Abstract
It has been suggested that systemic pentoxifylline may be beneficial in the treatment of olfactory dysfunction. The postulated mechanism of action involves nonselective competitive phosphodiesterase inhibition, leading to increased intracellular cyclic adenosine monophosphate and consequent increased olfactory neuron activity. This should in theory lead to improved olfactory function. We describe a pilot case series from our tertiary referral center of patients treated with oral pentoxifylline for olfactory dysfunction. Six patients with post-traumatic impairment who were treated with systemic pentoxifylline were included. Patients were treated with 200 mg of oral prolonged release pentoxifylline, 3 times a day for 21 days. Olfactory function was tested pre and post-treatment for odor threshold (T), discrimination (D), identification (I) and composite 'TDI' score using a psychophysical test battery, the “Sniffin’ Sticks.” Oral pentoxifylline was well tolerated and all patients completed the treatment period. There was a small improvement in odor threshold and identification scores, but these did not reach statistical or clinical significance. There were deteriorations in discrimination and composite TDI score, which did not reach significance. While our case series was small, systemic pentoxifylline did not appear to be beneficial in the treatment of hyposmia in this patient group.
Introduction
Olfactory dysfunction is an increasingly recognized condition, with an estimated prevalence of up to 20% of the general population. 1 It carries with it significant impact on quality of life, through associated depression, weight disturbances, social anxiety, and social isolation. 2 Despite this health-care burden, effective long-term treatments for olfactory dysfunction are lacking.
For proper olfactory function, both an intact peripheral sensory organ and higher processing cerebral pathways are required. At the peripheral level, olfactory neuroepithelium can be found in the upper third of the nasal cavity, including the superior turbinate and superior nasal septum. 3 Olfactory sensory neurons (OSN) are found within this neuroepithelium, with dendritic cilia that extend into the superficial mucus layer. These cilia contain olfactory receptors (OR) that bind volatile chemicals and thereby transmit smell. Through complex combinatorial coding, approximately 400 active OR genes lead to the detection of thousands of distinct odors. 4 During recent years, the downstream signaling cascades involved during OR activation have received attention as potential drug targets for olfactory dysfunction.
Olfactory receptors are G-protein-coupled receptors with 7 transmembrane domains. When an odorant binds an OR, this leads to increased intracellular cyclic adenosine monophosphate (cAMP) through G protein-mediated activation of type III adenylyl cyclase. Increased intracellular cAMP levels activate a cyclic nucleotide-gated (CNG) cation channel in the surface membrane. The ensuing cation influx leads to depolarization of the OSN, action potential propagation along its axon, and corresponding signal transduction to the olfactory bulb. 5
Phosphodiesterase inhibitors should theoretically modulate the olfactory signaling cascade by preventing cAMP degradation. By maintaining high intracellular cAMP, this may lead to enhanced peripheral olfactory function through increased activation of CNG channels.
Pentoxifylline is a competitive nonselective phosphodiesterase inhibitor derived from xanthine. It is used clinically for the symptomatic relief of muscular pain caused by peripheral artery disease. In Germany, it is additionally recommended for use in patients with tinnitus or sudden-onset sensorineural hearing loss. Previous work from our center suggested that pentoxifylline treatment for the latter purposes causes incidental improvement in olfactory function. 6 Following on from this, we now report results from a pilot case series of patients who were treated in our center with pentoxifylline for post-traumatic olfactory dysfunction.
Case Reports
Participants
Six patients who were treated for olfactory dysfunction at our center were included. There were 4 male and 2 female patients, of mean age 43 years. All patients had post-traumatic olfactory dysfunction.
Treatment and Olfactory Testing
Each of the patients underwent treatment with 200 mg of prolonged release pentoxifylline (Agapurin retard) orally, 3 times a day, for 21 days. Patients were monitored for medication side effects throughout this period. Olfactory function was tested before and after treatment using “Sniffin’ Sticks.” A period of 21 days was chosen as this was felt to be sufficient time for effects of modification of the peripheral olfactory system to become evident. Previous work investigating other medications targeting the OR signaling cascade has demonstrated significant effects following one off administration. 7 Furthermore, we felt that a treatment period longer than this would expose patients to unnecessary risk from side effects.
“Sniffin’ Sticks” are a validated psychophysical test in which olfactory threshold (T), discrimination (D), identification (I) and composite 'TDI' scores are assessed, with discrimination and identification being measures of suprathreshold olfactory function. 8,9 For threshold testing, a 16-step staircase forced-choice paradigm is used whereby patients are asked to choose which of 3 presented pens contains a rose-like odor (phenylethylalcohol, PEA), where the other 2 pens are odorless distractors. Triplets are presented with an increasing concentration of PEA (where 16 = lowest concentration and 1 = highest concentration) until the patient can reliably identify the odorant pen. The lower the detectable concentration of odorant, the lower the threshold level and the higher the score. Discrimination is tested using a 16-step forced-choice paradigm whereby 2 of the 3 presented pens have the same odor, and 1 has a different odor. The patient is asked to identify the pen they believe to contain the different odor. Finally, odor identification is tested using a 16-step forced-choice identification task. Here, the patient is provided with a choice of 4 visual and written cues (eg, peach, lemon, orange, pineapple) and then asked to identify which of these odors are contained within the presented pen. For threshold and discrimination testing, triplets were presented in random order, and the patient was blindfolded. Individually, and as a composite score (TDI), higher scores indicate better olfactory function.
Statistical Analysis
Statistical analysis was performed using GraphPad Prism (version 6, GraphPad Software, LaJolla, California). Comparisons between pre and post-treatment scores were performed using Wilcoxon matched-pairs signed rank test (nonparametric test used due to small n number). Statistical significance was assumed where P < .05.
Ethical Considerations
Written consent was obtained from each of the patients involved in this case series. Treatment with pentoxifylline was approved by the ethics committee of our hospital.
Results
Full TDI scores before and after treatment were obtained (see Table 1). Before treatment, the mean TDI was 17.33, in keeping with hyposmia. There was a small decrease in TDI after treatment, with a mean of 17.08. Using a Wilcoxon matched-pairs signed rank test, this difference was not significant (P = .81; see Table 2).
Mean Olfactory Scores Before and After Treatment, With Standard Deviation and Standard Error of the Mean.a
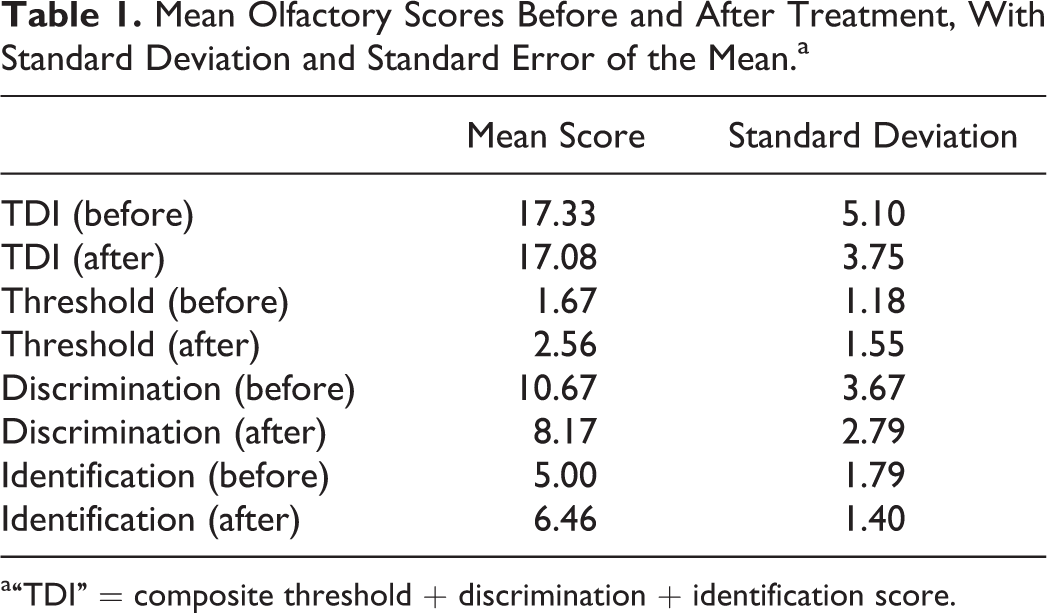
a“TDI” = composite threshold + discrimination + identification score.
Mean Difference in Olfactory Score With Results From Paired Wilcoxon Matched-Pairs Signed Rank Test.a
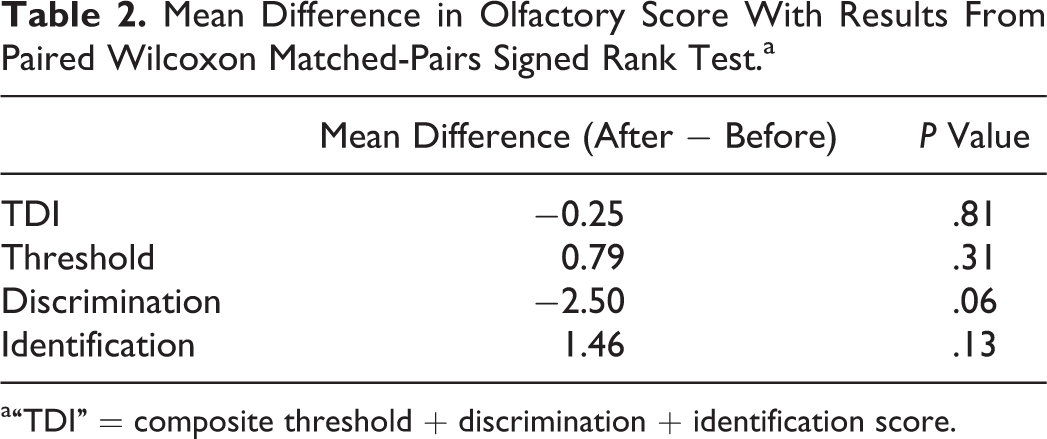
a“TDI” = composite threshold + discrimination + identification score.
There was a small increase in threshold score (corresponding with decreased odor threshold) after treatment (mean increase 0.79 points). This result did not reach statistical significance (P = .31).
The discrimination score worsened after treatment, with a mean change of −2.50. Again, this did not reach statistical significance (P = .06).
There was a small improvement in identification scores (mean increase 1.46 points), however, again this did not reach statistical significance (P = .13).
Discussion
In this group of patients treated with oral pentoxifylline for post-traumatic olfactory dysfunction, we found a small improvement in threshold, which did not reach statistical significance. Previous work using “Sniffin’ Sticks” has defined the minimal clinically important change in odor threshold as an increase of 2.5 dilution steps. 10 We saw a mean increase of only 0.79 dilution steps, in keeping with no clinical improvement. This was reflected anecdotally in this case series, with no patients reporting subjective improvement.
With regard to suprathreshold olfaction, there was a small improvement in mean identification scores, and a deterioration in discrimination scores, neither of which reached statistical significance. Overall, there was a small decrease in composite TDI score of −0.25 points, which did not reach statistical significance (P = .81).
Given the small patient number in this pilot case series, our results should be interpreted with caution. Phosphodiesterase inhibitors are postulated to increase peripheral olfactory sensitivity through modulation of OR downstream signaling cascades. This should lead to increased threshold sensitivity, and while a trend toward improved thresholds was seen, this was too small to reach statistical or clinical significance.
Post-traumatic olfactory dysfunction produces severe impairment compared to post-infectious, sinonasal, and neurodegenerative causes. 11 It is thought to be due to transection of olfactory fila as they pass through the cribriform plate and/or central damage due to contusions, intraparenchymal hemorrhage, or resultant gliosis. 12 Phosphodiesterase inhibitors were piloted in this patient population with the intention of enhancing any residual function at the peripheral level (assuming that injury had not caused complete transection of the olfactory nerve). Previous work has suggested that olfactory training may be advantageous in post-traumatic dysfunction. 12 Such training (which involves patients smelling different odors in a standardized regimen) is likely effective, at least in part, due to peripheral stimulation. Therefore, the efficacy of this treatment supports our hypothesis that modulation of the peripheral olfactory system at the level of the neuroepithelium may be of benefit. However, we acknowledge that the possibility of axonal transection and central damage may confound the peripheral effects of pentoxifylline in this cohort and that this oral medication should be further tested in patients with dysfunction due to other causes.
Our results contrast with those reported by Gudziol and Hummel in 2009, who showed that pentoxifylline improved olfactory threshold sensitivity but did not affect suprathreshold olfactory performance (discrimination and identification). 6 In this study, 19 patients (13 normosmic and 6 hyposmic) received either intravenous or oral pentoxifylline for otological conditions and their olfaction was assessed with the “Sniffin’ Sticks.” Following treatment, there was a statistically significant decrease in threshold level, which reached clinical significance in 2 patients. While this study had a larger overall patient number, there was heterogeneity in drug administration and preexisting olfactory function. As such, only 4 patients received medication orally, of whom 2 showed increased and 2 decreased threshold scores. It is known that the bioavailability of pentoxifylline is 30% to 40% less for oral administration. It is possible therefore that the discrepancy in results seen could be due to this reduced bioavailability. However, the utility of intravenous treatment for olfactory dysfunction is debatable.
In 2009, Hienkin et al., reported results from their unblinded clinical trial of theophylline, another nonselective phosphodiesterase inhibitor, for treatment of hyposmia. 13 Three hundred twelve patients with reduced cAMP/cyclic guanosine monophosphate (cGMP) levels in nasal mucus and saliva were included in the study and were treated with an increasing dose of theophylline, depending on subjective response. Drug blood levels were monitored and olfaction was assessed using subjective reporting and 4 odor psychophysical testing for detection threshold, recognition threshold, magnitude estimation, and hedonic value. Henkin et al found that at each drug concentration used, detection thresholds improved compared to pretreatment scores. This was statistically significant but was independent of subjective improvement. Suprathreshold scores also improved post-treatment, including subjective ratings, recognition thresholds, magnitude estimates, and hedonic scores. Overall, 50% of the study participants were considered responsive to treatment.
Whereas Henkin et al., observed suprathreshold improvement and subjective clinical benefit in their study, we did not. It is possible that this could be due to subtle differences in the pharmacological profile of theophylline and pentoxifylline, especially given the complex cerebral processing of olfactory stimuli and widespread physiological effects of phosphodiesterase inhibitors. It could also be due to the unblinded design used in Henkin’s study, whereby patients who had not responded well to the medication were given a higher dose to try to maximize response. Arguably, this may have influenced subjective ratings, magnitude estimates, and hedonic scores. Furthermore, Henkin’s inclusion of patients with low nasal mucus and saliva cAMP/cGMP levels may have created a systematic difference in patient populations. Importantly, Henkin et al used a mixed patient cohort, including those with postinfectious or post-traumatic olfactory dysfunction and allergic rhinitis. The difference in underlying etiological cause of dysfunction could therefore lead to the discrepancy in results seen.
In an effort to characterize the effect of phosphodiesterase inhibitors on ON activation, Gudziol et al recorded electro-olfactograms (EOG) in supravital mouse olfactory epithelium following topical theophylline application. 14 They found that EOG amplitude decreased following drug application. Typically, increased EOG amplitude is seen with increasing concentrations of odorant and previous work has shown that amplitude is proportional to odor intensity ratings in humans. 15 These results suggest that changes seen in olfactory function with phosphodiesterase inhibitors might not be due to modulation of the downstream OR signaling cascade, as originally theorized. Furthermore, one may argue that these results could support lack of effect of this class of drugs on olfaction.
The effects of other nonselective phosphodiesterase inhibitors on olfaction have also been disappointing. In a double-blinded study of 76 hyposmic patients, there was no significant difference in short-term odor threshold and discrimination scores, as assessed using “Sniffin’ Sticks,” following administration of 65 mg of caffeine, compared to placebo. 16 Finally, during double-blinded, placebo-controlled administration of 100 mg sildenafil (a cGMP type V phosphodiesterase inhibitor), odor threshold and discrimination scores were significantly decreased in comparison with control, indicating worse olfactory function. 17 Reduced olfactory function was attributed to possible nasal congestion, which is a known side effect of sildenafil.
Conclusion
We have shown that a short course of oral pentoxifylline, a phosphodiesterase inhibitor, did not result in significant improvement of olfactory function in patients with post-traumatic impairment. We would suggest that further work into the efficacy of this medication in patients with dysfunction due to other causes should be undertaken.
Footnotes
Authors’ Note
Katherine L. Whitcroft and Volker Gudziol contributed equally. Whitcroft contributed to data analysis and write up. Gudziol contributed to data collection and help with write up. Hummel contributed to draft the study, administrative support, data analysis, and help with write up.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
