Abstract
A retrospective review of children with confirmed hearing loss identified through universal newborn hearing screening (UNHS) in Virginia from 2010 to 2014 was conducted in order to compare the incidence of Joint Committee on Infant Hearing (JCIH) risk factors in children with unilateral hearing loss (UHL) to bilateral hearing loss (BHL). Over the 5-year study period, 1004 children (0.20% of all births) developed a confirmed hearing loss, with 544 (51%) children having at least one JCIH risk factor. Overall, 18% of children with confirmed hearing loss initially passed UNHS. Of all children with risk factors, 226 (42%) demonstrated UHL and 318 (58%) had BHL. The most common risk factors for UHL were neonatal indicators (69%), craniofacial anomalies (30%), stigmata of HL syndromes (14%), and family history (14%). The most common risk factors in BHL were neonatal indicators (49%), family history (27%), stigmata of HL syndromes (19%), and craniofacial anomalies (16%). Children with the risk factor for positive family history were more likely to have BHL, while those with craniofacial anomalies were more likely to have UHL (P < .001). Neonatal indicators were the most commonly identified risk factor in both UHL and BHL populations. Children with UHL were significantly more likely to have craniofacial anomalies, while children with BHL were more likely to have a family history of hearing loss. Further studies assessing the etiology underlying the hearing loss and risk factor associations are warranted.
Introduction
Congenital hearing loss has a prevalence ranging from 0.97 to 2.0 per 1000 newborns. 1 –3 Unilateral hearing loss (UHL) is more prevalent than bilateral hearing loss (BHL) among children aged 6 to 19 years. 4 Children with UHL often went undiagnosed and unnoticed until they reach elementary school. Universal newborn hearing screening (UNHS) was adopted to identify children with hearing loss at an early age. Early detection allows for early intervention, which is shown to play a key role in cognitive and verbal development.
Research has shown that as many as 22% to 35% of children with hearing loss fail at least one grade. 5,6 Additionally, up to 20% are identified as having behavioral attention-deficit/hyperactivity disorder-type problems. 7,8 Progression of hearing loss has been documented in up to 33% of children with hearing loss, emphasizing need for comprehensive early detection and intervention strategies. 9
In 2007, the Joint Committee on Infant Hearing (JCIH) released an updated position statement regarding benchmark ages and guidelines for hearing screening, audiological evaluation, and intervention as well as identifying risk factors for congenital hearing loss. 10 Previous studies have looked at the association between JCIH risk factors and hearing loss. Neonatal indicators such as neonatal intensive care unit stay greater than 5 days, ototoxic medication exposure, and mechanical ventilation have been associated with hearing loss. 11 –13 The association between in utero Toxoplasmosis, Other (syphilis), Rubella, Cytomegalovirus, and Herpes infections and hearing loss is well known. 11,14 Syndromes associated with hearing loss and craniofacial anomalies are strongly predictive of postnatal hearing loss, with craniofacial anomalies inversely related to the risk of progressive hearing loss. 15,16
The prevalence of JCIH risk factors in children with UHL or BHL has been studied independently. 12,17 To our knowledge, no studies have compared JCIH risk factors among groups of children with UHL or BHL. Previous work in this department compared the incidence of JCIH risk factors and comorbid birth defects occurring in children born between January 1, 2002, and December 31, 2010, with UHL or BHL. In UHL, craniofacial anomalies were the most commonly reported risk factor. In BHL, family history of hearing loss was the most frequent risk factor. Cardiovascular anomalies were the most common co-occurring birth defects in infants with either UHL or BHL. 18 The objective of this study is to analyze and compare the JCIH risk factor profiles of infants with UHL or BHL using updated, population-based data in order to better understand the etiology of UHL and BHL.
Materials and Methods
Data were extracted regarding newborn hearing screening and confirmatory diagnoses from the Virginia Early Hearing Detection and Intervention program database for children born between January 1, 2010, and December 31, 2014. Since 1999, all hospitals with neonatal intensive care units or those with infant nurseries must perform newborn hearing tests prior to discharge, per Code of Virginia §32.1–64.1 in Virginia regulations 12 VAC 5–80. The screening method differs across hospitals, but the most common protocol consists of automated auditory brain stem response testing.
All newborns with confirmed BHL and UHL, including those newborns who passed their initial hearing screening and required follow-up due to a known risk factor, were analyzed. Children were grouped into categories regarding status of their newborn hearing screen, confirmatory test outcome, and risk factor status. Hearing loss was defined as having one of the following lnternational Classification of Diseases, Ninth Revision, Clinical Modification: 14 codes reported in both ears at a follow-up assessment by a licensed audiologist: 389.0 (conductive hearing loss), 389.1 (sensorineural hearing loss), 389.2 (mixed hearing loss), and 389.9 (undetermined hearing loss).
Joint Committee on Infant Hearing risk factors were categorized as follows: (a) craniofacial anomaly, (b) family history of permanent childhood hearing loss, (c) head trauma requiring hospitalization, (d) in utero infections, such as cytomegalovirus, herpes, rubella, syphilis, and toxoplasmosis, (e) neonatal indicators for hearing loss such as neonatal care of more than 5 days or extracorporeal membrane oxygenation (ECMO), assisted ventilation, exposure to ototoxic medications (gentamycin or tobramycin) or loop diuretics (furosemide), and hyperbilirubinemia that requires exchange transfusion, and (f) stigmata of syndrome with known hearing loss.
The loss to follow-up rate was calculated by dividing those patients in the Virginia Department of Health who failed to return for a confirmatory result (ie, either no hearing loss on follow-up testing or a diagnosis of confirmed hearing loss on follow-up testing) by those who required follow-up screening (ie, those who failed their UNHS or had a JCIH risk factor). The authors provided a minimum of 2 years for follow-up from birth to ensure all follow-up data would be recorded into the system. This certifies that there is no missing data on any patient who followed up on any time from birth to 2 years of age.
Results
From January 1, 2010, to December 31, 2014, there were 504 654 infants born in the Commonwealth of Virginia who underwent UNHS. Overall, only 0.2% (1004 patients) were ultimately diagnosed with hearing loss. Of these, 581 (0.12%) patients had BHL, while 423 (0.08%) patients were diagnosed with UHL. This equates to a total rate of hearing loss in our study of 0.21%, which is comparable with previous work we have done and rates reported in the literature. 1 –3
In this study, 36 917 (7.3%) infants required follow-up due to either a failed UNHS or because they had the presence of a risk factor. In this cohort, 21 882 infants required follow-up secondary to the presence of JCIH risk factors. Of these, 125 (<1%) infants ultimately developed a confirmed hearing loss, with unilateral and BHL rates of approximately 46% and 54%, respectively. There was a loss to follow-up of 23 645 (64%) infants who never returned for confirmatory testing either after failing UNHS or passing with risk.
In the cohort of infants requiring follow-up, 13 544 (2.7%) initially failed their UNHS, and of those, 3.1% eventually were diagnosed with hearing loss. In this group, at least one JCIH risk factor was identified in 168 (6.2%) infants with confirmed UHL and 251 (9.3%) infants with confirmed BHL.
When analyzing all patients with documented hearing loss (regardless of whether they initially passed their UNHS), the most common risk factors for UHL were neonatal indicators (69%), craniofacial anomalies (30%), stigmata of HL syndromes (14%), and family history (14%). The most common risk factors in BHL were neonatal indicators (49%), family history (27%), stigmata of HL syndromes (19%), and craniofacial anomalies (16%; Table 1). The fact that neonatal indicators were the most commonly identified JCIH risk factor in both cohorts is likely secondary to the broad nature of the category. Family history of hearing loss was significantly more common in infants with confirmed BHL when compared to those with confirmed UHL, while craniofacial anomalies were more common in infants with confirmed UHL compared to those with BHL, P < .001 (Figure 1).
Joint Committee on Infant Hearing Risk Factors Present in Infants Diagnosed With BHL Versus UHL.a
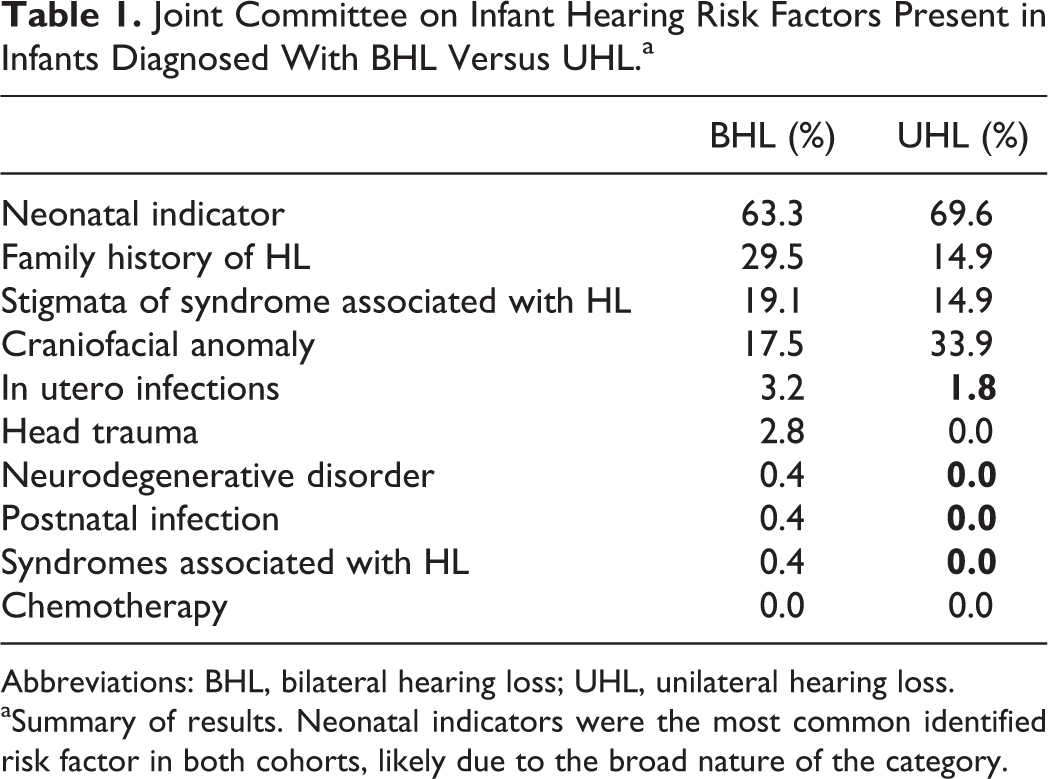
Abbreviations: BHL, bilateral hearing loss; UHL, unilateral hearing loss.
aSummary of results. Neonatal indicators were the most common identified risk factor in both cohorts, likely due to the broad nature of the category.
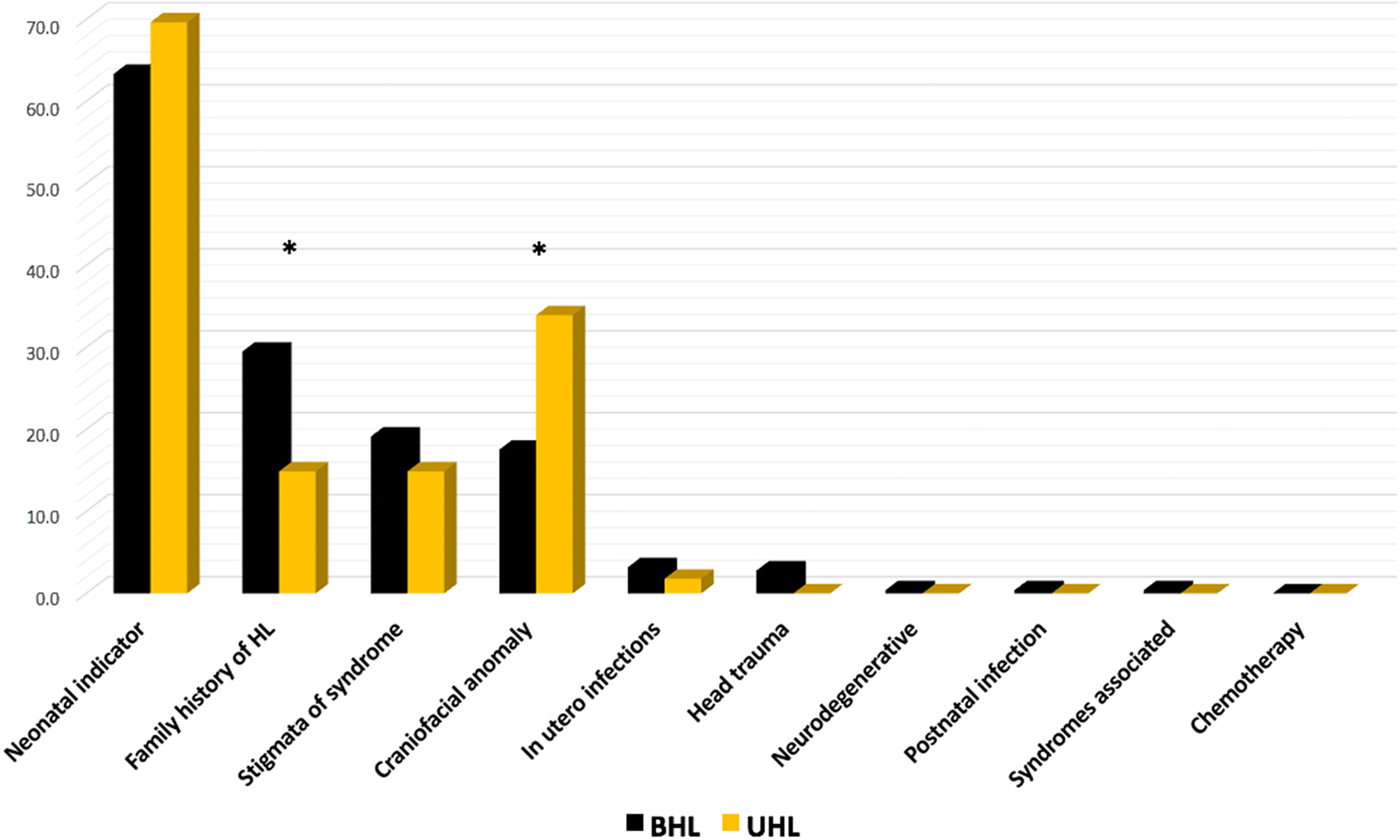
Joint Committee on Infant Hearing risk factors present in infants diagnosed with BHL versus UHL. Graph comparing rates of JCIH risk factors present among infants undergoing UNHS in the Commonwealth of Virginia 2010 to 2014. Family history of hearing loss was significantly more common in infants with BHL (30%) than UHL (15%), while craniofacial anomalies were more common in infants with UHL (34%) rather than BHL (18%), P < .001. BHL indicates bilateral hearing loss; UHL, unilateral hearing loss; UNHS, universal newborn hearing screening.
Discussion
Our study presents the results from the statewide analysis of associated JCIH risk factors in a cohort of infants with confirmed UHL and HL undergoing UNHS from January 1, 2010, to December 31, 2014. Identification of JCIH risk factors for hearing loss is an important part of diagnosis and needs to be integrated into an etiologic focus to newborn hearing screening. Research has shown that up to 30% of infants with confirmed UHL had a JCIH risk factor.17 In addition, this study confirms earlier work showing distinct differences in risk factor associations between BHL and UHL cohorts.18 Additionally, the most common risk factor identified among our entire study cohort was a constellation of neonatal intensive care–related issues that could potentially cause hearing loss, that is, “neonatal indicators.” This is a diverse grouping of interventions and physiologic processes related to neonatal intensive care. Increase precision in defining the causative neonatal indicator (eg, ECMO, hyperbilirubinemia) may be more beneficial in identifying those infants at greatest risk of hearing loss. Nevertheless, the ability to distinguish risk factors most commonly associated with the 2 types of hearing loss can significantly help clinicians identify those at risk and thereby implement compensatory strategies earlier. This is particularly important in those infants with the JCIH risk factors that also pass their newborn hearing screen. Previous studies from the current authors have shown that of this particular cohort of patients, 0.8% were found to have hearing loss at a voluntary rescreening 1 month later. 19 Knowledge of the relationship between these risk factors and hearing loss may prompt providers to advocate for retesting sooner, which can help identify those who would significantly benefit from early aural rehabilitation.
Our study is not without weaknesses; however, the data from which we extracted our results are reliant upon provider-reported input. Thus, risk factors may be more commonly reported due to their ease of diagnosis rather than true incidence. Underreporting or erroneous reporting of birth defects and other risk factors for hearing loss would lead to a skewed association with any existing hearing loss. Additionally, there was a high rate of patient loss to follow-up, which almost certainly has led to an underestimation of the true incidence of hearing loss, as the authors do not have confirmation of hearing loss or the absence of hearing loss in a large proportion of children. Further research is ongoing by the current authors regarding the rates of infants lost to follow-up and those ultimately diagnosed with hearing loss.
Conclusion
In conclusion, approximately 51% of children with confirmed BHL and confirmed UHL had a JCIH risk factor. For those infants with BHL, the most commonly reported risk factors were family history of hearing loss and neonatal indicators. Conversely, for those patients with UHL, the most commonly reported risk factors were craniofacial anomalies and neonatal indicators. Undoubtedly, it is important to recognize children at risk for hearing loss and to perform confirmatory testing in a timely manner in order to expedite the initiation of intervention strategies. Our study demonstrates that the absence of JCIH risk factors does not preclude the development of hearing loss. Further studies are needed to define the underlying etiology of congenital hearing loss and better define the associated risk factors.
Footnotes
Authors’ Note
Triological Society Poster, Combined Otolaryngology Spring Meetings, San Diego, California, April 26-30, 2017.
Acknowledgment
The authors would like to thank Dr Elizabeth Musser of the Virginia Department of Health for her contributions to this original article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
