Abstract
Gaze-evoked nystagmus is not rare among those who have acute balance problem and may indicate a cerebellar dysfunction that is associated with a broad spectrum of disorders. The aim of this study is to analyze optokinetic response in those patients. Eleven males and 7 females (age range: 25-60, 42.5 [9.75]) with gaze-evoked nystagmus were analyzed with optokinetic test (Micromed Inc). Nystagmus was elicited by a stimulator light spot moving across the patient’s visual field at a target speed of 30 degree/second. Ten age-matched healthy participants served as controls. The gain and slow-phase velocity difference in oculomotor response from left and right stimulus was compared in patients and the control participants. One-way analysis of variance test was used for multiple variance analysis of the groups. Statistical significance was set at P < .05. Slow-phase velocity of gaze-evoked nystagmus was ranging between 6 and 19 degree/second. The mean slow-phase velocity of gaze-evoked nystagmus to the right and left was 8.1 (3.81) and 6.8 (4.67) degree/second, respectively. Optokinetic gain was out of normal limits in 10 (55.5%) patients. Comparison of mean gain difference between the patients and the normal participants was statistically significant (P = .025). No statistical difference was found in mean slow-phase velocity difference in optokinetic nystagmus between control participants and patients (P > .05 [.099]). An acute-onset balance problem may be associated with dysfunction of separate populations of neurons in the brainstem and cerebellum even if there is no radiological neuropathy since gaze-evoked nystagmus is a sign of neural integrator dysfunction. Patients with gaze-evoked nystagmus and optokinetic abnormalities may have disruption of cerebellar pathways and should be followed closely.
Introduction
The study of eye movement abnormalities in patients with acute-onset balance disorders presents an ideal tool to investigate the function of the network controlling eye movements. The quantitative analysis of eye movement helps to document various kind of abnormalities. Gaze-evoked nystagmus is not rare among those who have acute balance problem without any initial neurologic sign. 1 It is characterized by drifts moving the eyes away from the target and a null position where the drifts are absent. Failure of maintaining eccentric gaze position is a central phenomenon. Once the eyes reach a given target, brainstem oculomotor integrator utilizes feedback from various sources to maintain steady gaze. The main source of information is the cerebellum that conveys information related to eye movements. Therefore, gaze-evoked spontaneous nystagmus that occurs at each lateral gaze is the manifestation of cerebellar dysfunction, which indicates defective gaze-holding mechanisms associated with alteration of the function of the vestibulocerebellar structures. 2 Degeneration of neurons can result in an increased time constant and instability of the vestibular velocity-storage mechanism. 3,4
We present 18 patients having acute vertigo and imbalance with gaze-evoked nystagmus which are presumably caused by damage to the central vestibular structures and by disruption of the neural pathways responsible for eye-position stabilization. We investigated the clinical features and distinct ocular motor abnormalities. The study was principally designed to quantitatively characterize specific optokinetic changes that may help to define the pathology and contribute to better characterize cerebellar involvement in the control of eye movements.
Material and Methods
Eighteen patients with gaze-evoked spontaneous horizontal nystagmus during primary gaze position were recruited among patients with acute balance problem between 2009 and 2016. Gaze-evoked nystagmus was confirmed by videonystagmography (VNG; Micromed Inc) in all patients. An informed consent was obtained from all individual participants included in the study. The procedures were in accordance with the ethical standards of the declaration of Helsinki and of institutional review board of the hospital. The study was completed in a regional community health center. All patients had normal otoscopic examinations and visual acuity. None of the patients had a story of neurological, muscular, or vascular disease. Magnetic resonance imaging or computed tomography of the temporal bone and brain were normal in all patients. All patients were first tested with VNG for spontaneous nystagmus in the seated primary gaze position before starting the test battery. The direction of nystagmus, slow-phase velocity, and optical fixation was recorded. Patients were asked to hold the eyes at eccentric gaze (right and left). The direction and the mean slow-phase velocity of pathologic gaze-evoked nystagmus were documented. Rebound nystagmus (primary position nystagmus that is provoked by prolonged eccentric gaze holding) was also analyzed. Associated problems that may be related to gaze-evoked nystagmus were documented.
Optokinetic nystagmus was elicited by a stimulator light spot moving across the patient’s visual field at a target speed of 30 degree/second. Lower target velocity was selected since velocities over 40 degree/second may lead to optokinetic asymmetry including declining gains with increasing frequency in healthy participants. 5 A commercially available system is used to generate optokinetic stimuli (Micromed Inc). The participant was seated in a completely dark room 1 m away from visual target at the level of his eyes. Testing procedure was explained, and the participants were instructed to follow the moving target without moving his head, keeping the stimulus in focus. Test conditions were performed randomly (visual stimulus; rightward or leftward). A blank, black wall was presented between trials for 5 seconds. Each participant was studied during one session that lasted for about 15 minutes. The gain of the oculomotor response was computed as the ratio of the mean slow-phase velocity of the eye and the target velocity. Mean horizontal gains and slow-phase velocity of optokinetic nystagmus were averaged for each patient. Saccadic abnormalities, the abnormalities of slow-phase velocity of optokinetic nystagmus and optokinetic asymmetry, were documented. Mean values and standard deviations (SDs) were calculated for each condition. Data were compared to 10 age-matched healthy participants. One-way analysis of variance test was used for multiple variance analysis of the groups (Statistical Package for the Social Sciences 17.0 version, IBM, Chicago, Illinois). Statistical significance was set at P < .05. Gains in patients with gaze-evoked nystagmus were plotted. Level of normal data for the gain was set between 1 and 0.85.
Results
Eleven males and 7 females with gaze-evoked nystagmus were included in the study group. Age range was 25 to 60 (42.5 [9.75]). None of the patients had any spontaneous nystagmus at primary gaze position. Three patients had very mild up-beating nystagmus during vertical gaze position. Positional tests (head-hanging, head-roll, head bending, etc) were normal. Five patients had migraine. One of those had hypertension. Two patients had a history of traffic accident 4 months before the onset of balance problem. One patient had diabetes and another one had diabetes and hypotroidism. One patient had alcohol addiction. Two patients were using antidepressant drugs on regular basis for more than a year. Bilateral high-frequency hearing loss was documented in 4 patients during audiometry. Twelve patients had magnetic resonance imaging and 6 patients had computed tomography which were normal. Three patients had angiography and 1 patient had electroencephalography which revealed nothing abnormal. Slow-phase velocity of gaze-evoked nystagmus was ranging between 6 and 19 degree/second. The mean slow-phase velocity of gaze-evoked nystagmus to the right and left was 8.1 (3.81) and 6.8 (4.67) degree/second, respectively. Asymmetric velocity of gaze-evoked nystagmus was documented in 10 (55.5%) patients.
Responses quantified by optokinetic nystagmus slow-phase velocity and gain were compared with those of 10 healthy age-matched participants. All control participants have normal gain. When cutoff line of the gain was set between 1 and 0.85, gain was out of normal limits in 10 (55.5%) patients. Seven (%38,9) patients had reduced gain and 3 (%16,6) patients had elevated gain (Figure 1). Mean gain difference in optokinetic nystagmus between right and left stimulus in the study group was ranging between 0.11 and 0.01 (mean: 0.035 [0.026]). Mean gain difference in optokinetic nystagmus between right and left stimulus in the control group was ranging between 0.01 and 0.03 (mean: 0.015 [0.007]). Comparison of mean gain difference between the patients and the normal participants was statistically significant (P = .025). Six patients had abnormal saccadic distortions even if they have normal gain (Figure 2). Mean slow-phase velocity difference in optokinetic nystagmus between right and left stimulus in the study group was ranging between 1 and 12 degree/second (mean: 3.88 [3.02] degree/second). Mean slow-phase velocity difference in optokinetic nystagmus between right and left stimulus in the control group was ranging between 1 and 3 degree/second (mean: 2.20 [0.91] degree/second). No statistical difference was found in mean slow-phase velocity difference in optokinetic nystagmus between control participants and patients (P > .05 [.099]; Table 1).
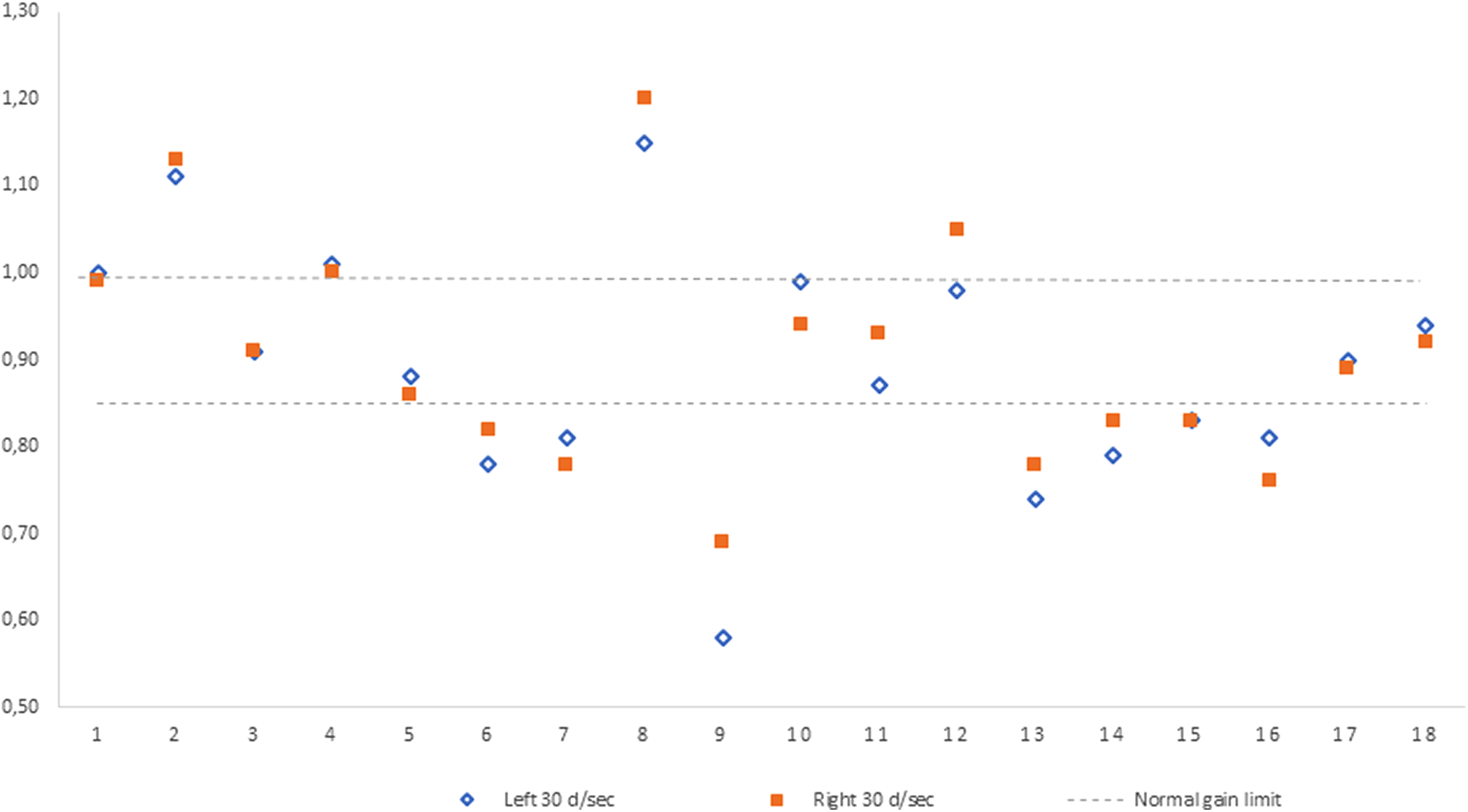
Distribution of gain of the optokinetic nystagmus following leftward and rightward visual stimulus in patients with gaze-evoked nystagmus.
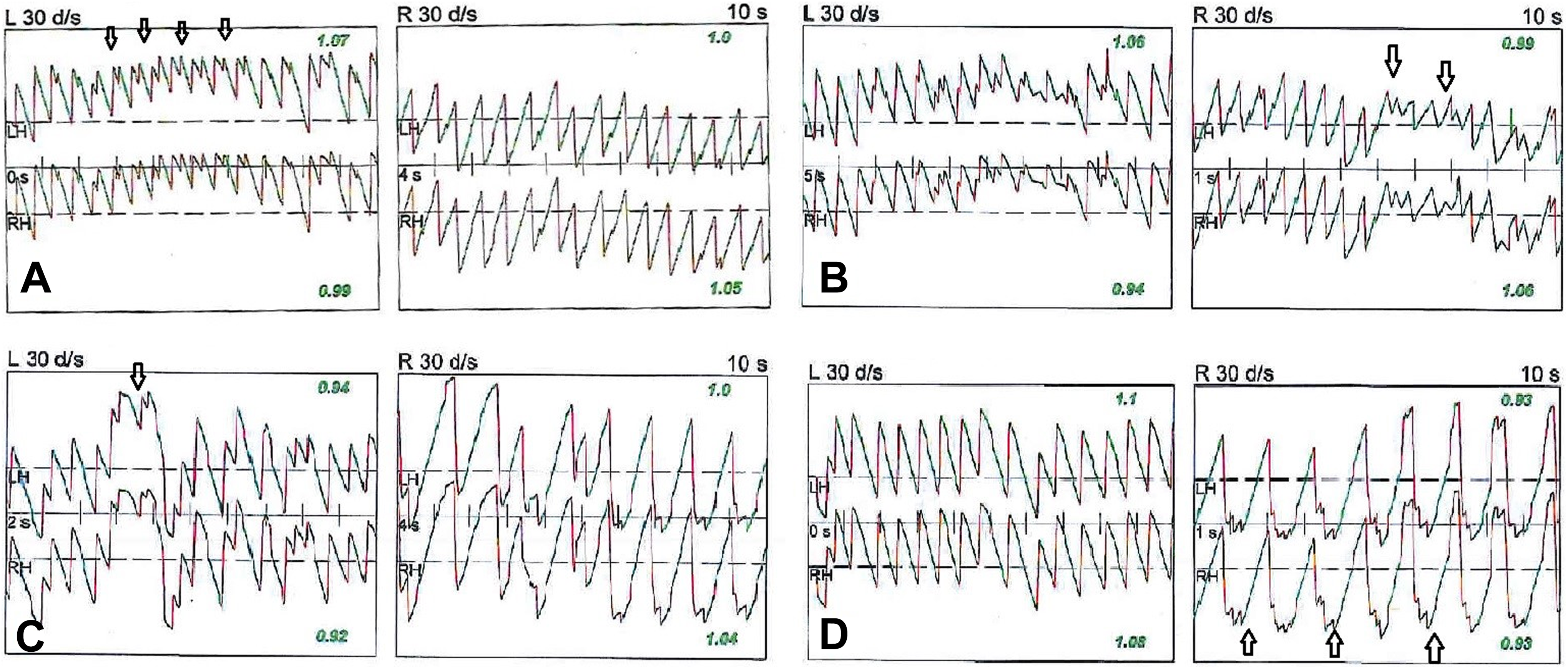
Examples of asymmetric optokinetic recordings (A-D). Distortion of nystagmus is marked with an arrow. Note instantaneous (B and C) or clear asymmetry between right and left recordings (A and D).
Comparison of Mean Optokinetic Gain and Slow-Phase Velocity Asymmetry Between the Groups.
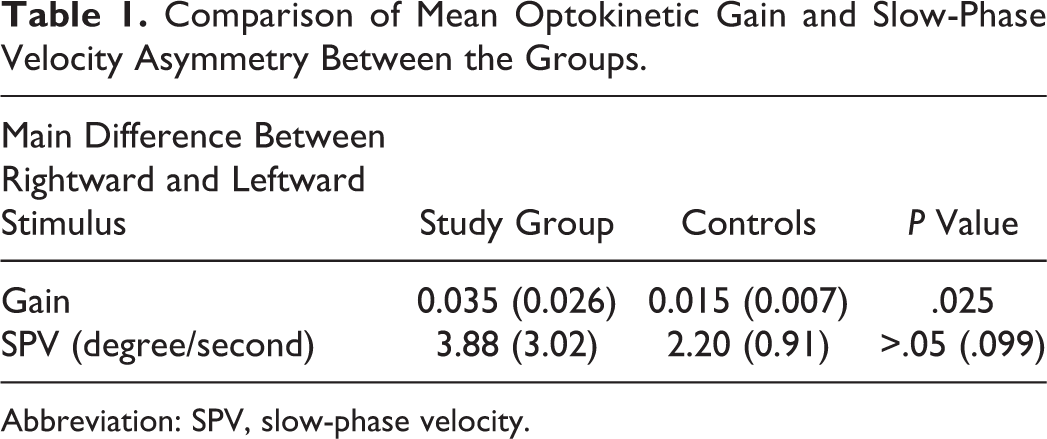
Abbreviation: SPV, slow-phase velocity.
Discussion
The ability to keep the eyes on a moving target is controlled by the oculomotor neurons which require a neural mechanism supplied by eye-velocity and eye-position signals. Eye-position holding at the eccentric gaze is provided by a neural integrator which is a neural network located largely in the medulla for horizontal movements and in midbrain for vertical movements. The horizontal gaze neural integrator consists of the nucleus prepositus hypoglossi and medial vestibular nuclei which are interconnected via the oculomotor nucleus. Other components of neural integrator include the flocculus and nodulus of the cerebellum. 4,6 Neural integrator takes velocity commands from the conjugate eye movement systems and creates a position command to hold the eye steady. When brainstem neural integrator fails to send tonic-step commands for eccentric gaze, eyes drift back to central position that is followed by corrective saccades and defective gaze-holding manifests as nystagmus.
Optokinetic nystagmus is an involuntary reflex eye movement responsible for stabilizing retinal images in the presence of visual object motion or self-motion which is an integral part of dynamic spatial orientation provided by a neural framework from the retinal input to the visual cortex and from the visual cortex to the motor neurons through the connections between nucleus of optic tract, dorsolateral pontin nucleus, nucleus prepositus hypoglossi, medial vestibular nucleus, and dorsal cap of the inferior olive. 7,8 It consists of 2 basic components, a slow tracking movement in the direction of motion and a rapid recovery saccade producing a characteristic irregular sawtooth waveform. Some forms of saccadic oscillation, such as square-wave jerks can be seen during optokinetic recording of healthy participants. However, there is strong evidence that horizontal OKN is symmetrical in normal healthy adults. OKN gains can be influenced by a variety of different factors including target size, shape, contrast, and velocity. 9 On the other hand, involuntary, sporadic, and horizontal square-wave such as saccadic intrusions that take the eye away from the intended fixation position off the moving target is an important finding in the presence of gaze-evoked nystagmus and smooth pursuit deficit even if the gain is within normal limits. An example of some patients with distortion during optokinetic response is presented in Figure 2.
Cerebellar dysfunction indicates a broad spectrum of disorders. The unique feature of our patients was that the symptoms were transient and they had no other radiological and neurological findings. Studies indicate diagnostic importance of eye movement analysis with greater accuracy than early magnetic resonance imaging. 10 Gaze-evoked nystagmus associated with acute-onset balance problem without any neurological sign could be one of the very early manifestations of inherited disorders such as spinocerebellar ataxia, familial hemiplegic migraine, and episodic ataxia. Genetic abnormalities include mutations in a potassium channel gene that is expressed throughout the nervous system but is especially common in the Purkinje and granule cells of the cerebellum. 2,11 Therefore, close follow-up with neuroimaging, molecular, and genetic testing are required for early diagnosis of an episodic but a progressive problem. Acquired cerebellar ataxia such as immune mediated and paraneoplastic, sensorial dysfunctions associated with loss of proprioception due to neuropathies are manifested long after neurological symptoms. 12
Gaze-evoked nystagmus could be the only main and early manifestation of lateral or medial portion of medullar or cerebellar ischemia due to thromboembolic events or autoimmune vasculitis especially in the territory of cerebellar arteries. 13,14 Particularly, the dorsal medulla contains vestibular nuclei, nucleus prepositus hypoglossi, and inferior cerebellar peduncle that are involved processing the vestibular and ocular motor signals. Even if these are progressive, radiological investigation at the early period may be negative. Demyelinating neuropathies such as multiple sclerosis, Guillain-Barre syndrome may present with gaze-evoked nystagmus. 15 Other causes of transient gaze-evoked nystagmus are alcohol consumption, migraine, intoxication (lead, aluminum, etc), pharmacological inactivation of ocular motor integrator in patients using sedatives, barbiturates, anticonvulsants, or marijuana which usually involve neurofunctional changes in Purkinje cells at the level of anterior and superior vermis and paravermal parts of anterior lobe in more advanced cases. 16 -18 Patients with head trauma, cerebellar tumors, cranial surgery, and viral infections may also exhibit gaze-evoked nystagmus. 19,20 Five patients had migraine, 2 patients had a story of trauma, and 1 patient was using antidepressant drug in our series.
We have focused on gain and slow-phase velocity difference in optokinetic response following rightward and leftward visual stimulus. Gain was abnormal in 55% of patients and comparison of mean gain difference between the patients and the normal participants was statistically significant. However, no statistical difference was found in mean slow-phase velocity difference in optokinetic nystagmus between control participants and the patients. Quantitative evaluation of directional asymmetry of velocity may provide some information on the mapping of lesions although conflicting results have been reported. Kato et al have reviewed optokinetic nystagmus, caloric response, and pursuit eye movements in 28 patients with unilateral brainstem lesions. Gain was lower in the affected side in 22 patients. The relationship of pursuit gains to percentage reduction in fixation–suppression of caloric nystagmus showed no correlation. However, combined use of optokinetic and pursuit analysis was useful for lateralizing the lesion in the brainstem. 21
In conclusion, gaze-evoked nystagmus is a sign of neural integrator dysfunction. Advanced age can cause degenerative impairment of the neural integrator, leading to an often symmetric, horizontal gaze-evoked nystagmus. Gaze-evoked nystagmus of short duration, with a low amplitude and frequency, elicited at extreme horizontal fields of gaze, especially in an elderly patient, is usually physiologic and does not require further investigation. In contrast, primary position nystagmus, sustained more than 20 seconds, or asymmetric gaze-evoked nystagmus are usually pathologic and should prompt further investigation. Patients with balance problem in the emergency setting usually have a heterogenous group of disorders. Gaze-holding deficits may be seen early from onset of cerebellar pathology and functional problems of neural integrator to provide stable gaze holding is probably nonlinear. 22 An acute-onset balance problem may be associated with dysfunction of separate populations of neurons in the brainstem and cerebellum even if there is no radiological neuropathy. An association between gaze-evoked nystagmus and optokinetic abnormalities may indicate disruption of cerebellar pathways and should be handled as such.
Footnotes
Authors’ Note
This study was conducted at Anadolu Medical Center (Affl. Johns Hopkins Medicine), Department of ORL & HNS, Kocaeli, Turkey. The study was not transmitted to any other journal.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
