Abstract

Case Report
A 29-year-old woman presented with a chief complaint of progressively increasing swelling on palate since 2.5 years. Intraoral examination revealed nontender, soft to firm, well-circumscribed mass measuring 3 × 4 cm on the junction of soft and hard palate. The overlying mucosa was intact and normal in color and appearance (Figure 1). No regional lymphadenopathy or any systemic disease was reported. Contrast enhanced computed tomography revealed enhancing soft tissue lesion involving palate measuring 5 × 4.6 × 2.3 cm.
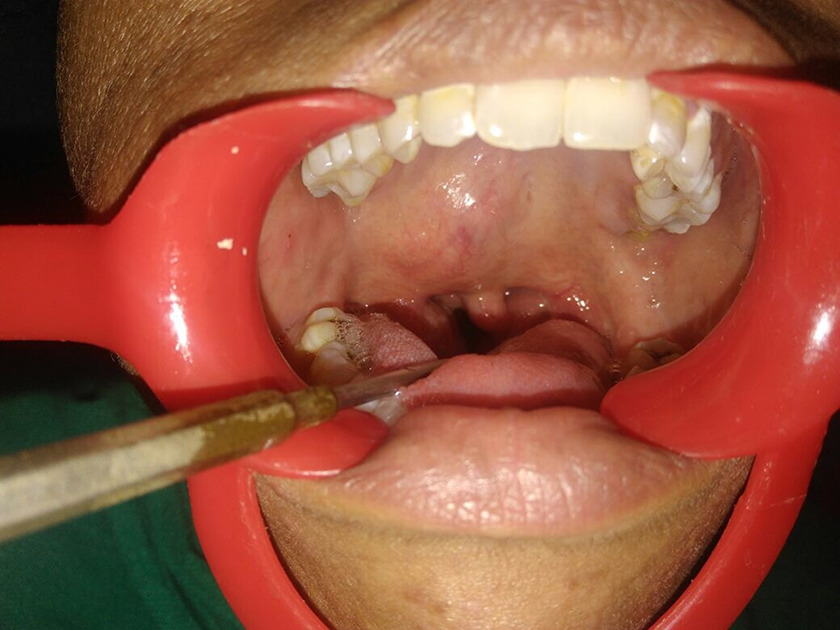
Intraoral photograph revealing the tumor mass.
Fine-needle aspiration cytology (FNAC) revealed monomorphic plasmacytoid cells. Based on clinical, imaging, and FNAC findings, it was provisionally diagnosed as pleomorphic adenoma (PA). Surgical excision was done under general anesthesia.
On histopathological examination, sections from the hematoxylin and eosin–stained slides showed an extremely cellular lesion with scanty connective tissue stroma surrounded by a thin capsule, which was infiltrated by tumor cells (Figure 2A). The tumor mass was composed of plasmacytoid (predominantly) and spindle-shaped cells arranged in the form of sheets and large nests (Figure 2B). Necrosis and hemorrhagic foci were also evident among these neoplastic cells. Few tumor cells around these necrotic and hemorrhagic areas show pleomorphism and hyperchromatic nuclei (Figure 2C). Ductal structures and acinar differentiation constituted less than 1% of all of the microscopic fields analyzed (Figure 2D).
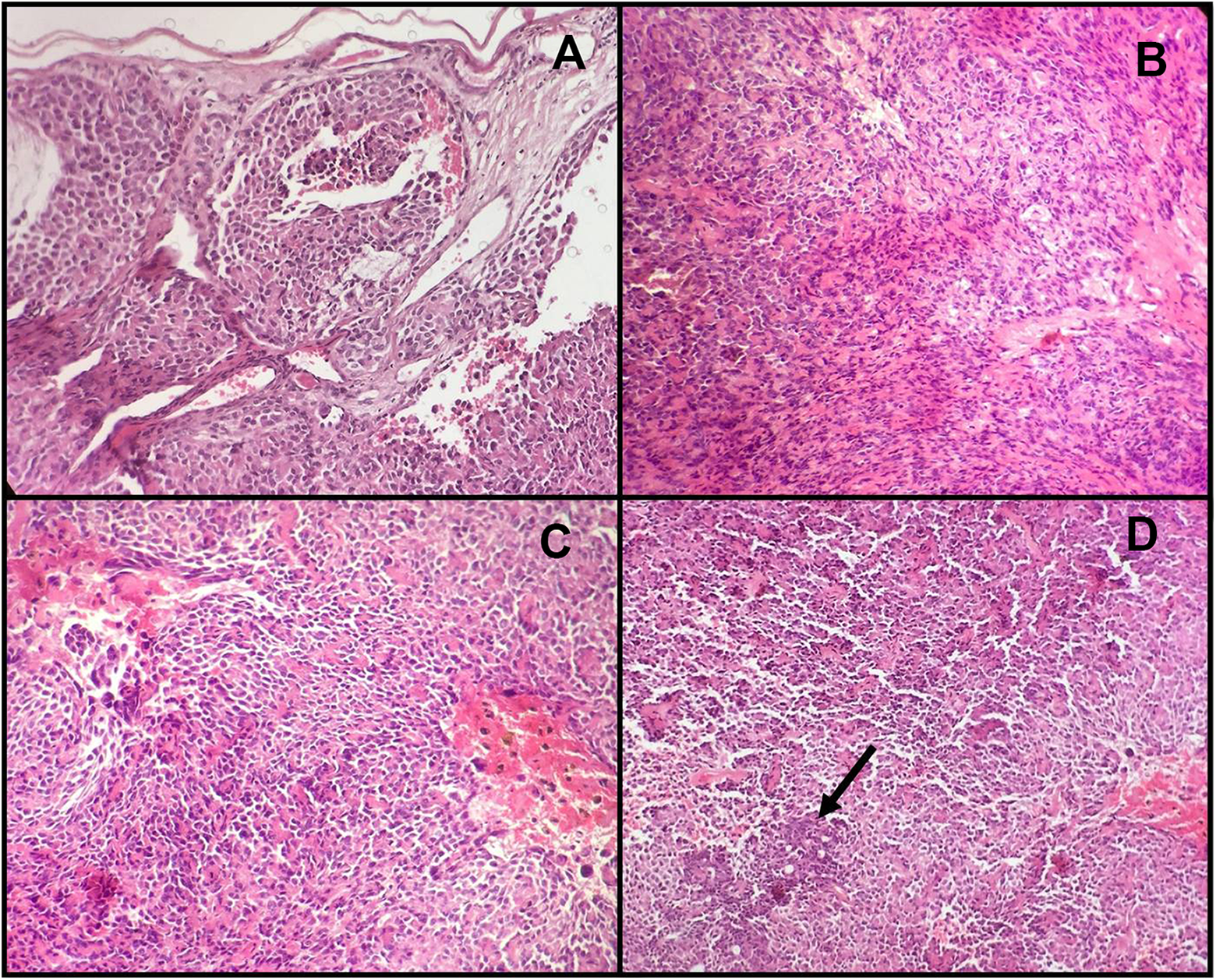
A, Plasmacytoid tumor cells infiltrating thin capsule. B, Areas of plasmacytoid cells intermixed with spindle shaped cells. C, Few pleomorphic tumor cells with hyperchromatic nuclei around necrotic and hemorrhagic areas. D, Ductal structures constitute less than 1% of all of the microscopic fields (hematoxylin and eosin stain).
With the objective of a better understanding of this neoplasm and to corroborate the myoepithelial nature of the neoplastic cells, immunohistochemical analysis was done. The antibodies used were Glial fibrillary acidic protein (GFAP), anti-S100 protein, CD138, and P63. Plasmacytoid and spindle cells showed intense cytoplasmic positivity for S-100 protein (Figure 3A). Plasmacytoid cells show diffuse cytoplasmic positivity for GFAP (Figure 3B), membranous positivity for CD138 (Figure 3C), and scant cytoplasmic positivity for p63 (Figure 3D). Considering the histopathological and immunohistochemical findings, final diagnosis of plasmacytoid myoepithelioma (ME) was established.
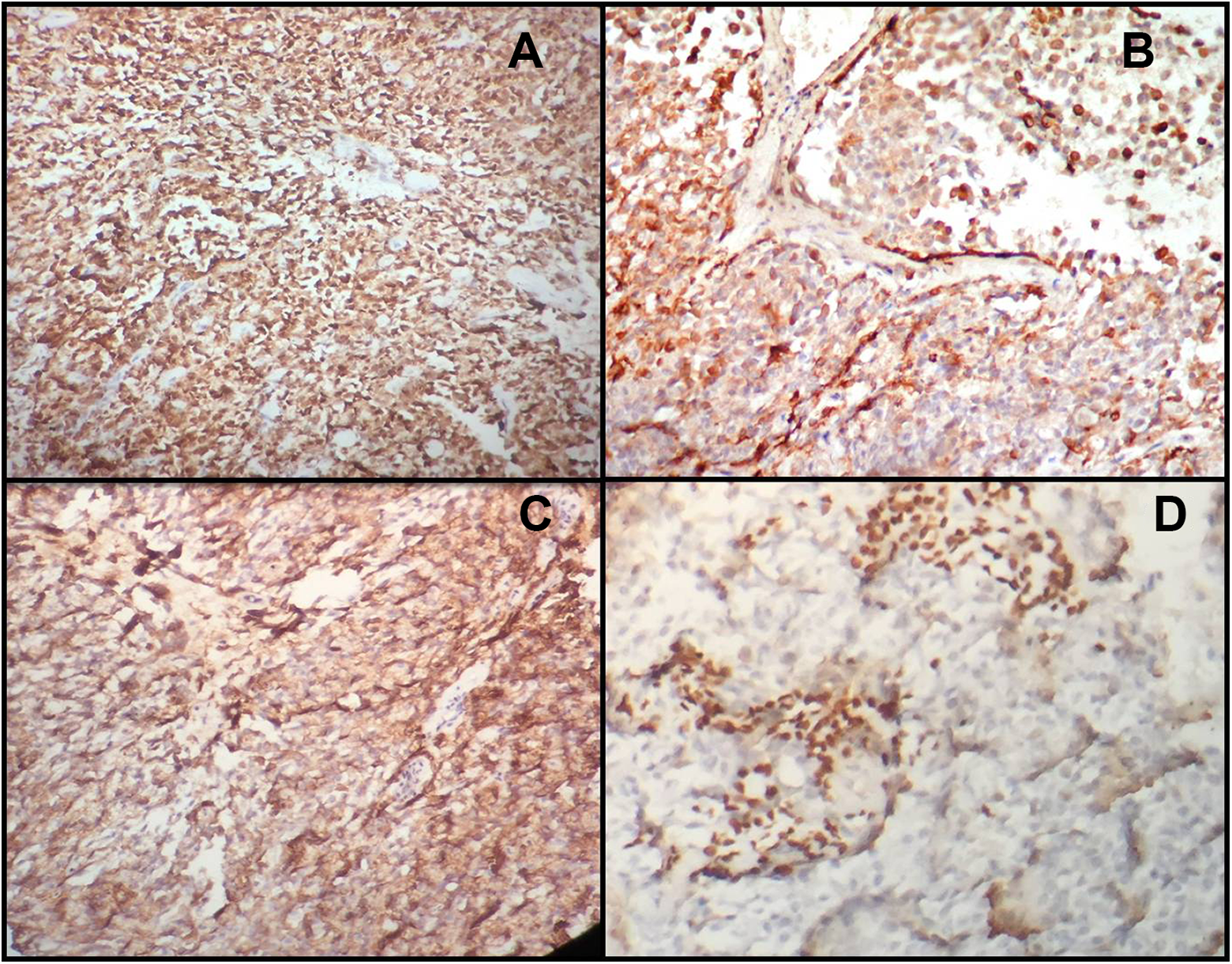
Immunohistochemical study of tumor plasmacytoid cells revealed strong positivity for CD138 (A) and S-100 protein (B), moderate immunoreactivity for Glial fibrillary acid protein (GFAP) (C), and focal positivity for P63 (D).
Keeping in mind, the histopathological picture revealing cellular atypia, necrotic, and hemorrhagic areas, infiltration of the capsule by tumor cells, and also the possibility of recurrence of this tumor as mentioned in the literature, the patient is followed up regularly.
Myoepithelioma is a benign neoplasm of the salivary glands, which accounts for 1% to 1.5% of all glandular tumors. 1 Depending upon the component, cell type ME can be classified as spindle, plasmacytoid (hyaline), reticular, epithelioid, and clear; additionally, mixed histological forms are also described. Plasmacytoid ME arising from palatal minor salivary glands is considered to be a very rare entity. 2 Microscopically, these tumors show various cellular patterns such as spindle, plasmacytoid, clear, or epithelioid cell, and the neoplastic cells are arranged in sheets, irregular collections, nests, interconnecting trabeculae, or ribbons. The cytoarchitecture of myoepithelial plasmacytoid neoplastic cells includes round cells with eccentric nuclei and large eosinophilic cytoplasm. 1,3
Diagnostic dilemma arises while reporting ME due to its histopathological similarity with PA. Myoepithelioma is exclusively composed of myoepithelial cells with absence of chondroid or osteoid changes in the matrix and <5% of acinar and ductal differentiation, as was seen in our case. Myoepithelioma must be definitely differentiated from mixed tumor as they may present a more aggressive behavior and undergo malignant transformation. 1,3,4
It is also important to separate benign from malignant variants of MEs. Absence of solid pattern, infiltrating growth, necrotic areas, mitotic figures, hyperkeratotic nuclei, cellular polymorphism, cellular atypia, and metastases differentiates benign ME from malignant ME. Also the proliferation index (>10%) is highly suggestive of malignant ME. 2 As interstitial hemorrhage, minimal cellular atypia and infiltration of tumor cells into the surrounding thin capsule were evident in the present case. Hence, current case supports the concept of malignization of this neoplasm.
Finally, myoepithelial cell predominant PA, plasmacytoma, lymphoma, skeletal muscle, and rhabdoid tumors must be excluded. Presence of myxohyaline stroma and cohesive clusters of plasmacytoid cells favors ME over other diagnosis. 4 The tumor markers recommended for diagnosing ME are S-100, Smooth muscle actin, cytokeratin, p63, vimentin, CD138/Syndecan-1, and GFAP protein antigen. 4,5
Surgical excision with tumor-free margins should be the treatment of choice as ME is reported to have more aggressive behavior than PA. Radiation therapy is used only in cases where surgical operation is not feasible. However, in base of our findings of few features of malignancy in this benign neoplasm of salivary glands, we suggest that plasmacytoid ME also can undergo malignant transformation. So timely diagnosis and early management can help prevent this transformation. The patient is followed up regularly and no recurrence noted till date.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
