Abstract

A 63-year-old woman with suspected sleep apnea was referred to our facility for right-sided tinnitus and hearing loss. She had first noticed the hearing loss 3 years earlier but reported that it had gotten worse recently. She also reported constant right-sided, pulsatile tinnitus for at least 3 years, although it was initially intermittent. She denied otalgia or otorrhea. She also denied vertigo but had experienced episodes of disequilibrium for almost 2 years with frequent falls, always to the right side.
The patient's medical history was significant for frequent otitis media as a child, mild hypertension, type 2 diabetes, and gastroesophageal reflux. Although she had not undergone a formal sleep study, she had symptoms consistent with obstructive sleep apnea, including loud snoring, restless sleep, and daytime somnolence. She had lived near sea level her entire life, never at a high altitude.
Otoscopy of the left ear was clear. Otoscopy of the right ear revealed a red, vascular mass filling the inferior half of the middle ear space, abutting the medial surface of the tympanic membrane and creating a visible meniscus (figure 1). On tuning fork testing, the Weber test lateralized to the right, and the Rinne test was positive bilaterally. The rest of her head and neck examination was normal, including the cranial nerve exam. Audiometry revealed a right-sided, downsloping mixed hearing loss.
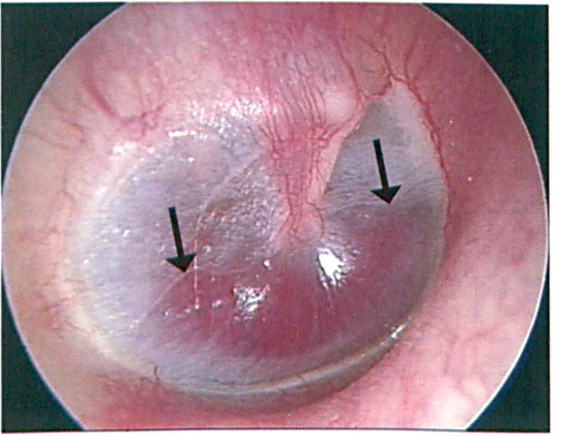
The vascular mass fills the inferior half of the middle ear space, abutting the medial surface of the tympanic membrane and creating a visible meniscus (arrows).
Computed tomography (CT) of the temporal bone showed a right middle ear mass on the promontory, biased toward the hypotympanum (figure 2). The carotid canal was intact with no evidence of an aberrant carotid artery. The jugular bulb was relatively low, with a slightly moth-eaten quality to the cortex. The patient was diagnosed with a right tympanic paraganglioma (commonly referred to as a glomus tympanicum tumor). After discussing the options of observation versus surgical removal, the patient chose to proceed with surgery. Radiation therapy was discussed but not advocated as primary treatment.
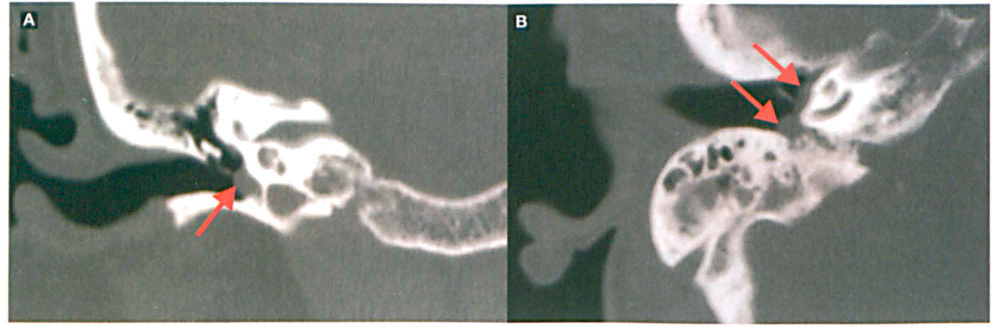
Coronal
The tumor was resected via a transmeatal hypotympanic approach. A type I tympanoplasty was required to extend the tympanic membrane to the lowered floor of the bony canal because of the hypotympanic approach.
Tympanic paragangliomas are neuroendocrine neoplasms located in the middle ear and arising from chromaffin cells of the parasympathetic paraganglia. 1 Patients classically present with pulsatile tinnitus and a red mass medial to the tympanic membrane. Some patients may have findings of a red mass that blanches with pneumatic otoscopy, called Brown's sign. Hearing loss and aural fullness are other common symptoms. However, even with such a classic presentation of a glomus tympanicum, an aberrant carotid artery, high-riding jugular bulb, and glomus jugulare must also be considered. 2 The best diagnostic tool to investigate pulsatile tinnitus with a visible retro- or intratympanic mass is a high-resolution CT scan of the temporal bones, since it can precisely define the location and extent of the tumor. 2
Treatment options include observation, radiation therapy, or surgical excision with or without preoperative angioembolization. However, radiation therapy is not considered optimal treatment for small paragangliomas, which can be Safety resected from the middle ear.
As many as 30% of paragangliomas can be caused by inheritance of mutations in protein subunits of succinate dehydrogenase, a critical enzyme within complex II of the mitochondrial respiratory chain. 3 Although the exact mechanism of tumorigenesis from such mutations is not known, it has been demonstrated that these familial mutations impair complex II function and cause activation of hypoxia-inducible pathways. 3 This is consistent with expression of hypoxia-inducible angiogenic growth factors such as vascular endothelial growth factor in sporadic paragangliomas 4 and with the increased incidence of paragangliomas in patients with chronic hypoxia, such as those living at high altitudes.
Since chronically hypoxic patients often develop paragangliomas arising from the carotid body, a critical site of oxygen sensing, activation of hypoxia-inducible pathways appears to be essential to the pathogenesis of these tumors and may greatly contribute to their hypervascularity. Therefore, when evaluating patients with paragangliomas, it is important to take a careful history and document all risk factors for chronic hypoxia, including living at a high altitude, chronic obstructive pulmonary disease, and sleep apnea.
