Abstract

Hersh and Hersh in their recent article “Red ear syndrome: Perspectives for the otolaryngologist” explicitly discussed about the Red ear syndrome (RES) and related otorhinological conditions. 1 Ara-C, an anticancer drug, is known to cause multiple nonhematological toxicities such as cerebellar dysfunction, conjunctivitis, skin toxicity, and liver dysfunction. 2 Skin toxicity in the form of palmoplantar-erythrodysesthesia (PPE) is well reported. Here in, we describe a dermatological manifestation similar to PPE but limited only to ears. 3 Through the following case, we would like to discuss “RES” from hematologist’s standpoint as an important dermatological side effect.
A 36-year-old female was started on induction chemotherapy (Ara-C 200 mg/m2/day × 7 days and daunorubicin 60 mg/m2/day × 3 days) for newly diagnosed acute myeloid leukemia (AML). On the fourth day of therapy, she started developing blanchable, discrete, nonpruritic, nonpalpable, erythematous discrete over the skin of both ears (Figure 1, panel A). There were no similar lesions in other parts of the body. She denied headache, excluding the possibility of trigeminal autonomic cephalalgias. These skin lesions gradually coalesced by the fifth day to appear as diffusely erythematous “red ear.” Her kidney and liver function tests were normal and blood counts suggested chemotherapy-induced myelosuppression (hemoglobin—9.1 g/dL, leukocyte count—0.8 × 109/L, and platelet count—90 × 109/L). Viral serologies for cytomegalovirus, epstein–barr virus, herpes simplex virus etc. were negative ruling out the common viral exanthems as the cause of ear rash. Ara-C was continued as scheduled for seven days as drug toxicity was not life-threatening (Figure 1, panel B showing red ears). By 14th day (ie, after seven days of discontinuing Ara-C), ear erythema faded completely (Figure 1, panel C). On 34th day, she was started on consolidation chemo regimen for AML requiring high-dose Ara-C therapy (HiDAC—18 g/m2/phase). On reexposure, she redeveloped similar dermatological manifestations over skin of both ears. A repeat evaluation was negative for infectious etiology. Patient did not give consent for skin biopsy. Based on strong temporal correlation between the drug exposure and appearance of skin rash, she was diagnosed with “RES secondary to Ara-C.”
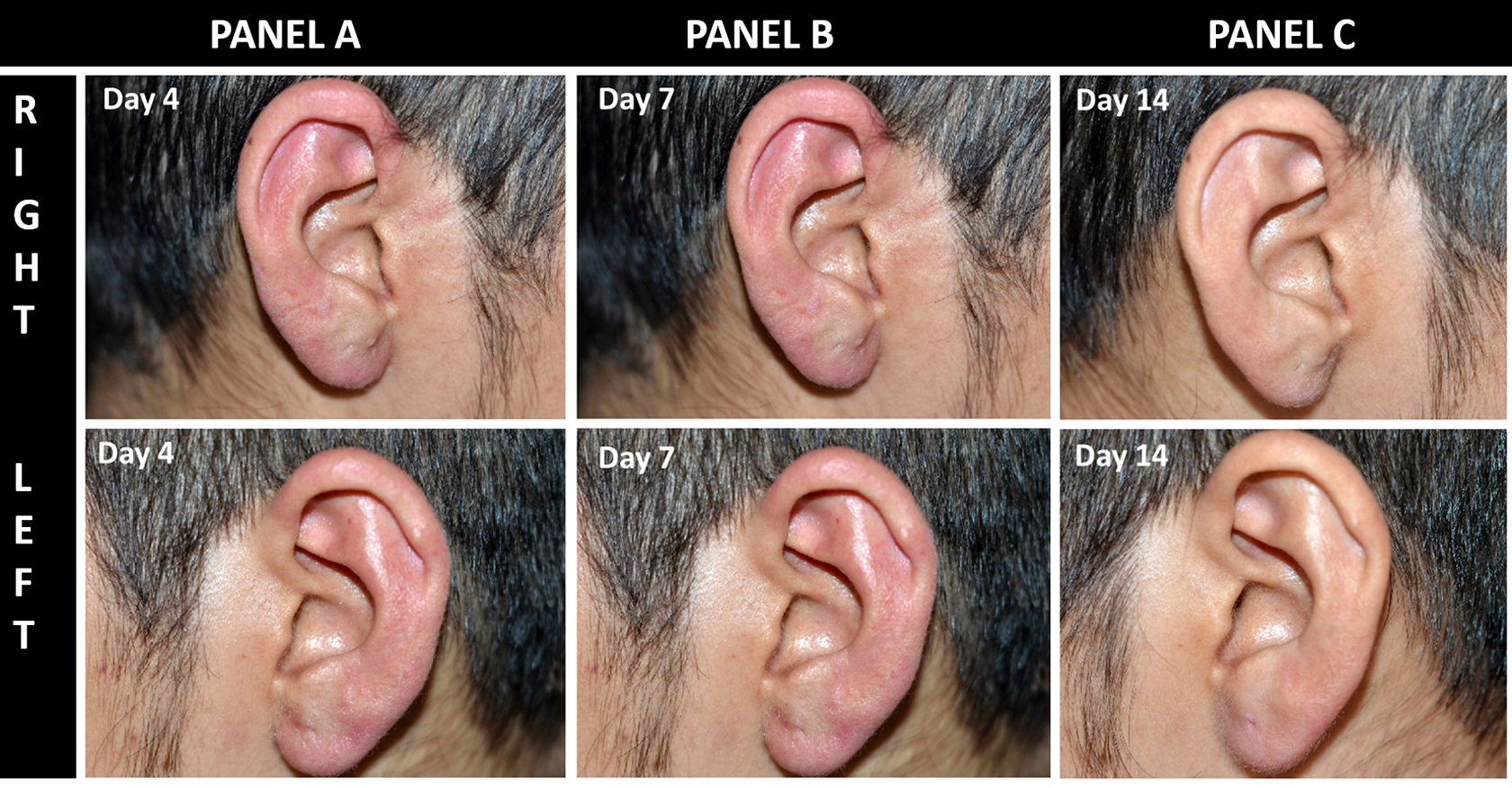
Day-wise comparison of redness of ears during chemotherapy (panel A, day 4 and panel B, day 7) and after chemotherapy (panel C, complete resolution by day 14).
Ara-C, an antimetabolite agent, is commonly used in many hemato-oncological conditions. Hence, it is important to be aware of its side effects for prompt diagnosis and appropriate treatment. Constellation of symptoms such as fever, bone pains, myalgias, maculopapular rash, conjunctivitis, and chest pain are collectively termed as Ara-C syndrome which can potentially be prevented by prophylactic steroids. 4 Skin toxicity secondary to Ara-C is usually seen after HiDAC therapy (high dose), with a reported incidence of 53% in one series. 5 Rash involving palmoplantar regions is known as Bergdorf reaction that requires only symptomatic therapy. 6 Incidence of Bergdorf reaction has been variably reported, ranging from 14% to 33% particularly with higher doses of Ara-c (>1 g/m2) and rarely even with lower doses. 7,8 Other related skin manifestations such as Steven-Johnson syndrome, sweet's syndrome, pyoderma gangrenosum, and so on, usually require use of high doses of steroid or intravenous Immunoglobulin therapy. Morio et al described lack of steroid prophylaxis and younger age (<50 years) as the potential risk factors for Ara-C-induced skin toxicity. RES is a transient phenomenon of redness of ears which commonly occurs as a part of migraine/trigeminal autonomic cephalalgias. 9 Nowadays, newer targeted therapies and novel anticancers are increasingly being used, thereby mandating clinicians to be aware of the pertinent side effect profile of anticancer drugs. 10 –15 To the best of our knowledge, only two cases of “Ara-C-related RES” have been reported in the past. 16,17
To conclude, RES is a rare Ara-C-related skin toxicity and physicians should be aware of its presentation, differential diagnosis, and management. It is essential to rule out infective process especially viral illnesses while evaluating possible cases of RES. RES rarely requires treatment and mostly responds to conservative management only.
Footnotes
Authors’ Note
The article doesn’t contain participation of any human being and animal. All authors have seen the manuscript and agree to the content and data.
