Abstract
Nasopharyngeal carcinoma (NPC) is the most common head and neck malignancy in Indonesia. Overall, it ranks fourth in males and sixth in females as the most prevalent type of cancer in that country. The data show that in the year 2011, NPC incidence was considered to be intermediate (6.2/100,000 population per year). Through histopathologic examination, about 70 to 80% of these cases were found to be type HI according to the WHO classificaton. NPC carries an excellent prognosis if treated early, but most patients presented with stage III to IV disease, which negatively affected the cure rate and increased the mortality rate. Epstein-Barr virus (EBV) IgA serology has been established as an effective marker for NPC. Therefore, biologic markers, DNA, and/or antibody-based diagnosis is needed to decrease NPC cases. A screening program needs to be developed that will identify people at high risk of NPC and those who are in the early stage of the disease. In this study, 20 samples were collected from posttherapy patients. An otolaryngologic examination, histopathology of nasopharyngeal tissue, and blood testing for serologic markers were performed. IgA anti-EBNA1 + VCA-p18 enzyme-linked immunosorbent assay showed positive impact as a tool for confirming the diagnosis of NPC, but it still has to be combined with other specific diagnostic tools for post-therapy monitoring and for determining prognosis.
Introduction
Nasopharyngeal carcinoma (NPC) is rare in most parts of the world, but it is relatively common in Southeast Asia, including Indonesia. Chinese populations, living either in mainland China or elsewhere, have the highest incidence of this tumor worldwide: 10 cases/100,000 population. 1
In Indonesia, NPC is the most prevalent head and neck cancer and among the most prevalent cancers overall with an estimated incidence of 6.2 in males and 4.6/100,000/year in females. 2 The data show that in the year 2011, NPC incidence was considered to be intermediate (6.2/100,000/year). 3 It is different than in Singapore, which has an age-standardized rate of 18.1 per 100,000 in males and 7.4 in females. 4
Patients with NPC have high-level anti-Epstein-Barr virus (EBV) antibodies, especially immunoglobulin A (IgA), compared to regional healthy carriers and patients with other head and neck diseases. 5 The association between specific serologic responses to EBV and NPC has been exploited to develop serologic tumor markers for this cancer. The antiviral capsid antigen IgA antibody (IgA-VCA), measured by indirect immunofluorescence or enzyme-linked immunosorbent assay (ELISA), is one of the most widely used antibody markers for assisting in diagnosis and for screening. 6
Assessment of IgA antibody responses to various EBV antigen complexes, usually involving multiple serologic assays, is important for early diagnosis of NPC. Through the combination of two synthetic peptides representing the immunodominant epitopes of EBNA1 and the viral capsid antigen VCA-p18, only a one-stop ELISA is needed for the specific detection of EBV-reactive IgA antibody in NPC patients. 7 Using either EBNA1 or VCA-p 18 peptide alone, IgA ELISA correctly identified 88.5% and 79.8% of Indonesian NPC patients, with specificities of 80.1% and 70.9%, respectively, combined single-well coating with both peptides yielded sensitivity and specificity values of 90.1% and 85.4%, respectively. 7
The objective of the present study is to determine the role of serology assay IgA anti-EBNA1 + VCA-p18 for detecting NPC as a biomarker for post-therapeutic monitoring and prognosis.
Materials and Methods
Serum samples were obtained from Indonesian (non-Chinese) people with histologically confirmed NPC, and the sera were taken from 16 post-therapy patients at the Otolaryngology–Head and Neck Surgery (ORL-HNS) Oncology Clinic of the Dr. Soetomo General Hospital, Surabaya, Indonesia, during November and December 2009. Tumor staging was based on routine oncologic evaluation, including physical examination, anterior rhinoscopy, indirect (with or without direct) nasopharyngoscopy, blood biochemistry, chest x-ray, and computed tomography (CT). The results were classified according to the 1996 Union International Cancer Control (UICC) classification.
Samples were examined by ELISA (Greiner Labortechniek; Frickenhausen, Germany) coated with EBNA1 and VCA-p18 peptide using 135 single peptide (1 mg/ml) or combined peptides (1 mg/ml EBNA1 plus 0.5 mg/ml VCA-p18) in 0.05 M Na2CO3, pH9.6 (Merck; Darmstadt, Germany). The procedures were done according to the previous test. 7 The cut-off value was 0.3526, determined by the value of sera from healthy donors and NPC patients. 7 All data were analyzed using the Statistical Package for the Social Sciences (SPSS) version 13.0.
Results
Among 16 samples, the male-to-female ratio was 1.7:1 (10:6), and age distribution ranged between 24 and 63 years. Ten patients were between 31 and 50 years. The first-time visitors to the ORL-HNS clinic were mostly patients at stage IV (81.25%), followed by 12.5% at stage II and 6.25% at stage III. No patients came at stage I.
Comparison between NPC Histopathology Results and Level of Serology IgA Anti-EBNA1 and VCA-p18
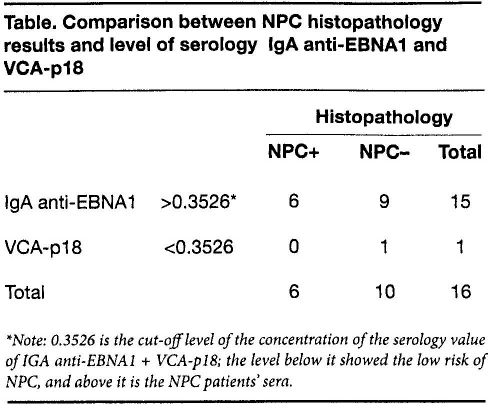
Note: 0.3526 is the cut-off level of the concentration of the serology value of IGA anti-EBNA1 + VCA-p18; the level below it showed the low risk of NPC, and above it is the NPC patients’ sera.
The histopathology results among post-therapy patients showed that in 62.5% there were no malignancies and in 37.5% there were still malignant cells. All histopathology results from the first biopsy indicated undifferentiated carcinoma (WHO type III). IgA anti-EBNA1 + VCAp-18 serology results showed that 93.75% were positive (>0.3526), and the rest were negative. The comparison between serology and histopathology results is shown in the table. The correlation between IgA anti-EBNA1 + VCA-p 18 and histopathology results was not significant (p > 0.05).
Discussion
NPC patients’ ages range between 46 3 and 50 1 years. In 1997, Chia and Lee found that the incidence was low among people less than 20 years of age, but that there have been several cases involving patients less than 5 years old. 8 The incidence begins to rise in the second decade of life and reaches a peak in the fourth decade. 8 Our male-to-female ratio of 1.7:1 differed from previous research that showed the ratio to be 2 or 3:1.1,3
Almost all of our patients came in for their first visit at a late stage of NPC; this might have been because in the early stage, symptoms and signs are minimal. It also could have been because of the nonspecific nature of the aural symptoms and the inconspicuous nature of painless cervical lymph nodes. 1
EBV serology is commonly used to facilitate the diagnosis of suspected NPC and is proposed for large field-screening and epidemiology survey studies. NPC risks are associated with the elevated responses of IgG, and particularly IgA antibodies to certain EBV antigens. Most people in Southeast Asia are first infected by EBV in early childhood, reflected by a nearly 100% seropositivity for IgG to EBV VCA and EBNA1. 3
Currently, options for NPC screening consist of serologic markers for EBV infection, such as antibodies to EBV IgA or the presence of EBV DNA gene products. Once a person is infected, the IgA antibody remains positive throughout the his or her lifetime. Although higher levels can suggest active infection, they cannot reliably discriminate past from present infection. 5
Elevated total and EBV-specific serum IgA levels are indicative of NPC stage and can precede tumor development by 1 to 5 years, which suggests that a reactivation of EBV infection plays a role in tumor development. In addition, a decline in anti-EBV antibody responses after radiotherapy may have prognostic value. 9 A study in Indonesia by Paramita et al in 2009 revealed the serologic abnormalities of positive EBV IgA responses 2 to 3 years before the onset of NPC. 10 These abnormalities are particularly applicable for screening in high-risk groups, such as family members of NPC patients and patients with suspicious head and neck symptoms. 10 In 2004, Lo et al showed that the risk of developing NPC after an initial normal biopsy result in Hong Kong patients with a positive EBV IgA VCA titer was 2%. 11 Although there was a significant proportion of patients with a positive EBV IgA VCA titer that reverted to normal, some of them later developed NPC. The overall incidence of seronegative subjects subsequently developing NPC was previously reported to be 0.15%, and they were 22 times less likely to develop NPC compared to patients with a seropositive EBV IgA VCA titer. 11
The presence of IgA antibodies is associated with increased NPC risk in the general population. Therefore, it has been suggested that field screening for EBV IgA might be useful to identify patients with early-stage NPC. Also, in established NPC patients, longitudinal monitoring of EBV IgA reactivity levels might be used for prognosis because declining reactivity is associated with remission, and stable or increasing responses are associated with persistent or recurrent disease and the development of metastasis. 7
The IgA anti-EBNA1 + VCA-p18 combination peptide ELISA permits discrimination of NPC patients from patients with acute or chronic active EBV infection, who may manifest similar nonspecific symptoms. This is relevant for diagnostic screening in populations with symptoms in the head and neck region that are suggestive of NPC. The 85.3% sensitivity was lower than EBNA1-only ELISA, but it showed higher activity. 7
EBNA1 IgA responses appear to be more specific for NPC and may be related to the release of EBNA1-DNA complexes from dying NPC tumor cells. However, not all NPC patients have a detectable EBNA1 IgA response, and the combination with VCA-p 18 in an IgA ELISA clearly improves diagnostic usefulness.
In NPC patients, IgG antibodies typically react with a broad range of EBV lytic proteins. By combining IgA ELISA with IgG immunoblot analysis of samples, specificity/sensitivity and positive predictive value (PPV)/negative predictive value (NPV) for NPC detection increased to >95%. It could show an initial positive ELISA result. Therefore, to increase the specificity and sensitivity of the serologic diagnosis of NPC, an IgG immunoblot test should be performed as a confirmatory assay. 7
Overall, the combined IgA anti-EBNA1 + VCA-p 18 ELISA showed promise as a tool for supporting a diagnosis of NPC in the Indonesia population compared to a single-antigen IgA ELISA. The lack of significant IgA responses to EBNA1 and VCA-p 18 alone without combination peptides in acute and chronic EBV infection further supports the potential use of EBV IgA combination peptide ELISA as an NPC-specific screening tool. IgA anti-EBNA1 + VCA-p 18 could predict the onset of NPC, but for relapses and post-therapeutic monitoring, it should be combined with another method, e.g., EBV DNA load from nasopharyngeal tissue biopsy.
The conclusion of this research is that IgA anti-EBNA1 + VCA-p 18 is useful for diagnosing NPC, but it cannot be used as a post-therapy monitoring tool, and further examination will be required.
