 mg/kg cisplatin was administered on the seventh day of the experiment. The DPOAE measurements were performed on the 1st and 10th days of the study.
6
Cisplatin + 10 mg/kg astaxanthin group. Cisplatin + 20 mg/kg astaxanthin group. Cisplatin + 40 mg/kg astaxanthin group.
In cisplatin + astaxanthin groups, 3 different doses of astaxanthin were administered as 10, 20, or 40 mg/kg through orogastric gavage from the first day to the seventh day of the experiment. Additionally, a single intraperitoneal injection of 14 mg/kg cisplatin was administered on the seventh day of the experiment in these groups. The DPOAE measurements were performed on the 1st, 7th, and 10th days of the study. The purpose of DPOAE measurement on the seventh day was to control astaxanthin effect on hearing. Anesthesia was administered only during DPOAE measurements. However, it was not administered during orogastric gavage administrations and intraperitoneal injections.
Otoacoustic Emission Measurements
The baseline DPOAE measurements were performed on the first day of the study. Only animals demonstrating a normal otoscopic examination and the ones determined to have a normal hearing test during DPOAE measurements were included in the study. The DPOAE measurements were performed by inserting a newborn probe into the rats’ right outer ear canal. After ensuring the device was in the appropriate measurement position, measurements were begun to be performed. The calibration was made automatically by the device before evaluation of each frequency. The rate between f2 and f1 was fixed at f2/f1 = 1.22. The DPOAE measurements were performed at frequencies of 6000, 8000, 12 000, 16 000, 24 000, and 32 000 Hz, and the results were recorded as distortion product gram.
Measurement of TAC
On the 10th day of the study, an intracardiac blood sample was taken from each animal after completion of the final auditory assessment. Blood samples were collected into evacuated tubes containing heparin and then centrifuged at 3000 rpm + 4°C for 10 minutes. Thereafter, the supernatant plasma was separated, and it was studied with cupric ion reducing antioxidant capacity (CUPRAC) method. The value of the TAC of each plasma sample was calculated in mM/Trolox equivalent (mM/TE).
Histopathological Examination of Cochlea
After taking intracardiac blood samples, the animals were killed by using a guillotine with a sharp blade under ether anesthesia. Then the head of the animal was divided into 2 pieces in the sagittal plane, and the soft tissues around the temporal bone were removed under an examination microscope. The cochlea was reached by opening the tympanic bulla, and it was removed by dissecting from the surrounding tissues.
After tissue processing for light microscopic investigations, the samples were embedded in paraffin. The initial sections obtained from tissue blocks were examined and stored under light microscope, and they are mainly the cross sections from the basal turn of the cochlea. Damage assessment in the samples was performed with scoring system defined by de Freitas et al. The outer hair cells were scored according to cell integrity, arrangement, nucleus, and cytoplasm characteristics as follows: normal: 0; mild damage: 1; intermediate damage: 2; and severe damage: 3. Evaluation of stria vascularis was performed according to marginal cell blebbing, cytoplasmic vacuolization, and atrophy of the intermediate cells (shrinkage) as follows: normal stria vascularis thickness and absence of marginal cell blebbing, cytoplasmic vacuolization, and atrophy of intermediate cells: 0; mild changes: 1; moderate changes: 2; severe changes: 3. Also, spiral ganglion cells were assessed according to vacuolization and findings of nuclear damage (no change: 0; mild changes: 1; moderate changes: 2; severe changes: 3). 7
Statistical Analysis
IBM SPSS Statistics 22 program was used for the statistical analysis. Conformity of the parameters to the normal distribution was assessed by the Shapiro-Wilk test. Kruskal-Wallis test was used for intergroup comparisons of parameters not normally distributed. Dunn test was used for binary comparisons of the groups. Significance was evaluated at a level of P < .05.
Results
The Results of Otoacoustic Emissions
When the mean signal to noise ratios (SNRs) of otoacoustic emissions of all study groups on the first day of the study were compared, no significant difference was observed between groups at all frequencies (P > .05). It showed that there was no difference between the groups according to hearing levels at the beginning of the study.
Otoacoustic emission results of the control group were statistically significantly higher compared to all the other groups in terms of the mean SNRs on the 10th day of the study (P < .05). There was no statistically significant difference between the results of otoacoustic emission of astaxanthin 10 mg/kg + cisplatin and astaxanthin 20 mg/kg + cisplatin groups and the results of the cisplatin group (P > .05). While otoacoustic emission results of astaxanthin 40 mg/kg + cisplatin group were numerically higher compared to the cisplatin group at all frequencies, the difference was statistically significant at only 24 kHz and 32 kHz frequencies (P < .05; Figure 1).
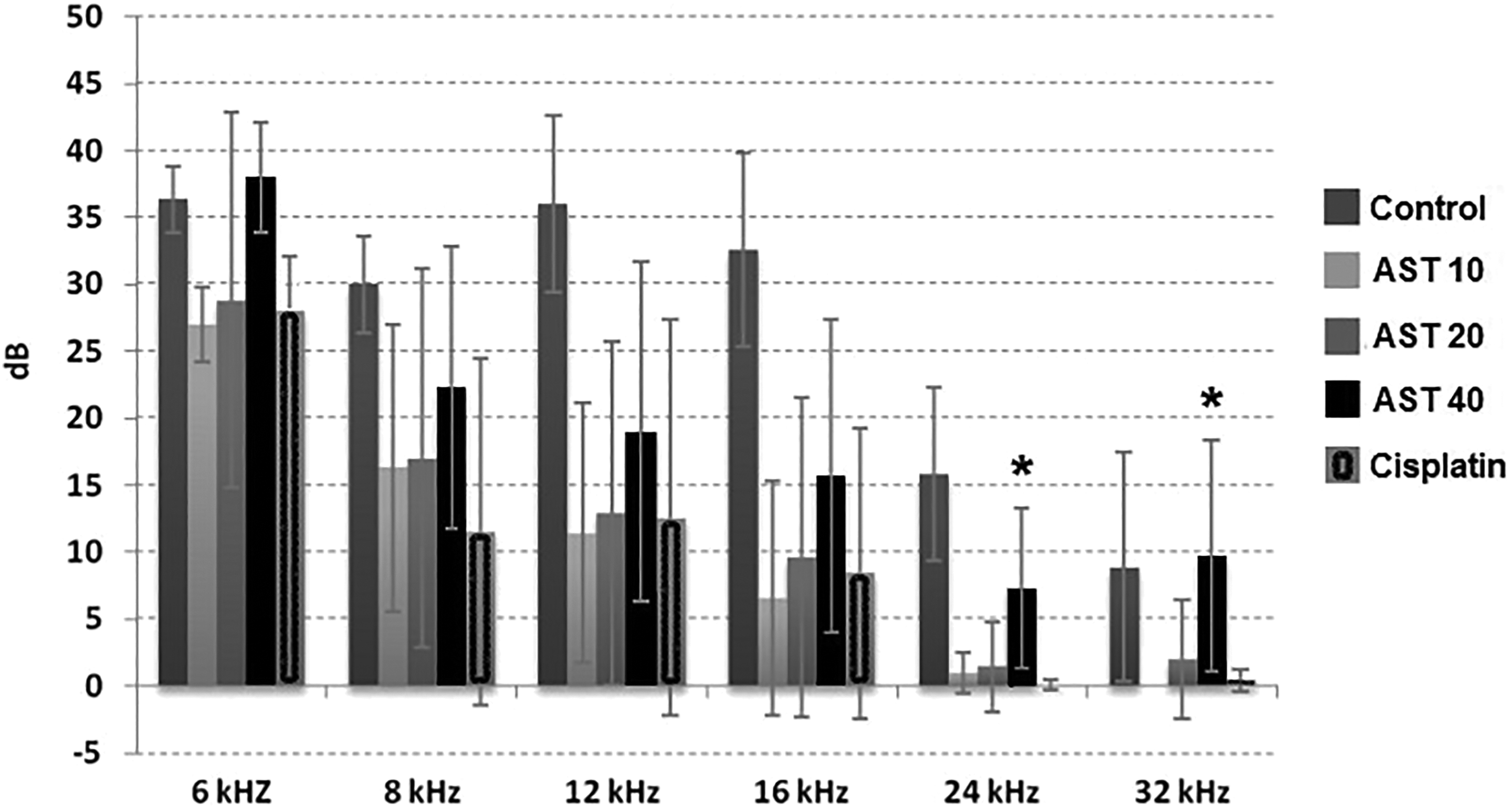
The results of otoacoustic emission measurements of all study groups on the 10th day of the study (ASTA10: Astaxanthin 10 mg/kg + Cisplatin group, ASTA20: Astaxanthin 20 mg/kg + Cisplatin group, ASTA40: Astaxanthin 40 mg/kg + Cisplatin group. Statistically significant values are shown as “*.”
Another assessment performed regarding study results was the calculation of measurement differences in the SNR of otoacoustic emissions between the 10th and the 1st day. Within the scope of this assessment, the difference between the 10th day SNR value and 1st day SNR value was calculated at each frequency for each animal, and the average of these differences was taken. So, the average of the differences between the 10th and the 1st day at each frequency was determined for each group. Accordingly, while the maximum difference was at 12 and 16 kHz frequencies, SNR differences of all other groups at frequencies of 12, 16, and 24 kHz were found to be statistically significantly higher compared to the control group (P < .05; Figure 2).
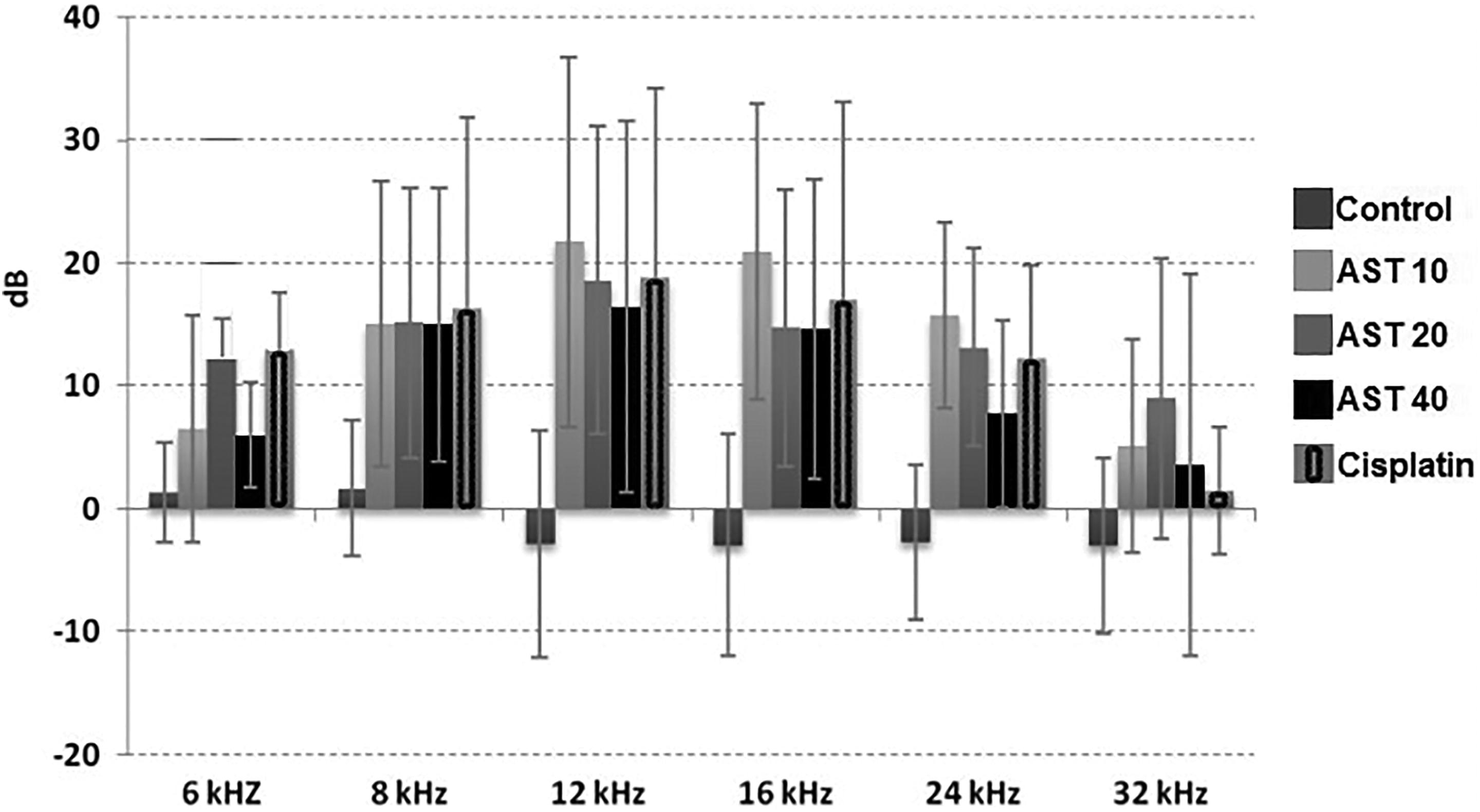
Average of otoacoustic emission signal to noise ratio (SNR) differences between the 10th and the 1st day.
The Results of TAC
While mean TAC value of the control group was 0.095 ± 0.021 mM/TE, the mean TAC values of cisplatin + astaxanthin 10 mg/kg group, cisplatin + astaxanthin 20 mg/kg group, cisplatin + astaxanthin 40 mg/kg group, and cisplatin group were measured to be 0.090 ± 0.022 mM/TE, 0.090 ± 0.010 mM/TE, 0.099 ± 0.021 mM/TE, and 0.065 ± 0.011 mM/TE, respectively. The TAC value of the cisplatin group was significantly lower than the control group. Similarly, there was statistically significant difference between the cisplatin group and astaxanthin 40 mg/kg + cisplatin group (P < .05).
Although TAC values between astaxanthin groups and control group were not significantly different, mean TAC value in the cisplatin + astaxanthin 40 mg/kg group was numerically higher than the control group, and the TAC values of low-dose astaxanthin groups were lower than the control group. According to these results, astaxanthin may have some effect to prevent decrease in TAC caused by cisplatin in a dose-dependent manner.
The Results of Histopathology
In light microscopic examination of the sections taken from the cochleas of all animals, averages of outer hair cell, stria vascularis, and spiral ganglion scores in the control group were found to be statistically significantly less than the averages of the astaxanthin 10 mg/kg + cisplatin, astaxanthin 20 mg/kg + cisplatin, and cisplatin groups (P < .05). While the astaxanthin 40 mg/kg + cisplatin group is the one coming very close to the control group numerically, the difference between this group and the other groups receiving cisplatin was not found to be statistically significant (P > .05). There was no statistically significant difference between the 3 groups receiving astaxanthin (P > .05; Figures 3 –5).
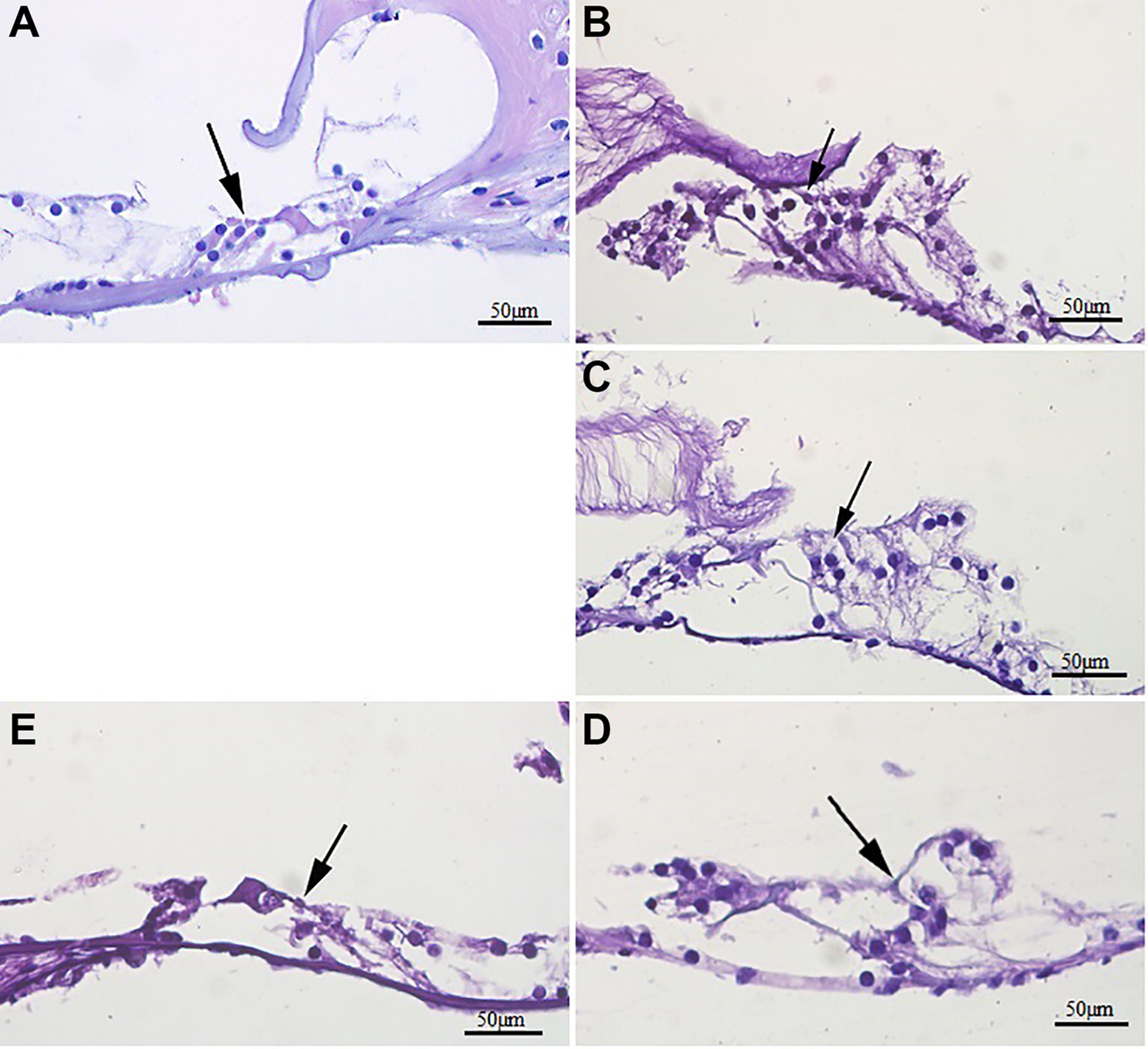
Light microscopic appearance of organ of Corti of 1 animal from each group. Normal histology of Corti is seen in the control group. In the cisplatin + astaxanthin and only cisplatin groups, cell integrity is deteriorated, and severe damage is seen at nucleus and cytoplasm of the cells (Arrow: outer hair cells). A, Control group, example of score 0. B, Astaxanthin 40 mg/kg + cisplatin group, example of score 1. C, Astaxanthin 20 mg/kg + cisplatin group, example of score 2. D, Astaxanthin 10 mg/kg + cisplatin group, example of score 2. E, Cisplatin group, example of score 3 (hematoxylin and eosin stain).
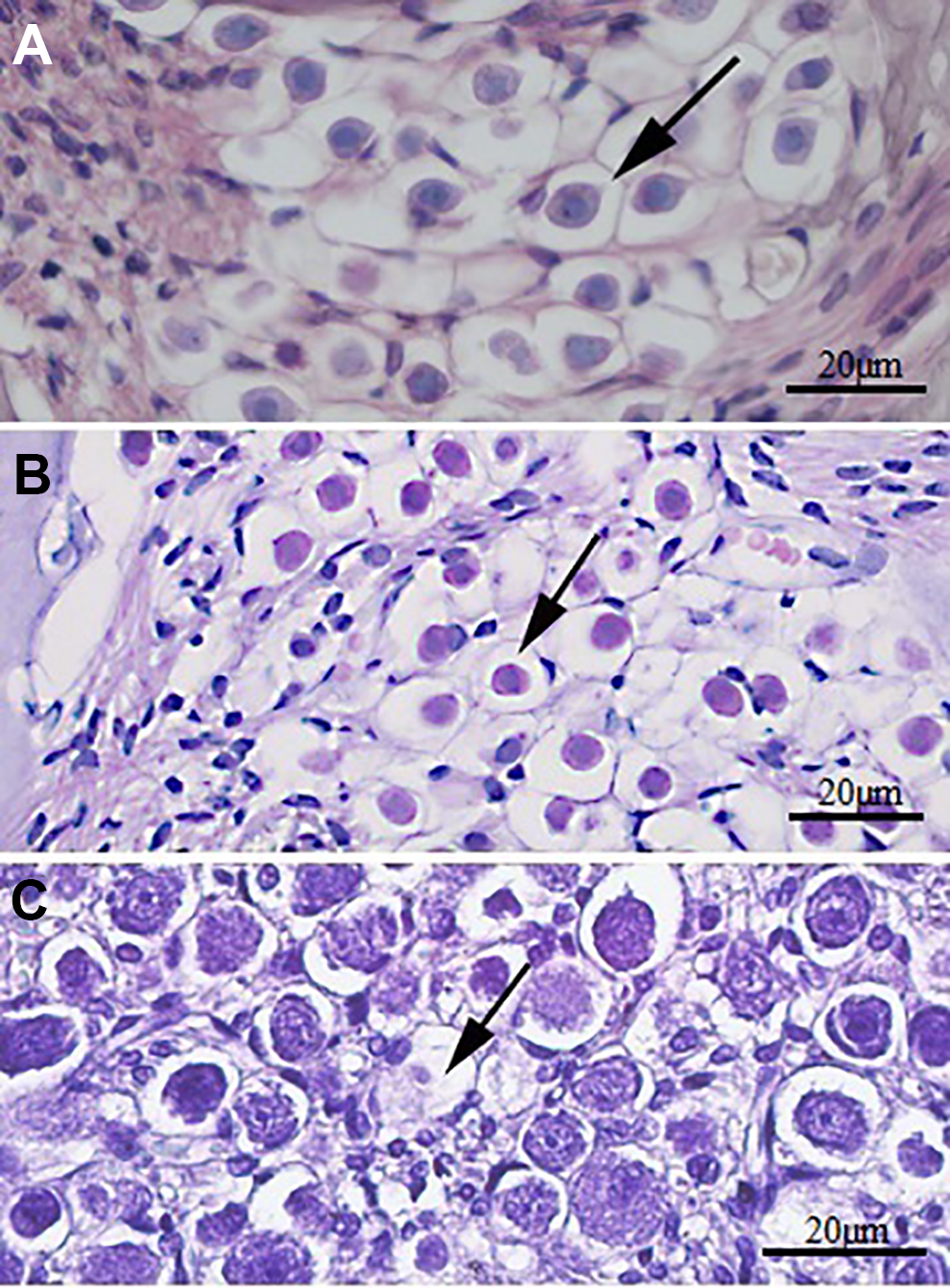
Histopathological appearance of spiral ganglion cells. Normal histology of spiral ganglion cells is seen in the control group. Cisplatin groups have vacuolization and findings of nuclear damage. A, Control group, example of score 0. B, Astaxanthin 40 mg/kg + cisplatin group, example of score 0. C, Cisplatin group, example of score 3. Hematoxylin and eosin stain.
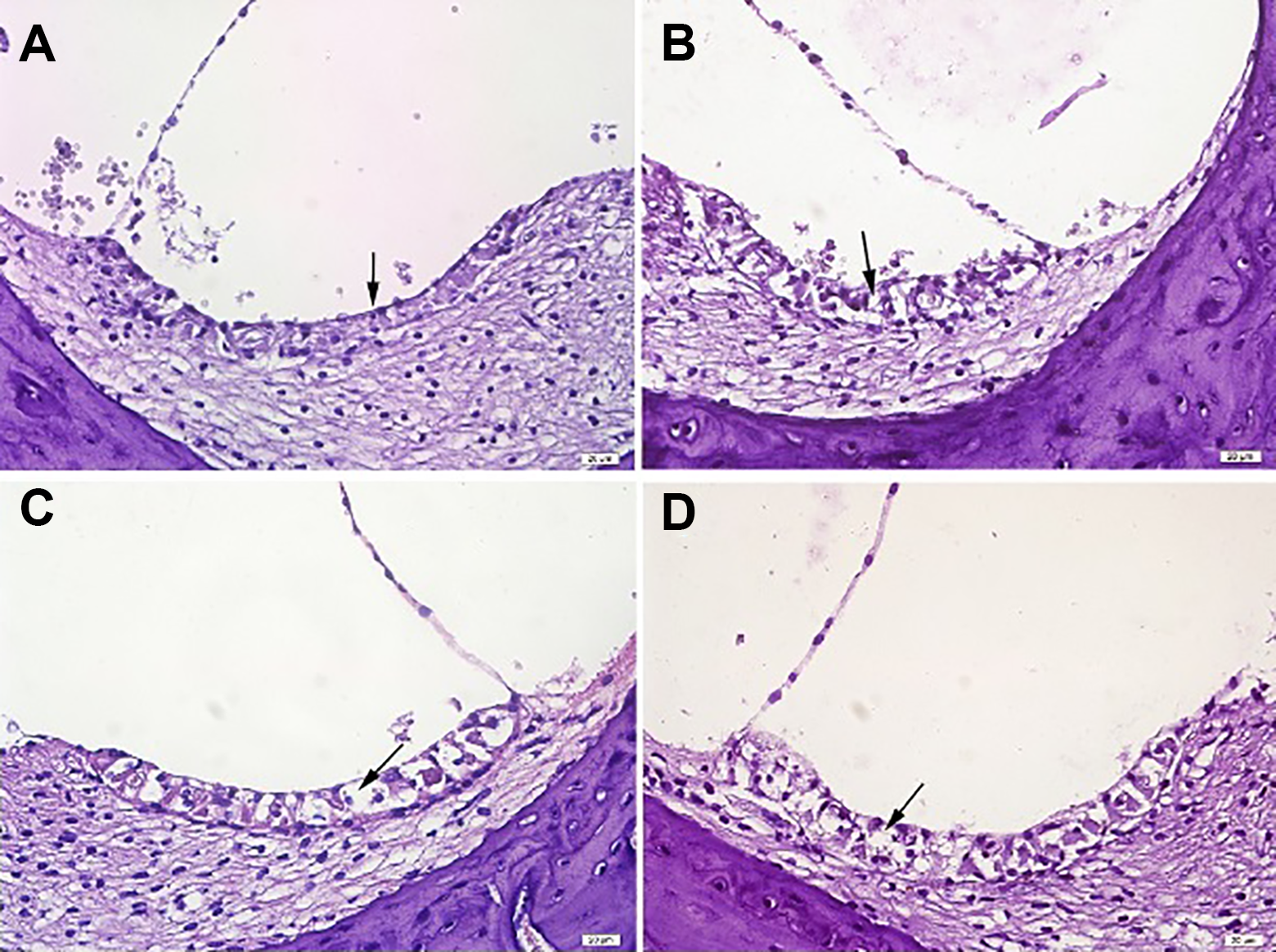
Histopathologic appearance of stria vascularis cells. Control group’s cochlea sections show normal histology. Cytoplasmic vacuolization and atrophy of intermediate cells are seen at the astaxanthin + cisplatin groups. A, Control group, example of score 0. B, Astaxanthin 10 mg/kg + cisplatin group, example of score 2. C, Astaxanthin 20 mg/kg + cisplatin group, example of score 1. D, Astaxanthin 40 mg/kg + cisplatin group, example of score 2. Hematoxylin and eosin stain.
Discussion
Cisplatin-induced ototoxicity is usually manifested by bilateral and irreversible sensorineural hearing loss. It can also be observed as a sudden and profound hearing loss after high-dose chemotherapy. Cisplatin-related hearing loss appears first only at high frequencies. However, when the therapy continues, lower frequencies may also be affected. 6,8
Although the molecular mechanism of cisplatin-induced ototoxicity has not been demonstrated exactly, there are various theories suggested on this subject. First, a simple explanation of the mechanism for cisplatin ototoxicity is the acute and chronic generation of ROS in the cochlea in addition to DNA damage caused by the agent. This excessive generation of ROS leads to the depletion of the cochlear antioxidant enzymes. Since ROS cannot be neutralized sufficiently in this condition, lipid peroxidation increases. Oxidative modifications of proteins occur. Pro-inflammatory cytokine formation and superoxide generation increase in the cochlea. This uncontrolled increase in generation of ROS causes the activation of proapoptotic pathways. Consequently, cell death occurs through cell damage and apoptosis. 9
Cisplatin dose administered to produce ototoxicity in the rats was determined to be a single dose of intraperitoneal cisplatin in a dose of 12 to 14 mg/kg in many studies available in the literature. 10,11 Also in our study, cisplatin was administered intraperitoneally in a single dose of 14 mg/kg.
Numerous studies have been published in the literature related to cisplatin-induced ototoxicity. In these studies, a lot of different antioxidants were studied against cisplatin-induced ototoxicity. Some of these antioxidants are N-acetyl cysteine (NAC), sodium thiosulfate (STS), amifostine, lipoic acid, resveratrol, intratympanic administration of dexamethasone, intratympanic administration of Ringer lactate, erdosteine, α-tocopherol, chrysin, and quercetin.
1,12
-15
Especially, antioxidants such as NAC, α-tocopherol, lipoic acid, STS, salicylate, ebselen,
Astaxanthin used in our study is a red carotenoid pigment belonging to the xanthophyll group of carotenoids. Astaxanthin was used in many studies in the literature and is known to be one of the most powerful antioxidants in nature. However, there is no study in the literature regarding the use of astaxanthin in ototoxicity. To the best of our knowledge, our study is the first one in the literature investigating the efficacy of astaxanthin in cisplatin-induced ototoxicity.
In the study performed by Zhang et al, the authors compared all kinds of mitochondrion-permeable antioxidants (edaravone, idebenone, lipoic acid, carotenoids, coenzyme Q, MitoQ, and SkQ) and proposed astaxanthin to be the best antioxidant effective in ROS-mediated acute diseases. 3
People usually use antioxidants by oral administration. Oral route is the safest and the most cost-effective administration. In a previous study investigating the protective effect of astaxanthin against arsenic-induced nephrotoxicity, astaxanthin was administered to the rats for 5 days through gavage at low dose (10 mg/kg), normal dose (20 mg/kg), and high dose (40 mg/kg) once a day. 17 Also in our study, astaxanthin was administered in the same doses and same route for 7 days once a day.
It is known that drug toxicities affect primarily the outer hair cells in the organ of Corti. While normal otoacoustic emissions show that the outer hair cells are healthy, deterioration in otoacoustic emissions reflect injuries to the outer hair cells. The most commonly used otoacoustic emissions in clinical practice are transient-evoked otoacoustic emission (TEOAE) and DPOAE. It has been demonstrated by the studies performed that DPOAE was more sensitive than TEOAE for early detection of cisplatin-induced hearing loss. 18 In our study, DPOAE was used for audiologic monitoring of ototoxicity, and high frequencies were included in the assessment.
Evaluation of the results of otoacoustic emissions was based on SNR in our study. The SNR value is obtained by subtracting the ambient noise amplitude from the DPOAE amplitude. Studies show that the SNR value is more reliable than DPOAE amplitude in the assessment of DPOAE responses. 19 -21
According to “The Guideline for the Audiologic Management of Individuals Receiving Cochleotoxic Drug Therapy” published by the American Speech-Language-Hearing Association in 1994, prospective studies using high-frequency audiometry have demonstrated that cochleotoxicity can occur initially or solely in the high frequencies ranging between 9 and 20 kHz. 22 In many studies performed, it was demonstrated that ototoxic agents affected high frequencies above 8000 Hz before affecting the speech frequencies below 8000 Hz, and the importance of high-frequency audiometry and DPOAE tests in the early diagnosis was highligted. 23 When the studies related to ototoxicity in the literature are investigated, it is seen that the frequencies below 8000 Hz are examined in most of these studies. In our study, the frequencies between 6000 Hz and 32 000 Hz were examined using DPOAE test, and it was observed that hearing loss occurred at high frequencies due to cisplatin consistent with the literature. One of the important aspects of our study is the evaluation of high frequencies beginning from 6000 Hz to 32 000 Hz.
Dreschler et al reported that early diagnosis of ototoxicity can be made with high-frequency audiometry, and most sensitive frequencies for ototoxicity diagnosis were frequency range of 12 000 to 14 000 Hz. 24 Similarly, in our study, we found that maximum SNR differences in otoacoustic emission measurements on the 1st day and the 10th day occurred at 12 kHz and 16 kHz frequencies in the groups administered cisplatin.
We also performed otoacoustic emission tests on the seventh day in the groups administered astaxanthin before cisplatin administration, and the evaluations confirmed that astaxanthin exerts no negative effect on hearing.
Measurement of TAC is a rational and reliable way in order to reveal oxidative stress changes. The TAC can be used to evaluate the conditions affecting in vivo oxidative status such as exposure to ROS and galenic antioxidant supplementation. Furthermore, changes in the plasma TAC values may provide information on the absorption and bioavailability of antioxidant supplement. 25
According to the results of measurement of TAC in our study, it was observed that oral astaxanthin exerted its effect on the circulation. We detected that while TAC level of cisplatin group was significantly lower than other groups, TAC levels were not different between the control and cisplatin + astaxanthin groups (P > .05). We can say that astaxanthin could prevent decrease in TAC level when it was administered with cisplatin.
In our study, while SNRs between the low-dose astaxanthin (10 and 20 mg/kg) + cisplatin and cisplatin groups were not different, numerically higher SNR levels in the astaxanthin 40 mg/kg + cisplatin group than the cisplatin group at all frequencies were remarkable (P > .05). In addition, SNRs in the highest dose astaxanthin + cisplatin group were significantly higher than the cisplatin group at 24 and 32 kHz (P < .05). According to these results, we can say that oral high-dose administration of astaxanthin may have protective effect against cisplatin ototoxicity in high frequencies.
Cisplatin exerts its ototoxic effect by targeting 3 structures in the cochlea. First, the hair cells of the organ of Corti in the basal turn of the cochlea; second, the spiral ganglion cells; and third, the lateral wall tissues (stria vascularis and spiral ligament). All of the outer hair cells, cells in the stria vascularis, and spiral ligament undergo apoptosis, and platinated DNA immunoreactivity occurs in the nuclei of these cells. 12,26 In our study, although decrease in cisplatin-induced cochlear degeneration with increasing doses of astaxanthin was detected, it was not statistically significant.
Conclusion
We have several results in our study: * Astaxanthin has no negative effect on hearing at ≤40mg/kg doses. * Astaxanthin can prevent the decrease in plasma TAC level when it is administered together with cisplatin. * The most susceptible frequencies to cisplatin ototoxicity are 12 to 16 kHz frequencies. * Astaxanthin may have protective effect against cisplatin ototoxicity when it is administered only at higher doses.
Although these results are promising, we can say that new studies with longer study periods and larger number of animals using higher doses of astaxanthin are necessary.
Footnotes
Authors’ Note
This study was presented at the 39th National Turkish Otolaryngology and Head & Neck Surgery Congress, Antalya, Turkey, November 8-12, 2017.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
