Abstract
We describe 2 cases of bilateral Ménière disease with features resembling autoimmune inner ear disease in patients who were found to be carriers of human leukocyte antigen (HLA) B27. For immunohistochemical analysis, mouse inner ear sections were used as the tissue substrate for reaction with serum. Both patients demonstrated an increased immunofluorescence reaction compared with a normal control. We suggest that an antibody-mediated mechanism may be responsible for HLA-B27–associated Ménière disease.
Introduction
The etiology of Ménière disease is thought to be multifactorial, involving both genetic and environmental factors. 1 In several studies of temporal bones, researchers have attempted to correlate pathologic findings with a known clinical history in Ménière disease, suggesting congenital, infectious, inflammatory, traumatic, allergic, and autoimmune causes. 1
The idea of an autoimmune etiology of inner ear disease was first introduced more than 30 years ago, 2 and it still remains an active area of research today. Autoimmune inner ear disease is defined by many authors as a bilateral progressive hearing loss that responds to a short course of high-dose oral steroid treatment. 3 Studies have demonstrated that 20 to 50% of patients classified as having autoimmune inner ear disease have vestibular symptoms that are correlated with Ménière disease.3-5
It is assumed that autoimmunity occurs via one of three basic pathways to cause tissue damage in Ménière disease, which may or may not be organ-specific: (1) autoantibodies directed against antigens found upon or within tissue cells,6–10 (2) circulation and deposition of antigen-antibody complexes with activation of the complement system and resultant inflammatory tissue destruction,8,10,11 or (3) an inflammatory reaction mediated by sensitized T lymphocytes.10,11
The association between the human leukocyte antigen (HLA) complex and Ménière disease has been explored, but results have varied among different populations worldwide.12-16 Specifically, no association between HLA-B27 and Ménière disease or autoimmune inner ear disease has been described.
We report 2 cases of HLA-B27-associated Ménière disease, and we describe their clinical and immunocytologic features as determined by the patients’ serum reactions with mouse inner ears.
Case Reports
Patient 1
A 64-year-old man presented with episodic vertigo, aural pressure, tinnitus, vertebral arthritis, and a progressive bilateral sensorineural hearing loss. His medical history was significant for chronic low back pain. He also related a strong association of episodes with salt and caffeine intake. The patient had earlier been treated for 2 episodes of sudden sensorineural hearing loss—one in 1984 and one in 1991. Both episodes resolved after a course of steroid therapy. Starting in 1993, the patient began experiencing regular episodes (every 6 mo) of bilateral sudden sensorineural hearing loss and vertigo lasting 3 to 4 hours. These episodes were very responsive to intramuscular steroid treatment.
In March 2003, the patient was referred to our clinic. Laboratory tests revealed that his erythrocyte sedimentation rate (ESR), antinuclear antibody (ANA) level, rheumatoid factor (RF) value, and fluorescent treponemal antibody absorption (FTA-ABS) titers were within normal limits. Findings on magnetic resonance imaging (MRI) of the internal auditory canals were normal. Pure-tone audiometry was obtained during an episode of vertigo and again after treatment with 40 mg of methylprednisolone given intramuscularly (figure 1). In light of the discovery of HLA-B27 in a family member, the patient underwent HLA testing, which revealed that he, too, was a carrier of HLA-B27.
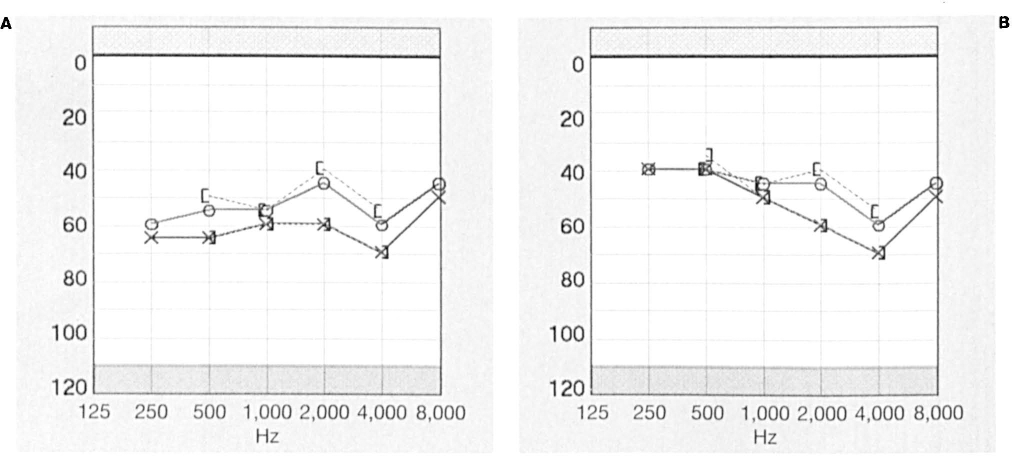
Patient 1.
Patient 2
A 71-year-old man presented with a 10-month history of fluctuating bilateral hearing loss and occasional vertigo, aural pressure, and tinnitus. He had had a previous diagnosis of cervical arthritis. His ESR, ANA, RF, and FTA-ABS values were within normal limits. Findings on a previously obtained MRI of the internal auditory canals were normal. Audiometry revealed a sensorineural hearing loss down to 70 dB on the right and 60 dB on the left across all frequencies. On questioning, the patient also reported recurring pink eye about every 4 months. Cogan syndrome was initially suspected in view of the characteristic interstitial keratitis, fluctuating sensorineural hearing loss, and episodic vertigo.
The patient was treated with 60 mg of oral prednisone every day for 1 week with a taper, but there was no change in symptoms. When a review of the patient's ophthalmologic examination revealed that he suffered from recurrent iritis and not interstitial keratitis or uveitis, the diagnosis of Cogan syndrome was questioned. Additionally, we explored his complaint of cervical arthritis and found ankylosing spondylitis. At this point, the idea of an association with HLA-B27 was introduced, and subsequent blood tests revealed that the patient was indeed a carrier of the HLA-B27 allele. With this working diagnosis of an HLA-B27 genotype, the patient continued treatment with intratympanic dexamethasone. The following week, he reported no change in symptoms, and audiometry revealed a worsening of his hearing loss to 80 dB at 250 to 500 Hz on the right and to 90 dB at 250 to 500 Hz on the left.
Intratympanic injections were performed again bilaterally in the office, and the hearing in the right ear improved from its lowest threshold of 75 dB at both 250 and 500 Hz to 60 dB (figure 2). Similar improvement was seen on the left side, with increases from 95 to 75 dB at 250 Hz and from 85 to 70 dB at 500 Hz (figure 2). After having received a total of five intratympanic dexamethasone injections, the patient reported a subjective stabilization of his hearing bilaterally and satisfactory control of his vertigo with meclizine. At his final follow-up, he reported no change in hearing and no episodes of vertigo.
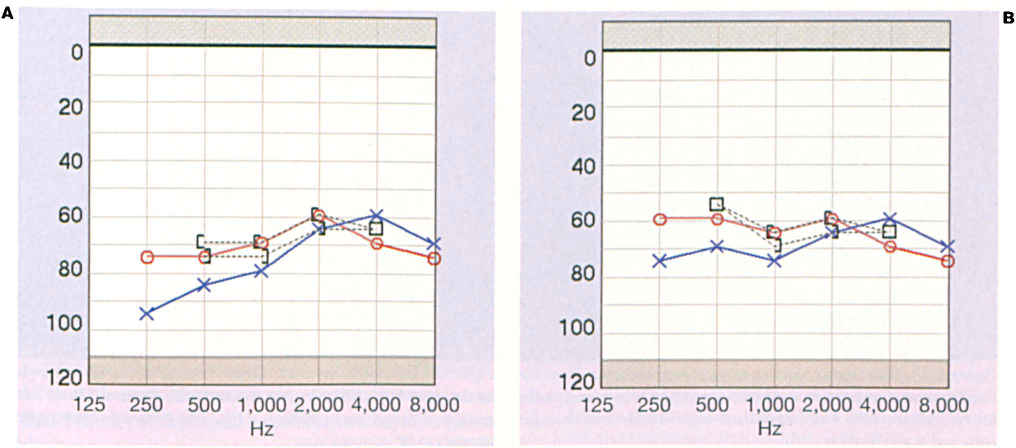
Patient 2.
Experiment
Protocol
In order to examine the immunocytologic features of the proposed association between HLA-B27 and Ménière disease, we devised a protocol involving the fluorescence-labeled reaction of human serum with mouse inner ear sections. This protocol received institutional review board approval. Serum samples were provided by both patients and by a healthy control. These samples represented the primary antibody. The secondary antibody was a fluorescein isothiocyanate-labeled goat antihuman IgG. Procedural controls consisted of sections incubated in secondary antibody only. Preparation of the tissue substrate entailed fixation of the mouse inner ear sections in either paraformaldehyde or acetone. These sections were then rehydrated in phosphate-buffered saline incubated in 1% Blotto for 15 minutes, and then incubated in 1:1 or 1:25 dilutions of primary antibody for 60 minutes. A dilution of 1:100 secondary antibody was then incubated with the sections for an additional 60 minutes for fluorescent labeling. Finally, the sections were mounted and covered with a coverslip and viewed under x10, x20, and x40 magnification. The sections were graded for location and the amount of immunofluorescence by two blinded observers who had a thorough knowledge of temporal bone histopathology.
Results
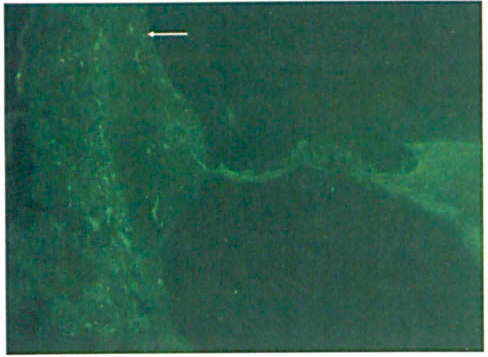
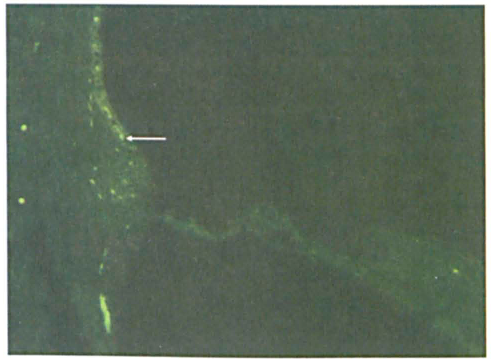
The serum from patient 1 exhibited a diffuse fluorescence pattern that was mainly localized to the nuclei of all cells in the cochlea on both paraformaldehyde (figure 3) and acetone (figure not included) preparations. Cytoplasmic immunofluorescence was not observed in patient 1. The sample from the healthy control (figure 4) and the secondary antibody-only preparation (figure 5) did not show any nuclear or cytoplasmic immunofluorescent reaction. Fluorescence reactions seen in the stria vascularis of patient 1 and patient 2 were also seen in the healthy control and in the secondary antibody-only samples. In patient 2, a greater fluorescence reaction was seen in the hair cells and in the spiral ganglion cells compared with the control and antibody-only samples (figure 6). There was no difference noted in the pattern or amount of immunofluorescence between the paraformaldehyde preparation and the acetone preparation. These results suggest that a humoral immune response— specifically an antigen-antibody reaction—may underlie the development of Ménière disease symptomatology in carriers of HLA-B27.

Patient 1. This immunofluorescence pattern with para-formaldehyde-fixed sections shows the reaction of patient 1's serum with the mouse cochlear sections. A diffuse fluorescence pattern is seen in the nuclei of all cells in the cochlea with the exception of the hair cells (dotted arrow), in which fluorescence is weaker (original magnification × 10). The solid arrows point to nuclear fluorescence patterns.
The fluorescence reaction seen between the serum from the healthy control and the mouse inner ear section is minimal in the lateral wall (arrow), which appears to he primarily intravascular (original magnification × 10).
Minimal fluorescence (arrow) is seen in the stria vascularis in the secondary antibody-only reaction with the mouse inner ear section (original magnification x10). The minimal fluorescence appears to be intravascular.
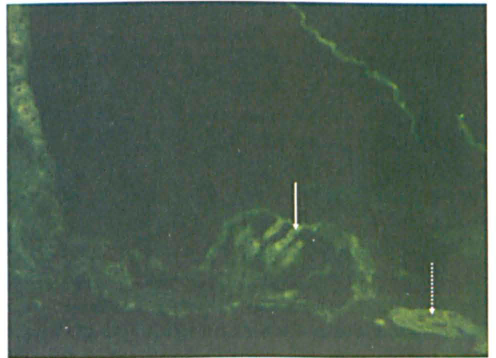
An increase in fluorescence is seen in the hair cells (solid arrow) and in the spiral ganglion cells (dotted arrow) in the reaction between patient 2's serum and the mouse cochlear section (original magnification × 20).
Discussion
We have described the cases of 2 patients with signs and symptoms of autoimmune Ménière disease who were found to be HLA-B27 carriers. Reaction of the serum of these patients with mouse cochlear sections showed a greater antigen-antibody reaction compared with control specimens. However, no common pattern of immune reaction was seen in the 2 patients. The results of our investigation indicate a possible role of HLA-B27 in the development of autoimmune bilateral Ménière disease. The relationship between autoimmune Ménière disease and HLA-B27 carriage is worthy of further study.
Our patients’ bilateral progressive asymmetrical hearing loss supports an autoimmune etiology. Both of our patients had symptoms of episodic vertigo, aural pressure, tinnitus, vertebral arthritis, and progressive sensorineural hearing loss—a hearing loss primarily in the lower frequencies that over time involved the higher frequencies. Patient 2 demonstrated a more rapid progression over a short period of time, whereas patient 1 had a more protracted course. Patient 1 also related a strong association of episodes with salt and caffeine intake. It is possible that the cochlear and vestibular symptoms in both patients were related to their HLA-B27-positive serology.
Since 1979, researchers have been studying the potential autoimmune etiology of Ménière disease. 2 More specifically, its. association with particular HLAs has been explored, but results among different populations worldwide have varied.12-16 Many authors have reported a high prevalence of Ménière-type symptoms in carriers of the HLA-Cw*07, 12 -Cw*0602, 13 -DRB1*1602, 14 -A2, 15 and -B44 15 alleles. Yet a group from Spain reported no association between Ménière disease and HLA-A, -B, -C, or -DR alleles. 16 To date, no link has been shown between HLA-B27 and Ménière disease. Our patients represent the first reported cases of Ménière disease associated with HLA-B27.
The mechanism of HLA-associated autoimmunity is complex. HLA-B27 antigen plays a role in several non-organ-specific autoimmune disease states, including ankylosing spondylitis, acute anterior uveitis, and Reiter syndrome. In white populations and most other groups, 90% of patients with ankylosing spondylitis carry HLA-B27, as opposed to 8% of the general population. 17 The various mechanisms of an autoimmune response in these patients include T-cell-mediated autoantibodies and natural killer cells. 18 Most commonly, HLA molecules that are involved in disease predisposition act by preferential peptide presentation to T cells. However, for each target tissue, the mechanism of the autoimmune reaction is different. Wang et al studied a group of 34 patients with HLA-B27–associated ankylosing spondylitis and found that 9 (26%) of them had circulating autoantibodies to the inner ear, which may be nonspecific in nature. 17 Our investigation of 2 patients with HLA-B27–associated Ménière disease also found evidence of the presence of autoantibodies against inner ear structures in the serum of both of patients. This was evidenced by the greater fluorescence reaction of the patients’ serum with mouse cochlear sections compared with control samples. The goal of our investigation was twofold: (1) to illustrate a possible association between carriage of the HLA-B27 allele and the presentation of autoimmune inner ear disease or autoimmune Ménière disease symptomatology in 2 patients and (2) to investigate the immunocytologic features of this association—namely the presence of a circulating autoantibody reaction as demonstrated by the patients’ serum reactions with mouse inner ear tissue substrates. Based on our findings, we believe there is a possible role of a humoral immune response in the development of Ménière disease in HLA-B27–carrying individuals.
Our investigation has a few limitations. First, the small sample size obviously prevents a generalization of the data to the entire population. A link between HLA-B27 and autoimmune Ménière disease will require a much larger sample. Second, only cochlear sections from mice were used as substrates for immune reaction of the serum. Use of human temporal bones and the inclusion of the vestibular organ would enhance such an investigation.
