Abstract
This study was conducted to address the rock burst problem caused by serious stress concentrations in coal seams. Based on the principle of inducing agent corrosion affecting coal components to soften coal, different inducers were selected to study the desensitization effect of acid leaching induction on coal components. The microscopic behavior characteristics of coal under acid leaching were analyzed, and the qualitative and quantitative laws were obtained for different acid leaching times and microscopic behavior characteristics. The results show the following: (1) The mixed acid solution as an inducer has the effect of immersion, dissolution, and fracture on coal. During the reaction process, the internal pores of the coal expand, and the height of the external liquid decreases. The mixed acid solution had the best effect in terms of the removal of minerals in coal. (2) Under microscopic conditions, the internal crack width of the coal body expanded several times after mixed acid soaking; the difference in the unevenness before and after compression fracture was stable at 25–27%, and the mixed acid expanded the gap in the coal. (3) This practical study shows that the characterization changes of the mixed acid solution, injected into the borehole, combines the characteristics of the citric acid and phosphoric acid solution. The minerals around the borehole are dissolved, and the debris fuses with the solution. The liquid is viscous and forms a film on the surface of the solution. The mixed acid has a more obvious effect on the immersion and dissolution of coal.
Keywords
Introduction
China is currently the country with the largest coal mining depth in the world at 1500 m and has more than 40 pairs of ultra-kilometer mines. The geological conditions of coal seams are complex, with fault structures and steeply inclined coal seams being common. The consequent rock burst problem is complex and of varying nature, and the prevention and control of rock bursts in deep mines is a problem that urgently requires a solution.
In recent years, many scholars have carried out studies on softening the coal body to relieve pressure, so as to reduce the impact of rock bursts in deep mines (Ni et al., 2019; Guerue et al.,2008; Guo et al., 2021; Xu et al., 2021). Deep coal seams usually have obvious characteristics of high pressure, strong adsorption and low permeability (Zhou et al., 2016). Shi (2017) applied coal seam water injection to mines affected by strong mine pressure, which could reduce the softening coefficient of coal and prevent damage from the strong mine pressure. Li et al. (2021) used reasonable parameters for water injection and pressure relief of the coal mining face, which can reduce the occurrence of large energy vibration events, reduce impact risk, and ensure mining safety. Kang and Feng (2017), Cao (2020), and Lin et al. (2020) found that hydraulic fracturing had remarkable effects for hard roof weakening and high stress roadway surrounding rock pressure relief. Zhang (2021) conducted pre-hydraulic fracturing on the key roof strata of the upper layer of the track transportation roadway in the east wing of Qinan Coal Mine, which not only protected the roadway, but also increased the amount of resource recovery. Lu et al. (2017) used viscoelastic surfactant fracturing fluid and water treatment to increase the permeability of coal samples by nearly 178%. Qi and Geng (2012) noted that water has no softening effect on coal, but rather fractures, loosens, and damages its internal structure. He proposed a softening agent to soften coal, which can addressed the problems of hard coal mining, dust removal, and gas dilution. Xu et al. (2019) and Lu and Xu (2020a, 2020b) used a chelating agent and surfactant combination made of composite wetting agent for coal seam water injection; its “humidifying pore”‘ effect was remarkable. Nabeel, Khan, and Sharma (2009) showed that under mild conditions, using NaOH and H2SO4, continuous extraction can remove more than 75–80% of the minerals in coal. Liu (2012) conducted a test on the hydraulic properties of coal under the action of fracturing fluid, and they found that the hydraulic fracturing softening of coal and rock mass was conducive to coal mine disaster control. Several studies have shown that coal can be induced to achieve a softening pressure relief effect.
In previous studies, water and liquid softeners were often used to soak and soften coal to assist in hard coal mining. Acid–base continuous extraction (sodium hydroxide–sulfuric acid) has been used to remove minerals and ash from coal. In this study, two kinds of acids (low concentration citric acid and high concentration phosphoric acid) were used to leach coal and remove minerals, effectively increasing the internal pores and fissures of coal, which leads to softening and cracking of the coal, so as to reduce rock bursts in coal mining. Based on the principle of softening and pressure relief of acid leaching on coal, the desensitization effect of acid leaching induction on coal components was studied, and the coal surface was observed. Tests were conducted from the microscopic and macroscopic perspectives using scanning electron microscopy (SEM) and engineering simulations to analyze the desensitization effect of coal components under the influence of chemical induction at room temperature (20°C). The microscopic behavioral characteristics of coal under acid leaching effects were analyzed, and the qualitative and quantitative laws of different acid leaching times and microscopic changes were obtained. This study can provide important engineering guidance for chemical depressurization technology in deep mining.
Experimental study on chemical-induced desensitization of coal components
Working mechanism of chemical inducer
Coal is a heterogeneous, anisotropic, porous medium containing a variety of minerals. The main mineral components in coal are calcite, dolomite, and kaolinite. The content of internal compounds are Al2O3, SiO2, SO3, TiO2, and Fe2O3 (Lu et al., 2020; Lv and Ji 2021; Xu and Lu, 2020). Coal comprises numerous pores and cracks, where water and gas migrate and are stored. Therefore, determining one or more solutions to remove the internal minerals of coal, through pore immersion, can achieve the effect of coal softening and gap expansion, which is conducive to the coal mining process.
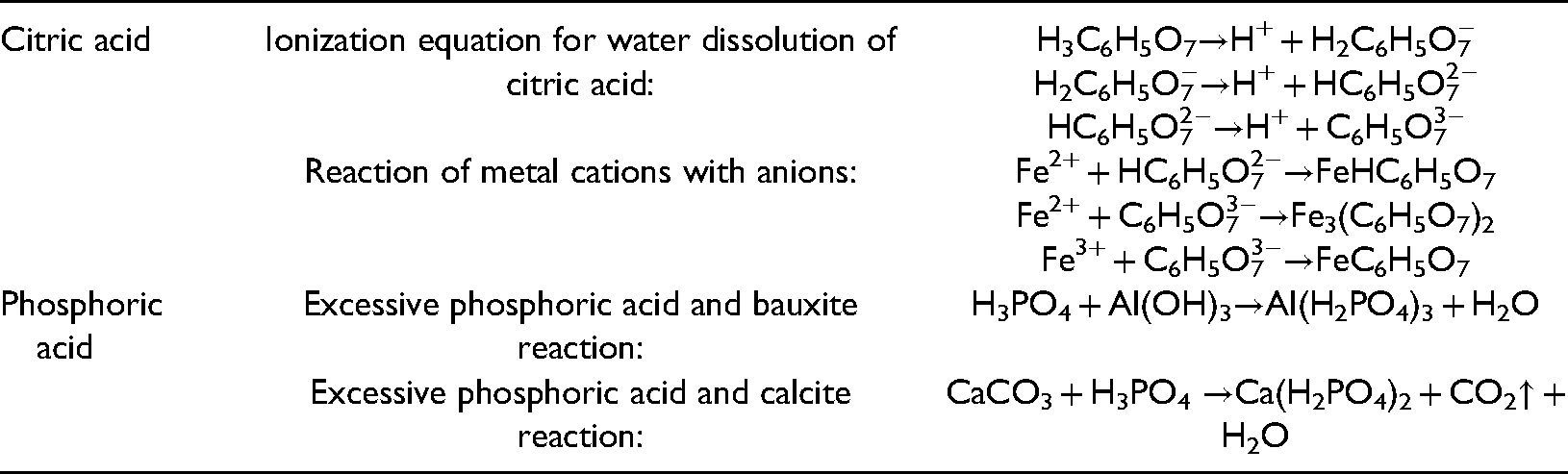
Citric acid is commonly used in industrial desulfurization, with better results than other desulfurizers (Qiao, 2018). Additionally, citric acid reacts with iron ions and dissolves iron compounds in coal, which produces iron citrate. Through the phosphate dissolution method (Guo and Zhong, 1957), it was found that more than 90% of minerals can be dissolved by phosphoric acid. The reaction is shown in Table 1. Phosphoric acid and citric acid do not react with each other, therefore, a mixture of phosphoric acid and citric acid is selected to configure the inducer for the study of the removal effect of coal components.
Chemical equation.
Softening and pressure relief induced by coal chemistry is realized through the migration and complex chemical changes of the inducer in many of the interconnected pores in coal. The inducer is applied to the coal seam. Under pressure, the water first permeates along the pores and fractures of the coal body, and the moisture content of the coal body increases. At the same time, the inducer enters the pores of the coal body, and the decomposition or dissolution of the minerals occurs, breaking the combination of various elements in the minerals, leaching the mineral ions. The number of pores in the coal increases and the pore connectivity improves, creating suitable conditions for the inducer to continue leaching into the coal body. With the increasing contact area between the inducer and the coal body, the mineral components in the coal body are completely removed.
Design of chemically induced desensitization test
The analytically pure citric acid particles and 14.63 mol/L concentrated phosphoric acid solution were selected to configure three inducers for the chemical induction desensitization test at room temperature. Combined with 2.1, three inducers were determined. Due to the relatively low presence of sulfur compounds in coal (Xu and Lu, 2020), low concentration citric acid reagent A was used to remove sulfur related compounds in coal (hereinafter referred to as citric acid); Coal contains different kinds of minerals, more than 90% of which can be dissolved by phosphoric acid (Guo and Zhong, 1957), and the dissolution of most minerals requires excessive phosphoric acid environment. Combined with various factors, high concentration phosphoric acid reagent B was used to remove a variety of minerals in coal (hereinafter referred to as phosphoric acid); Reagent A and B were mixed with a 1:1 volume ratio to form mixed acid reagent C (hereinafter referred to as mixed acid). The composition configuration is presented in Table 1.
The coal samples from a coal mine in the Yinchuan area were selected, as shown in Figure 1. Approximately regular-shaped coal blocks with a volume of roughly 150–200 cm2 were selected for the induction immersion test The coal samples were internal pieces of Yinchuan coal, minimizing the loss of coal quality.

Coal samples from yinchuan.
We soaked the coal blocks with the acid inducers, as specified in Table 2, at room temperature (20 °C), following the principle of single variable testing. The characterization changes during the soaking process were observed. Additionally, the apparent characteristics of the coal after soaking were observed and the SEM electron microscope scanning test was carried out. The effects of the inducer under room temperature induction was analyzed from macroscopic and microscopic perspectives (Table 3).
Reagent components.

Specific configurations of reagent soaking.

Effect analysis of desensitization test
Effect analysis of coal surface characteristics
The prefabricated specimens were soaked with three kinds of acid inducers and observed at 0, 3, 6, 18, and 42 h, respectively. The changes of coal surface over time were observed with a focus on the amount of debris produced by coal decomposition, the intensity of the reaction and the change of the coal surface. Different types of coals were observed to display different changes, and thus representative coals were analyzed.
Figure 2 shows the characterization of coal soaked in pure water with pH = 7 and without impurities in an acid-free environment. In the early stages, fine dust was washed off of the coal blocks, both externally and internally, this was suspended in the liquid, and the liquid was slightly turbid. Pure water entered the pores of the coal body, and the gas in the pores was discharged. There were occasionally small bubbles on the surface of the coal block, which floated up to the liquid's surface. The original coal cuttings at the bottom of the beaker and some of the coal cuttings on the surface of the coal block floated on the liquid's surface. After soaking for 3 h, the liquid showed a turbid state. The dust particles at the bottom of the beaker had increased and the results of pure water soaking became more pronounced. There was a small collection of bubbles attached to the surface of the coal block. Over time, the gas was slowly discharged, and the bubbles increased in size. The buoyancy was less than the water pressure, thus the bubbles could not float to the surface and became attached to the surface of the coal block. In short, the coal block was immersed in pure water and the gas within the pores was discharged, leaving bubbles on the surface of the coal block. Additionally, the soaking in pure water made some particles and dust on the surface of the coal fall off. After 6 h, coal debris on the liquid surface was clearly observed to have increased. A small section of the upper coal block was exposed. After soaking in pure water, the fine debris on the coal surface broke into smaller pieces along the coal block bedding, these particles had a very low density. This fine debris then floated to the surface of the liquid due to buoyancy. At 42 h, it was observed that the upper part of the coal block was clearly exposed. As time increases, the soaking effect of pure water increases

Change of coal soaked in acid-free environment.
Figure 3 shows that characterization of a coal block soaked in 0.5 mol/L citric acid solution as the inducer. In the initial stage of soaking, small bubbles were released continuously, which floated to the surface. Citric acid solution reacted with minerals inside the coal block and the coal body was immersed, but the reaction was not intense, and the solution was still clear. With the increase of soaking time, the coal debris scattered at the bottom of the beaker also increased. At 3 h, the solution became yellow, and the color gradually became a bright yellow. According to the analysis of the mineral composition in the coal body, it can be seen that the citric acid reacted with iron ions in the coal, and the resulting ferric citrate water was bright yellow. After 42 h, rust red minerals were more apparent on the surface of the coal. It can be inferred that after soaking, the citric acid solution chemically reacts with the coal, and the citric acid is immersed into the coal.

Change of coal soaked in citric acid solution.
Figure 4 shows the characterization of coal soaked in 7 mol/L phosphoric acid solution as an inducer. At the initial stage of soaking, a violent reaction occurred, the liquid was turbid, and a large number of bubbles continued to emerge from the surface of the coal body after contact with the acid solution, these quickly rose to the surface, and the reaction gradually weakened after 5 min. After 3 h, with the reaction gradually complete, the gas was discharged slowly. The bubble buoyancy was now less than the liquid pressure and was uniformly attached to the surface of the coal block. At the bottom of the beaker, there were exfoliated coal blocks and dispersed fine coal chips. After the induction of the acid solution, the coal structure was clearly broken. White mineral precipitation could be observed on the surface of the coal body at 6 h. Mineral precipitation increased at 18 h, being densely attached to the surface of the coal body and showing bright surface characteristics. After 42 h, the precipitation had disappeared. Soaking in phosphoric acid led to an intense reaction, accompanied by gas production. Coal blocks and chips fell off and white mineral precipitation formed on the coal. The coal blocks soaked in phosphoric acid showed rapid results.

Change of coal soaked in phosphoric acid solution.
Figure 5 shows the characterization of coal soaked in a mixed acid solution prepared with 0.5 mol/L citric acid solution and 7 mol/L phosphoric acid solution with a 1:1 volume ratio. Initially, there was an intense reaction, and upon surface contact with the mixed acid a reaction occurred, resulting in a large number of bubbles. However, the quantity of bubbles was less than that of the phosphoric acid-soaked coal samples, with a more dense shape. The bubbles quickly rose to the surface of the liquid and the reaction gradually weakened after 5 min. At 3 h, the bubbles produced by the intense reaction did not disappear but stuck to the surface of the liquid, scattered at the edges of the beaker. There was a small distribution of pulverized coal at the bottom of the beaker. The size of the particles was much smaller than that of the phosphoric acid-soaked coal chips. A small number of bubbles was produced after the reaction had weakened, these were attached to the surface of the coal block due to their low buoyancy. After 6 h, the bubbles on the surface of the coal block and at the edge of the beaker gradually disappeared, and at the same time, the liquid level decreased, and the coal block showed became exposed. At 42 h, it could be observed that the coal block was clearly exposed, and the mixed acid reacted violently with the coal block to dissolve some minerals within the coal. The mixed acid solution dissolved the minerals in the coal with the combined action of the citric acid and the phosphoric acid, and a large amount of gas was generated. The number of pores in the coal increased and the pore connectivity became better. The mixed acid entered the coal body, resulting in a decrease in the liquid level of the mixed acid.

Change of coal soaked in mixed acid solution.
Comparing the effects between four kinds of liquid soaked coal blocks, it was found that they reacted differently. There was no chemical reaction after pure water immersion, and only physical changes occurred when the internal coal pores were immersed in pure water. Coal soaking in the citric acid solution did not lead to a strong reaction, but it did react with iron elements in the coal, which achieved the effect of soaking the internal pores of the coal. Compared with the first two solutions, phosphoric acid and mixed acid solutions had much stronger reactions and higher gas production. After soaking in the phosphoric acid solution, coal blocks and coal chips fell off. The phosphoric acid solution can produce fractures in the coal blocks, and phosphoric and citric acids in the mixed solution do not react with each other. On the contrary, the dilution and heat release of the two acid solutions after mixing can improve the reaction. The coal blocks soaked in mixed acid solution combine the immersion, dissolution, and fracture effects of both acids and expand the internal pores of coal blocks. The external liquid immersed the pores and the liquid height decreased significantly. Therefore, the mixed acid solution has the best effect on the demineralizing of coal components.
Analysis of coal microstructure characteristics
There are many methods for studying microscopic characteristics (Chen et al., 2022; Yang et al., 2022a, 2022b; Zhu et al., 2022). In this study, SEM was used to observe the microstructure characteristics of coal. The SEM experimental instrument was a KYKY-2800B scanning electron microscope manufactured by China Science and Technology Instrument Co., Ltd The SEM can observe the phase in a magnification range of 15–250,000 times, with a resolution higher than 4.5 nm. The electronic optics system is a fork type tungsten wire cathode electron gun, and the accelerating voltage range is 0–30 kV. During the test, we opened the instrument into standby state, selected the appropriate coal sample fixed on the tray, and then placed it in the instrument cavity. We then vacuum sealed the cavity. We initialized the instrument, setting the appropriate focal length and selecting the appropriate contrast and gray parameters. From the analysis of the apparent characteristics of coal soaked in section 3.1, it was found that the mixed acid has the best induction effect on coal. According to the test requirements, the coal samples soaked in mixed acid were scanned at a magnification level of 100 and 300 times, and the quantitative changes under the microscope were observed. (Ma et al., 2020; Yang et al., 2019; Ye et al., 2019; Yu, 2014; Zhao et al., 2015)
To quantify the microscopic changes in coal using the SEM image, we introduced unevenness S. We used MATLAB to binarize the SEM image. Owing to the differing flatness of the section, the black and white regions produced by the binary pixel images were different. The black area represents a smooth flat interface, and the white area represents a rough interface, such as faults, cracks, and ravines. S is calculated as the proportion of pixels in the black area, and the roughness of the section is evaluated using the unevenness as follows:
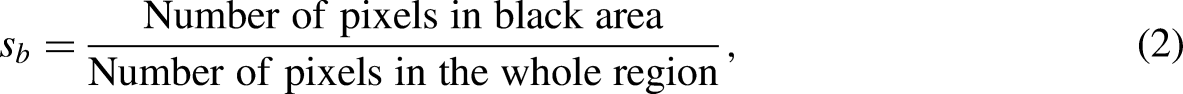
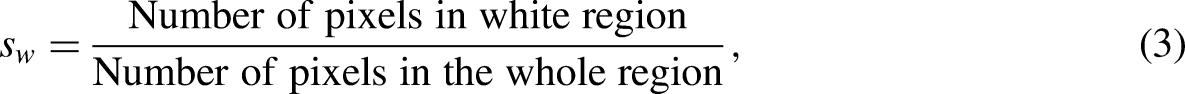
where

Coal 100 × SEM images and binarized images. (a) Raw coal sample SEM image; (b) Binary processing image of raw coal sample; (c)Coal samples with mixed acid immersion SEM image; (d) Binary processing image of coal sample soaked in mixed acid.
Figure 6 shows a big difference between the raw coal sample and the coal sample soaked in mixed acid (Pan et al., 2016). The raw coal sample under 100 times scanning had consistent uniformity and a high integrity. In addition, pores were observed on the surface of the intact coal body but only a few fine cracks were observed. The pore morphology was diverse and irregular, with varying sizes and uneven distribution. The pore sizes ranged from 10–40 μm. There were many small fractures, with a length distribution of 100–800 μm. These pores were primary pores without external influence, which were the weak links for acid leaching, and provided a migration route for the acid within the coal block. Under 100 times scanning, the coal sample soaked in mixed acid changed compared to the raw coal sample, with the fine cracks becoming penetrating cracks, and the crack widths expanded several times. The coal body showed microscopic fractures after mixed acid soaking. Microscopically, granular clastic minerals attached to the coal surface could be observed. The coal body was immersed in the mixed acid solution to dissolve the minerals within the coal body. With the dissolution of minerals in the pores, the number of pores increased and the connectivity between them improved. The mixed acid expanded the size of the pores/cracks of the coal body.
The smooth part of the coal surface can be observed more clearly under 300 times scanning, with Figure 7 showing that many cracks and holes are distributed on the surface of the coal body, like those of the 100 times scanning image, and some stripe-like white substances were also distributed on the surface of the coal body. These white substances are the result of acid solution etching on the weaker surface sections of the coal block. The observation of coal samples under 300 times magnification after soaking in mixed acid showed that the microscopic fracture effect was more prominent. In addition to the particle adhesion and cracks observed under 100 times magnification, there were also holes distributed over the coal body, that is, the mixed acid soaking destroyed the integrity of coal body. The fine grained minerals on the surface of the coal body reacted with the acid, which affected the uniformity and integrity of the coal and made the surface uneven. Additionally, the clastic mineral particles were attached to the coal body. The appearance of small holes indicated that under the action of mixed acid, the internal pores of coal samples increased in number and the pore connectivity improved. The mixed acid also had an impact on the composition of the coal samples.

Coal 300 × SEM images and binarized images. (a) Raw coal sample SEM image (b) Binary processing image of raw coal sample; (c) Coal samples with mixed acid immersion SEM image (d) Binary processing image of coal sample soaked in mixed acid.
The SEM observation of the apparent characteristics of coal showed that the coal body changed from a smooth state to a broken state, and the mixed acid had a microscopic fracture effect on the coal body. The unevenness of coal in different soaking environments is shown in Table 4.
Unevenness of coal in different immersion environments.

Table 4 and Figure 8 show that the unevenness of coal samples before and after soaking changed significantly, and the percentage difference between the final and initial unevenness of coal samples was stable at 25%–27%. Additionally, the SEM image observations showed that fractures occur in the coal, with an uneven concave and convex shape. The soaking of mixed acid led to changes in the microstructure of the coal, leading to an ideal and stable end result.
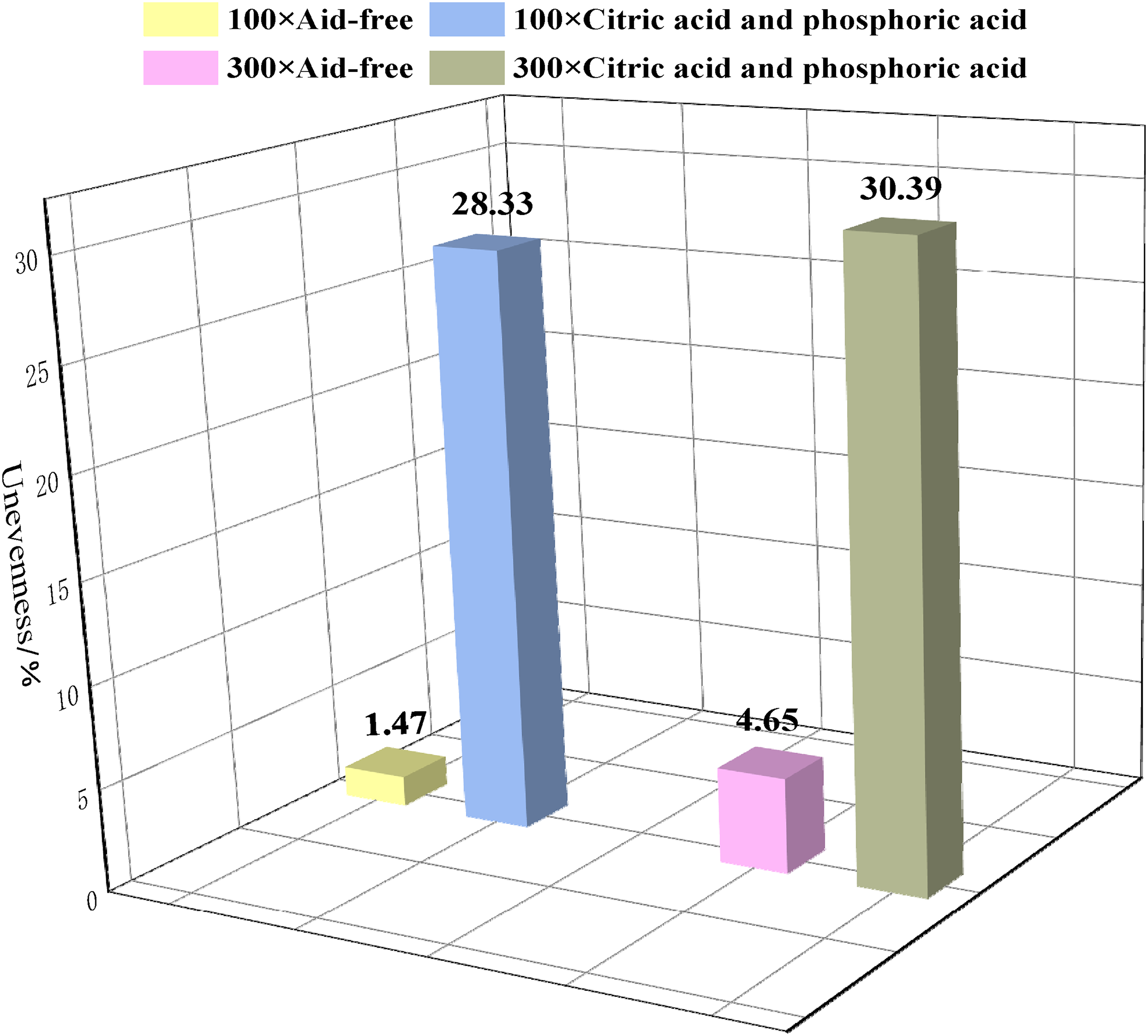
Comparison of unevenness of coal samples in acid-free and mixed-acid environments.
Engineering site verification
Small-scale tests found that different acid solutions had different effects on coal components. To confirm that the acid solutions also had the effects of immersion, dissolution and fracture of coal on the engineering site, the engineering site simulation test was carried out. We select the coal block mold to have a diameter of 50–80 cm. Using a rapid solidification concrete injection mold to fix the coal block, we then proceeded with drilling(drilling diameter of 42 mm), as shown in Figure 8. The borehole was grouted to observe the changes in the borehole over time. Borehole 1# was injected with 0.5 mol/L citric acid solution, borehole 2# was injected with 7 mol/L phosphoric acid solution, and borehole 3# was injected with the phosphoric acid, citric acid mixed solution with a volume ratio of 1:1.
Figure 9 shows that the coal gloss at the bottom of borehole 1# is good, and the ring-shaped traces left by the bit are obvious. Owing to the cracks in the coal body, the acid liquid was slowly lost after grouting. After the grouting had been standing for 6 h, the coal body at the bottom of the borehole lost its luster, and the coal surface showed a sand-grinding form. The circular traces left by the drill had eroded. After 12 h, there was no significant change and the bottom of the borehole was smooth. A chemical reaction then occurred with the iron compounds (FeO, Fe2O3) within the coal, and a liquid film formed after the fusion of the acid liquid and the coal. The citric acid solution injected into the borehole had a clear result in the removal of coal ash inside the borehole. Next, the phosphoric acid solution was injected into the borehole 2#, with a strong reaction. At 6 h, the coal body at the bottom of the borehole had eroded, and the circular coal produced by the drill had disappeared, along with the gloss. The quick-setting concrete used to hold the coal block in place had been exposed, and the acid solution remained unchanged after grouting and soaking. In other words, the phosphoric acid soaking reacted with the minerals in the coal and lead to significant erosion within 6 h. After the fusion of the acid solution and the coal, the stratification was clear and the coal debris had precipitated at the bottom.

Engineering site drilling.
When the mixed acid solution was injected into borehole 3 #, the erosion at the bottom of the borehole could be observed at 6 h. When grouting was continued until 12 h, the mixed acid solution dissolved the minerals in the borehole, and the debris and solution fused with each other. Unlike the phosphoric acid solution injection drilling, the mixed acid liquid is viscous and creates a film on the surface. The change characterization of mixed acids combines the characteristics of citric acid and phosphoric acid solution etching. Compared with boreholes 1 # and 2 #, the solution in borehole 3 # was more viscous, that is, the mixed acid was more thorough for the immersion, dissolution, and fracture of coal debris. Further, the coal debris shedding effect was the best Overall, the effect of mixed acid on coal leaching and dissolution was more obvious (Figure 10).

Drilling grouting changes. (1) 1# borehole injection of citric acid over 18 h; (2) 2# borehole injection of phosphoric acid over 18 h; (3) 3# borehole injection of mixed acid over 18 h.
The engineering field simulation tests show that acid leaching influences drilling. Compared with the existing technology, the drilling and high-pressure grouting technology can be optimized on the engineering site, which is simple to operate and easy to implement. High-pressure injection of chemical reagents softens the coal under the premise of determining the number of pressure-relief boreholes, enhances the pressure-relief effect, and ensures the stability of the surrounding rock. High pressure slurry injection breaks the weak parts of the coal body, resulting in new cracks, which not only ensures the full reaction of the chemical reagent and the coal, but also ensures that the chemical reagent can dissolve some substances within the coal, as well as loosening the coal, further increasing the efficiency of pressure relief. Chemical reagents inhibit coal dust, improve the working environment of coal mine workers, and reduce the threat of a coal dust explosion. Additionally, the mixture of citric acid and phosphoric acid as a chemical inducer has a significant effect on the coal component, and this study can provide a basis for the engineering site application.
Conclusion
In this paper, the desensitization effect of acid leaching induction on coal components is studied, and the microscopic behavioral characteristics of coal under acid leaching are analyzed. The qualitative laws of different acid leaching times and microscopic behavioral characteristics are obtained. The conclusions are as follows:
Under the apparent conditions, the pure water leaching of coal only induces a physical reaction, whereas acid leaching coal induces a chemical reaction. Among the acidic leaching solutions, the mixed solution of citric and phosphoric acids as the inducer portrayed the effects of immersion, dissolution, and fracturing on coal. Through the reaction process, the internal pores of the coal expands and is immersed in the liquid. This is observed through a significant drop in the liquid surrounding the soaked coal. The mixed acid solution has the best effect on the removal of mineral components in coal. Under the microscopic conditions, the fine cracks in the coal, after mixed acid soaking, develop into penetrating cracks, and the crack widths expands several times. The mixed acid has a microscopic fracture effect on the coal. Microscopic observation of granular clastic minerals attached to the surface of the coal also occurred, showing an uneven shape. The change in evenness stabilized at 25%–27%. The mixed acid solution in the coal dissolved the minerals within the coal body. With the dissolution of minerals in the pores, the number of pores increased, which improved the connectivity between them. Additionally, the size of the pores also increased. The engineering field-study, on the effects of the three kinds of acid solutions, shows that the characterization changes of the mixed acid solution injected into the borehole combines the characteristics of the citric acid solution and the phosphoric acid solution. The minerals around the borehole are dissolved and the debris fuses with the solution. The resulting liquid is viscous and the surface of the solution has a filmed layer. The mixed acid has a more obvious effect on the immersion and dissolution of coal. In this work, Yinchuan coal was used to analyze the effects of an inducer, with good component desensitization effects being obtained, which shows significant viability for application. Different coal mines would still need to adjust the concentration of the inducer to achieve the best desensitization effect.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fundamental Research Funds for the Central Universities, National Key R&D Program of China, (grant number FRF-TP-19-009B1, 2021YFC3001301).
