Abstract
Objective:
To examine differences in outcomes after tonsillectomy in children who received outpatient narcotics prescriptions compared to those who did not.
Methods:
The MarketScan database was analyzed for claims made for 14 days following tonsillectomy/adenotonsillectomy between 2008 and 2012 for privately insured children 1 to 17 years. Post-op bleeding, dehydration, emergency department (ED) visits, readmissions, and mean total costs for the 14 days after tonsillectomy were compared.
Results:
Of the 294 795 patients included, 60.9% received a narcotic prescription. Acetaminophen/hydrocodone bitartrate was received by 53.2% of the group receiving narcotic drugs, 42.5% received acetaminophen/codeine phosphate, 3.0% received acetaminophen/oxycodone hydrochloride, and 0.5% received oxycodone hydrochloride alone. Children who had been prescribed narcotics had significantly higher percentages of bleeding complications (2.7% vs 2.5%, P < .001), and ED visits (6.8% vs 6.6%, P < .001) within 14 days, but a lower percentage of readmissions (1.0% vs 1.5%, P < .001). No significant difference was observed between groups for dehydration. There were some age-related differences. The mean total health-care costs for 14 days post-op were the same in each group, except for the 4- to 6-year-olds, where the narcotic group had higher costs (US $7060 vs US $5840, P = .006).
Conclusion:
In this large-scale study, we found small but statistically significant differences in outcomes related to use of narcotics. The only outcome that benefitted the narcotics group was a lower readmission rate.
Introduction
Pain control following pediatric tonsillectomy is a difficult and controversial problem. With obstructive sleep apnea (OSA) being the most common indication for tonsillectomy, 1 accounting for almost 70% of pediatric patients, there are concerns about respiratory complications related to use of narcotics. Children with OSA have intermittent hypoxia, which has been shown in animal studies to increase respiratory sensitivity to opioids. 2 Children with OSA undergoing tonsillectomy have an increased sensitivity to opioids and thus a decreased postoperative opioid requirement 3,4 as well as a blunted ventilatory response to carbon dioxide. 5 This results in an increased risk of apnea because opioids decrease ventilatory response to hypercarbia and hypoxemia. 6 Tonsillectomy does not resolve OSA immediately; in fact, these children may have worse respiratory function on the first night after tonsillectomy, 7 so caution using narcotics with this group is necessary. 6
There are many otolaryngologists who avoid narcotics completely in children undergoing tonsillectomy who have been diagnosed with OSA, whereas others use narcotics routinely. Because small studies can fail to show relatively small differences or be influenced by a single outlier, our goal was to use a large population to look for general differences in outcomes—bleeding, dehydration, emergency department (ED) visits, and readmissions—for children post-tonsillectomy who received narcotic analgesics compared to those who did not. This report utilizes an insurance database comprised of the 5 years available prior to the time of the study, being aware that this predated the Food and Drug Administration (FDA) boxed warning about codeine use post-tonsillectomy.
Methods
The MarketScan Commercial Claims and Encounters (Truven) database 8 was used for this study. The database contains information on reimbursed medical claims submitted by more than 130 payers for over 56 million individuals covered annually under private insurance plans. It includes fully integrated patient-level data (inpatient, outpatient, drug, laboratory, health risk assessment, health and productivity management, dental, and benefit design) from all regions of the United States.
The database was queried for patients aged 1 to 17 who had an inpatient or outpatient claim indicating that a tonsillectomy or adenotonsillectomy was performed in years 2008 to 2012. These surgeries were identified using International Classification of Diseases, 9th revision, Clinical Modification (ICD-9-CM) procedure codes for tonsillectomy (28.2) with or without adenoidectomy (28.3) and Current Procedure Terminology (CPT) codes of 42820, 42821, 42825, and 42826. 9 Patients were required to be covered by a private insurance plan from 1 day prior to surgery to 14 days following the date of discharge and to have a length of surgical stay ≤14 days. Longer lengths of stay (>14 days) were excluded because these patients likely had significant morbidity at the time of tonsillectomy. The first claim for tonsillectomy, by date, was used for the analysis when multiple tonsillectomies were found.
The primary comparison of interest for this study was between patients who received a narcotics prescription at the time of their surgery and those who received no narcotics prescription. Outpatient pharmaceutical claims were searched to determine whether patients had a claim for a narcotics prescription from 1 day prior to surgery to the day of discharge from hospital. These patients were considered to have been prescribed a narcotic related to their surgery. The following generic drug names were classified as narcotics: acetaminophen/hydrocodone bitartrate, acetaminophen/codeine phosphate, acetaminophen/oxycodone hydrochloride, and oxycodone hydrochloride. Patients with a first claim for a narcotics prescription from the day after discharge to 14 days after discharge were excluded because these patients may have received a narcotics prescription as a result of an ED visit or some other reason.
Clinical outcomes for this analysis included bleeding complications, treatment for bleeding complications, dehydration complications, ED visits, and readmissions. All were based on claims that occurred within 14 days of discharge. A patient was considered to have a bleeding complication if an inpatient or outpatient claim had an ICD-9-CM diagnosis code of 998.11 or 784.8, a CPT code of 42960-42962, or an ICD-9-CM procedure code of 28.7. For patients with a bleeding complication, control of the hemorrhage was determined by CPT codes 42960 (“simple” control), 42961 (“complicated, requiring hospitalization”), and 42962 (“secondary surgical intervention”). A patient was considered to have a dehydration complication if a claim had an ICD-9-CM diagnosis code of 276.50 or 276.51. An ED visit was based on the last 2 digits of the service subcategory code being coded as 20 for an inpatient or outpatient claim. A readmission was defined as any inpatient claim (for any reason) within 14 days of discharge from surgery.
Total health-care costs for each patient were calculated from all inpatient, outpatient, and pharmaceutical claims that occurred within 14 days postdischarge. The gross payments for each claim, which included copayments, deductibles and any applicable discounts, were inflated to 2012 dollars using the Consumer Price Index. The total health-care cost for each patient was calculated as the cumulative sum of all inflated gross payments. This value represents the total cost to all payers for all services during the 14-day period following discharge from surgery. Total health-care costs therefore represented the perspective of third-party payers.
For statistical analysis, χ2 tests were used for differences among clinical outcomes by narcotics groups. T tests were used for differences in mean costs between narcotics groups for the subgroups of patients with an ED visit, with a readmission, and with a bleeding complication. Each subgroup was tested separately. Clinical outcomes and costs were also tested separately between narcotics groups within each age-group (1-3, 4-6, 7-10, and 11-17) using the same tests. No adjustment was made for the large number of statistical test conducted because this study was intended to be largely descriptive and we did not want to overlook potentially interesting differences.
Ethical Considerations
This study was reviewed by the Penn State Milton S. Hershey Medical Center Institutional Review Board and deemed to be exempt (#2457).
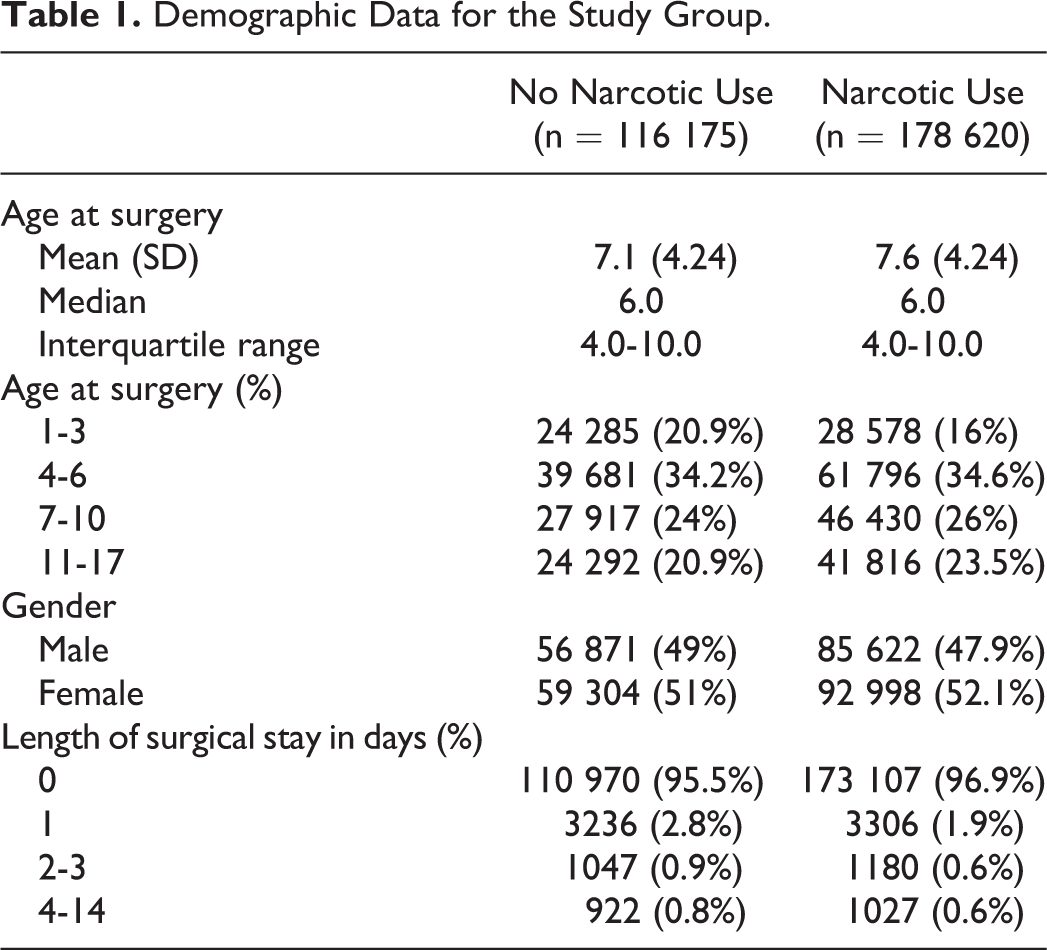
Results
The database contained 294 795 pediatric patients aged 1 to 17 years who met all inclusion/exclusion criteria. A total of 178 620 (60.6%) patients had a prescription for a narcotic drug from 1 day before surgery through the day of discharge, while 116 175 (39.4%) patients had no narcotic prescriptions from 1 day prior to surgery through 14 days after discharge. Table 1 shows demographic details and length of stay for the sample. The median age was 6 years, and the sample contained a slightly higher percentage of females (51.7%). Most patients (98.6%) had outpatient surgery. Patients who did not receive a narcotics prescription were more likely to stay more than 1 day post-op (4.5% vs 3.1% stayed longer than 1 day, P < .001). Figure 1 shows that the percentage of children who received narcotics increased through age 3 years, then plateaued.
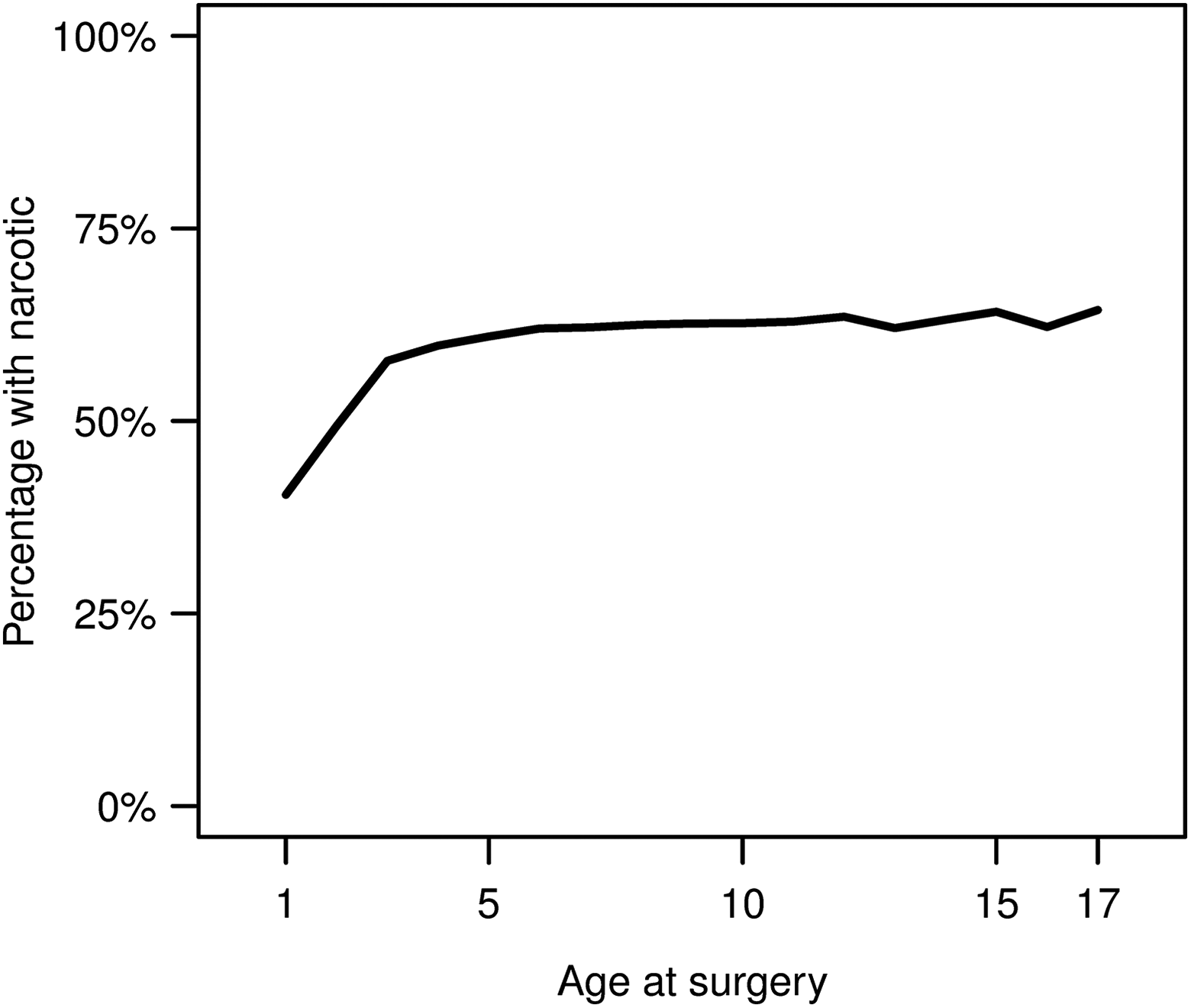
Percentages of patients who received a narcotics prescription after tonsillectomy by age at surgery.
Demographic Data for the Study Group.
Acetaminophen/hydrocodone bitartrate was most commonly prescribed (53.2% of the group receiving narcotic drugs), followed by acetaminophen/codeine phosphate (42.5%), acetaminophen/oxycodone hydrochloride (3.0%), and oxycodone hydrochloride alone (0.5%). Less than 1% received a combination of these medications.
For the entire group, bleeding occurred in 2.6%, dehydration in 2.6%, an ED visit in 6.7%, and readmission in 1.1%. Among patients with a bleeding complication, 55.5% did not require any treatment, 28.6% required surgical intervention, 10.2% required nonsurgical treatment, and 5.6% required hospitalization without surgical treatment.
Children who had been prescribed narcotics had significantly higher percentages of bleeding complications (2.7% vs 2.5%, P < .001), and ED visits (6.8% vs 6.6%, P = .05) within 14 days but a lower percentage of readmissions (1.0% vs 1.2%, P < .001). No significant difference was observed between groups for dehydration.
Table 2 shows bleeding complications, dehydration, ED visits, and readmissions stratified by narcotics use separately for each age-group. Children aged 1 to 3 years with narcotics prescription were significantly more likely to be diagnosed with dehydration within 14 days of surgery than children with no narcotics (4.1% vs 3.7%, P = .028), but they were less likely to be readmitted (1.5% vs 2.0%, P < .001). No significant differences were found for children aged 4 to 6 years. Children aged 7 to 10 years with a narcotics prescription were significantly more likely to have a bleeding complication or dehydration within 14 days after surgery (2.7% vs 2.4%, P = .019). Children aged 11 to 17 years with a narcotics prescription were more likely to have a bleeding complication (4.7% vs 4.4%, P = .043) and an ED visit (8.3% vs 7.9%, P = .036) within 14 days of surgery.
Post-Tonsillectomy Complications, by Age, for Children Who Received Narcotics Prescriptions or Did Not Receive Narcotics Prescriptions by the Time of Discharge After Surgery.
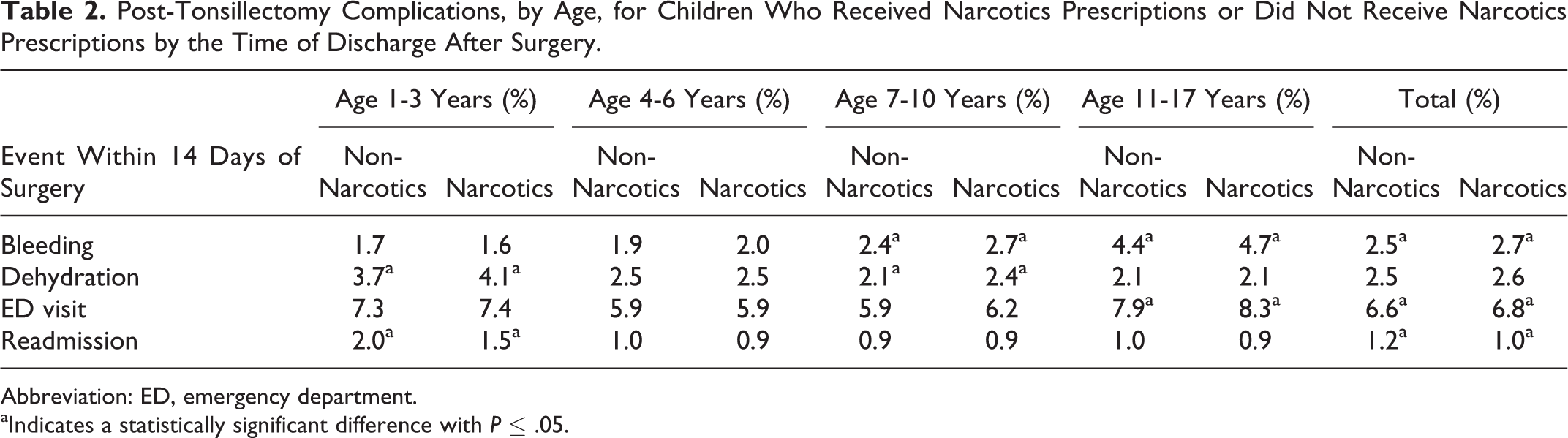
Abbreviation: ED, emergency department.
aIndicates a statistically significant difference with P ≤ .05.
For the full sample, among patients with an ED visit, mean total health-care costs within 14 days after discharge were US $2920 for patients with no narcotics and US $3000 for patients with narcotics (P = .35). Among patients with a bleeding complication, mean total health-care costs were US $6620 and US $6240 for patients with and without narcotics, respectively (P = .09). When conducting the analysis separately for each age-group, the only significant difference observed was for children aged 4 to 6 years old who had a bleeding complication, in which the mean cost for patients receiving narcotics was US $7060 compared to US $5840 for patients without narcotics, a difference of US $1220 (P = .006). One possibility that may explain this difference is that among children aged 4 to 6 who had a bleeding complication, a higher percentage who received narcotics required hospitalization (6.7% vs 4.1%, P = .014). No significant differences with respect to treatment of bleeding complications were observed in the other age groups.
Discussion
There are several options for pain control post-tonsillectomy. Codeine was one of the mainstays of the management paradigm until the FDA issued a warning against using codeine following pediatric tonsillectomy on August 15, 2012. 10 This was in response to a number of deaths associated with anomalies of codeine metabolism present in a minority of patients. 11 Some surgeons continued to use codeine, but others turned their attention to ibuprofen, morphine sulfate, oxycodone, and hydrocodone. Unfortunately, there was little information in the literature regarding the safety of these medications in this particular patient population at that time. This study includes patients who were prescribed codeine.
Comparisons With Other Studies
St Charles et al looked at 110 children undergoing tonsillectomy and compared use of acetaminophen with codeine versus ibuprofen. 12 There were no significant differences in pain levels, pain relief by the medication used, post-op bleeding, readmission, or fever post-op. Ibuprofen users had less nausea. The goal of this study was to evaluate the safety of ibuprofen, but these results are informative because they showed no advantage to using this particular narcotic.
There are 2 recent studies of limited narcotic use along with baseline prescription of an acetaminophen alternating with ibuprofen regimen. Syed et al looked at 125 children undergoing adenotonsillectomy discharged on acetaminophen and ibuprofen and 3 doses of morphine sulfate with instructions to give it to the child on the morning of days 3, 4, and 5. Seventy-four percent of parents were compliant with these instructions, and in this group, pain scores were better and the number seeking help “outside of hours” was lower. 13 Oremule et al compared children getting acetaminophen together with ibuprofen (N = 24) with those who received morphine as needed in addition (N = 50). 14 They found no difference in mean worst pain score on day 4 but a higher score on day 7 for the morphine group. The number seeking advice post-op or being readmitted was not different, but more patients without morphine received an antibiotic when they sought outside help post-op (24% vs 5%). The authors believed that the general practitioners who evaluated these children felt the morphine group already had an “as needed” medication available to use for pain so they didn’t feel the need to add another medication to their regimen. It also seems likely that nonsurgeons were reluctant to prescribe narcotics if the surgeon had not, as they did not choose this option for the non-narcotic group.
Kelly et al ran a randomized controlled trial to compare acetaminophen with morphine (0.2-0.5 mg/kg) versus acetaminophen with ibuprofen (10 mg/kg) for pain control in children after tonsillectomy. 15 All patients had some degree of sleep disordered breathing. They measured first post-op night oxygen saturation via a home pulse oximeter and compared it to levels on the night prior to surgery. They found that 68% of the ibuprofen patients showed improvement in nocturnal oxygen saturation compared to 14% of the morphine group. The morphine group had more desaturation events than they did preop, a mean of 11.2 events per hour more. There were no differences in analgesic effectiveness, post op bleeding or adverse drug reactions in this small group. Only 91 children were enrolled because their Data Safety Monitoring Board discontinued the study after the interim analysis. The obvious conclusion from this study is that morphine sulfate use in children who snore has potential risks.
Clinical Applicability of the Study
It is difficult to run a prospective study using narcotics in children who undergo tonsillectomy because of the perceived risks and subsequent ethical concerns, and retrospective chart reviews are extremely difficult to publish, so even if our colleagues have looked at this question, the information is not currently in the literature. The goal of our study was to examine general factors that comprise outcomes after tonsillectomy for patients who received narcotics early in their post-op course. With such a large scale study, even small differences can be revealed to be statistically significantly different. Overall, narcotics recipients were slightly more likely to bleed post-op and had more ED visits, but fewer readmissions. It’s difficult to say whether such small differences are clinically important. What we can say is that narcotics do not confer a large difference with respect to post-op bleeding, dehydration, ED visits, or readmissions in children after tonsillectomy. A 2011 survey showed that only 0.3% of otolaryngologists prescribed nonsteroidal anti-inflammatory medications after pediatric tonsillectomy 16 so presumably, most of the patients in the comparison group in our study received acetaminophen for pain control.
Only 16% of children under the age of 3 in this study group were prescribed narcotics for home use. Kelly et al showed mean pain scores (measured by Faces Scale and Objective Pain Scale) were not significantly different between their morphine and ibuprofen groups. 15 For surgeons willing to use ibuprofen in young children after tonsillectomy, there is likely no argument to employ narcotics for pain control.
Children who receive outpatient narcotic prescriptions have shorter hospital stays after tonsillectomy. Whether these medications are used in hospital and result in better early recovery or whether surgeons who prescribe outpatient narcotics are more confident that their patients will be able to tolerate their post-op pain outside the hospital is not known from our data. This database does not include in-patient medications. We know that some children have oxygen desaturations with narcotics, but there must not be enough of them to alter the length of stay when averaged over almost 295 000 patients. It would be a logical non sequitur to assume that prescribing outpatient narcotics in children allows earlier discharge.
We did not find significant differences with costs of postoperative management between the narcotic and non-narcotic groups, except for children aged 4 to 6 years. This may be explained by the fact that children in this age-group who had received narcotics prescriptions were more likely to require hospitalization when they bled post-op. There were no differences between the narcotics and non-narcotics groups for treatment of bleeding (no treatment, ED management, hospitalization, or surgery) or for mean costs in the other age groups. This could indicate that preschoolers using narcotics and suffering a post-op bleed together require more medical care.
Study Limitations
Our study is limited by the fact that data are culled from coding done for billing purposes. It will underestimate complications for which no medical billing occurred—for example, bleeding that stops spontaneously and no medical opinion is sought or dehydration that is not recognized. The database does not contain information about mortality. We know that these patients received a prescription for narcotics and that it was filled, but we don’t know if the child took any of the medication. In the study by Syed et al, only 74% of parents provided with a narcotic prescription reported that they had given their child the medication. 13 In fact, there are no published studies looking at narcotics post-tonsillectomy that used objective evidence that the subjects actually took the medications under study; at least in this study, we know that the parents bought them. As well, due to the delay in production of large-scale databases, the information we had access to was dated. This study group included children who had received codeine, and in 2013, the FDA issued a boxed warning against the use of codeine following tonsillectomy in children; this was related to a small number of deaths attributed to polymorphisms in cytochrome P450 genes producing ultra-rapid codeine metabolism and subsequent elevated blood morphine levels that increased the risk of overdose. 17,18 Even with this warning, by 2015, 1 in 20 children undergoing tonsillectomy still received a prescription for codeine in the 7 days following their surgery. 17 This study also found that as codeine prescriptions dropped, prescriptions for higher potency narcotics increased, so the overall percentage of children receiving an opioid medication after tonsillectomy did not change significantly. 17 Many clinicians have turned to ibuprofen after tonsillectomy, particularly since the recent American Academy of Otolaryngology Guidelines advocate its use. 19 Despite these changes in practice, the information is useful because it can act as a springboard for more analysis of narcotic use in children after tonsillectomy. Clinicians who look to the otolaryngology literature to determine whether narcotics should be employed in their pediatric post-tonsillectomy patients will not currently find a clear answer there; this is a topic deserving of more study.
Conclusion
About 70% of these children undergo tonsillectomy because of sleep disordered breathing, 1 and narcotics have been shown to suppress respiration in this group. There are clear risks to using these medications in children with OSA, particularly in the early post-op period. In this study, we found that about 60% of children were prescribed (and parents received the medication from a pharmacy) narcotics for home use post-op. Although narcotics recipients had statistically significantly higher percentages of bleeding, dehydration, and ED visits, depending on the age, and lower percentages of readmission, these differences were all less than 1%. We cannot say that narcotics are safe or not safe in all children post-tonsillectomy from these data. We can say that there are small advantages and disadvantages to using them in a large outpatient pediatric population, but these may not be clinically significant in the average pediatric practice.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant from the HG Barsumian MD Memorial Fund.
