Abstract
Objective:
Sinonasal inverted papilloma (SNIP) is a benign tumor characterized by an aggressive growth, a tendency to recur, and an association with malignancy. However, the precise etiology of SNIP is still unknown. The objective of this study was to identify the expression pattern of speckled protein 100 (Sp100) in the malignant transformation (MT) of SNIP and its correlation with human papillomavirus (HPV)-16 and HPV-18 infections and other clinical features. This would further help in understanding the possible mechanisms for the development of SNIP.
Methods:
Individual nasal mucosa specimens from 40 patients (25 males and 15 females) and 10 inferior turbinate specimens as controls were included in the present study. The samples were divided into several sections for histopathological examination, HPV DNA detection, and immunohistochemical staining.
Results:
We observed that as SNIP progressed, the Sp100 protein expression was gradually downregulated, and SP100 localization changed from nucleus to the cytoplasm. Positive rate of HPV infection in the SNIP with MT group was higher than that in the other groups, and Sp100 expression was correlated to HPV infections and SNIP with MT. However, no correlation was observed between Sp100 expression and clinical features, such as age, gender, and smoking.
Conclusion:
Positive rate of HPV infection is high in the SNIP with MT and has a correlation with Sp100 expression. In addition, the expression of Sp100 is downregulated in SNIP with MT, and Sp100 may play a role in the progression of SNIP.
Keywords
Introduction
Sinonasal inverted papilloma (SNIP) is a benign epithelial tumor that occurs in the nasal cavity and paranasal sinuses 1 of adults, usually during the fifth decade of life. The tumor is characterized by a local aggressive growth, tendency to recur, and an association with malignancy. 2 Sinonasal inverted papilloma has been observed in 0.4% to 7% of patients with sinonasal cavity tumors 3,4 Malignant transformation (MT) has been found in 5% to 15% of the inverted papilloma lesions. 5 Although the precise etiology of SNIP is still unknown, high-risk human papilloma virus (HPV) is regarded to be a potential etiological agent in the MT of SNIP. 1 Especially, high-risk HPV subtypes—HPV-16 and HPV-18—are more frequently found in dysplastic and malignant transformed SNIP. 6,7
Speckled protein 100 kDa (Sp100) is a major constituent of promyelocytic leukemia protein nuclear body (PML-NB) with a stimulatory effect on p53-dependent gene expression, 8 which has been reported to inhibit the invasion of breast cancer cells, 9 human glioma cells, and laryngeal cancer cells. 10 The Sp100 is also believed to have implications in inhibiting HPV replication and transcription in maintenance and differentiation-dependent replication. 11 However, little is known about the role of HPV in SNIP.
In the present study, we aimed to identify the expression of Sp100 in the MT of SNIP, and its relationship with HPV (HPV-16 and HPV-18) infections and other clinical features.
Materials and Methods
Participants
A total of 40 individual nasal mucosa specimens (25 males and 15 females, age: 26-72 years, mean age: 51.17 years) were obtained from patients treated in our hospital between September 2009 and August 2016. The histopathological examination confirmed the diagnosis of SNIP. None of the patients underwent chemotherapy or radiotherapy prior to the surgery. Ten inferior turbinates diagnosed with normal nasal mucosa as control specimens, 18 specimens diagnosed as SNIP, 15 specimens diagnosed as SNIP with dysplasia and 7 specimens diagnosed as SNIP with MT were included in the study.
Samples were divided into several sections for histopathological examination, HPV DNA detection, and immunohistochemical staining. Written informed consent was obtained from all the patients. All the study protocols were approved by the ethics review committee of the first Hospital of China Medical University.
DNA Extraction and HPV DNA Detection
DNA was extracted from SNIP using TIANamp DNA Mini Kit (Tiangen, China). Fluorescence-based PCR analysis of HPV DNA was performed using High-risk Human Papilloma Virus PCR Kit (DAAN Gene Co., Ltd, Guangzhou, China) according to the manufacturer’s protocol. The Kit included a pair of HPV-specific primers, 1 single-stranded HPV-specific fluorescence probe, PCR reaction solution, and Taq DNA polymerase and deoxynucleoside triphosphates. It was capable of detecting 2 types of high-risk HPV DNA (HPV-16, HPV-18).
Immunohistochemistry
Sections of formalin-fixed, paraffin-embedded tissues were prepared for immunohistochemical analysis. Immunohistochemistry method was performed using the peroxidase-conjugated biotin–streptavidin method (SABC kit, Maixin, Wuhan, China) per manufacturer’s instructions. Following deparaffinization and rehydration, the specimens were incubated with goat anti-human Sp100 primary antibody (1:400; Santa Cruz Biotechnology, USA), and then incubated with the biotin-conjugated anti-goat secondary antibody (Maixin, Wuhan, China). Sections were counterstained with hematoxylin and cover slipped. Negative control was performed by incubation with phosphate-buffered saline instead of the primary antibody.
Two independent pathologists performed the procedure who were blinded to the clinical outcomes. The Sp100 protein expression is localized in the nucleus and cytoplasm and usually stains as granules (yellow to tan colored granules). The Sp100 expression was assessed semi-quantitatively as the percentage of cells with positive staining: 0, complete negative; +, less than 25%; ++, 25% to 50%; and +++, more than 50%. The final immunoreactive score was calculated by multiplying 2 parameters. A score of 5 or lower was considered to be a low expression of Sp100. The staining intensity was ranked from 0 to 3 with an increasing trend indicating absent, weak, moderate, and strong reactivity, respectively.
Statistical Analysis
Data were analyzed using SPSS 25 software. Statistical analysis was performed using χ2 test and Fischer exact test. A P value of .05 was used as the threshold for statistical significance.
Results
Human Papillomavirus DNA Detection
Based on HPV DNA detection, the HPV-positive rate in normal nasal mucosa was 0 of 10 (0%), 0 of 18 (0%) in SNIP, 1 of 15 (6.7%) in SNIP with dysplasia, and 3 of 7 (42.9%) in SNIP with MT (Table 1). On comparing the positive rate of HPV infection, statistical significant difference was observed between the 4 groups (P < .05, χ2 test) and the SNIP with MT was found to be highly HPV infected than the other groups (P < .05, Fischer exact test)
Presence of HPV in the Group of Normal Nasal Mucosa, SNIP, SNIP With Dysplasia, and SNIP with MT.

Abbreviations: HPV, human papillomavirus; MT, malignant transformation; SNIP, sinonasal inverted papilloma.
Sp100 Protein Expression
From immunohistochemistry staining, Sp100 expression was mainly observed in the nucleus with clustered distribution in normal mucosa and SNIP group, while cytoplasmic expression with diffuse distribution was seen in SNIP with MT. Figure 1 shows the staining pattern of Sp100 protein expression in the 4 groups. The Sp100 expression was highly intense in the normal nasal group, moderately expressed in the SNIP, low expressed in the SNIP with dysplasia group, while the lowest expression was seen in the SNIP with MT group. Thus, significant downregulation of Sp100 was found in SNIP with MT.
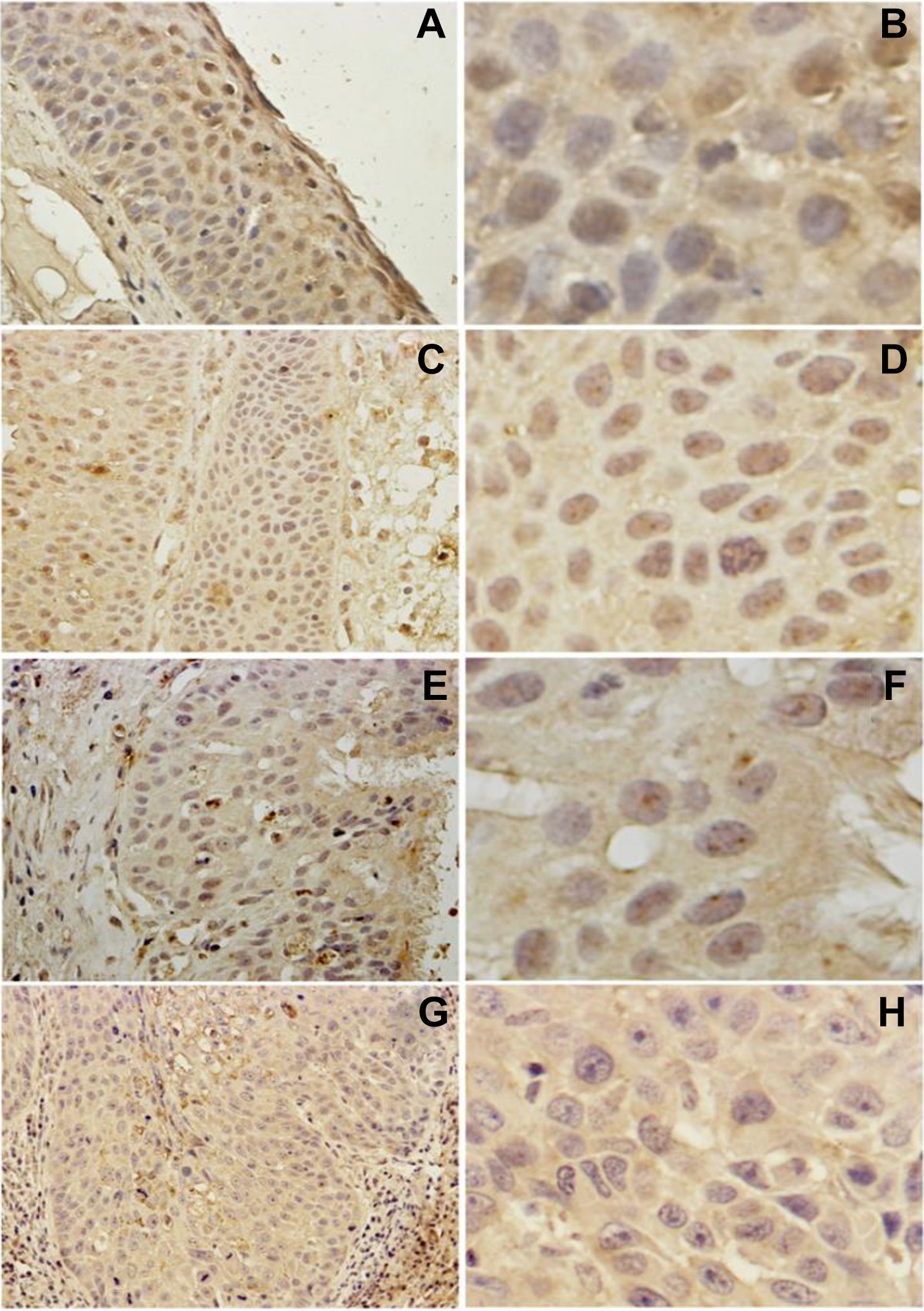
Differences of Sp100 protein expression among the 4 groups. Distribution of Sp100 in normal mucosa, ×100 magnification; (B) Distribution of Sp100 in normal mucosa, ×400 magnification; nuclear staining with clustered distribution (C) Strong expression in SNIP cell, ×100 magnification;(D) Strong expression in SNIP cell, 400× magnification; nuclear staining with clustered distribution (E) Moderate expression in SNIP with dysplasia cell, ×100 magnification; (F) Moderate expression in SNIP with dysplasia cell, ×400 magnification; (G)Weak expression in SNIP with MT, ×100 magnification; (H) Weak expression in SNIP with MT, ×400 magnification and diffuse distribution in cytoplasm. SNIP indicates sinonasal inverted papilloma; Sp100, speckled protein 100; MT, malignant transformation.
Correlations Between Sp100 Protein and SNIP Clinicopathological Features
Table 2 shows the relationship between Sp100 and clinicopathological features of SNIP. We found that Sp100 expression was correlated with HPV infections and SNIP with MT; however, no correlation with respect to age, gender, and smoking was observed.
Correlations Between Sp100 Protein and SNIP Clinicopathological Features.
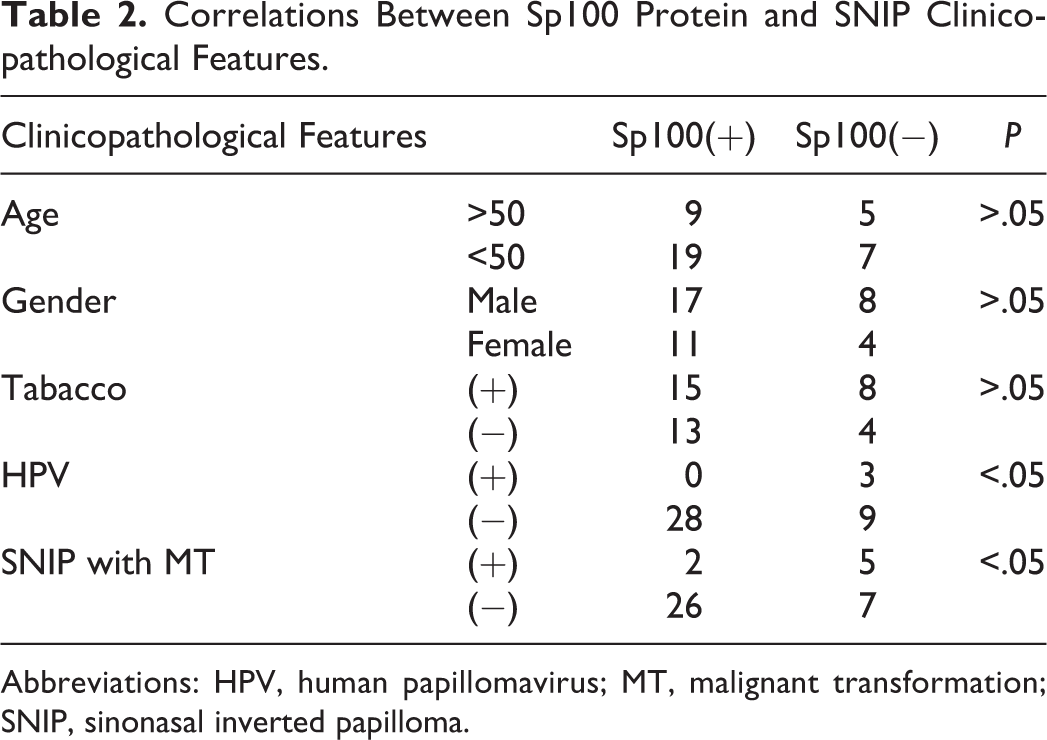
Abbreviations: HPV, human papillomavirus; MT, malignant transformation; SNIP, sinonasal inverted papilloma.
Discussion
Many investigations have been reported to understand SNIP; however, its etiology and causes for MT are yet to be elucidated. 12 In the present study, we observed that the expression of Sp100 was considerably varied among the groups indicating that lower level of Sp100 might play a role in the progression of SNIP.
The HPV DNA was first reported in SNIP in the year 1987. 13 Since then, several studies have shown widely varying type and frequency of HPV DNA detection. The HPV type 16 was the predominant strain found in the previous studies while examining HPV and MT of SNIP followed by HPV type 18 and type 11. 1 A recent review illustrates that HPV infection, especially HPV-18, is significantly associated with malignant SNIP. 12 An earlier study found varied HPV detection rates ranging from 0% to 63% on considering different detection methods, geographic origin, and histologic differences. 2 Katori et al reported that among inverted papilloma with severe dysplasia and inverted papilloma with carcinoma, HPV 16/18 are present in 50% tumor. 14 In our study, the presence of HPV was found to be 42.9% in SNIP with MT group, 0% in SNIP group, and 6.7% in SNIP with dysplasia group. The HPV-positive rate was observed especially higher in the SNIP with MT indicating that the high-risk HPV played a major role in MT of the SNIP. However, it cannot be conclusive as it is just one of the etiologies contributing toward disease progression. The mechanism underlying the malignant conversion of high-risk papillomavirus types, especially HPV-16 and HPV-18, involves the integration of virus DNA into the host genome. 15
The Sp100 proteins were also recently shown to have a role during virus infection. The Sp100 represses transcription of incoming HPV-18 genomes in human keratinocytes 11 and functions as a host restriction factor in early as well as late phases of the HPV lifecycle. 16 Reduction of Sp100 levels results in increased viral transcription and enhanced replication. The present study also demonstrates that Sp100 expression has a correlation with HPV (HPV-16 or HPV-18) in SNIP, and the downregulation of Sp100 may be caused by HPV infection thereby inducing MT of the SNIP. However, the HPV infection rate was not high in SNIP. One possible explanation is the role of other factors that may be contributing to the decreased expression of Sp100 protein.
The PML-NBs are composed of PML, transcriptional regulator ATRX, Death associated protein-6, and Sp100 protein and have been implicated in the regulation of diverse cellular functions such as the cell cycle, apoptosis, senescence, stress, and DNA damage responses. 10 The Sp100 as a potent tumor suppressor has been identified in several research however the underlying mechanisms are different. The Sp100 is induced by interferon (IFN) by modulating ETS1-dependent biological processes to inhibit the invasion of breast cancer cells. 9 In another study on brain tumors, Sp100 expression stimulated by IFNs reduced the malignancy of brain tumors. 10 The Sp100 is also reported to be a coactivator of HIPK2-mediated p53 activation. 17 and knockdown of Sp100 triggers CD95-activated apoptosis. 18 In our study, the MT of SNIP has a correlation with Sp100 expression and from immunohistochemical results, the degree of Sp100 staining was observed to be gradually downregulating with the localization pattern changing from the nucleus to cytoplasm with diffuse distribution. The Sp100 is usually localized in nuclear bodies while it might migrate outside the nucleus and bind with the targeted protein in the cytoplasm to participate in the cell signaling pathways that could be induced by IFN, p53, and apoptosis. However, the exact functional mechanism of Sp100 in the pathogenesis and progression of SNIP needs to be further validated.
There were several limitations in the present study including small sample size. Second, we only detected 2 types of high-risk HPV and detection of other types of HPV needs to be investigated to confirm the association between HPV and SNIP. We are planning further studies to overcome our limitations and understand the functional mechanism of Sp100 in SNIP.
Conclusions
We conclude that positive rate of HPV infection is high in the SNIP with MT than other types of SNIP. We also showed that HPV infection is correlated to Sp100 protein expression which is upregulated in SNIP with MT. From the obtained data, we hypothesize that Sp100 might be playing a functional role in the progression of SNIP.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
