Abstract
The comprehensive review presents multi-faceted biofuel applications in various sectors and prospects, highlighting state-of-the-art technologies and potential changes in triglyceride-based biofuels. From the first-generation biofuel produced from food crops to the fourth generation of engineered microbes, there is an improvement in technologies and sustainability. Transesterification synthesis of biodiesel with different chemical catalysts and biocatalysts shows continuous efforts toward making the process more efficient and economically viable; emerging technologies like hydrodeoxygenation and pyrolysis open new routes for optimized biofuel production with less environmental impact. With a focus on biodiesel, bioethanol, biobutanol, and biohydrogen, this paper deals with their contributions to energy generation in a sustainable manner and environmental stewardship. AI and machine learning in the production of biofuels could provide new revolutions in efficiency and sustainability directly from feedstock selection to product recovery. Bioethanol is effectively used in gasoline blends with several advantages, reducing carbon dioxide emissions. Biobutanol is a good transport fuel alternative, with high energy content, easily used with the existing infrastructure. Biological processes yield Biohydrogen and may have bright prospects for producing clean energy, specifically in the case of bioelectrochemical systems. Prospects hinge on addressing the challenges, promoting research and development, and fostering collaboration and investment in biofuel innovation. This review underscores the importance of continued efforts to advance biofuel technologies, recognizing their pivotal role in achieving a sustainable energy future while addressing global energy and environmental challenges.
This is a visual representation of the abstract.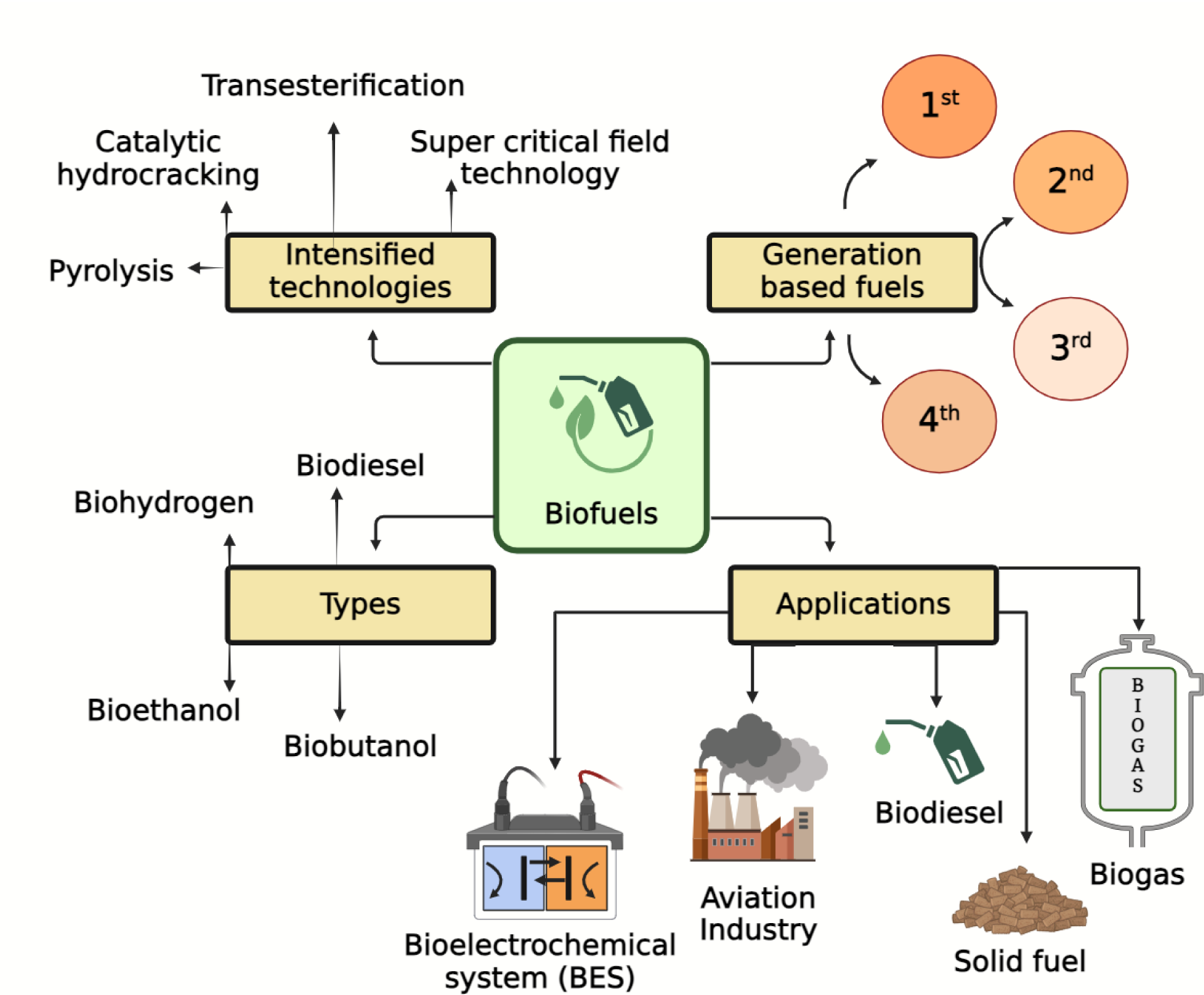
Introduction
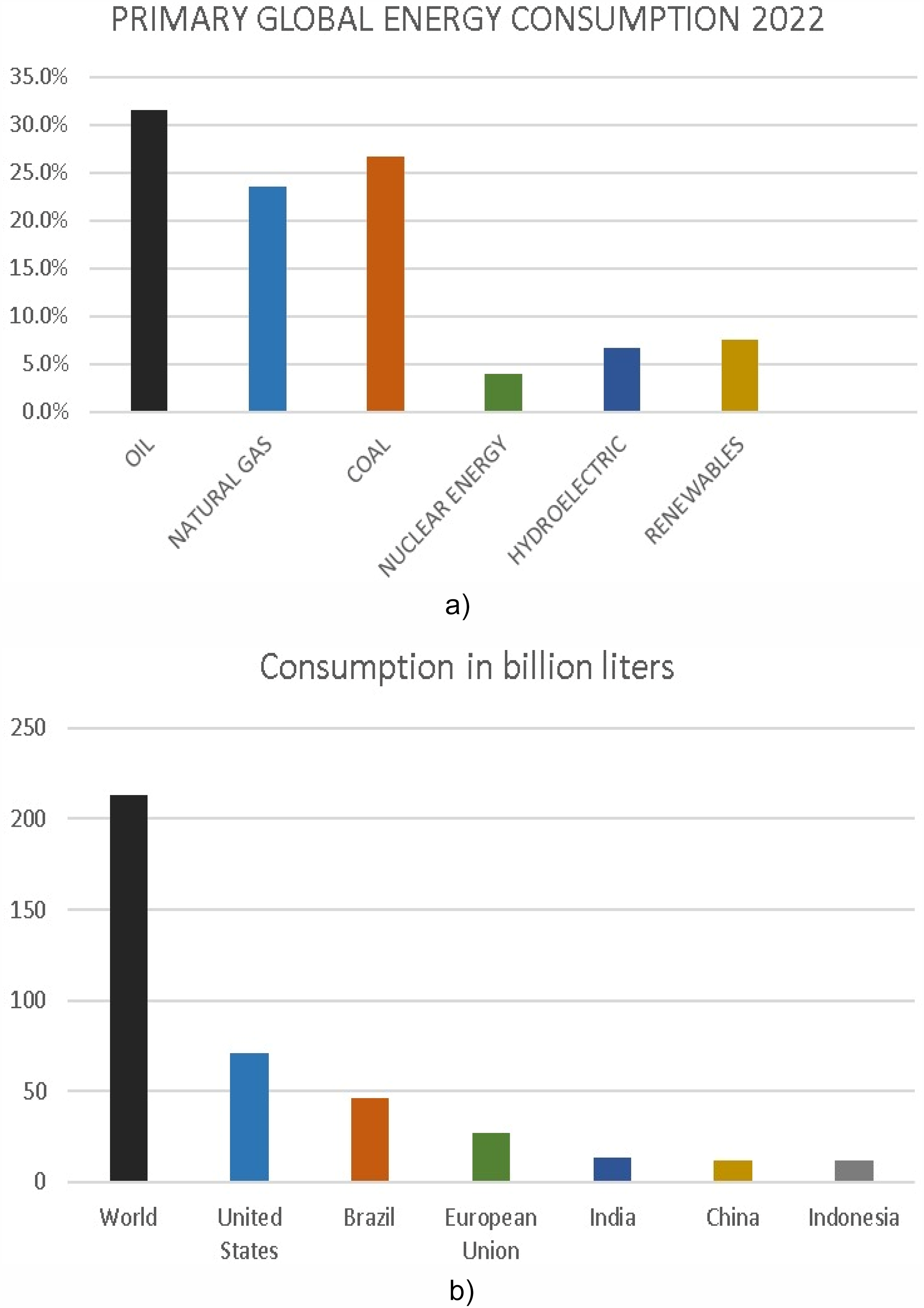
These days, many countries are making extraordinary efforts to decarbonize by reducing anthropogenic greenhouse gas (GHG) emissions, significantly impacting the energy sources they use (Estevez et al., 2022). Transportation of goods and people accounts for about 20% of the total primary energy consumption, approximately 23% of carbon dioxide emissions, and around 14% of overall global GHG emissions when considering other gases like methane (U.S. Energy Information Administration, 2020). Globally, social development with an increasing rate of population has resulted in an unprecedented rise in energy consumption (Xu et al., 2016). The estimated energy requirement will be 50% greater than the current level by 2030 (Shelare et al., 2023). According to EIA, there will be a 47% rise in worldwide energy demand in the next 30 years, owing to population and economic expansion, especially among developing Asian countries. The global energy consumption in 2022 is illustrated in Figure 1(a). Most of the petroleum reserves are situated in the mid-west regions, due to political conflicts the petroleum economy, which has fulfilled the energy demands for more than 150 years, is currently facing many challenges (World Economic Forum, 2016). Fossil fuels, which are responsible for more than three-quarters of usage and have a bad effect on the ecosystem through climate change and global warming, are also a major concern of this decade (Bwapwa et al., 2017). The rate at which crude oil reserves are depleting is 4 billion tons per year, and at this rate, they are projected to be exhausted by 2052 (Baloch et al., 2016).
(a) Global energy trends: insights from 2023 statistical review of world energy. (b) Global projected biofuel consumption volume by country in 2030. Data sourced from Statistica 2024.
Consequently, the demand for environmentally friendly and sustainable energy sources has become increasingly important in addressing the current energy shortage in both industrial sectors and all forms of transportation (Liu et al., 2014). In pursuit of sustainable and renewable energy sources, biofuels have been proven as a substitute for traditional fossil fuels. Biomass, a major renewable energy resource, holds significant potential for meeting future energy demands by producing liquid biofuels (Pattanaik and Misra, 2017). By 2050, it is predicted that between 10% and 50% of the energy used worldwide will come from biomass (Kumar et al., 2015). The expected global biofuel usage by country or region in 2030 (in billion liters) is displayed in Figure 1(b). The United States is expected to continue to be the biggest user of biofuels, followed by Brazil, the EU, China, and India. Biofuels can be produced in all three states including gaseous, solid, or liquid (Gómez-Castro et al., 2023). When using liquid biofuels in engines, the emissions of greenhouse gases are reduced substantially (Pearson and Turner, 2014). Triglyceride-based biofuels known as biodiesel have gained the limelight because of their compatibility with existing diesel engines and their ability to lower GHG emissions. The major biofuels produced for the global market include green diesel, bioethanol, biodiesel, and bio-butanol (Sharma et al., 2020).
The physio-chemical characteristics of feedstock are crucial considerations when choosing a raw material for biofuel production. Biofuels are produced from animal or vegetable fats, sugar, cellulose, starch, and oils supplied by many animal or agricultural feedstock. The distinction between first, second, third, and fourth-generation biofuels is using original feedstock provenance (Callegari et al., 2020). First-generation biofuels (use food sources) gained popularity mostly in developed nations (Anwar, 2021). Second-generation biofuels, which depend on nonfood materials comprising animal manure, cellulosic biomass, spent cooking oil, straw, and other non-conventional sources, that typically end up in dumps after their essential component have been utilized, were developed as an interim solution to many problems with first-generation biofuels (Zuorro et al., 2020). Due to the complex nature of biomass and the challenges related to its synthesis, storage, and transportation, second-generation biofuels are not commercially viable (Chowdhury and Loganathan, 2019). Biofuels derived from algae mostly referred to as third-generation biofuels, offer a better replacement because of their capability to produce biofuel with significantly higher yields up to 9000 liters per hectare, or 10 times higher than the best conventional feedstock. Recent interest in algae as a biofuel feedstock has been fueled by energy price uncertainties and the potential advantages of microalgae over conventional terrestrial crops, leading to substantial investments in this area (Quinn and Davis, 2015).
Biodiesel is often created via the process of transesterification to produce esters and glycerol as a byproduct, using a reaction in which lipid reacts with alcohol essentially, the act of alcohol which replaces an ester known as alcoholysis or alcohol-cleavage (Maheshwari et al., 2022). Many processes for biofuel production are discussed in this article, each having its benefits and some limitations. Transesterification is a cheap and time-saving way of producing biofuel that yields biofuel with an increased cetane number and enhanced performance (Sajjad et al., 2022). The homogeneous catalysts exhibit strong catalytic performance in transesterification reactions, their inherent corrosive nature towards equipment and their challenging extraction from the mixture result in additional operational costs (Khan et al., 2021). In substitute of this, heterogeneous catalysts such as combined acid-alkali catalysts, solid alkali catalysts (hydroxides, hydrotalcite, metal oxides, dolomites, and supported earth elements), acidic solid catalysts (zeolites, sulfated metal oxides, hetero-poly acids, sulfonic ion-exchange resins, VOPO4), or biological catalysts can be utilized in the reaction (Joshi et al., 2017). Because lipases can break the ester link in tri-acyl glycerides (TAGs) and transform them into free fatty acids, monoglycerides and diglycerides, and the glycerol byproduct they are regarded as biocatalysts (Cavalcante et al., 2021). Nano-catalysts with high activity and ease of recovery, are revolutionizing biofuel production processes such as transesterification and pyrolysis, promising higher yields and reduced costs.
Pyrolysis is another promising method for turning biomass into biofuels at extreme temperatures (250–600°C) in an inert state (Chong et al., 2019). It is a relatively new technique to produce bio-based fuels and chemicals using biomass. Triglyceride hydrotreating processes including decarboxylation, hydrodeoxygenation, and hydrogenation are all part of the catalytic conversion process. The catalytic deoxygenation (DO) process works well for turning TGs into bio-jet fuel (Chehadeh et al., 2023), machine learning (ML) and artificial intelligence are converting triglyceride-based biofuel production by optimizing growth conditions for biofuel sources like algae, accelerating strain selection, and genetic engineering. These technologies enhance efficiency through advanced algorithms that analyze genomic data to identify high-lipid-producing strains. As AI manages and interprets production data, it supports decision-making, predicts maintenance needs, and optimizes resource allocation, driving innovation and sustainability in the biofuel industry (Okolie, 2024). Each type of biofuel holds great potential and offers various applications. This review focuses on triglyceride-based biofuel production, shedding light on a transformative aspect, and intensified technologies.
Generation-based biofuels produced from different feedstock
The type of feedstock needed for the conversion of biofuel/chemical compounds into higher alcohols, bioethanol, butanol, isoprenoids, hydrocarbons, and molecules produced from fatty acids varies and can be either sugary/starchy or lipid enriched (Zada et al., 2018). To produce biofuel, different kinds of biomass, including vegetable and residual oils and animal fats, can be utilized. Vegetable oils and most animal fats share a similar chemical makeup, primarily containing triglycerides with lower amounts of diglycerides and monoglycerides (Banković-Ilić et al., 2014).
First-generation biofuel
Over 95% of the world's first-generation biofuels have been made from edible vegetable oils, which are widely accessible and generated on a massive scale by the agriculture sector (Mat Aron et al., 2020). Vegetable oils are typically derived from different plant seeds, consisting of corn, sunflower, soybean, coconut, peanut, and palm. Whereas canola (known as rapeseed) and palm oil are the most prevalent sources of biodiesel in the EU and other tropical nations, soybean oil is the most preferred food source in the United States for biodiesel (Kim et al., 2018; Umana et al., 2020) while countries like Malaysia, Thailand, and Indonesia mainly utilize palm oil for the production of biodiesel (Pikula et al., 2020). However, since edible vegetable oils are derived from crops, cultivated for food, and used to produce biofuel, this raises many ethical questions (Ramos et al., 2019). Moreover, the cost of edible vegetable oils has increased, resulting in a 70–92% increase in production costs (Hanif et al., 2022). Additionally, their use resulted in significant environmental consequences, including contaminated water and soil, increased greenhouse gas emissions, a decline in biodiversity, the transmission of infestations and crop diseases, and sustainability problems like insufficient energy consumption (Agarwal et al., 2017; Correa et al., 2017).
Second-generation biofuel
Alternative, less expensive oils that don't compete with food sources, like sea mango, jatropha, castor, P. pinnata, rubber seed, and jatropha, have recently been considered as possible options for future feedstock. Second-generation biofuel is made from cellulosic biomass (cellulose, hemicellulose, and lignin) in a more environmentally friendly way (Joshi et al., 2017). These feedstocks don't need agricultural land and don't depend on food plants (Bhuiya et al., 2014). Palm has the highest oil content both among edible and non-edible feedstock while Jatropha comes second (Babazadeh, 2017). The most beneficial non-food crop, jatropha plantation, has drawn a lot of attention due to its high oil content for producing biofuel, ability to withstand drought for a prolonged amount of time, soil reclamation, reduction of desert areas, rural development, and environmental advantages (Ewunie et al., 2021). The jatropha plant has minimal nutritional needs, however as it prefers alkaline soil, it needs additional minerals like calcium and magnesium when grown in acidic soil. Jatropha oil is inedible because the seeds contain the poisonous substance curcin. Oil content is between 35%–40% in seed and 50%–60% in kernel, with the main fatty acids being oleic acid and linoleic acid (Issariyakul and Dalai, 2014).
Since waste cooking oil is about half as expensive as virgin oil, using it to make biodiesel is another effective method of reducing the cost of raw materials. The majority of waste cooking oil is dumped on soil, in garbage, or in a drainage system. Their dumping also causes a lot of environmental problems so utilizing waste cooking oil eliminates problems between the availability of food and land. There are two types of waste cooking oil (WCO) namely yellow grease and brown grease. WCO may include animal fat or vegetable oil which has been heated and used to prepare a large range of dishes. Yellow grease has a free fatty acid content of less than 15% and is produced from waste frying oil, fats, and oils gathered from restaurants or commercial cooking processes, has a significant amount of potential as an inexpensive raw source for the manufacturing of biofuel (Adewale et al., 2015). The properties some of edible and non-edible oils are mentioned in Table 1.
Properties of edible and non-edible oils.
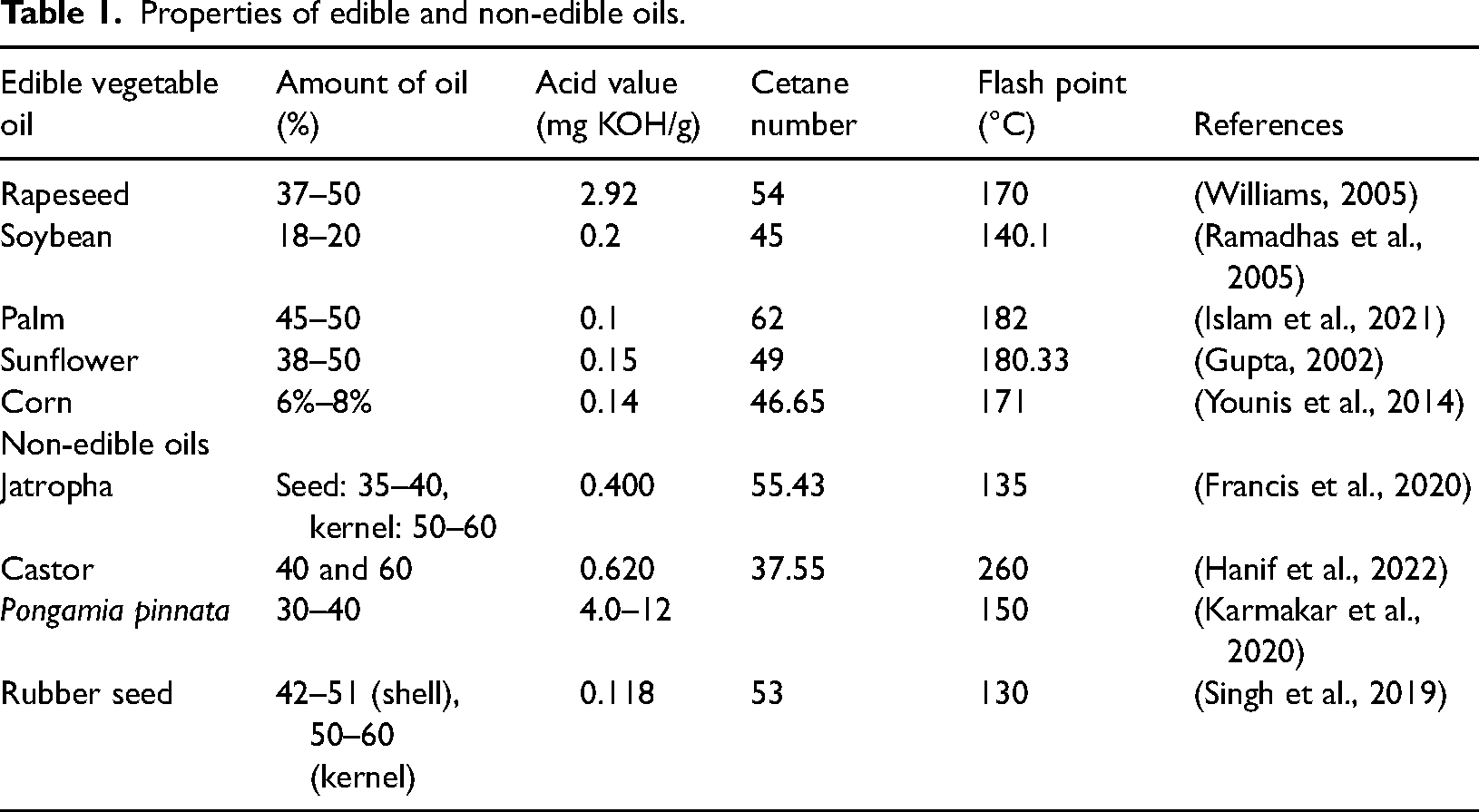
Due to its lower proportion of unsaturated fatty acids, biodiesel manufactured from animal fats has a higher cetane number than the others from most vegetable oils and diesel fuel (Adewale et al., 2015). The calorific value of biofuel derived from animal fats is also higher. Various animal waste lipids can be utilized as feedstock to produce biodiesel. They are: rendered pork fat, chicken fat, grease, and beef and mutton tallow (Banković-Ilić et al., 2014). Meat from cattle or mutton, either as edible or non-edible tallow, is one of the most commonly utilized animal fats used as a feedstock for biofuel. The raw animal fats are typically solid at room temperature, so heating them to 45°C is necessary before using them as diesel engine fuel (Cernat et al., 2020). Additionally, a higher percentage of saturated fatty acids produces biodiesel that is generally more stable and has an elevated cetane number (over 50 for lard and tallow) (Toldrá-Reig et al., 2020). In the United States, animal fats accounted for 8.4% of the feedstock in 2019, in the EU, over 13 million tons of vegetable oil and animal fat were used as feedstock for biodiesel. Of these, 6% (800,000 tons) were animal fats. This percentage has been consistent since 2014 (US EIA, 2020).
Third-generation biofuels
Given its many benefits, algal-based biofuel namely third-generation biofuel can be a viable alternative energy source (Yaşar, 2020). Algae are freshwater species that absorb carbon dioxide from the atmosphere and transform it into oxygen, also due to the large quantity of lipids they have, algae produce oil by breaking down their cell walls (Adeniyi et al., 2018). Microalgae are one of the many types of algae and have several benefits over plant-based biofuels, including (i) a high rate of growth, (ii) the ability to grow on non-arable land, (iii) the ability to grow in wastewater, (iv) increased CO2 consumption and (v) the ability to use the production of microalgae to synthesize several compounds of commercial interest (Quintero-Dallos et al., 2019). Microalgae typically have a lipid concentration between 20% and 50% of their dry weight, but under some circumstances, that content can rise to 80% (Sun et al., 2018). Other than lipids, the two primary components of algal biomass that can be transformed into varied by-products, such as dietary supplements, feed for livestock, or biofertilizers, are carbohydrates and proteins (Raheem et al., 2018). Due to their small size, microalgae are often grown by submerging them in water and monitoring several key factors, including temperature, pH, carbon dioxide levels, photoperiod, availability of nutrients, and light penetration into the water body. Then, using different mechanical, chemical, and biological methods, matured microalgae are collected and dried to create the biomass feedstock needed for other conversion processes (Maliha and Abu Hijleh, 2023). Although algae-based biofuels are very promising, there are several obstacles to overcome, algal collection and oil extraction are challenging technological tasks (Dixon, 2013).
Life cycle assessment (LCA) studies use several indicators to quantify energy use in biofuels, with an emphasis on fossil energy consumption since policies focus on energy security and reduced dependence on fossil fuels. Most studies estimated fossil energy consumption for first- and second-generation biofuels to be under 0.5 MJ MJ-1 of energy produced, though results vary widely. First-generation biofuels vary between 0.04 and 0.86 MJ per MJ and second-generation from −0.57 to 0.87 MJ MJ-1, with negative values representing energy credits. Most variations are due to the feedstock productivity, farming practices, and conversion methods. The range of estimates for fossil fuel demand in the LCA of algal biodiesel is even wider, from 0.15 to 40.5 MJ MJ−1. These differences highlighted technological uncertainties, a variety of possible feedstocks, and production systems. However, most reports agree that algal biofuels are usually not energy-intensive. This is mainly because of energy requirements such as pumping, dewatering, lipid extraction, and thermal drying processes (Jeswani et al., 2020).
Fourth-generation biofuels
The disadvantages of the above three generations were thought to be overcome by genetically altering microorganisms (known as the fourth generation) to increase efficiency and product production (Mehmood et al., 2021). Fourth-generation biofuels are obtained from bioengineered microorganisms such as bioengineered algae, fungi, yeast, and cyanobacteria or bioengineered crops whose natural characteristics have been modified (Jiang et al., 2014; Shin et al., 2016). This covers a wide range of characteristics, such as the use of different sugar forms (pentoses and hexoses), increased production of lipids, enhanced carbon fixation, and photosynthesis (Cavelius et al., 2023). The successful manufacture of ethanol and other fuel products including butanol, iso-butanol, and modified fatty acids has been achieved by the use of genetically modified cyanobacteria (Nozzi et al., 2013). These microorganisms have undergone genetic modification to augment their carbon dioxide intake for photosynthesis, generating an artificial carbon sink and augmenting the biofuel generation process (Abdullah et al., 2019).
A variety of fuel molecules, including alcohols, free fatty acids, molecular hydrogen, and alkanes, have been produced from cyanobacteria. Examples of these model strains are Synechocystis sp. PCC6803, which was shown to produce 5.5 g L−1 (212 mg L−1 day−1) of ethanol and 4.8 g L−1 (302 mg L−1 day−1) butanol, as well as other cases where titers were shown to be high due to strong promoters that drive the expression of pathway enzymes. However, more productive production would require a better understanding of native metabolism as well as the construction of highly sophisticated genetic tools. Transcriptomics has thrown light on the role of small RNAs in cellular regulation, while genome-scale models have helped couple production with growth. Nonetheless, cyanobacterial productivity is still inferior to that of model heterotrophic hosts (Liu et al., 2019a). The amount and quality of biofuels can be managed and enhanced by genetic engineering technologies, but widespread adoption will require political acceptance and support (Malode et al., 2021).
Intensified technologies
Transesterification (alcoholysis)
Transesterification is a method by which lipids react with alcohol to manufacture different kinds of biofuels (Salaheldeen et al., 2021). In the presence of a catalyst, vegetable oil (triglycerides) reacts with primary alcohol to produce biofuel-like fatty acid alkyl esters, usually fatty acid methyl esters (FAME) (Baskar et al., 2019). This reaction is illustrated in Figure 2. Another byproduct produced is glycerol. Methanol is the alcohol that is most frequently used for transesterification due to its lower cost and other advantageous qualities such as polarity and short-chain molecular structure (Akram et al., 2022). Because methanol has a higher equilibrium conversion rate than ethanol and ethanol has great solvent performance, using a methanol/ethanol mixture for transesterification could be advantageous (Martinez-Guerra and Gude, 2014). Reaction time, temperature, pressure, catalyst type and concentration, feedstock oil type, alcohol-to-oil molar ratio, and mixing are some of the variables influencing biodiesel yield in transesterification reactions (Mandari and Devarai, 2022). To boost reaction speed and yield, intensified technologies concentrate on improving these reaction parameters, for example, temperature and pressure. Various type of catalysts is employed in the transesterification process which can be chemical or biological in origin. There are the following types of transesterification conversion processes: acid-catalyzed, base-catalyzed, and enzyme-catalyzed conversions (Long et al., 2021). The chemical catalyst includes homogeneous catalysts (base or acid), heterogeneous catalysts (solid acid or solid base catalysts), and non-catalytic supercritical fluids (SCFs) while enzymes are used as biocatalysts.
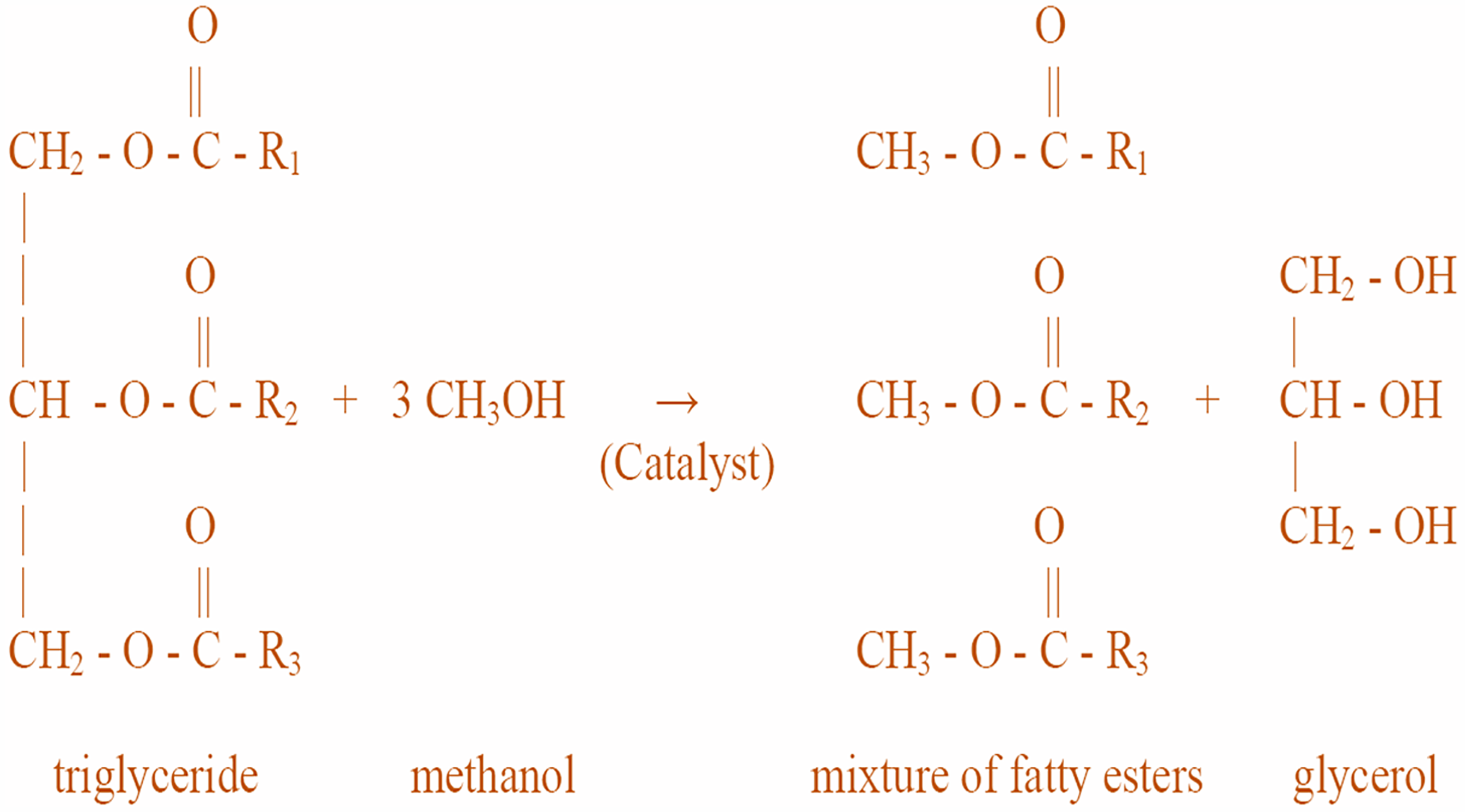
Transesterification reaction of triglycerides with methanol.
Homogeneous catalysts
Homogeneous catalysis refers to a sequence of steps employing a catalyst from a similar phase as the reactants, either liquid or gaseous (Neupane, 2022). The biofuel yield of different homogeneous catalysts at particular temperatures, reaction time, and in the addition of a co-solvent such as ethanol or methanol is given in Table 2. So far, the most frequently employed catalysts for acid-catalyzed transesterification have been HCl and H2SO4 (Farobie and Matsumura, 2017). A study showed that in addition to methanol 0.5% H2SO4 was used to transform algal oil which contained 90% water to biodiesel and at 120°C and 180 min, 93.2% biodiesel yield was produced (Cao et al., 2013). However, acid-catalyzed processes are not more cost-effective or faster than alkali-based catalysis because of their higher energy consumption (Rizwanul Fattah et al., 2020). Alkali metal hydroxides, like sodium or potassium hydroxide; alkoxides, and carbonates are examples of basic catalysis (Chen et al., 2020). In a base-catalyzed process, a homogeneous catalyst such as KOH or NaOH is typically utilized to make biodiesel fuel.
Different homogeneous catalysts.
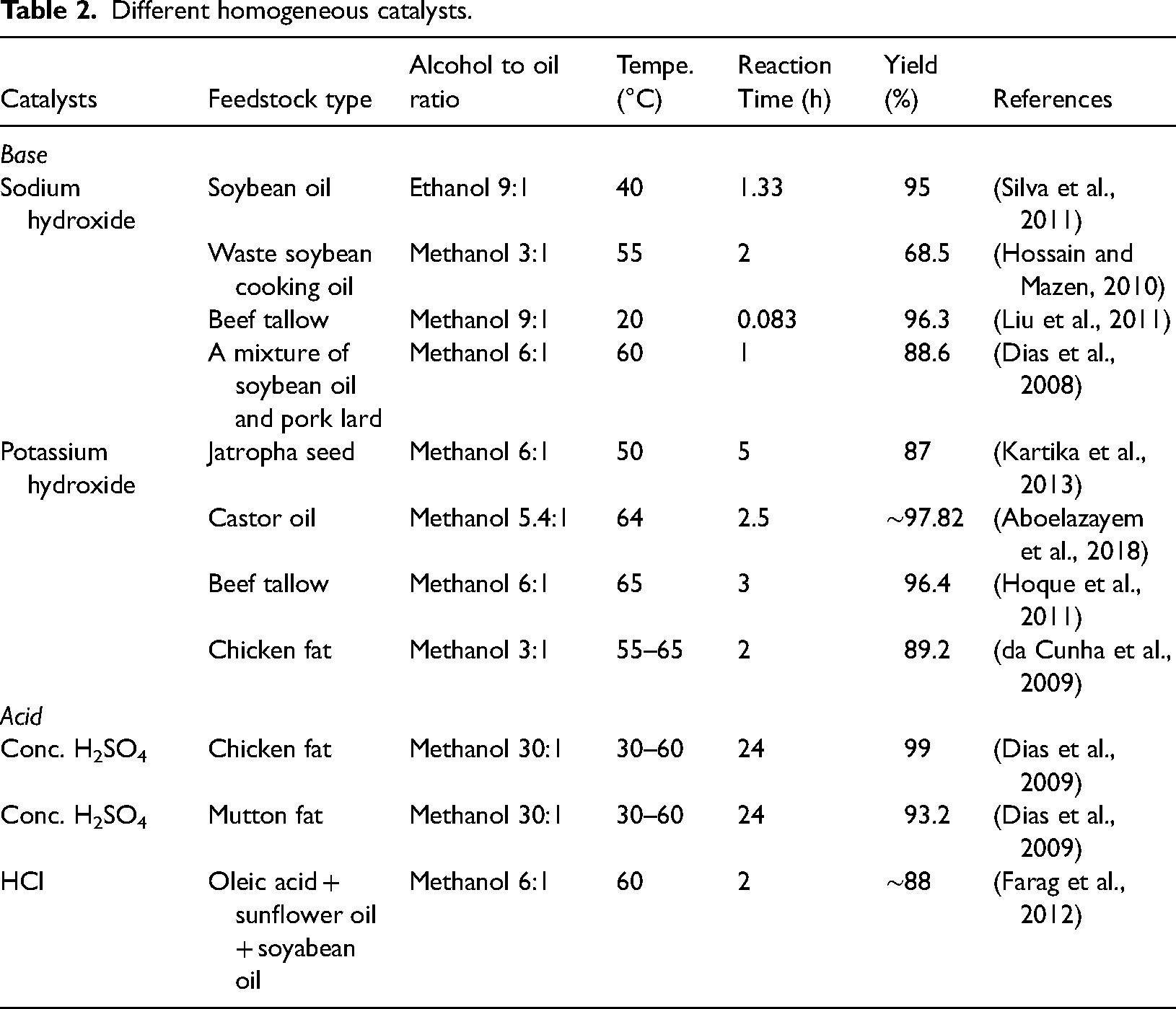
Under mild conditions; atmospheric pressure, 60°C temperature, and roughly 1 h of reaction time can create significant amounts of methyl ester. NaOH is taken into consideration for transesterification because of its high purity, inexpensive cost, and comparatively low requirement when compared to KOH. However, alkali metal alkoxides are found to be more active than hydroxides (Saydut et al., 2016). One well-known disadvantage is that oil with high-fat content cannot be entirely transformed into biodiesels; instead, it mostly remains as soap. When we use extra-pure virgin oils with a free fatty acid content of less than 0.5% and acid values of less than 1 mg KOH/g, homogeneous alkali catalysts produce better results (up to 97%) and are purer (Mandari and Devarai, 2022). Another major drawback is that the catalyst utilized in this procedure cannot be recovered and is therefore not reusable. It needs to be neutralized and disposed of as an aqueous salt waste stream as a result, in addition, the process of purifying biodiesel produces a significant amount of wastewater, which harms the environment (Selvakumar and Sivashanmugam, 2017).
Heterogeneous catalysts
Heterogeneous catalysts are more promising as transesterification process catalysts than homogeneous catalysts because they are recyclable and separable, which eliminates the need for purification and separation steps and lowers costs and pollutants. For instance, 88 tons of sodium hydroxide may be required to produce 8000 tons of biodiesel, yet only 5.6 tons of supported MgO are needed to produce 100,000 tons of biodiesel (Romero et al., 2011). These catalysts tolerate higher free fatty acid and moisture content (Baskar et al., 2019). Additionally, heterogeneous catalysts can be employed in fixed-bed reactors and have a less harsh nature, making their operation safer, more affordable, and eco-friendly. Some of the heterogeneous catalysts and their biodiesel yields are listed in Table 3.
Various heterogeneous catalysts.
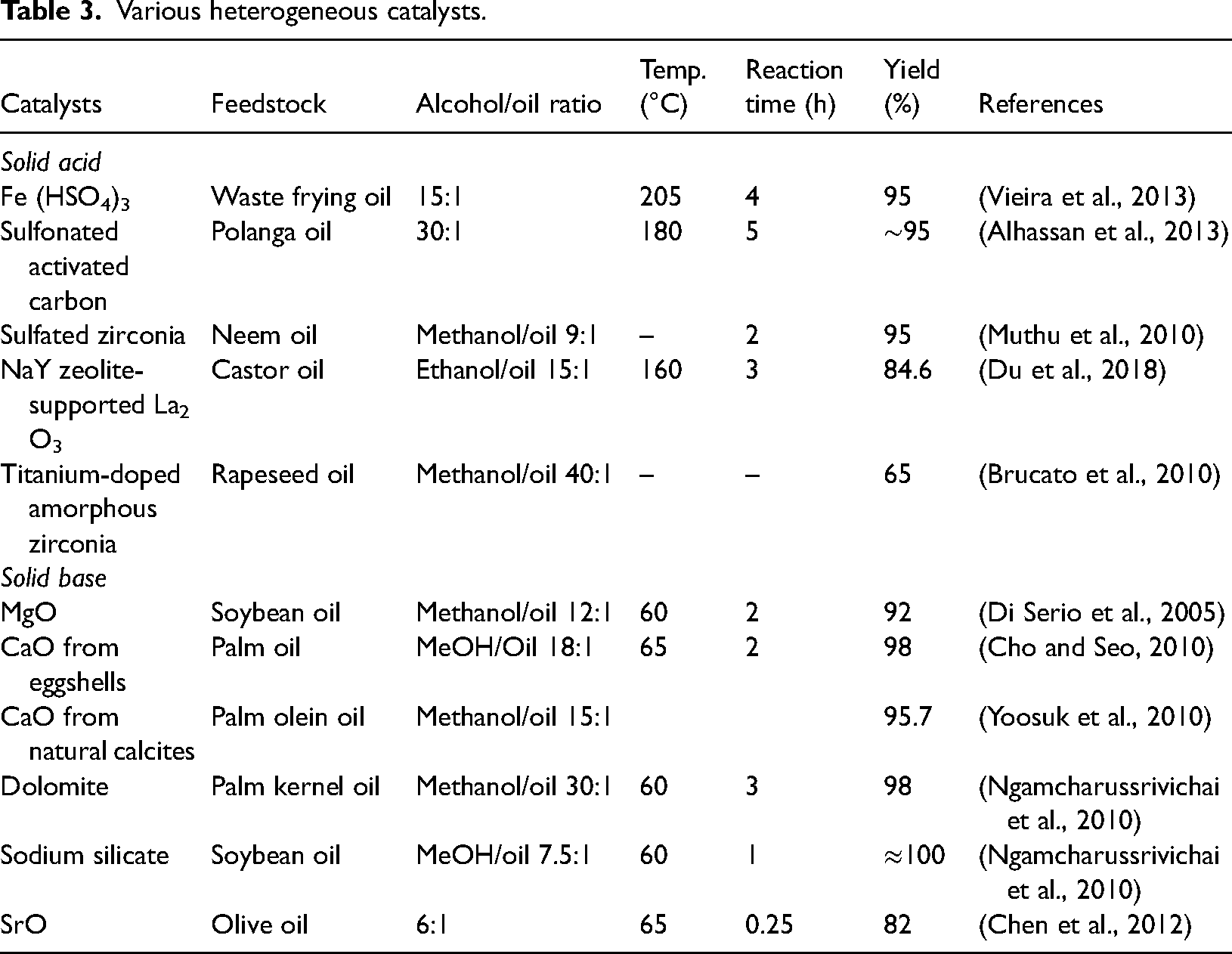
Since heterogeneous solid acid catalysts have Lewis and Bronsted acid active sites, they are regarded as a significant catalyst in the industrial setting compared to homogeneous acid catalysts because they support both esterification and transesterification simultaneously for the production of biodiesel from oils with high free fatty acid content (Guldhe et al., 2017). The production of biofuels has made extensive utilization of several solid acid catalysts such as zeolites, cation exchange resins, zirconium dioxide (ZrO2), tin dioxide (SnO2), and titanium dioxide (TiO2) (Carvalho et al., 2017). Sulfated and supported polyanions, however, are the catalytic systems that have been researched the most. Under ideal reaction conditions, NaY zeolite-supported Lanthanum tri oxide catalysts made via physical mixing for the transesterification of castor oil produced an 84.6% biodiesel yield (Du et al., 2018). According to a report, the yield of biodiesel from palm oil increased from 64.5% to 90.3%, and from coconut oil, it increased from 49.3% to 86.3%, respectively, following the addition of zirconium dioxide and sulfuric acid. The alteration of metal oxides’ surface acidity is the cause of the rise in the volume of biodiesel produced (Pikula et al., 2020). The manufacturing process is complex; this results in increased costs, excessive reaction temperature (∼100°C) and pressure (∼5 bar), high alcohol-to-oil molar ratio, and prolonged reaction time. Also, contamination may occur from catalyst active sites due to leaching (Batalha and da Silva, 2021).
For the Trans-E method, the heterogeneous base catalyst is preferable because of its short reaction time, lower temperature, and economic viability (Jayakumar et al., 2021). Additionally, depending on the catalyst's lifespan, regeneration, reuse, and construction may result in higher selectivity and longer activity (Talha and Sulaiman, 2016). The most frequently studied solid material-based catalysts are hydrotalcite, metal oxides (CaO), supported base catalysts, metallic salts (MgO), and zeolites (Faruque et al., 2020). Along with the other alkaline earth metals, which have a rather heterogeneous character, SrO and magnesium oxide are widely used as catalysts in biodiesel production (Yoo et al., 2010). Researchers are more interested in using calcium oxide (CaO) as a heterogeneous catalyst because of its high basicity and long catalyst life. It requires only moderate reaction conditions. Despite their high conversion efficiency, these catalysts are unsustainable for biodiesel production because of their complex preparation methods, dependency on non-renewable sources, and associated environmental and disposal issues. In recent years, agricultural wastes have been transformed into eco-friendly and cheap catalysts for the production of biofuels from different low-grade feedstock (Awogbemi et al., 2021a, 2021b). Some of the heterogeneous catalysts having catalytic activities obtained from agricultural wastes are eggshells (Piker et al., 2016), chicken bones (Farooq et al., 2015), coal fly ash (Manique et al., 2017), banana peel (Fan et al., 2019), banana trunk ash (Rajkumari and Rokhum, 2020), Musa balbisiana peel ash (Gohain et al., 2017), etc. The disadvantages of using heterogeneous catalysts are catalyst loss as a result of leaching throughout the operation, and their susceptibility to glycerol soap and FFA in oil (Naeem et al., 2021; Nath et al., 2019).
Enzyme-catalyzed Trans-E
Enzymes are the foundation of the biofuel industry, and employed effectively to facilitate the synthesis of different kinds of biofuels through their unique biochemical processes (Singh et al., 2022). Lipase known as triacylglycerol acyl hydrolase (EC 3.1.1.3), is the type of enzyme used in the manufacturing of biodiesel. It transforms oil into fatty acid alkyl ester and glycerol, as a byproduct (Norjannah et al., 2016). Enzyme-catalyzed reactions like non-catalyzed methods, offer simple biodiesel purification processes but due to their moderate operating conditions, they require significantly less amount of energy (Amini et al., 2017). When compared to chemical catalysts, lipases are a more appealing option for biocatalysts since they produce better quality biodiesel, need mild reaction conditions, don't make soap, use minimum energy, are easier to recover from products, and can be recycled (Brun et al., 2011). Although lipase enzyme takes the role of an acid or basic catalyst, the process of enzyme-catalyzed transesterification is identical to that of simple transesterification. The essential elements of the enzyme-catalyzed transesterification reaction for the manufacture of biodiesel are oil feedstock, lipase, and acyl acceptor (Pasha et al., 2021). The catalytic performance of some enzyme catalysts in the production of biodiesel is displayed in Table 4.
Different enzyme catalysts and their efficiency in biofuel production.
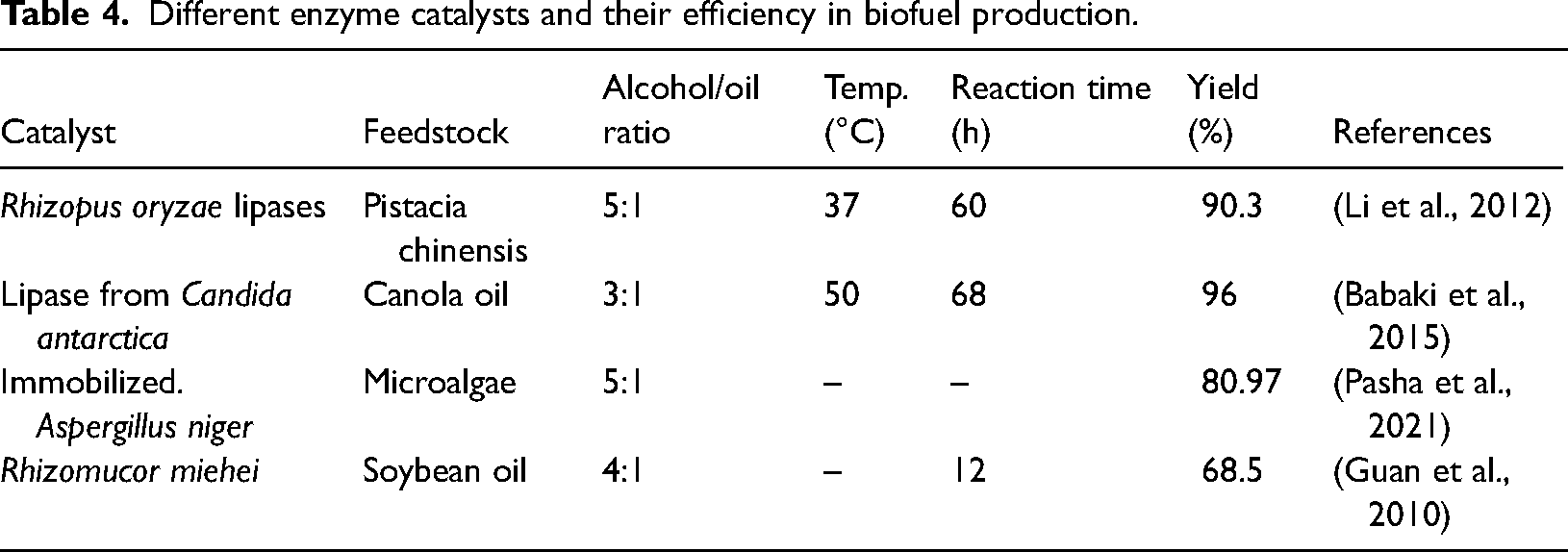
Generally speaking, free or immobilized enzymes and microbial cells can be used for biological catalysis. Enzyme immobilization makes product recovery easier and allows costly enzymes to be recycled for various cycles without losing their activity (Kalita et al., 2022). There are different types of immobilization of lipases and these include cross-linking, covalent binding, entrapment, adsorption, and encapsulation (Sandoval et al., 2017), as shown in Figure 3. Since whole-cell systems have greater benefits like simple preparation and purification, they are currently the most widely recognized and utilized biocatalyst for biological transesterification as compared to free or immobilized enzymes (Osorio-González et al., 2020). Yan et al. (2014) reported that the enhanced strain's enzyme produced an 82% transesterification efficiency, which helped with the oil conversion. The many reactor designs used to produce biodiesel are packed-bed reactors (PBR), stirred tank reactors (STR), fluidized bed reactors (FBR), and bubble column reactors (BCR) (Zhao et al., 2015). Because membrane bioreactors enable continuous product separation and eliminate enzyme inhibition, their use in enzymatic processing is becoming more and more appealing. Each method for the production of biofuel offers its own advantages; however, different case studies show us that the mostly widely used as well as effective method is transesterification by using enzymes. This technology has several advantages over other methods. Low reaction temperatures are achieved because reactive molecules are activated by energetic electrons, hence eliminating chemical catalysts and streamlining the process. It is not affected by impurities, hence making it more reliable. The short reaction time minimizes saponification, which means minimal glycerin production, simplifying the separation process and increasing the efficiency of overall production.
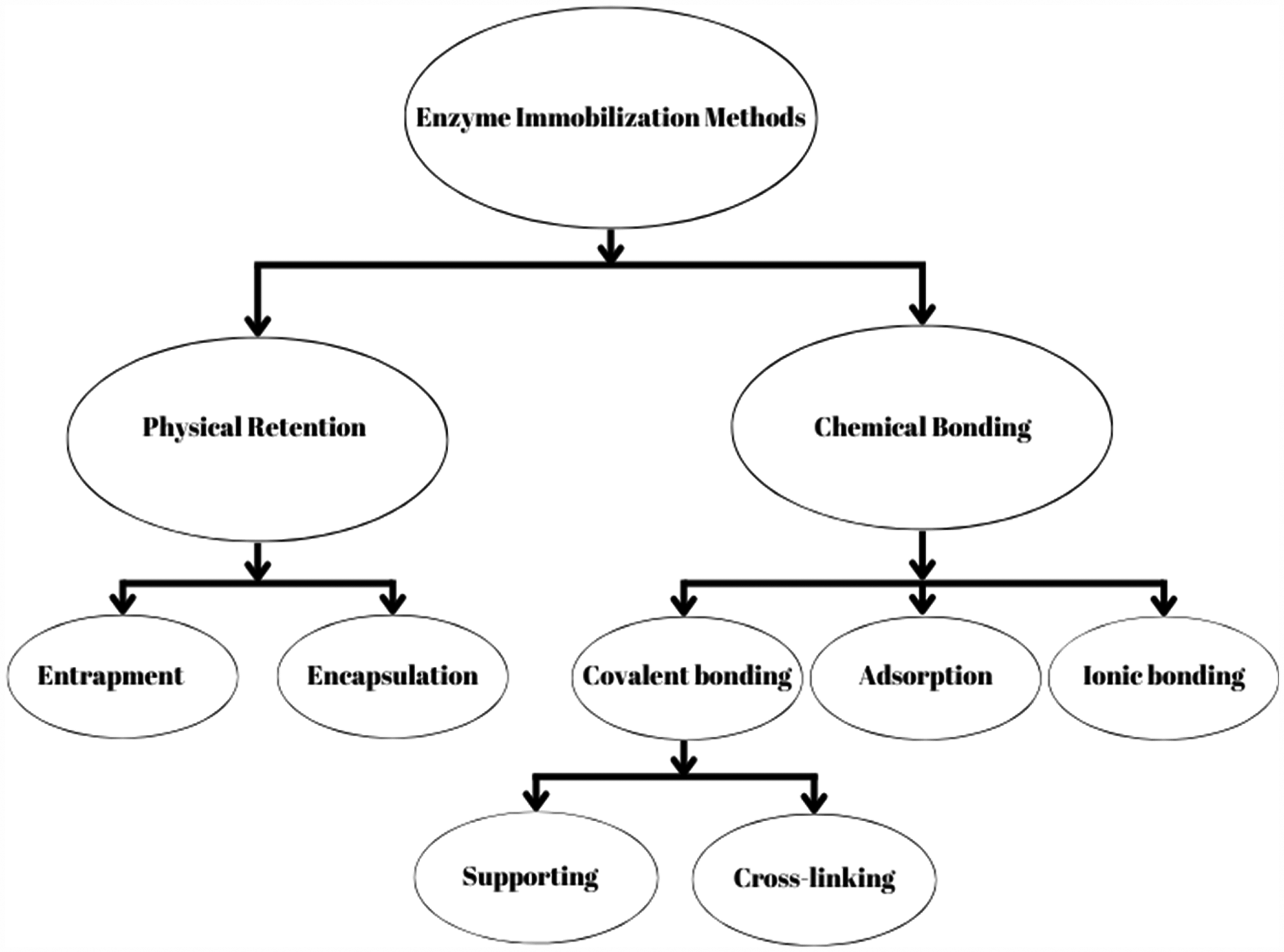
Different methods for enzyme immobilization.
Nano-catalysts
Nano-catalysts have revolutionized biofuel production by offering significant advantages over traditional catalytic processes. The hallmark of the use of nanoparticles in this field has been attributed to their higher surface area/volume ratio, enhanced catalytic activity, and stability as well as recyclability (Esmaeili et al., 2021). Recent studies have brought to light several nano catalysts used for biofuel synthesis such as metal nanoparticles magnetic, and metallic oxide. Metals such as silver, platinum, and gold display excellent catalytic ability due to high numbers of active sites for reaction from the accompanying metallic nanoparticles. Metal oxides like zinc oxide (ZnO), magnesium oxide (MgO), and titanium dioxide (TiO2), are valued for their stability and high catalytic activity (Pramanik et al., 2023). In parallel such iron oxide (Fe3O4) magnetic nanoparticles enable simple separation/re-utilization from the reaction mixture, a highly economic step to make this process cost-competitive (Ahmed et al., 2024). The reaction rate and yield of biodiesel have been significantly improved in the process by nano-catalysts. Most significantly scientists have managed to use CaO nano catalysts for converting non-edible oils and waste cooking oils into biodiesel. Possible reasons could be that the CaO nano-catalysts with strong basicity would suppress side reactions, leading to high purity and improved yield only for conversion of triglycerides into biodiesel (Saoud, 2018). The main issue of the solid catalysts is their separation from the reaction mixture. Another challenge is reusing them after filtration. With magnetic nano-catalysts, it seems to be possible; magnetic separation enhances the possibility of recovery and reuses of catalysts than normal filtration. For example, the KF/CaO-Fe3O4 nano-catalyst can be recycled and reused up to 14 times with a loss that's minimal and about 90% recovery (Wu et al., 2023). Additionally, Amini et al. (2013) prepared nano-sized amorphous alumina powder via the sol-gel method that showed a conversion rate of 95.8% biodiesel after being reconstructed at 700°C for four hours after the experiment. Transesterification without a catalyst led to a conversion of 21.6%. With a conversion efficiency of more than 99%, the biodiesel from the X-Li-CaO nano-catalyst, there is no chance for reuse of it. In contrast to this, the CZO nano-catalyst that was synthesized using the method of sedimentation can be reused up to six cycles with 73.95% of efficiency in the sixth cycle (Gurunathan and Ravi, 2015). Future prospects in the field involve advancing towards more economical and environmentally sustainable nano-catalysts. Researchers are also focusing on exploring new nano-materials and combinations to enhance the production of biofuel (Arya et al., 2021).
Supercritical fluid technology
The catalytic transesterification is a multi-step process, and thus it includes not only the transesterification reaction itself but also several other additional steps: recycling of the unused reactants, purification of the esters, separation of glycerol, and separation of the catalyst from the reactants and products (Nayab et al., 2022). It yields biodiesel with fast reaction rates a larger yield of FAMEs and reduced operational stages. In these supercritical conditions, this biodiesel production process offers a promising alternative to conventional methods like catalytic transesterification. This technique allows the concomitant production of biofuel through TG transesterification and FFA esterification. The flow chart for supercritical (SC) vegetable oil transesterification (TE) into biofuel is shown in Figure 4. Any substance having the nature of both gases and liquids under values of temperature and pressure above their critical point is considered a supercritical fluid. Kinetics is faster, higher fuel production rate, ease of handling, and no catalyst addition are some of the advantages the SCF technology has over conventional techniques for biofuel production (Pavlova et al., 2022). Carbon dioxide is the most popular supercritical solvent (Duarte et al., 2017; Patel et al., 2020) however acetone, methanol, ethanol, and water are also utilized in supercritical circumstances to produce biofuel (Akalın et al., 2017; Marcus, 2019).
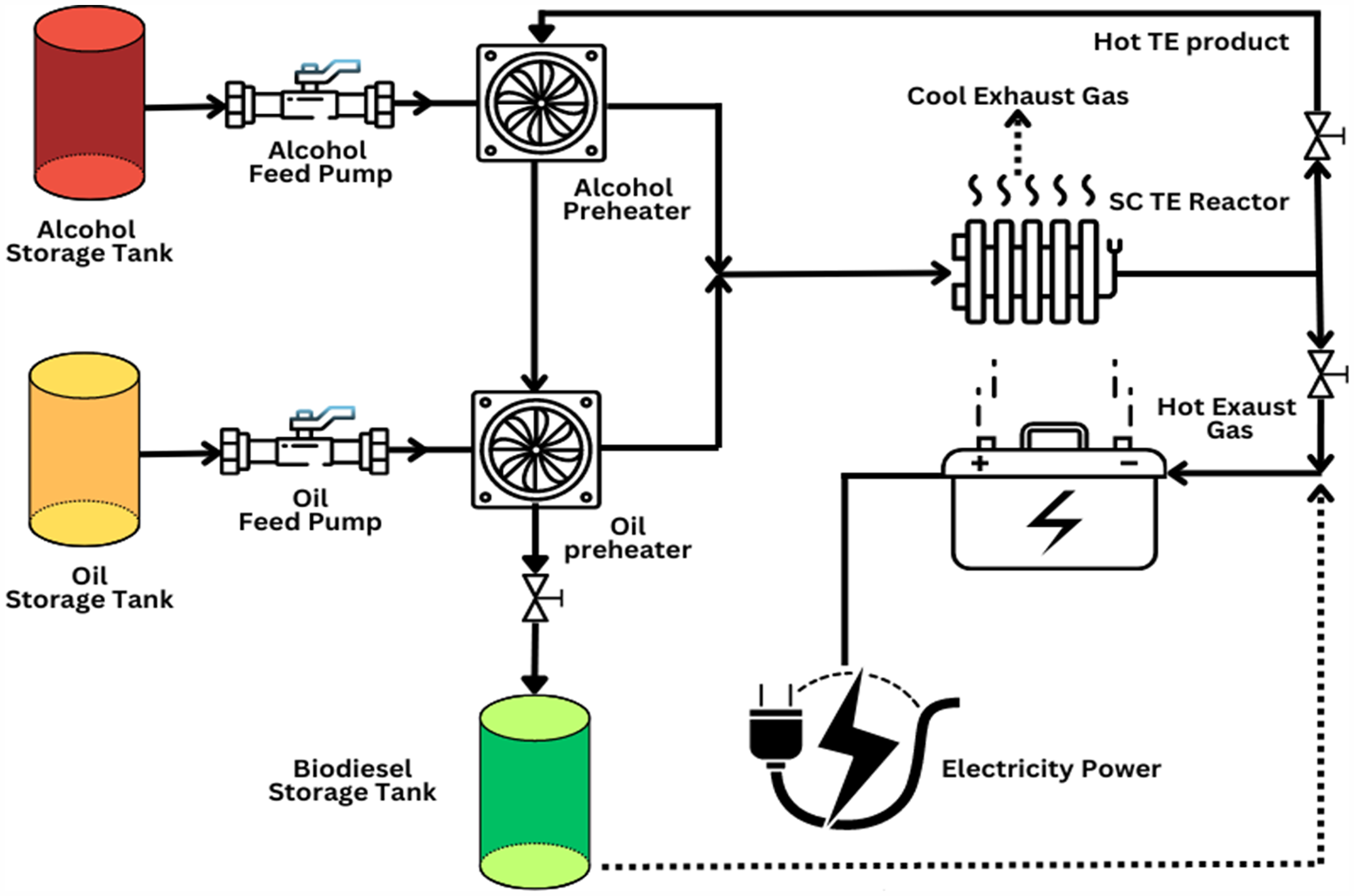
Flow chart for supercritical (SC) vegetable oil transesterification (TE) into biofuel. The setup includes storage tanks, pumps, and preheaters for reactants, a reactor heated by diesel generator exhaust, and energy-efficient preheating by the transesterification product stream.
Supercritical CO2 can easily dissolve nonpolar chemicals, and the addition of acetyl chloride, methanol, and ethanol as polar co-solvents can improve the recovery of polar compounds (Molino et al., 2018). For various oil feedstock, higher alcohol-to-oil ratios, temperatures above 300°C, and short residence durations are utilized to optimize biodiesel yields with methanol/ethanol (Reddy et al., 2018). Triglyceride and methanol combine into a single phase in supercritical methanol because of an increase in methanol's density and a fall in its dielectric constant, which ultimately lessens methanol's polarity because of H-bonding (Farobie and Matsumura, 2017). Therefore, in supercritical circumstances, non-polar triglycerides solubilize more readily in methanol and generate a homogenous phase. However, when using the non-catalytic SCTE technology, high temperatures (supercritical conditions, T = 250–400°C) must be employed to achieve a high conversion reaction, as this will cause the biodiesel fuel to thermal breakdown. Some researchers have attempted to address this issue by introducing heterogeneous or homogeneous catalysts to finish the reactions at lower temperatures and short residence times to prevent breakdown reactions (Zeng et al., 2014). Since heterogeneous catalysts require fewer stages in the separation process than homogenous catalysts, they are typically used to increase conversion rates (Andreo-Martínez et al., 2020).
Pyrolysis or thermal cracking
Pyrolysis, which is also known as thermal cracking, is a method of transforming triglycerides into hydrocarbons using external heating, with or without a catalyst. The reaction temperature is dependent upon different factors like biomass composition and proportion of solid, liquid, and gaseous products. It typically varies between 250 and 650°C (Hoang et al., 2021). Non-catalyzed cracking of triglycerides at a temperature of 300–500°C yields complex products with high acid values which are unsuitable for fuel use (Palanisamy and Gevert, 2016). Basic catalysts like CaO and Na2CO3 enable deoxygenation, lowering acid values, while molecular sieve catalysts like ZSM-5 and MCM-41 can further transform linear hydrocarbons into aromatics and isomeric hydrocarbons, enhancing fuel characteristics (Li et al., 2018; Long et al., 2020). However, a major problem is the molecular sieve catalyst fails to remove the O atom sometimes which yields pyrolysis oil with having high acid value. So composite catalysts combining basic metals and their oxides on mesoporous molecular sieves are used which enhance liquid fuel production by reducing acid values while maintaining high yields (Li et al., 2013).
Depending upon their heating rate pyrolysis is divided into three types; slow, fast, and flash pyrolysis. Slow pyrolysis, utilized for centuries, provides solid char and liquids due to longer vapor residence durations; however, its technological limits hinder high-quality bio-oil production, requiring additional energy input (Aboelela et al., 2023). Many primaries, and non-equilibrium liquid and gas outputs including useful chemicals, petrochemicals, chemical intermediates, and fuels derived from biomass, have been reported in the latest studies on fast or flash pyrolysis (Patel et al., 2020). Fast pyrolysis rapidly heats biomass with a heating rate between 10 and 200°C/s, producing mostly liquid products (60–75%), making it ideal for bio-oil production (Tripathi et al., 2016). This approach reduces heat exposure duration, favoring liquid formation over char, and its adaptability extends beyond energy production to applications in the food and chemical sectors, providing a low-cost investment with excellent energy efficiency (Al-Rumaihi et al., 2022). Flash pyrolysis achieves up to 75% bio-oil yield with a heating rate greater than 1000°C/s (Ighalo et al., 2022). Since the pyrolysis process produces green diesel at atmospheric pressure and doesn't require hydrogen as a co-reagent, it is far easier and cheaper than other methods, even if the cost of raw materials accounts for around 80% of the entire cost of bio-oil synthesis (Abdulkareem-Alsultan et al., 2019). In summary, any of the three forms of pyrolysis have lower costs than traditional transesterification when it comes to producing green diesel using triglycerides.
Catalytic hydrocracking
Apart from pyrolysis or cracking, another very appropriate technique for transforming fats and oils into fuels akin to fossil diesel is hydrocracking or hydrodeoxygenation (Yu et al., 2022). The unsaturated carbon-carbon bonds of the triglycerides need to be added with significant hydrogen for the unsaturated carbon–carbon bonds to generate saturated triglycerides. This needs to result in total hydrogenation. Decarboxylation and dehydration then remove oxygen. The only items that are left over that can be used as fuel are hydrocarbons and water (Khan et al., 2019). This process uses heterogeneous catalysts to convert vegetable oils into alkanes that have boiling points roughly comparable to those of gasoline and diesel and hence may be used as fuels without further processing (Hájek et al., 2021). These green fuels, such as naphtha, jet fuel, and diesel, which are created in existing hydro-treating units within the refinery from triglycerides, have generated interest in optimizing catalytic systems and operating conditions for economic viability (El-Sawy et al., 2020). Hydrocracking or hydro decarboxylation converts all O atoms in triglycerides to CO2, resulting in hydrocarbons with an odd number of carbon atoms from FFA (Hermida et al., 2015). This technique, including acid sites of amorphous supports, modifies catalytic acidity to achieve hydrocarbons with appropriate boiling temperatures, improving attributes like lower pour point and higher-octane number (Žula et al., 2022).
Blends of vegetable oils and heavy vacuum gas oils are treated in hydrogen streams to produce liquid alkanes with acceptable properties, with byproducts primarily consisting of different gases. Despite producing byproducts, the yield of liquid hydrocarbons remains high, making this process a possible option for getting high-quality diesel biofuel from renewable sources (Venkateswaran et al., 2022). Research in this field has grown significantly, particularly in the manufacture of biofuels for aircraft, motivated by the need for greener alternatives in the aeronautical industry. UOP Honeywell Co. and Haldor Topsøe have developed techniques to manufacture renewable green liquid fuels from non-edible vegetable oils and fats. This contributes to reducing greenhouse gas emissions and lesser pollutant production without requiring prominent alterations in existing aviation infrastructure (Sotelo-Boyás et al., 2012; Verdier et al., 2019). Several hydro-processing plants across the world are increasing jet biofuel production by co-processing methods, combining biofuels with petroleum feedstock to generate low-sulfur fuel products, hence reducing the carbon footprint of aviation fuel (Alherbawi et al., 2021).
Types of biofuels
Biodiesel
One promising renewable and low-carbon alternative to fossil diesel for the transportation sector is biodiesel, a blend of alkyl esters made of fatty acids. Due to the close similarity in the physical and chemical properties of biodiesel to fossil fuel, biodiesel is ranked as one of the most prospective substitutes for these fossil fuels. From the findings of the current research, biodiesel is similar to traditional fossil fuels but with slightly reduced emission levels (Abomohra and Elshobary, 2019). The biodiesels can be used in a currently available diesel engine without the need for modification, and there is minimal production of dangerous gaseous emissions such as Sulphur oxide. Biodiesel performance is subjected to the temperature as well as the ratio between the bio-diesel and the conventional diesel. Biodiesel with a higher blend of FAME has a lower cloud point, so the smaller the percentage of biodiesel in the blend, the better the blend will perform in colder temperatures (ASTM Biodiesel Specifications, 2018). The biodiesel blends are represented as Bxx, where the xx represents the percentage of biodiesel. Hence, B100 is similar to biodiesel and a B80 mixture indicates that the composition of the biodiesel is 80% by volume, and that of petroleum diesel is 20% (Ramos et al., 2019). The key drawback of biodiesel is its cost, which is higher compared to petroleum fuel. It has been estimated that the raw material cost accounts for varying ranges from between 70% and 95% of the total cost of production (Banković-Ilić et al., 2014).
Bioethanol
Currently, bioethanol is by far the most popular alcoholic biofuel type and stands as the leading biofuel in the market (Chia et al., 2018). Even though ethanol is classified as an octane booster for gasoline, it is also a performance-enhancing additive, and fuel blends containing up to 40% ethanol can help lower gasoline consumption by 3%–4.4%, improve the efficiency of internal combustion engines, and reduce carbon dioxide emissions by 19–35 metric tons every year. Bioethanol can be manufactured from microalgae via three processes: hydrolysis and fermentation of biomass, dark fermentation, and photo fermentation (de Farias Silva and Bertucco, 2016). Bioethanol is an oxygenated fuel consisting of about 35% oxygen and this curtails particulate matter and nitrous oxides, NOx released during combustion (Sarlak et al., 2024). Another advantage of bioethanol over regular gasoline is that it has a higher-octane number; it has a higher degree of energy per unit volume compared to regular gasoline, i.e., greater heat of evaporation, higher flame speed and the flammable range is larger as compared to gasoline (Manzetti and Andersen, 2015). These properties cause higher compression as well as shortened burning time and neat burning engines (Vohra et al., 2014). Over the last decade, interest in biofuels has increased due to the introduction of policies aimed at mitigating climate change and reducing greenhouse gas emissions from the transportation sector. In the last 10 years, global bioethanol production increased by a significant margin of 67% following the implementation of such policies; it rose from 67 billion liters in 2008 to 110.4 billion liters by 2018. Production of biodiesel increased by more than three times between the periods from 12 billion liters to 41 billion liters. Bioethanol is also significant in a large number of countries including the United States, Canada, and Brazil as a reformed transport fuel or as an enhancement for fuel (Liu et al., 2019b).
Biobutanol
Butanol is the emerging biofuel having higher resistance power as well as has lower octane number as compared to ethanol because of less volatile, less hygroscopic in nature, compatible with pipelines that can be mixed thoroughly with gasoline, and is less toxic in nature than ethanol due to its higher caloric value (Kumar and Banerjee, 2018; Phuengjayaem et al., 2020). Efforts to enhance butanol tolerance have been made through several approaches; one of the approaches being metabolic engineering (Generoso et al., 2015). Some of the genetically modified microorganisms include the Clostridium spp. (Zhang et al., 2018), E. coli (Dong et al., 2017), and Saccharomyces cerevisiae (Generoso et al., 2015) have been used to improve the production of butanol. Dong et al. (2017) constructed a wholesome chromosomal knock-in E. coli strain with the optimizing butanol yield of 20 g/L starting from E. coli strain EB243; they knocked out 33 indigenous genes as well as introduced 5 exogenous genes. It has close energy content to that of gasoline and in terms of energy density per volume, it is known to have 25% more energy than ethanol which means it will cover more distance per gallon than ethanol. Like the case with ethanol, butanol has the added advantage of being less corrosive than ethanol, and thus easy to handle. Moreover, it can be mixed and delivered with the existing gasoline infrastructure and can be pumped through pipelines as it is, ethanol, on the other hand, requires a separate delivery system and is usually blended at the “Point of Dispensing,” which is normally a filling station. Biobutanol does not require engine modifications to support it and requires fuel-to-air ratios, similar to both gasoline and ethanol but not ethanol. Furthermore, it is believed to be an environmentally friendly material because it has been identified that during combustion it does not release any toxic compound (Dahman et al., 2019).
Biohydrogen
Hydrogen is a renewable energy source that produces water and energy, it counteracts the social impact arising from the burning of fossil fuels (Poleto et al., 2016). Biohydrogen (H2) is a fuel energy that is generated in bio-logged processes where it is reported that Biohydrogen is one of the clean fuels that can be produced by biosynthesis from special types of biomasses. Knowledge of algal biohydrogen has been increased in the later years as gaseous fuel or for the generation of electricity. It finds application in processes such as bio photolysis whereby water in the algal biomass produces hydrogen and oxygen molecules when exposed to light (Anisha and John, 2014), and photo fermentation where through the use of sunlight hydrogen is produced hydrogen from biomass enriched with organic acids (Sağır and Hallenbeck, 2019). The speculative and economical method of biohydrogen production is by using microbes under a dark or photo fermentation process (Argun and Dao, 2017).
Advancements in triglyceride-based biofuels
It is worth mentioning that the process or/and technology under biofuel production are those of complex nonlinearity. In this respect, a quick and accurate modeling technique can turn out to be important to design and optimization of relevant systems. Furthermore, it is only through the revealing of relationships between input parameters, like reaction conditions, and product yield that the synthesis of biofuel can be done effectively (Jeon et al., 2023). AI has several subfields like speech processing, vision, and robotics but for the biofuel market, the most pertinent areas are machine learning and deep learning. Machine learning, in the form of artificial intelligence, addresses real-world issues by building statistical models out of specialized datasets (Okolie, 2024). Artificial intelligence, associated with machine learning, is also revolutionizing the production of triglyceride-based biofuels associated with the opening up of new ways for process streamlining and efficiency improvement. One of the most famous applications is in the optimization of growth conditions for sources of biofuel, such as algae (Mafat et al., 2024). Advanced algorithms in genomic data produce the most productive strains of algae and other microorganisms, thus aiding in strain selection and genetic engineering. This delineates traits that lead to higher lipid production and quickens the development of efficient biofuel feedstocks (Cosgun et al., 2023).
There are several machine learning algorithms in wide use, including linear regression, decision tree regression, GA, random forest regression, gradient boosting machine, support vector machine regression, K-nearest neighbor regression, and ANN (Zhu et al., 2019). In another study a developed artificial neural network model trained by the Levenberg–Marquardt algorithm with backpropagation learning was used to predict the yield of biofuel in the transesterification process. Blends of jatropha and algae oils were used as input in their study. Their ANN presented a very good R-squared value of 0.9976, showing its good efficiency when compared with the experimental results. Another study presented the development of an artificial neural network model and a linear regression model using the Levenberg-Marquardt learning algorithm for predicting biodiesel yield from soybean oil transesterification (Kumar, 2020; Kumar et al., 2019).
The ANN outperformed LR in this context. The study explored various conditions of the soybean oil-to-biodiesel transesterification process to forecast biodiesel yield (Moradi et al., 2013). The ANN utilized a multilayer feed-forward neural network alongside kinetic models. Results indicated that the ANN model exhibited superior accuracy and clarity compared to the kinetic modeling approach (Li et al., 2023), which used four machine learning models to maximize the biomass waste gasification method, concentrating on product output predictions. The gradient boosting regression model showed high performance, with test R² values of 0.82–0.96, for tar, char, and syngas outputs and compositions. In general, it was observed that under conditions of temperature, greater than 800°C, more hydrogen-rich syngas from biomass with higher than 48 wt.% carbon, less than 0.5 wt.% of nitrogen, and 1–5 wt.% of ash was produced. Under optimized conditions, an up to 62 wt. % in syngas with a hydrogen production of 44.34 mol/kg was achieved. Again, this role that AI would have in making data-driven decisions further underlines its importance in the biofuel sector. By managing and interpreting large data sets generated during the production process, AI helps monitor system performance, forecast maintenance needs, and optimize resource allocation (Cosgun et al., 2023). As research continues to evolve, these technologies are expected to play an increasingly critical role in the biofuel industry, driving innovation and enhancing the sustainability of biofuel production.
Applications of biofuels
Biofuels have varying uses across sectors in building the sustainability of energy and reducing dependence on fossil fuels. Moreover, biofuels may be used to generate power, as transportation fuel, for heating, and powering new technologies. Biofuels are used in biomass-based power plants to convert organic matter, such as agricultural waste, forestry residues, or energy crops, into electricity. These plants have realized high-efficiency rates because technological improvements increased conversion rates while simultaneously lowering emissions. Biofuels can be used in the electrical grid to help power producers boost the renewable energy share in their production mix, thus achieving a cleaner mix of energy (Nedayali and Shirneshan, 2016; Rouhany and Montgomery, 2019).
Biogas is generated as the final product from the anaerobic digestion of organic waste materials. Principally rich in methane, it can be used to run combined heat and power plants, providing a renewable energy supply for both domestic and industrial needs (Awogbemi et al., 2021a, 2021b). Bioelectrochemical Systems (BES) represent an emerging technology where bacteria catalyze electrochemical processes at the anode and/or cathode to form biofuels (Chong et al., 2023). These systems have the ability for biosensing, treatment of wastewater, and restoration of groundwater—all versatile tools for environmental management (Srivastava et al., 2020). Biofuels in the form of biodiesel and bioethanol find huge applications in the transport sector, improving vehicle performance while reducing emissions (Su et al., 2019). Biodiesel, being a blend of fatty acid methyl esters (FAME), could be used in almost all diesel engines without modification, and it can lower SOx emissions drastically as well as particulate matter emissions (Callegari et al., 2018; Molognoni et al., 2018), an alcohol-based fuel, enhances gasoline by improving combustion efficiency and reducing carbon dioxide emissions (Cecconet et al., 2018; Pous et al., 2017).
The aviation industry continues to achieve huge strides in reaching out to biofuel as a substitute for conventional jet fuel, with an increase in the consumption of aviation fuel by 13% by the year 2050, biofuels can contribute to alleviating the environmental impacts and decreasing dependency on petroleum-based fuels (Chong et al., 2023). Solid biofuels, especially wood and agricultural residues, are mainly used for heating. In addition, huge amounts of such fuels are available and they are renewable; hence, they provide sustainable heat production both for residential and industrial applications. The techniques mainly applied are gasification and dry biomass combustion to produce heat energy efficiently (Argun and Dao, 2017). Processed forms of solid biofuels, such as pellets and briquettes, are used for heating in domestic stoves and industrial boilers. They exhibit a higher energy density and more consistent combustion compared to raw biomass (Chong et al., 2023). Biohydrogen generation is gaining increasing interest because of its huge potential to be used as a clean fuel. Hydrogen generation can occur biologically through either bio-photolysis or photo-fermentation by microorganisms, evolving hydrogen from water and organic material in the presence of light energy. This hydrogen can be used in fuel cells for the generation of power and heat with water as the only byproduct (Poleto et al., 2016). Biofuels provide a versatile and sustainable energy solution across various applications, from power generation and transportation to heating and emerging technologies. With technology development continuously improving efficiency and eventually reducing prices, biofuels can assume very significant proportions in the global energy transition toward cleaner and more sustainable sources.
The US biodiesel industry, utilizing feedstocks like soybean oil and waste oils offers an energy return on energy invested EROEI of between 3:1 and 4:1 and as much as 85% fewer emissions than petroleum diesel. The industry supports over 60,000 jobs, primarily in rural areas, and has helped stabilize agricultural markets by providing an additional revenue stream for farmers (NREL, 2019). In contrast, the advanced biofuels in Europe, such as hydrotreated vegetable oil (HVO), have a high production cost, but the savings on emissions are well above the target. It also supports the concept of a circular economy in the sense that agricultural residues and waste fats are utilized in its production. India is planning to enhance ethanol blending with petrol by up to 20% by 2025 as a means of reducing dependence on oil imports and stabilization of farmer incomes (Ministry of Petroleum and Natural Gas, Government of India, 2018). On the other hand, a lot of water is utilized in the production of sugarcane. LanzaTech, for instance, converts industrial waste gases into sustainable aviation fuel. This method can result in as much as 70% less emission than jet fuel, therefore potentially becoming a means through which biofuels may be incorporated into reducing the carbon footprints of industry.
Future perspectives
The future looks promising for biofuels since they can play a very important role in ensuring energy security, mitigating greenhouse gas emissions, and tackling climate change. Major areas that have to be tackled to overcome current challenges and fully exploit the potential of biofuels exist. Only inedible biomass feedstock can support the sustainable production of biofuels, such as lignocellulosic biomass and several organic waste products. Research and development activities are directed toward enhancing the efficiency of such feedstock through advanced pretreatment, enzymatic hydrolysis, and fermentation methods. Biofuels have enormous potential for GHG mitigation as compared to fossil fuels. For example, biodiesel reduces the emission of unburned hydrocarbons, carbon monoxide, and carbon dioxide. Further research will be required to overcome NOx emissions problems by certain biodiesels, such as those produced from microalgae and palm oil. Technologies are underway to perfect the means of increasing the yield of biofuel from feedstock while reducing the overall cost. This involves the development of genetically engineered microorganisms and enzymes that can convert biomass into biofuels. Metabolic engineering and synthetic biology are also investigated in enhancing lipid content, improving biofuel yields, and ensuring economic viability in the species of algae and other microorganisms. An emerging application in this respect is the development of integrated biorefineries, where one facility is capable of processing different types of biomasses to produce a diversity of biofuels and co-products. Byproducts will be put to effective use, hence reducing waste and increasing the efficiency of these biorefineries. For example, residues from algal biomass can be used for biogas generation to economically make the production of biofuel more viable. There is a bright future for biofuels that has enormous potential to contribute towards sustainable energy systems. Most importantly, how to overcome current challenges is linked to technological developments, supportive policies, and international cooperation. The top three strategies for the growth of the biofuel industry and sustainable contribution to mitigating climate change would be those including a focus on advanced feedstock, integrated biorefineries, and new waste-to-energy solutions that offer innovative means of production of biofuels. Biofuels will substantially contribute to global energy consumption, especially when research is continued in searching for next-generation biofuels and applications in different sectors.
Conclusion
This review identifies that there is enormous work regarding the development and application of biofuels in many sectors. Starting from power generation to transport, biofuels offer an alternative to fossil fuels to sustain the environment, develop the countryside, and secure energy. The prospect, therefore, remains that further technological advances and policy support from governments will help unlock even greater potential for biofuels through cooperative efforts at the international level. More importantly, biofuel can contribute considerably to solving global challenges of climate change and energy transition through continuous research in advanced feedstock, improved conversion efficiency, and integrated bio-refinery concepts. Soon, their applications will grow further towards new frontiers: aviation, maritime transport, and next-generation bio-fuel solutions. Moreover, the combination of AI and ML with circular economy models manifests their key role in the development of sustainable and resource-effective systems. In other words, there exists huge potential for the future of biofuels that serve as a pathway toward a much more sustainable and resilient future of energy. Innovation, collaboration, and strategic investments continued in this sector will make biofuels one of the cornerstones in the transformation toward a greener, more sustainable world.
Footnotes
Acknowledgments
This work is carried out with the help of prestigious material from the institute's libraries.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that they have no competing interests. We assure the integrity and quality of our research work. It is also stated that there is no plagiarism in this work and all points taken from other authors are well cited in the text. This study is completely independent and impartial.
Ethical approval
Our study did not require ethical board approval because it did not contain human or animal trials. This research did not involve human participants and/or animals.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
