Abstract
The objective of our study was to assess the long-term effectiveness of balloon catheter sinuplasty in the treatment of pediatric chronic maxillary sinusitis following failed first-line medical and surgical management with adenoidectomy. Pediatric patients younger than 12 years were reviewed for having failed previous adenoidectomy and undergone a balloon catheter sinuplasty between August 2006 and March 2011 for chronic rhinosinusitis. Demographic data and clinical characteristics were recorded in patients who met inclusion criteria. Outcomes were assessed by need for functional endoscopic sinus surgery and persistence of chronic infection within at least 5 years of follow-up. Failure was defined by any child who was rediagnosed with chronic sinus disease after balloon catheter sinuplasty or who required endoscopic sinus surgery within 5 years. Sixty-two children were reviewed, with 38 patients meeting inclusion criteria (prior adenoidectomy, a preoperative Lund-Mackay score of ≥5, and balloon catheter sinuplasty). The mean age (standard deviation) was 6.76 (2.27) years with an age range of 2 to 11 years. Eight children (21.1%, P < .01) continued to have chronic sinus complaints following balloon procedure, with 5 (13.1%) individuals requiring eventual endoscopic surgery within 5 years. Age, asthma, allergy, and gender did not have any statistically significant impact on outcome. Balloon catheter sinuplasty is an effective long-term alternative for the treatment of chronic rhinosinusitis in pediatric patients, hoping to avoid further infections and eventual endoscopic surgery.
Keywords
Introduction
Chronic rhinosinusitis (CRS) is one of the most common diseases afflicting the general population, estimated to affect up to 12% of the population in the United States. 1 Nearly $1.8 billion is spent on treating CRS in children younger than 12 years annually. 1 Pediatric CRS may start as a childhood viral upper respiratory tract infection initially, but up to 13% of these patients may have symptoms that progress to a chronic disease. 2 A majority of children will improve with medical therapy, such as oral antibiotics, allergy therapy, nasal saline irrigations, topical nasal steroids, and asthma control. 3 However, when symptoms continue despite maximal medical management, surgical intervention is indicated. 2
Current clinical guidelines recommend that adenoidectomy is the first-line surgical intervention for children with CRS. 3 However, even with adenoidectomy, sinus disease may persist in up to 50% of cases. 4 Success rate of first-line adenoidectomy declines even further with the presence of asthma in children aged 6 years or younger. 5 For patients who have failed both medical management and adenoidectomy, functional endoscopic sinus surgery (FESS) has been shown to be an effective next-step procedure for treating CRS. 2 In fact, in some cases, FESS is more effective than adenoidectomy alone for some patients with complicated sinus disease. 5 However, it can be technically difficult and has the potential for serious complications. 6
First described in the early 2000s, balloon catheter sinuplasty (BCS) has been shown to be an available treatment modality for pediatric CRS. However, its proven role in treating disease has not yet been elucidated. 2 In 2012, our institution looked at the success rate of BCS in the first year and found that 81% of children achieved significant symptomatic improvement based on the SN-5 questionnaire. 7 In this study, we evaluated the long-term effectiveness of BCS in the treatment of pediatric CRS following failed first-line surgical management with adenoidectomy. Currently, the most recently clinical consensus statements suggest that an FESS be performed after failure to control sinus disease with adenoidectomy. 2 Our aim is to assess the utility of BCS as a long-term, less invasive alternative to FESS for resolution of chronic disease in the hopes that endoscopic surgery can be avoided in the pediatric population.
Patients and Methods
This study was performed at a large, academic tertiary care facility, following institutional review board approval. A computational search of all pediatric patients referred to the otolaryngology clinic for CRS was performed. Search was limited to patients younger than 13 years from August 2006 to March 2011. All patients were managed by the same attending otolaryngologist. As per recent guidelines, CRS was defined by at least 90 continuous days of 2 or more symptoms of purulent rhinorrhea, nasal obstruction, facial pressure, cough, or endoscopic signs of mucosal edema, purulence, or polyposis. 2,8 To have been considered for balloon sinuplasty, patients would have had to have failed maximal medical therapy as defined by the European Position Paper on Rhinosinusitis and Nasal Polyps, which endorses 3 to 4 weeks of oral or intravenous antibiotics, topical nasal steroid sprays, and allergy management when appropriate. 8
This subset of patients was further reviewed to include only those who (1) previously underwent adenoidectomy, (2) had a computed tomography (CT) scan performed of their sinuses prior to BCS, and (3) were available for follow-up for at least 5 years postoperatively. Patients who were asymptomatic for at least 3 years postoperatively were discharged from clinic but still eligible as long as they did not return for sinus complaints within 5 years of BCS. The CT scans of the sinuses were graded using the Lund-Mackay system by the senior author. 9 Only children with a Lund-Mackay score of ≥5 were included, as this is consistent with a diagnosis of CRS that is unlikely to be controlled by adenoidectomy alone. 10 Most children had at least unilateral or bilateral maxillary sinus disease, although some had additional ethmoid disease that was not addressed at the time of BCS. Each BCS was performed following positive maxillary sinus disease on CT scan prior to surgery with diagnostic criteria for CRS. Exclusion criteria included children having underlying conditions predisposing for CRS, including immunodeficiency disorders, ciliary motility disorders, or cystic fibrosis.
Data extracted from the available notes within the electronic medical record included the following: gender, birthdate, prior adenoidectomy, prior CT scan, date of BCS, office visits for monitoring of sinus infections, number of antibiotic treatments required post-BCS, and subsequent surgeries after initial BCS with associated age and time frame between surgeries. Additional data into past medical history looking at asthma, allergies/allergic rhinitis, and aforementioned congenital immunodeficiency were also recorded. Diagnosis of medical conditions (allergies, asthma, etc) was based on their presence either in the medical notation by the treating otolaryngologist or within the medical record via International Classification of Diseases, Ninth Revision codification at the time of BCS.
For all patients included in this study, BCS was performed by the senior author in the following manner under general anesthesia. The nasal passage was decongested with oxymetazoline-soaked pledgets. Under direct visualization through a rigid endoscope, the sinus guide catheter was inserted behind the uncinate process. A flexible guidewire was passed through the catheter into the maxillary sinus. Confirmation that the guidewire was in the sinus was made with fluoroscopy or transillumination. The sinus balloon catheter was passed over the guidewire into the sinus and placed across the ostium. After positioning was confirmed, the balloon was inflated, thereby enlarging the natural ostium. The BCS was performed either to one maxillary sinus or bilaterally to both sinuses depending on the CT scan findings.
The children were followed at 3- to 6-month intervals post-BCS for up to 5 years. At each clinical visit, the patient or caregiver was asked about the major symptoms of nasal obstruction or stuffiness, yellow/green rhinorrhea, or postnasal discharge, cough, and facial pain or headache since their last visit. In addition, questions involving diagnosed sinus infection or need for antibiotic prescription for sinus disease were also documented. The questions were subjective because, when the study was initiated, no validated chronic sinusitis instruments were available for use. Any child who had documented sinus infections requiring antibiotics more than 5 times within a 12-month period (whether for acute recurrent or solitary recalcitrant sinus infections), who was rediagnosed with CRS as defined by the European Guidelines, 8 or who required an FESS was considered a treatment failure if the event occurred within 5 years of the initial BCS. Patients who met criteria to have subsequent endoscopic sinus surgery, but did not due to parental decision-making, were also considered for failure criteria.
Statistical evaluation was executed using IBM SPSS Statistics (version 24.0; IBM Corp, Armonk, New York) software. Failure criteria were analyzed using binomial significance testing, with a null test statistic for BCS set at 0.99. This test statistic was utilized as it is consistent with the clinical consensus that an FESS would otherwise be performed in CRS patients who failed medical management and prior adenoidectomy. Any success expected from BCS in preventing FESS in pediatric patients would counter current recommended guidelines, so a null test statistic for failure would be expected at 0.99. χ2 analysis was performed for nominal data with Pearson correlation, while independent samples t tests were performed for interval data (time between procedures and age characteristics). Statistical significance was defined as P ≤ .05.
Results
A total of 62 children initially met initial criteria for having undergone a BCS at our institution. After excluding patients who did not have a prior adenoidectomy and had a Lund-Mackay score <5, 38 patients (age range: 2-11 years old) met the inclusion criteria for our study. Table 1 summarizes the demographic and operative characteristics of these patients. Mean age (standard deviation [SD]) at the time of BCS was 6.76 (2.27) years. Male children (22; 57.8%) outnumbered females (16; 42.2%) in this patient population. Allergic rhinitis (15; 39.4%) and asthma (13; 34.2%) were also prevalent among these patients. The mean CT Lund-MacKay score was 8.63 (3.47) in all patients undergoing BCS, with 11 patients (28.9%) having findings indicative of ethmoid disease. The average time of follow-up was 3.84 years post-BCS.
Demographics and Clinical Characteristics of Pediatric Patients With CRS.
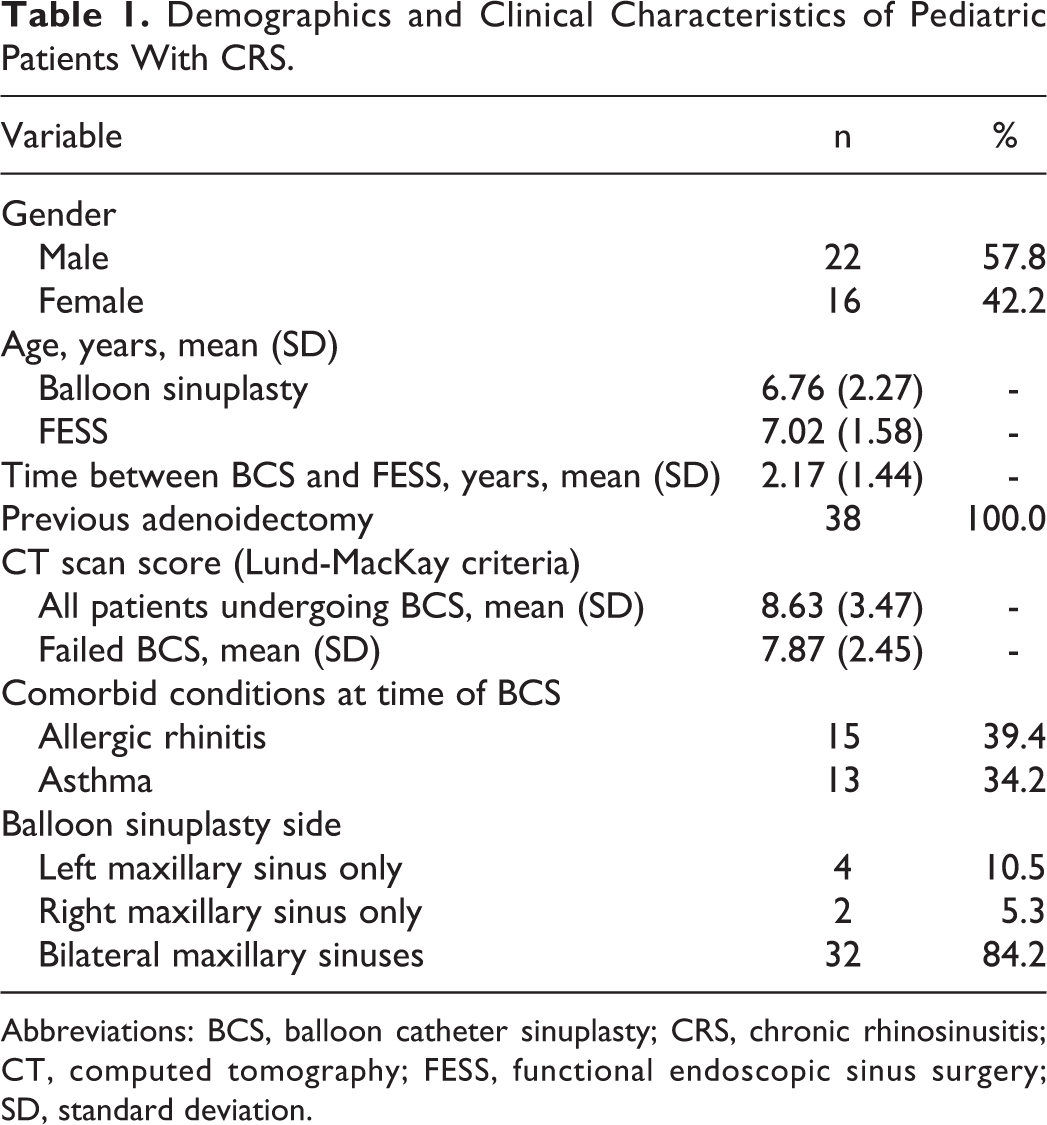
Abbreviations: BCS, balloon catheter sinuplasty; CRS, chronic rhinosinusitis; CT, computed tomography; FESS, functional endoscopic sinus surgery; SD, standard deviation.
Overall, BCS was a long-term success for 30 (78.9%) patients in preventing further sinus infections and subsequent sinonasal surgery. Eight (21.1%) children met failure criteria within 5 years after BCS due to continued symptoms of CRS despite medical therapy (P < .01). Of these 8 patients, 5 (13.1%) children underwent subsequent FESS to control their symptoms of CRS. The average time (SD) to surgical failure, that is, requiring FESS, was 2.18 (1.44) years. The mean CT Lund-MacKay score for patients who failed BCS was 7.87 (2.45).
Fortunately, 33 (86.9%) patients were able to avoid having further surgery within 5 years of their balloon procedure, although 3 did not meet success criteria due to subsequent infections. The majority of patients (26; 68.4%) had zero documented infections or need for sinus-related antibiotics within 5 years of BCS. Only 4 (10.5%) children had subsequent episodes of acute sinusitis but did not meet failure criteria as their disease was controlled with either single courses of antibiotics or minimal medical management. Although we did not address ethmoid disease intraoperatively, 9 (82%) patients with additional ethmoid opacification with the maxillary sinus disease on CT scan met success criteria following BCS. This success rate was similar to patients who only had maxillary sinus disease (21, 78%). No statistically significant data were found relating failure criteria to age, gender, asthma, or allergic rhinitis. Table 2 summarizes the statistical analysis of these patients.
Statistical Analysis for Balloon Sinuplasty Success in Pediatric Patients With CRS.
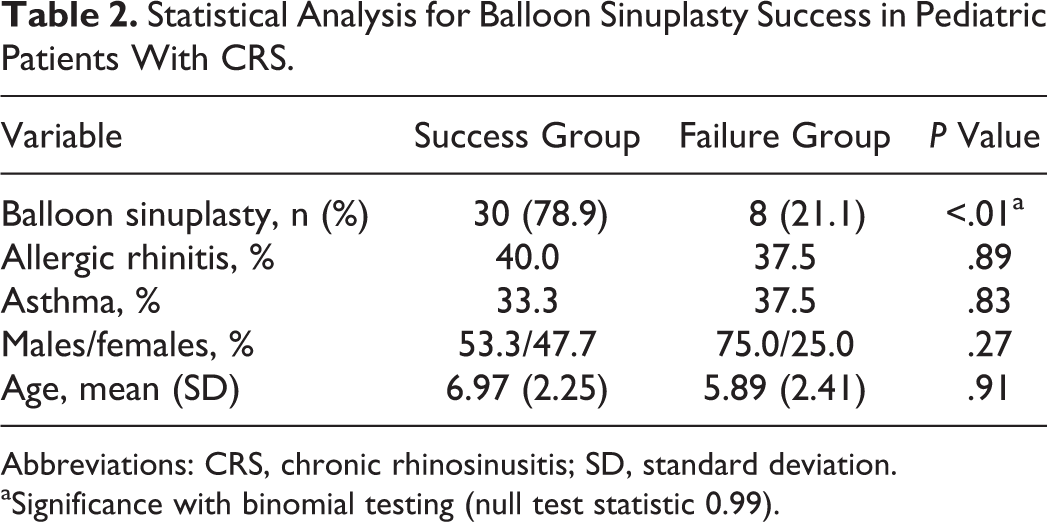
Abbreviations: CRS, chronic rhinosinusitis; SD, standard deviation.
aSignificance with binomial testing (null test statistic 0.99).
Discussion
In 2016, the International Consensus Statement on Allergy and Rhinology (ICAR) released a clinical consensus statement for the optimal diagnosis and management for pediatric CRS, which followed up on the American Academy of Otolaryngology (AAO) recommendations from 2014. 2,11 Concerning first-line therapy, the group determined that culture-directed antibiotic therapy improves outcomes in patients who did not respond to empiric antibiotic treatment. 2 Long-term appropriate antibiotic therapy and, in some situations, intravenous antibiotics may be necessary. 12 Nasal saline irrigations and intranasal corticosteroid sprays are also first-line medical therapy with documented benefits. 11 Children who did not respond to medical treatment might require alternative therapy. Although asthma and/or allergic rhinitis may be strong contributors to symptomology, negatively affecting up to 13% of children, 13 very few children will require surgical intervention for their disease.
Adenoidectomy reached consensus by the AAO and ICAR as an effective first-line surgical treatment for children up to 6 years with CRS and supported for kids up to 12 years of age. 2,11 Although previous research has shown the independent effectiveness of adenoidectomy varies among studies, the average success rate typically settles around 50% of patients with CRS. 4,7 Adenoidectomy is recommended as a better surgical intervention for a child with chronic adenoiditis without sinusitis, especially in children younger than 13 years. 2 When children have chronic adenoiditis and CRS, the success rate of adenoidectomy falls significantly. 14 In such situations, FESS is an effective procedure for the treatment of pediatric CRS only after medical therapy and adenoidectomy have failed. 2
Previous research suggests that BCS can offer an alternative to FESS that is effective over short-term periods in addition to being a less invasive operation comparatively. 7,14 With BCS, there is no sinonasal tissue removal compared to FESS, making the possibility of affecting facial development relatively low. While the AAO did consider BCS for the treatment of pediatric CRS, the consensus statement reached determined there was insufficient evidence to compare BCS to FESS at the time of publication. 2 In addition, the panel did not reach consensus regarding the effectiveness of balloon sinuplasty for the treatment of pediatric CRS alone, but did reach near consensus for its safety. 2 In 2016, ICAR also did not give any consensus regarding the role of BCS in the treatment to pediatric CRS due to lack of robust evidence, especially long-term effectiveness of this technology. 11
Our research represents one of the first studies evaluating the long-term effectiveness of BCS as an alternative step for FESS in pediatric patients less than 13 years. Over a 3-year period, 78.9% of patients who underwent BCS no longer had recurrent or persistent sinus infections (5 infections over 12 months), characterized by nasal obstruction, congestion, yellow/green discharge, cough, headache, facial pain/pressure, or need for antibiotics (P < .01). Based on current guidelines, since all these patients presented to our clinic following prior adenoidectomy and failed medical management for CRS, FESS would have been viable alternative for treatment. By performing a BCS instead of FESS, 86.9% of our pediatric patients did not require any further surgical intervention within 5 years of their balloon sinuplasty. Among those who met failure criteria, there was no difference in their gender, age, asthma, or allergy status (Table 2). If patients with any sinus symptoms or need for antibiotics were included in our failure group, 68.4% of children would still have benefited from balloon procedure as they did not have any documented symptoms or antibiotic requirements within their medical chart.
Balloon catheter sinuplasty may not be suitable for some children, as several patients were excluded from our study due to underlying immunodeficiency. In addition, some patients were not candidates for BCS, specifically those who have hypoplastic maxillary sinuses or children with significant ethmoid sinusitis from CT scan evaluation. Previous research found that children with a Lund-Mackay score ≥5 are more likely to have CRS as opposed to allergic sinusitis or adenoiditis, making children with Lund-Mackay scores <5 also not suitable for our study. 10,15 Therefore, the choice of surgical intervention should be individualized for every patient.
A significant limitation of this study, however, was the retrospective nature of data accumulation. Because no objective or validated study was utilized at the time of CRS diagnosis to determine symptom recurrence, documentation of sinus disease was entirely from subjective medical notation. In addition, as this is a 1-arm retrospective study with no comparison to other treatment modalities, BCS as a comparative therapy cannot be analyzed. However, as mentioned previously, this patient population would otherwise have undergone endoscopic surgery due to previously failed management of CRS. In addition, as this is a pediatric patient population, it is plausible that sinus development allowed some patients to “grow out” of their disease rather than direct effect from the BCS.
Balloon catheter sinuplasty is an effective long-term alternative for the treatment of CRS in pediatric patients who continue to have infections following adenoidectomy and medical management. For parents hoping to avoid further infections and eventual endoscopic surgery in their children, BCS is a viable solution in selected cases due to its efficacy and well-established safety profile compared to FESS. Further studies, particularly a prospective randomized trial comparing balloon sinuplasty to endoscopic surgery following adenoidectomy, would be important to evaluate in the future based on recent clinical guidelines.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
