Abstract
Use of rigid endoscopes has become widespread in middle ear surgeries, thereby attracting attention to the safety of antifog agents. However, few studies on the ototoxicity of antifog agents have been conducted. The purpose of this study was to evaluate hair cell damage and the underlying mechanisms caused by antifog agents using zebrafish larvae. We exposed zebrafish larvae at 3 days postfertilization to various concentrations of the antifog agent, Ultrastop (0.01, 0.02, 0.04, and 0.08%) for 72 hours. The average number of hair cells within 4 neuromasts of larvae, including supraorbital (SO1 and SO2), otic (O1), and occipital (OC1), in the control group were compared to those in the exposure groups. Significant hair cell loss was observed in the experimental groups compared to that in the control group (P < .01; control: 53.88 ± 4.85, 0.01%: 45.08 ± 11.70, 0.02%: 41.36 ± 12.00, 0.04%: 35.36 ± 16.18, and 0.08%: 15.60 ± 7.53 cells). Concentration-dependent increase in hair cell apoptosis by terminal deoxynucleotidyltransferase (TDT)-mediated dUTP-biotin nick end labeling assay (control: 0.00 ± 0.00, 0.01%: 3.48 ± 2.18, 0.02%: 9.64 ± 5.75, 0.04%: 17.72 ± 6.26, and 0.08%: 14.60 ± 8.18 cells) and decrease in the viability of hair cell mitochondria by 2-(4-[dimethylamino] styryl)-N-ethylpyridinium iodide assay (control: 9.61 ± 1.47, 0.01%: 8.28 ± 2.22, 0.02%: 8.45 ± 2.72, 0.04%: 7.25 ± 2.44, and 0.08%: 6.77 ± 3.26 percentage of total area) were observed. Antifog agent exposure can cause hair cell damage in zebrafish larvae, possibly by induction of mitochondrial damage with subsequent apoptosis of hair cells.
Introduction
Recently, the application of endoscopy in ear surgery has increased. It has many advantages, such as high magnification and the ability to identify corners that cannot be visualized with a microscope. 1,2 Although endoscopic sinus surgery has become a standard technique, endoscopic ear surgery has developed relatively late due to associated disadvantages, such as one-hand surgery, lack of depth, spatial limits of the ear canal, and potential of heat injury from the instruments. 1,3,4 However, recent advances, such as endoscope holders and increased resolution of endoscopic cameras, have overcome many technical problems. Moreover, successful outcomes of endoscopic ear surgery have been reported and compared to those of conventional surgery using a microscope. 4 Minimal invasiveness being one of the important requirements in almost all surgical fields, the demand for endoscopic ear surgery is expected to increase significantly in the future.
Antifog agents are widely used to improve viewing through the endoscope during the examination of the tympanic membrane and perform simple investigative procedures in otology in a short span of time. Moreover, the probability of the antifog agent to enter the middle ear cavity is low. Therefore, there has been little consideration toward their ototoxicity; to our knowledge, only one study has been conducted on its ototoxicity. 5 With the development of endoscopic ear surgery, use of antifog agents has gained importance because maintaining a clear view, despite the humidity and blood, is one of the important factors affecting the quality of the surgery. In particular, there is a possibility that the residence time or residual amount of the antifog agent in the middle ear may be increased, which may affect the inner ear homeostasis. 1,3
Zebrafish have neuromasts, which are composed of clusters of hair cells, on the surface of their lateral line system. 6 -8 Morphology and functions of hair cells of zebrafish are similar to those of the mammalian inner ear. 6 Furthermore, zebrafish has been used for investigating damage to hair cells after exposure to ototoxic chemicals owing to the easy assessment of damage to hair cells. 9 -11 The purpose of the present study was to evaluate the effect of various concentrations of the commonly used antifog agent, Ultrastop, on hair cells of zebrafish larvae.
Materials and Methods
Chemicals and Zebrafish
A well-known antifog agent, Ultrastop (MoNo chem-pharm Produkte GmbH, Vienna, Austria), was used. The solution consists of purified water, 70% ethanol, and less than 5% surfactant. 5,12 Wild-type and transgenic (Brn3c: EGFP) zebrafish larvae were produced by mating adult fish reared at 28.5°C ± 1°C in a zebrafish facility. 13 These transgenic larvae have hair cells that express green fluorescence under a fluorescence microscope without staining. The larvae were maintained in an embryo medium (15 mM NaCl, 0.5 mM KCl, 1 mM CaCl2, 1 mM MgSO4, 0.15 mM KH2PO4, 0.05 mM NH2PO4, and 0.7 mM NaHCO3) and staged by growth in days postfertilization (dpf). 14 The protocol used in this study was approved by the Institutional Animal Care and Use Committee of our hospital (Approval No.: KOREA-2018-0054). All protocols were performed according to the guidelines of the Animal Care and Ethics Committee of the Korea University and National Institutes of Health.
Chemical Treatments
The wild-type and transgenic larvae (3 dpf) in the experimental groups were exposed to the antifog agent added to the embryo medium for 72 hours. Considering the round window permeability of well-known agent, gentamicin, 2.5%, 15 the antifog agent was prepared as 4 different concentrations: 0.01%, 0.02%, 0.04%, and 0.08%. A control group with no treatment was also maintained. The larvae were washed thrice with embryo medium after treatment and anesthetized using tricaine (3-aminobenzoic acid ethyl ester, 0.4 g/100 mL; pH adjusted to 7.0 using Tris buffer) for 5 minutes as described in previous studies. 16,17 They were then mounted on a depression slide using methylcellulose and observed under a fluorescence microscope (LSM5 PASCAL, Carl Zeiss, Germany) at 40× magnification.
The number of hair cells within the neuromasts of the supraorbital (SO1 and SO2), otic (O1), and occipital (OC1) lines were counted in each zebrafish larva of the experimental and control groups (n = 25).
Estimation of Hair Cell Apoptosis by the Terminal Deoxynucleotidyltransferase (TdT)-Mediated dUTP-biotin Nick End Labeling Assay
Hair cell apoptosis was measured by the terminal deoxynucleotidyltransferase (TdT)-mediated dUTP-biotin nick end labeling (TUNEL) assay using an in situ cell detection kit (Roche Molecular Biochemicals, Mannheim, Germany) in accordance with the manufacturer’s protocol. The transgenic larvae in the experimental groups were exposed to the antifog agent for 72 hours. They were then washed with phosphate-buffered saline and fixed in 4% paraformaldehyde. Thereafter, they were incubated with 50 μL of the TUNEL reaction mixture (TdT and fluorescein-dUTP) at 37°C for 60 minutes in a humid atmosphere. The TUNEL staining intensity in the hair cells within the 4 neuromasts of larvae was assessed using a fluorescence microscope (AxioCam MRc5, Carl Zeiss). 18,19 The number of TUNEL-positive cells in each type of neuromast was counted and the averages of the 4 experimental groups (0.01%, 0.02%, 0.04%, and 0.08%) were compared to that of the control group (n = 25).
Analysis of Mitochondrial Damage Using 2-(4-[Dimethylamino] Styryl)-N-Ethylpyridinium Iodide Assay
The fluorescent dye, 2-(4-[dimethylamino] styryl)-N-ethylpyridinium iodide (DASPEI; Invitrogen, Carlsbad, California) was used to stain mitochondria within the hair cells of wild-type larvae in the experimental (72 hours antifog agent-exposed) and control groups. The transgenic larvae were exposed to antifog for 72 hours in experimental groups. After anesthetizing the larvae with tricaine, they were incubated in an embryo medium containing 0.005% DASPEI for 15 minutes. The fluorescence intensity of hair cells within the 4 neuromasts was analyzed under a fluorescence microscope, as described in previous studies, 16,17,19 and quantified based on the percentage of the total DASPEI-stained area within the cropped image using the ImageJ software (NIH, Bethesda, Maryland); the fluorescence intensities were compared between the groups (n = 25).
Statistical Analysis
Statistical analyses were performed with IBM SPSS version 22.0 for Windows (IBM, Armonk, New York). The 1-way analysis of variance and post hoc analysis using Tukey honestly significant difference test were performed to determine statistical differences (P < .05). All data were expressed as the mean ± standard deviation.
Results
Effect of the Antifog Agent on Hair Cell Numbers in Neuromasts
Wild-type larvae were exposed to different concentrations of the antifog agent for 72 hours (0.01%, 0.02%, 0.04%, 0.08%, and 0% control, n = 25). We observed that 5 larvae died in the 0.08% exposure group; therefore, hair cells were counted from 15 larvae in this group. Exposure to the antifog agent decreased the number of hair cells in a concentration-dependent manner (Figure 1). Fluorescence microscopy revealed significant hair cell loss in the center of the neuromasts. The number of hair cells in the 4 types of neuromasts of the lateral line was counted (Figure 2). In the control group, it was found to be 53.88 (4.85) cells per larva. However, exposure to the antifog agent decreased the number of hair cells in a concentration-dependent manner, which were observed in the 0.02%, 0.04%, and 0.08% exposure groups, 41.36 (12.00), 35.36 (16.18), 15.60 (7.53) cells per larva, respectively, to be significantly lesser than that in the control group (P < .01).
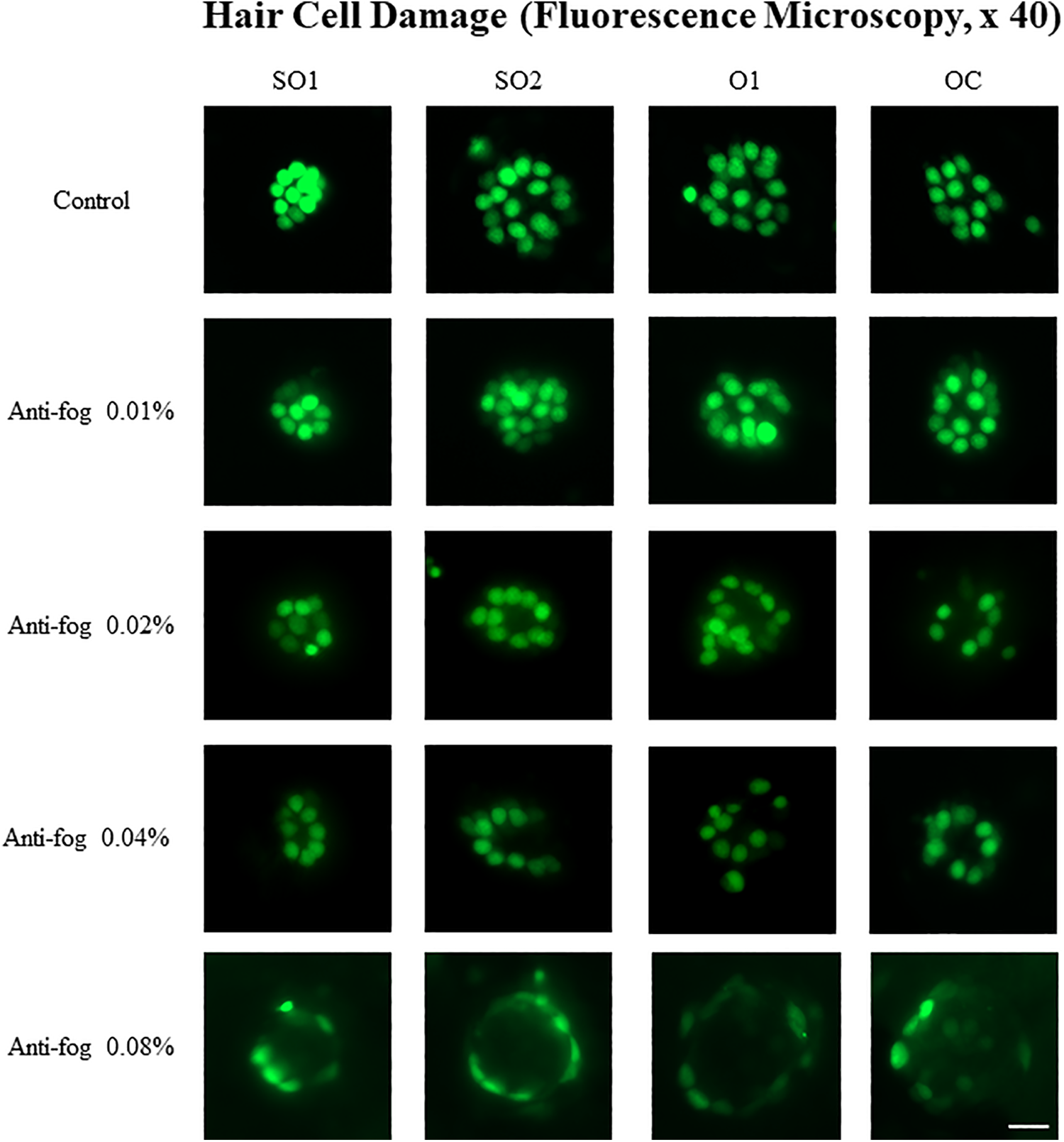
Hair cell damage according to the different concentrations of the antifog agent. Wild-type zebrafish larvae (3 dpf) were exposed to different concentrations of the antifog agent: 0.01%, 0.02%, 0.04%, and 0.08% for 72 hours. Compared to the control group, the experimental groups showed decreased number of hair cells. dpf indicates days postfertilization; O1, otic; OC, occipital; scale bar = 10 µm; SO1, supraorbital 1; SO2, supraorbital 2.
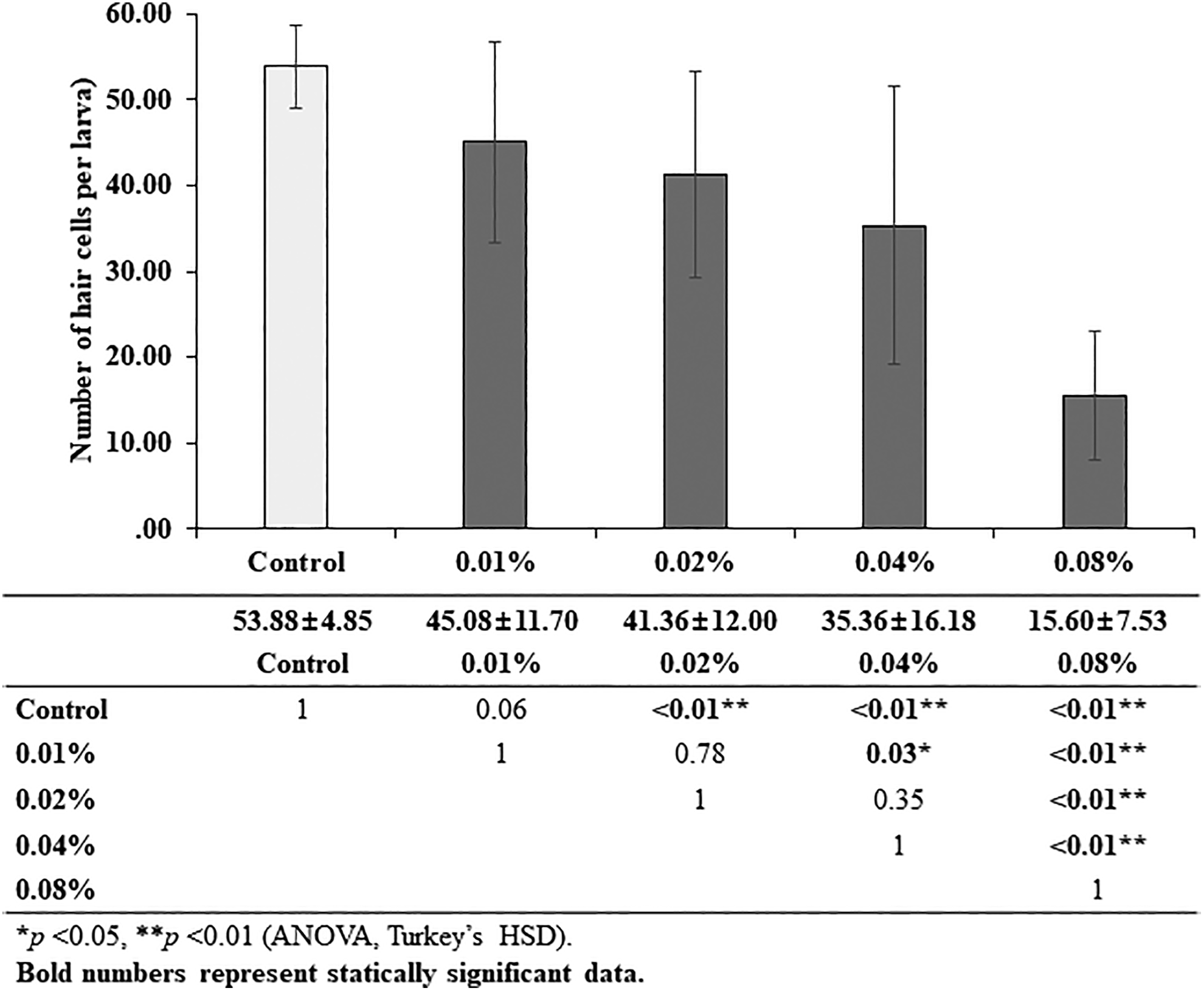
Quantitative assay for hair cell damage. Wild-type zebrafish larvae (3 dpf) were exposed to different concentrations of the antifog agent: 0.01%, 0.02%, 0.04%, and 0.08% for 72 hours. The number of hair cells in the 4 neuromasts (SO1, SO2, O1, and OC1) was analyzed in each larva. Each group consisted of 25 larvae, except for the 0.08% exposure group, which included 15 larvae because of the death of 10 larvae after the exposure. Hair cell loss occurred in a concentration-dependent manner (control: 53.88 [4.85], 0.01%: 45.08 [11.70], 0.02%: 41.36 [12.00], 0.04%: 35.36 [16.18], 0.08%: 15.60 [7.53] cells). dpf indicates days postfertilization; O1, otic; OC, occipital; SO1, supraorbital 1; SO2, supraorbital 2.
Effect of the Antifog Agent on Cellular Apoptosis
The extent of apoptosis in the neuromasts was examined by the TUNEL assay in transgenic zebrafish larvae. Due to the green fluorescence of neuromasts in the lateral line of the Brn3c: GFP larva model, apoptotic cells appeared red after TUNEL staining (Figure 3). Although larvae in the control group did not show any apoptotic cells, those exposed to the antifog agent showed few apoptotic cells in their neuromasts. The number of the TUNEL-positive hair cells showed the same tendency. The number of TUNEL-positive apoptotic cells per larva increased in the groups exposed to higher concentrations (except 0.08%) of the antifog agent (Figure 4). However, values for the 0.04% and 0.08% exposure groups were not significantly different.
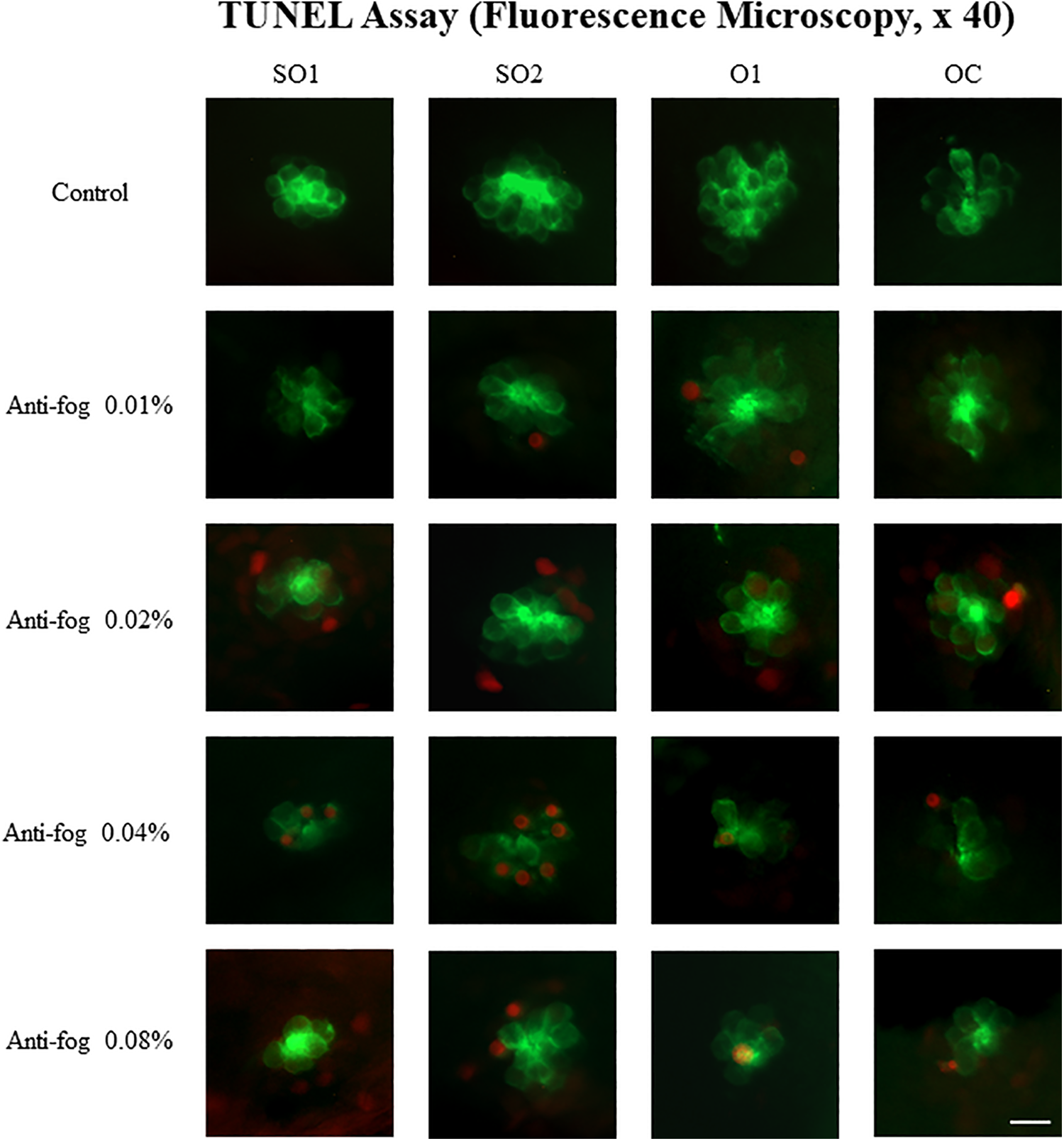
Terminal deoxynucleotidyltransferase (TdT)-mediated dUTP-biotin nick end labeling (TUNEL) assay using fluorescence microscopy. For the TUNEL assay, 3 dpf transgenic zebrafish (Brn3c: GFP) were exposed to different concentrations of the antifog agent: 0.01%, 0.02%, 0.04%, and 0.08% for 72 hours. TUNEL-positive apoptotic cells that were stained red, were not observed in the control group. However, the number increased in larvae exposed to higher concentrations of the antifog agent. dpf indicates days postfertilization; O1, otic; OC, occipital; Scale bar = 10 µm; SO1, supraorbital 1; SO2, supraorbital 2.
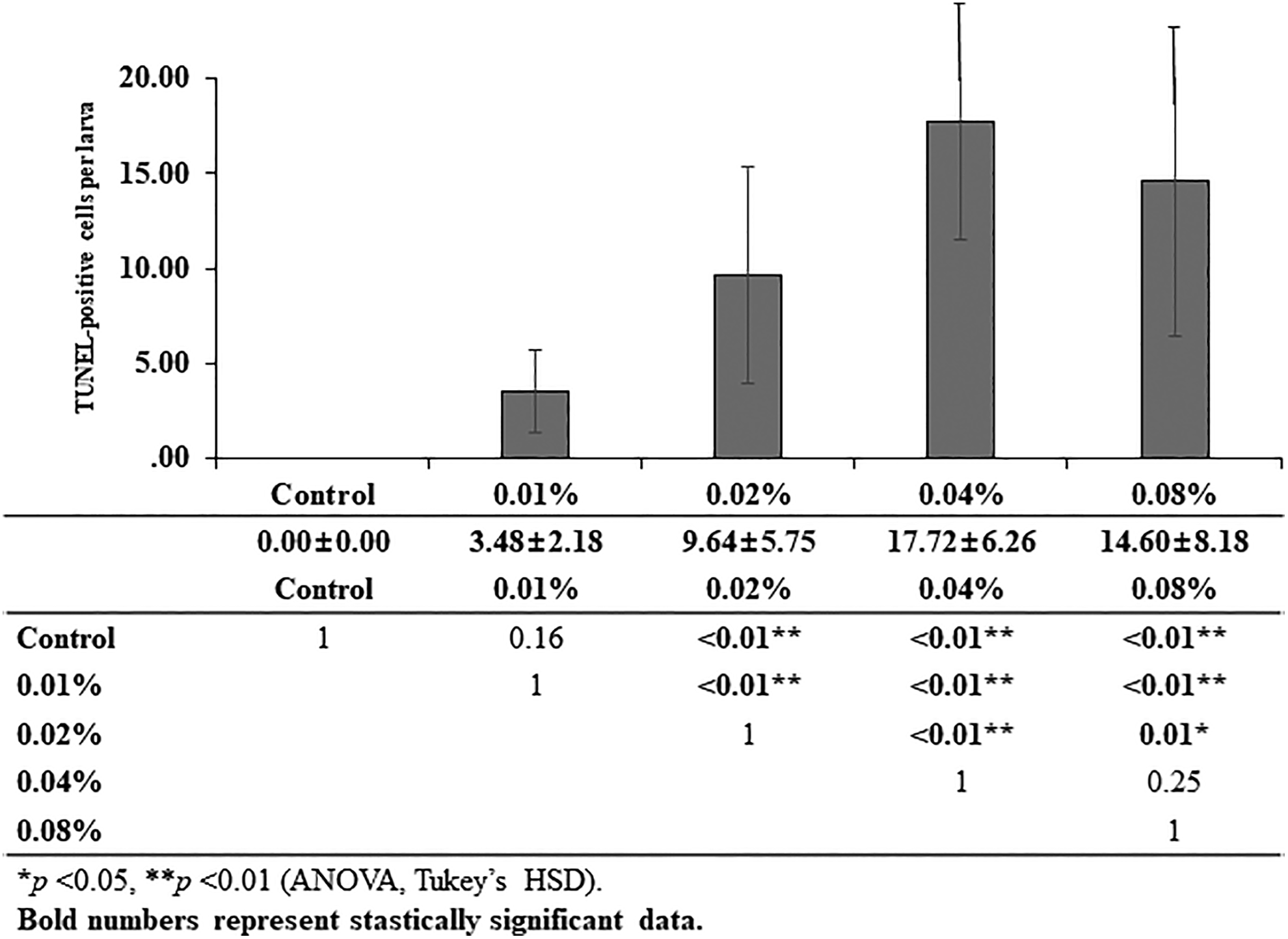
Quantitative analysis of results of the TUNEL assay. The number of TUNEL-positive cells per larva was counted in larvae exposed to different concentrations of the antifog agent: 0.01%, 0.02%, 0.04%, and 0.08% and in the control group. The number of TUNEL-positive cells increased in a concentration-dependent manner, except in the 0.08% exposure group (control: 0.00 [0.00], 0.01%: 3.48 [2.18], 0.02%: 9.64 [5.75], 0.04%: 17.72 [6.26], 0.08%: 14.60 [8.18] cells). TUNEL indicates terminal deoxynucleotidyltransferase (TdT)-mediated dUTP-biotin nick end labeling.
Effect of the Antifog Agent on Hair Cell Mitochondria
The DASPEI assay was conducted to visualize viable mitochondria in the hair cells. It was observed that the intensity of green fluorescence was stronger in the control group than in the experimental groups; it got fainter as the concentration of the antifog agent increased (Figure 5). The average extents of DASPEI-stained areas in the 4 neuromasts, calculated by the ImageJ software, were compared between the groups. There were no significant differences between areas of the 0.01% and 0.02% exposure groups and the control group (8.28 ± 2.22, 8.45 ± 2.72, 9.61 ± 1.47%, respectively). However, the areas of the 0.04% and 0.08% exposure groups were significantly lower than that of the control group (P = .01 and <.01, respectively), indicating a significant loss in the number of viable hair cell mitochondria in the neuromasts (Figure 6).
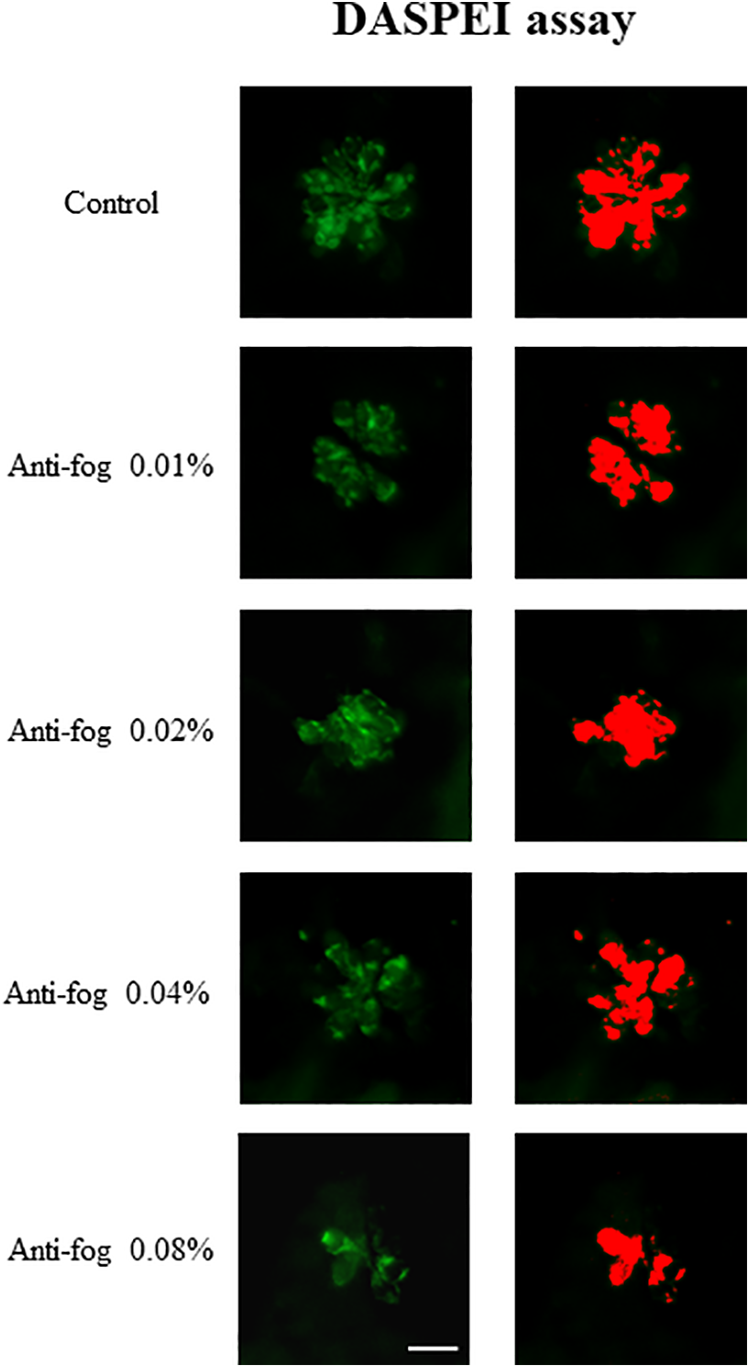
Analysis of viable mitochondria by the DASPEI assay in wild-type zebrafish larvae (3 dpf) exposed to different concentrations of the antifog agent. The green fluorescence images on the left represent the viable DASPEI-stained area under the fluorescence microscope (×40). Images in this figure represent the occipital area of the neuromast. The DASPEI-stained area (µm2), as quantified by the ImageJ software, is shown on the right. A decrease in the DASPEI-stained areas is observed for the higher concentration groups. Scale bar = 10 µm. DASPEI indicates 2-(4-[dimethylamino] styryl)-N-ethylpyridinium iodide; dpf, days postfertilization.
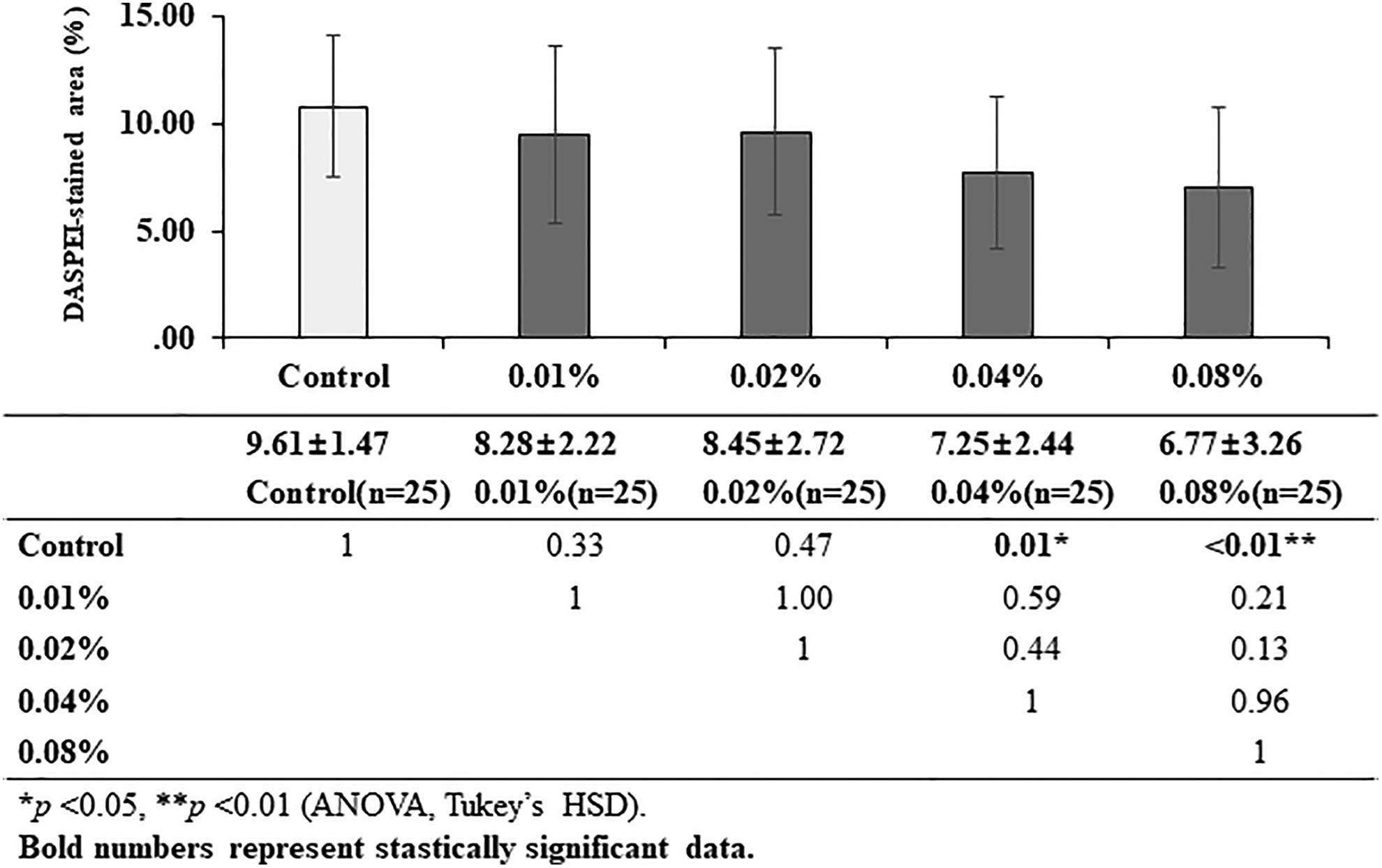
Quantitative analysis of results of the DASPEI assay. Average DASPEI-stained areas (%) were compared between the experimental and control groups. Compared to the control group, no significant changes were observed in the lower concentration groups, 0.01% and 0.02%, whereas a significant decrease in the DASPEI-stained areas was observed the higher concentration groups, 0.04% and 0.08% (control: 9.61 [1.47], 0.01%: 8.28 [2.22], 0.02%: 8.45 [2.72], 0.04%: 7.25 [2.44], 0.08%: 6.77 [3.26] area). DASPEI indicates 2-(4-[dimethylamino] styryl)-N-ethylpyridinium iodide.
Discussion
As one of the early attempts to investigate antifog exposure-induced histopathological changes in inner ear hair cells and the underlying mechanisms, we evaluated the ototoxicity of antifog agent exposure at 4 different concentrations in a model of zebrafish larva. When exposed to 0.01%, 0.02%, 0.04%, or 0.08% of the antifog agent, the number of hair cells decreased in a concentration-dependent manner. Although not statistically significant, the extent of apoptosis in the 0.08% exposure group was lesser than that in the 0.04% exposure group. This may be explained by the difference in the extents of reference area analyzed—the area occupied by green-colored neuromasts. Loss of hair cells led to a decrease in the reference area. Subsequently, the number of apoptotic cells might have been underestimated despite the relatively high apoptosis. Mitochondrial damage in the hair cells was also increased in the groups exposed to higher concentrations of the antifog agent.
Ethanol is one of the most widely used disinfectants and the major component of an antifog solution. It was also used in the form of ear drops after being mixed with boric acid to clean the ear canal in chronic suppurative otitis media. 20,21 However, ethanol-based ear drops induce significant changes in the auditory brain stem response that are different from those induced by water-based ear drops, suggesting ethanol-induced ototoxicity. 22 Ethanol (70%) administered into the middle ear cavity causes vestibular and cochlear ototoxicity, as reported in other studies that warned against the use of ethanol in conditions of the perforated tympanic membrane. 22,23
Ototoxicity of the antifog agent, Ultrastop, has been reported using a guinea pig model, 5 in which exposure to the antifog agent induced changes in the auditory brain stem response threshold and extensive middle ear inflammation equivalent to those caused by the established ototoxic agent, gentamicin. Although results of this reported study conclusively established its ototoxicity, changes in the inner ear hair cells and underlying mechanisms of ototoxicity were unclear. Moreover, accumulation of the antifog agent in the mastoid bulla employed in this study was beyond that generally expected after an endoscopic ear surgery.
In the present study, zebrafish larvae were exposed to the 0.01% to 0.08% of antifog. In the endoscopic ear surgery, the patient’s head may turn less to the opposite side than in microscopic surgery. Therefore, the facial recess is located much lower part of tympanic cavity in surgical position than in microscopic surgery. Most otologists pay attention to antifog agent not to enter the tympanic cavity. As mentioned in a previous study, a small amount of antifog agent that covered the lens may soon evaporated and have little effect on the inner ear. 24,25 However, it is still possible that a few missing droplets of antifog agent enter into the tympanic cavity and may stay in the facial recess because of the adverse anatomical condition. To our knowledge, there are no known data on the round window membrane permeability by antifog agents or ethanol, which is influenced by molecular size, concentration, lipophilicity, and electrical discharge. 26 Instead, it is known that 2.5% of administrated gentamicin can be detected in perilymph after round window irrigation. 15,27,28
Ethanol has a molecular weight lower than that of gentamicin and is expected to have a permeability of at least 2.5%. Therefore, it is possible that the antifog agent accumulated in the facial recess after the endoscopic ear surgery reaches to the perilymph at a concentration of 0.08% or more. In addition, significant hair cell apoptosis in the 0.01% exposure group suggests that attention should be paid to even a small amount of antifog agent accumulation.
Ethanol-induced ototoxicity occurs after a relatively short exposure. In a previous study linking ethanol concentration and ototoxicity, the endocochlear potential changed in the mastoid bulla after 10 minutes of exposure to 50% ethanol. 29 The effects of ethanol also appear earlier than those of other ototoxic agents. Exposure to isopropyl alcohol (70%) for 10 minutes changed the auditory brain stem response latency after 30 minutes. 30 When harvesting the cartilage, the temporalis fascia or trimming the perforation margin of the tympanic membrane, the surgery often proceeds without sufficient irrigation for more than a few minutes. If the surgery is performed under local anesthesia, excessive irrigation is often intentionally avoided to minimize patient’s discomfort. Therefore, it is necessary to pay attention to the inner ear damage due to antifog agent in terms of exposure time as well as the concentration of it.
This study has some limitations. First, the reversibility of ototoxicity was not studied. After serial measurement of auditory brain stem responses, reversibility of alcohol-induced ototoxicity after 2 weeks has been demonstrated in a rat model. 30 However, conflicting results have also been reported. 5,29,30 Further studies with serial measurement of cochlear and/or vestibular function and histopathological changes are needed to obtain clinically valuable information. Second, the effect of surfactant in the antifog solution was not investigated. Currently, there are no reports on ototoxicity induced by surfactants in antifog solutions. Surfactants account for less than 5% of the antifog solution composition. Although present in an extremely small amount compared to other ototoxic agents, surfactants used for various purposes have been reported to cause irritation or harm to the human body, suggesting that the safety of antifog surfactants should be considered. 31,32
In conclusion, exposure to the antifog agent can cause hair cell damage in zebrafish larvae. Based on the results, possible underlying mechanisms include induction of mitochondrial damage with subsequent apoptosis of hair cells.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was supported by Bumsuk Academic Research Fund in 2017.
