Abstract
A prospective, multicenter research study is under way to demonstrate long-term improvement in chronic rhinosinusitis symptoms after transantral balloon dilation of the ethmoid infundibulum. Trial results from an interim analysis of symptomatic status using the Sino-Nasal Outcome Test 20 survey demonstrate significant and sustained improvement through post-procedure 1-year follow-up. Additionally, these data provide evidence that the symptomatic improvements following balloon expansion within the ostiomeatal unit to treat medically refractory inflammation of the maxillary sinuses, either alone or with concomitant anterior ethmoid disease, are similar.
Introduction
In 2007, the study investigators began enrollment in a single-arm, prospective
clinical trial (
Historically, the standard surgery for treating medically refractory chronic sinusitis has required tissue removal under general anesthesia. Within the past 5 years, balloon catheters have been introduced for use in sinus surgery, and 2-year follow-up data have been published on the safety and outcomes of paranasal balloon antrostomy performed primarily under general anesthesia and with fluoroscopic guidance to deliver the balloon catheter to the planned treatment sites. 1
Recently, 6-month interim results from the first 30 subjects treated with FinESS Sinus Treatment under the BREATHE I protocol (NCT00645762) were reported. 2 The primary objectives of this study were to assess device and procedural safety during the treatment of maxillary sinus outflow tract narrowing and inflammation of the maxillary and anterior ethmoid sinuses, and also to evaluate the feasibility of remodeling the ethmoid infundibulum without the use of imaging equipment or general anesthesia. In addition to these safety outcomes, subjects also consented to long-term follow-up for post-procedure assessment of symptoms through 12 months.
Patients and Methods
The study was designed to prospectively treat as many as 100 subjects at as many as 40 study centers in the United States under a common protocol approved by an institutional review board. The study was sponsored and funded by Entellus Medical Inc. (Maple Grove, Minn.). Eligible for enrollment were adults ≥18 years of age with:
chronic rhinosinusitis,
isolated maxillary disease (with or without coexistent anterior ethmoid disease), and
documented computed tomography (CT) evidence of either OMU narrowing and ≥2 mm maxillary mucosal thickening or an air-fluid level in the maxillary antrum after maximum medical therapy.
Subjects were excluded if there was radiographic evidence of frontal, posterior ethmoid, or sphenoid disease or fungal sinusitis. To mitigate the likelihood of confounding analyses, FinESS Sinus Treatment was the only sinus surgery permitted in the study. Subjects requiring concomitant septoplasty, turbinate reduction, or any other endoscopic sinus surgeries were also ineligible.
This interim report provides long-term follow-up results for the first 30 subjects to undergo the study procedure. Study subjects underwent baseline assessment, including nasal endoscopy and CT evaluation, and completed the Sino-Nasal Outcome Test 20 (SNOT-20) 3 survey to document the severity of their sinus symptoms at the time of enrollment and before treatment. The subjects were required to return to the investigational site at 1 week and 3 months post-procedure for physician evaluation and completion of the SNOT-20 survey.
After 3 months, the subjects were not required to return to the study center, but SNOT-20 surveys were still required at the 6-month and 12-month follow-up. The preferred method of survey completion was via mail to reduce the likelihood of bias, but phone administration of the questionnaire was also permitted.
Baseline characteristics, procedural parameters, and safety results were previously reported. 2 The purpose of the present interim report is to compare symptomatic status at 12-month follow-up with the previously reported improvement in symptoms through 6 months, to demonstrate the durability of FinESS treatment.
Standard summary statistics were calculated for all study variables by an independent statistician. For continuous variables, statistics included mean, standard deviation, and 95% confidence intervals for the means, while categorical variables were summarized using frequency distributions. All statistical tests were two-sided, with p values <0.05 deemed significant. Statistical analyses were conducted using SAS software (version 9.1 or above; SAS Institute, Cary, N.C.).
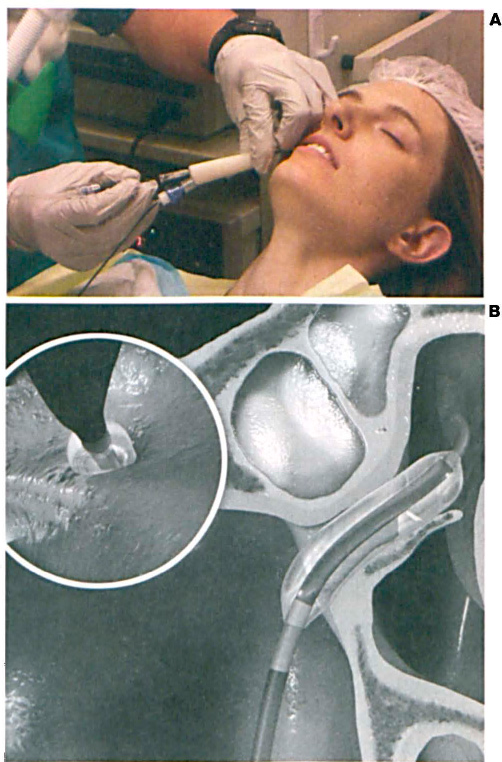
Results
Thirty patients (56 ostia) were successfully treated with FinESS alone. Follow-up compliance through 1 year was 97% (29 of 30 patients). One subject withdrew after completing the 6-month follow-up and after subsequently undergoing endoscopic sinus surgery to remove a large fungal ball in the maxillary sinus that was cultured and confirmed to be aspergillus. A second subject underwent revision surgery to treat sphenoid sinusitis. Twenty-nine subjects successfully completed 1-year follow-up and end-of-study requirements.
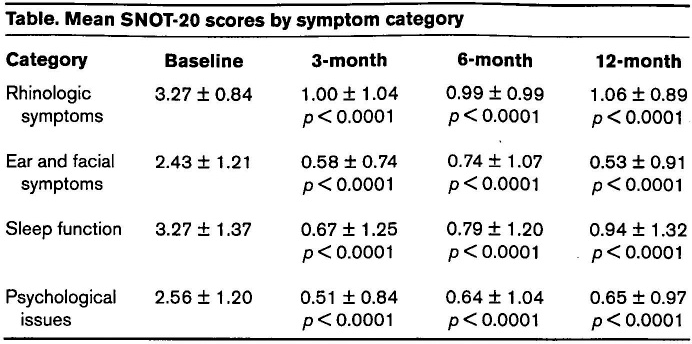
The SNOT-20 survey is a validated quality-of-life assessment in which subjects rank 20 unique symptoms related to their sinusitis on a scale from 0 (no problem) to 5 (problem as bad as it can be). Additionally, symptoms were grouped into the following 4 categories: rhinologic symptoms, ear and facial symptoms, sleep function, and psychological issues (i.e., sad, embarrassed, etc.). Figure 2 shows the mean overall SNOT-20 scores through 12 months, and the table provides these same data grouped by symptom category.
The aggregate data demonstrate clinically meaningful and statistically significant symptomatic improvement in sinus symptoms throughout the duration of the study. At baseline, 60% (n = 18) of this study cohort showed CT evidence of maxillary sinus disease only, while 40% (n = 12) also presented with partial or complete opacification of the anterior ethmoid sinuses. Comparison of the mean overall SNOT-20 scores at each follow-up interval between these two subgroups shows a similar trend in clinically and statistically significant improvement, and comparison of the p values between them at every time point provides statistical evidence to suggest that the treatment effect is similar whether the disease is isolated to the maxillary sinuses or includes the anterior ethmoid sinuses. Mean SNOT-20 scores for each subgroup are shown in figure 3.
Discussion
The keystone to functional endoscopic sinus surgery (FESS) is treating the natural sinus outflow tracts. Tissue preservation has been emphasized by many surgeons. The uncinate process is routinely removed in FESS but preserved in most maxillary ostium balloon dilations. With FinESS treatment, it is always preserved. This study shows that patients’ symptoms still improve when relatively small changes are made to the maxillary ostium and ethmoid infundibulum, and when the uncinate process is preserved. Also, this study shows not only that the procedure can be routinely performed without general anesthesia, but also that it has durable results at 1-year follow-up.
Mean SNOT-20 scores by symptom category
Medical management of chronic rhinosinusitis with antibiotics and topical/oral steroids has its limitations. In this study, subjects were required to present with radiographic evidence of mucosal thickening or an air-fluid level in the antrum of one or both maxillary sinuses after maximal medical therapy (as determined by the standard of care of the treating physician). As previously reported by Stankiewicz et al, 94% of this study population received at least a 1 -week course of antibiotics prior to the pre-enrollment CT scan, and 80% also received either a topical or oral steroid. 2
Additionally, during the most recent 12-month period prior to enrollment, the average number of sinus episodes requiring medical therapy was 4.0 ± 2.7, and antibiotics were used on average 4.3 ±2.0 times to treat these sinus problems. Despite the prevalent use of medications to manage sinusitis during the pre-enrollment period, the mean SNOT-20 score at baseline (2.86 ± 0.98) suggests that the symptoms were of moderate severity before treatment with FinESS.
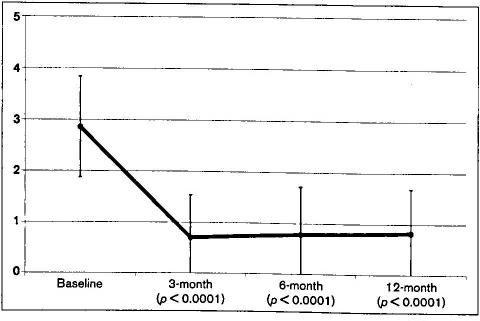
Graph shows the 1-year mean overall SNOT-20 scores.
Although the small sample size and absence of a prospective comparator are limitations of the study design, post-treatment medical management is also reported in an attempt to demonstrate that symptomatic improvement is unlikely to be solely attributed to an increase in the use of antibiotic therapy at the time of the 6-month and 12-month evaluations. Conversely, the lack of sustained medication management after FinESS Sinus Treatment suggests improvement in patient symptoms in the absence of antibiotic use for sinus infections. Post-treatment medication usage data at the 6- and 12-month follow-up indicate that only 1 subject was on an antibiotic for a sinus infection at 6 months. This subject remained on antibiotic therapy at 12 months, and one additional subject reported antibiotic usage at the 12-month follow-up, for a total of 2 subjects receiving antibiotic therapy. If these 2 subjects are excluded from the analysis, the reduction in the overall mean SNOT-20 score for subjects who were not on antibiotic therapy during long-term follow-up from baseline to 6 months and from baseline to 12 months is 2.1 for both intervals. These symptomatic improvements remain clinically and statistically significant.
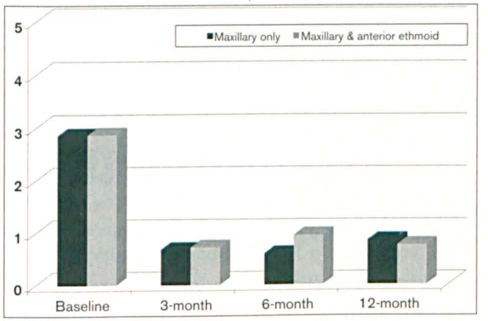
Graph shows the 1-year mean overall SNOT-20 scores by subgroup.
The role of catheter-based balloon technologies to treat chronic sinusitis continues to evolve as long-term outcomes data are reported, but it is widely acknowledged that balloon antrostomy is not intended to replace FESS. However, the results presented from this research study demonstrate an improvement in long-term symptomatic status that is similar in magnitude to the results previously reported by other balloon dilatation device manufacturers. The results further demonstrate that sinus disease of the anterior sinuses due to narrowing of the ethmoid infundibulum can be effectively treated with FinESS Sinus Treatment and without tissue removal.
Footnotes
Financial disclosure: This study was funded by Entellus Medical, Inc. (Maple Grove, Minn). Dr. Stankiewicz, Dr. Truitt, and Dr. Atkins are paid consultants to Entellus Medical for their work performed as members of the Entellus Medical, Inc., Scientific Advisory Board.
Previous presentation: Some content in this article was included on a poster that was presented during the Scientific Program Poster Reception (Scientific Program 160271; “Transantral Balloon Dilation Under Local Anesthesia”); October 6, 2009; San Diego, Calif., at the American Academy of Otolaryngology–Head and Neck Surgery Foundation's Annual Meeting & OTO EXPO.
