Abstract
Botulinum toxin is the most commonly performed facial cosmetic procedure and pain at the injection site is a frequent patient concern. While various topical interventions have been described for analgesia, there have not been any studies comparing different techniques. We compared the use of a vibratory stimulus, ice pack application, and no intervention on injection site pain for cosmetic botulinum toxin injection. A prospective-, randomized-, individual-controlled study was conducted using a visual analog scale to assess pain. Patients received bilateral glabellar injections, with randomization into unilateral vibration, unilateral ice application, or vibration and ice on either side. We analyzed 88 injections on 22 patients. Mean visual analog scores were 26.5 (standard deviation [SD]: 23.1) among injections with vibration, 24.4 (SD: 22.9) with ice, and 29.4 (SD: 27.1) without analgesia. There was no significant difference in pain scale scores with the use of vibration, ice, or no topical anesthesia (P = .737). Further, pain scale scores did not differ significantly between medial and lateral injections nor did patients have a reduction in pain on either side of the forehead regardless of which method was used. While there may be a role for topical interventions to improve injection site analgesia, we maintain that consistently proper technique plays a greater role in improving patient tolerance. Future studies will continue to investigate the role of topical anesthesia in cosmetic facial injections and address patient-specific factors contributing to discomfort.
Introduction
In 2016, an estimated 4.6 million botulinum toxin injection procedures were performed for cosmetic purposes in the United States. 1 Forehead and periorbital injections are a well-known source of patient discomfort and anxiety, 2 which can delay scheduling of treatments in 31% of patients. 3 Strategies to mitigate botulinum toxin injection pain include dilution with sterile saline, 4 utilizing a 30-gauge insulin syringe, 5 and incorporating topical analgesia during the procedure, such as ice, vapocoolant sprays, or anesthetic ointments. 6 The 2004 Consensus Recommendations on the Use of Botulinum Toxin Type A in Facial Aesthetics reported 65% of panel members supporting the use of topical anesthesia. 7 Although some patients find these strategies helpful to ease discomfort, a clear benefit has not been equivocally proven. 8
The application of a vibratory stimulus has been shown to improve patient discomfort associated with cosmetic botulinum toxin injections. 6,9 The “gate-control” theory, initially proposed in 1965, suggests that intensity of pain can be mitigated by concurrent non-noxious stimulation. 10,11 Studies have applied this theory to eyelid local anesthesia injections, 12 dermal filler injections, 13 and keloid steroid injections 14 with reduction in pain outcomes. Thermally anesthetizing the skin with topical ice has also been shown to be an effective method of reducing facial botulinum toxin injection pain. 15,16 Finally, the application of eutectic mixture of local anesthetics (EMLA - mixture of 2.5% lidocaine and 2.5% prilocaine) cream has reduced pain associated with botulinum toxin injections with favorable results. 2
Despite various options, a prospective comparison between topical analgesia techniques for botulinum toxin injections has not been investigated. This study compared the use of a vibratory stimulus with thermal analgesia on patient discomfort with cosmetic botulinum toxin injections. We utilized 2 well known and easily obtained topical therapies to determine which technique, if any, improved facial injection pain.
Patients and Methods
This prospective, randomized, self-control trial was approved by the institutional review board of SUNY Upstate Medical University . Men and women aged 18 to 65 years old were recruited to receive cosmetic injections of onabotulinum toxin A (Botox, Allergan, Inc, Irvine, California). Patients were included if they had no prior facial surgeries, no history of cosmetic facial injections, and had glabellar lines or furrowed brow lines amenable to Botox injection as determined by physical examination. Patients were excluded if they had prior facial surgeries or facial injections, were pregnant or breastfeeding, had a current or previous history of immunodeficiency, neuromuscular disorder, hypersensitivity or allergy to botulinum toxin, cardiovascular disease, inflammatory skin disorder, or an active skin infection near the intended injection sites.
Patients were randomized in blocks of 3 to 1 of 3 treatment arms: unilateral vibration analgesia, unilateral ice pack analgesia, or analgesia with vibration on one side of the forehead and ice pack on the other (Figure 1). The side to receive intervention(s) was also randomly determined. A fellowship-trained facial plastic surgeon (ACS) performed all injections utilizing a uniform technique. Briefly, an appropriate timeout was performed prior to each procedure and the forehead skin was prepped with alcohol. Using 2.5 mL of 0.9% sterile saline, we diluted 100 unit vials of Botox A so that each 0.1 mL injected contained 4 units of Botox A. A 30-gauge 1 mL 1/2 inch needle was used to inject 0.1 mL into 5 total injection points along the bilateral corrugator supercilii muscles. Pain levels were recorded on a visual analog scale (VAS), ranging from no pain to worst possible pain. Patients placed a mark along a 10-cm line and these were measured in millimeters to correspond to a VAS score. While injections were performed at the midline glabellar region, this was performed for cosmetic balance only. We did not include this in the final analysis since we were unsure how topical therapies applied unilaterally would affect the midline. A new needle was used for each injections site and the surgeon rapidly inserted the needle through the skin and slowly injected into the facial muscles. Patients received bilateral injections so that they could serve as their own control.
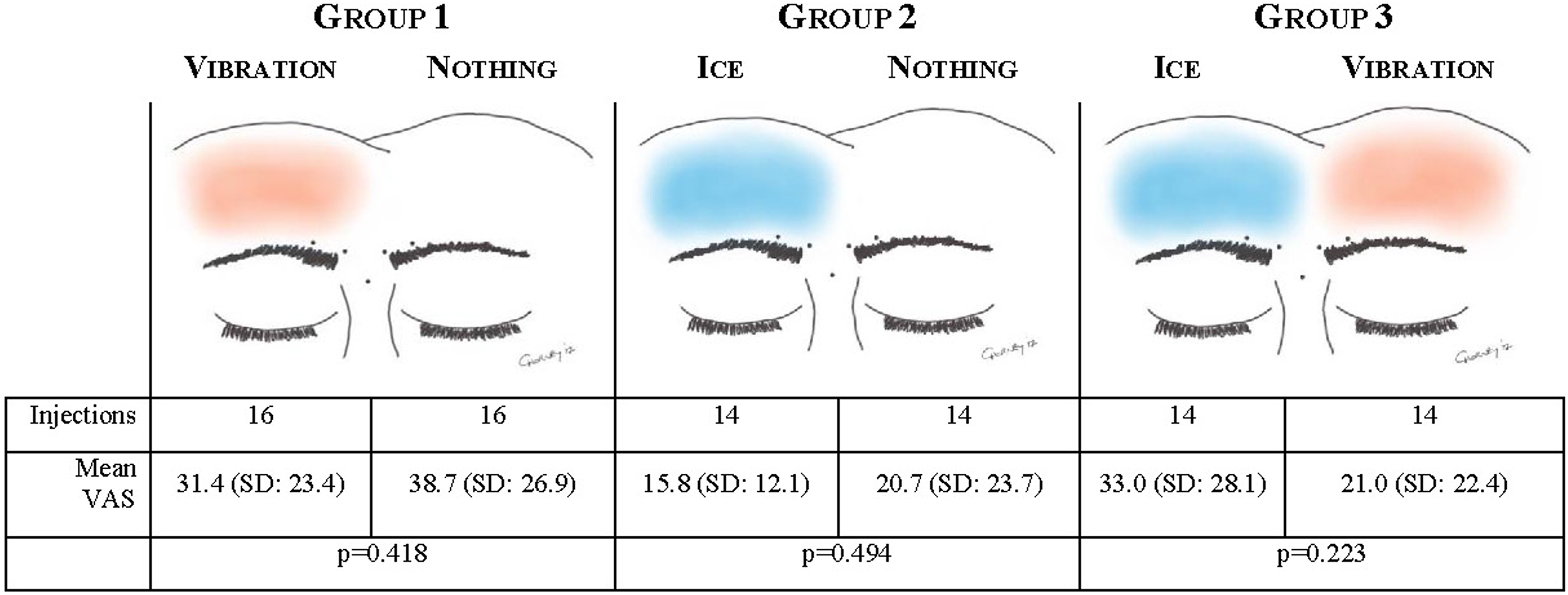
Comparative visual analog scale scores based on analgesia.
If a patient was in the vibration arm, a Buzzy (MMJ Labs, LLC, Atlanta, Georgia) vibratory distractor was applied and held within 2 cm of the injection sites. The ice group had a small reusable cold pack applied to the skin overlying the injection site for 60 seconds prior to injection. On the contralateral injections, no topical anesthesia was utilized and this served as an individual control. A patient could also be randomized to receive both interventions, in which case we randomized, which side had vibration, and which side had ice applied. In between each injection, the patient filled out a VAS identifying how much discomfort they had with the injection. Once all injections were completed, the patients were observed in the office for 15 minutes to assure no complications and discharge instructions were given. One week following the injection, patients were contacted by telephone to identify if any complications had developed and if they were to undergo repeat Botox injection in the future, which analgesia intervention, if any, they would want used.
Statistical analysis was performed using SPSS Statistics for Windows (version 22.0; SPSS, Inc, an IBM Company, Chicago, Illinois). A Student t test for independent samples (α < .05) was used for continuous variable comparison, while 1-way analysis of variance (ANOVA) testing was used with significance set at P < .05 for comparison of means between groups. The primary outcome was the difference in pain with Botox injection experienced in areas treated with the vibration device versus areas that were not pretreated on a VAS. The secondary outcomes were the difference in pain with Botox injection experienced in areas pretreated with the Buzzy device versus areas pretreated with ice, if patients had unilateral improvement in pain with the use of topical analgesia, or if the location of injections were impacted by analgesia.
Results
There were 22 patients who were successfully recruited for this study with a total of 110 botulinum toxin injections performed. As per the protocol, the midline glabellar injections were not included, yielding 88 analyzed injections: 44 lateral corrugator and 44 medial corrugator. Every patient was female, with an average age of 43.9 (standard deviation [SD] 9.89) years, and an average VAS score of 26.8 (SD: 24.3, 95% confidence interval [CI]: 21.7-31.9). Table 1 shows the average pain scores based on analgesia. A total of 8 patients were randomized to receive unilateral vibration, 7 patients received unilateral ice, and 7 patients received both interventions. There were 30 injections given with vibration analgesia, with a mean VAS of 26.5 (SD: 23.1, 95% CI: 17.9-35.2). There were 28 injections given with ice that resulted in a mean VAS of 24.4 (SD: 22.9, 95% CI: 15.5-33.3). Among the 30 injections given without any analgesia, patients had mean VAS scores of 29.4 (SD: 27.1, 95% CI: 19.3.-39.5). Based on 1-way ANOVA testing, there was no statistical difference between mean VAS scores between vibration, ice, and no intervention (P = .737).
Visual Analog Scale Scores Based on Analgesia.
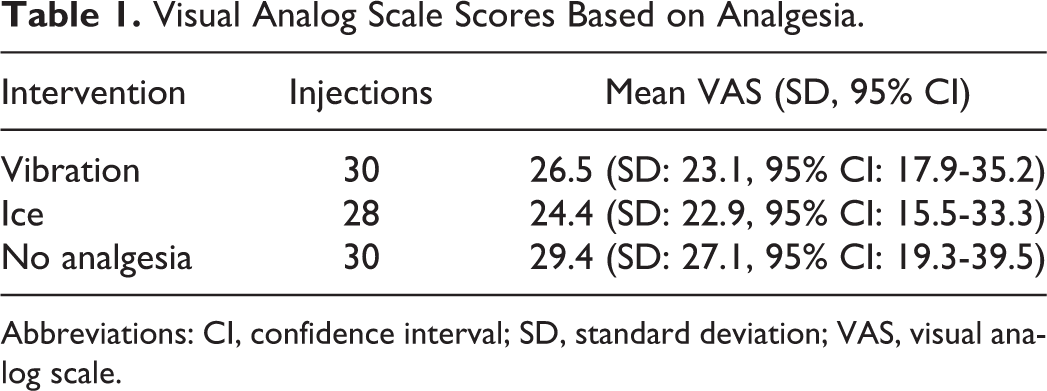
Abbreviations: CI, confidence interval; SD, standard deviation; VAS, visual analog scale.
There was no significant reduction in individual patient pain scores regardless of intervention utilized. As described in Figure 1, there was no statistically significant difference between mean scores. Among the patients receiving unilateral vibration, injection scores were 31.4 (SD: 23.4) with vibration compared to 31.4 (SD: 26.9) without vibration (P = .418). When ice was applied, visual analog scores changed from 20.7 (SD: 23.7) without analgesia to 15.8 (12.1) with the ice pack (P = .521). Finally, when patients received both interventions, ice was more painful than vibration injections (33.0 vs 21.0, P = .223) but this failed to meet statistical significance. Based on follow-up communication, patients did not have a clear preference for preinjection treatment.
When we compared medial corrugator versus lateral corrugator injections, we found no statistically significant impact on pain scores. Among lateral injections, mean scores for ice were 26.0 (SD 24.5), vibration scores were 24.3 (SD 24.7), and no analgesia scores were on average 31.1 (SD 28.3). These failed to meet significant difference (P = .762). Medial corrugator injections resulted in mean pain scores of 22.8 (SD 22.1), 28.7 (SD 22.1), and 27.7 (SD 26.8) for ice, vibration, and no analgesia, respectively (P = .775). There was no difference in pain scales among lateral or medial injections for ice (mean: 26.0 vs 22.8, P = .719), vibration (mean: 24.3 vs 28.7, P = .611), or no analgesia (mean: 31.1 vs 27.7, P = .742).
Discussion
Americans spend over US$1.7 billion dollars every year on the injection of botulinum toxin. 1 Worldwide, this is the most common nonsurgical cosmetic procedure performed 17 and a growing body of literature has been dedicated to reducing needle-associated discomfort. Patients report mild to moderate pain 39% to 69% of the time 3,6,13 and 65% of patients who never had injections expect the pain to be mild to moderate. 3 We were not able to identify a statistically significant difference in pain during cosmetic facial Botox injections between vibration, ice, or no analgesia. Further, there was no impact on whether injections were in the medial or lateral corrugator muscle and patients were not able to identify a strong preference for any particular methodology.
The theory behind vibration as an analgesic method originates from the “closed gate” pain model as described by Melzack and Wall. The Meissner’s corpuscles located in the superficial dermis and Pacinian corpuscles located in the deep dermis and primary endings of muscle spindles are key mechanoreceptors. In response to vibration, they stimulate Aβ fibers and close or “gate” noxious pain that stimulate small C fibers. 10 This same principle explains why pinching, stretching, pressing, or tapping near a needle stick can effectively block the brain’s perception of pain. 18 The principle of thermal analgesia is based on decreased nerve conduction velocity of the C and Aδ fibers that impede nociceptive inputs. 19 Both of these methods are well tolerated, easy to apply, and are inexpensive, making them of particular interest for common injection procedures. 20,21
Several groups have looked at the use of vibratory analgesia for reducing injection-related discomfort during cosmetic procedures. When a vibratory stimulus was applied during the injection of cosmetic botulinum toxin on the face, there was a statistically significant reduction in patient’s pain scores (1.3 ± 0.6 vs 2.4 ± 0.8, P = −0.000). Of the 50 patients, 82% noted that the vibration side of the face had lower pain than the side of their face injected without vibration. Further, 86% these patients preferred to utilize vibratory analgesia for subsequent injections. 6 This study, however, did not compare vibration with ice as was included in our protocol. Injection of the nasolabial folds with dermal filler has been shown to be better tolerated with vibratory stimulation. While 88% of patients found the injections moderate to severely painful without vibration, only 14% with vibration experienced moderate to severe pain. 13 Keloid triamcinolone injections also produced a significant decrease in VAS scores when vibration anesthesia was used. Intralesional injection therapy without vibration was yielded mean visual analog scores of 5.88 ± 2.34 compared to 3.28 ± 1.85 (P < .05) when vibration was used. 14 This study included a variety of located keloids, with only a third located head or neck. Notably, each of these studies used a control group of no intervention. Therefore, our results, though equivocal in our population, is unique in the comparison of 2 common interventions: vibration and ice.
Thermal analgesia with application of cold materials, notably ice, have also been commonly utilized. Botulinum toxin injections for hemifacial spasm or blepharospasm was significantly less painful in a group of patients who applied ice either 5 minutes before or 5 minutes after injection compared to those without ice application. However, after 5 minutes, the mean numeric pain scales were not statistically different in any of the 3 groups. Interestingly, there was a significant decrease in the rate of bleeding after the injections for the group who applied ice prior to injection (P = .0472) compared to those who applied ice afterward, but this same difference was not found when comparing to the control group. 16 Application of ice 5 minutes before cosmetic Botox on one side of a patient’s face yielded statistically significant reduction in injection-related pain compared to the side without ice application. Mean VAS scores dropped from 5.9 to 1.1 (P = .000) on the side of the face with ice applied. 15 Finally, application of crushed ice or a frozen gel mask before multiple botulinum toxin injections for blepharospasm or rhytids in the periocular region decreased the pain sensations by 45%. 22 We must point out that we only placed ice topically for 60 seconds, which is less than other protocols. However, we believe that this is more of an appropriate time frame for application among patients presenting to our practice.
Another common topical analgesic is the EMLA cream, which combines a eutectic mixture of lidocaine and prilocaine. When applied 30 minutes prior to facial botulinum toxin injections, EMLA cream 73% of injections were considered painless compared to 11% of injections on the control side of a patient’s face. In this group of 20 patients, the topical analgesia was preferred to the control side injections in each patient. 2 When EMLA cream was compared to ice periocular Botox injections, there was no difference in pain scores (P = .05). However, either method was considered more effective than no topical intervention (P < .001). 23 Of note, the use of Betacaine ointment prior to botulinum toxin A injections for blepharospasm has been reported to reduce patient-perceived efficacy compared to no intervention. While not formally proven, the authors suggested that temperature and neuronal firing, which affect botulinum toxin, are reduced by topical local anesthetics. 24
We found no complications from utilizing either of these 2 topical analgesia methods. Consistent with prior studies, both vibration and cold analgesia are largely well tolerated. The literature does mention transient paresthesia, bruising, or headaches in 10% of patients with vibration analgesia. 6 There was reporting of one patient who felt her keloid pain was worse with the use of vibration. 14 From the topical standpoint, ice packs are considered cumbersome and not completely effective at reducing or eliminating injection pain. 2 Extra time and cost, albeit small, have also been described as drawbacks. 15 Ice has also been shown as a means to constrict surface blood vessels and potentially decrease postinjection bruising. 22 Both the cost of the vibratory stimulus (US$60) and ice pack (US$4) are negligible compared to the estimated US$376 per botulinum injection procedure. 1
The main limitation of our study is the small sample size. This potentially reduces our ability to detect a difference between groups due to poor power. Nonetheless, previously published prospective-, randomized-, individual-controlled trials assessing analgesia for cosmetic botulinum toxin range from 14 to 52 patients (median 34.5, SD: 16.7). 2,3,6,15,22,25 Having 22 patients with 88 injections still keeps our data within one SD of the average reported patients. The strength of our study is the randomized protocol and the ability to maintain consistent and meticulous injection technique. There have been a number of key steps to mitigating injection site pain in cosmetic procedures. Notably, the use of 30-gauge 1/2 inch insulin syringes not used more than 4 to 6 injections. 5 We replaced the needle between each injection for an optimally sharp needle. Further, reducing needle wobble, perpendicular injection through the skin, and slow infiltration are important means to improve injection technique. 18 Each of these steps were followed in our study and may have been more important to establishing patient comfort. Therefore, the use of vibration or ice may be beneficial in injections with suboptimal technique or where prior patient experience dictates.
In conclusion, there was no difference in cosmetic botulinum toxin injection pain when vibration, ice, or no topical anesthesia was applied. While there may be a role for topical interventions to improve injection site analgesia, we maintain that consistently proper technique plays a greater role in improving patient tolerance. Future studies will continue to investigate the role of topical anesthesia in cosmetic facial injections and address patient-specific factors contributing to discomfort.
Footnotes
Acknowledgments
The authors would like to extend tremendous gratitude to Brenda Singer and Denise Luman for their administrative and clinical support throughout this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
