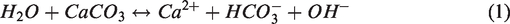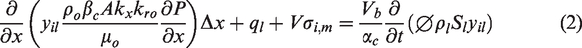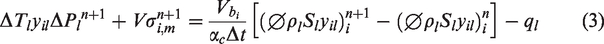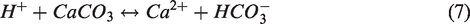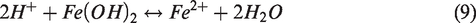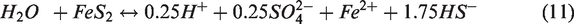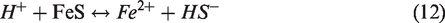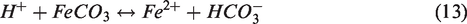Abstract
The low-salinity waterflooding is an attractive eco-friendly producing method, recently, for carbonate reservoirs. When ferrous ion is present in the formation water, that is, acidic water, the injection of low-salinity water generally with neutral pH can yield precipitation or dissolution of Fe-minerals by pH mixing effect. FeSO4 and pyrite can be precipitated and re-dissolved, or vice versa, while siderite and Fe(OH)2 are insoluble which are precipitated, causing permeability reduction. Particularly, pyrite chemically reacts with low-salinity water and release sulfate ion, altering the wettability, favorably, to water-wet. In this aspect, we analyzed oil production focusing on dissolution of Fe-minerals and Fe-precipitation using a commercial compositional reservoir simulator. From the simulation results, the quantities of precipitation and dissolution were enormously large regardless of the type of Fe-minerals and there was almost no difference in terms of total volume in this system. However, among Fe-minerals, Fe(OH)2 precipitation and pyrite dissolution were noticeably large compared to troilite, FeSO4, and siderite. Therefore, it is essential to analyze precipitation or dissolution for each Fe-mineral, individually. Meanwhile, in dissolving process of pyrite, sulfate ions were released differently depending on the content of pyrite. Here, the magnitude of the generated sulfate ion was limited at certain level of pyrite content. Thus, it is necessary to pay attention for determining the concentration of sulfate ion in designing the composition of injection water. Ultimately, in the investigation of the efficiency of oil production, it was found that the oil production was enhanced due to an additional sulfate ion generated from FeS2 dissolution.
Introduction
Low-salinity waterflooding (LSWF) is a simple and environmentally friendly enhanced oil recovery (EOR) method, which can improve the oil recovery by lowering the salinity of injected brine or by adjusting the ion concentration. The recovery of oil from carbonates is below 30% since more than 80% of carbonate reservoirs possess intermediate or oil wetness (Mahmoud and Nasr-El-Din, 2014; Yue and Wang, 2015; Zhou and Yang, 2017). Through various experimental studies, it has been verified that the injection of low-salinity water (LSW) to an oil-wet carbonate reservoir has been able to alter its wettability to water-wet (Morrow et al., 1998; Standnes and Austad, 2000; Yassin et al., 2015). Besides, the salinity of injection water and the concentration of potential determining ions (PDI) such as sulfate ion (SO42–), Ca2+, and Mg2+ have been reported to be major factors leading to a wettability alteration (Al-Shalabi et al., 2014; Jalilian et al., 2017; Salamat et al., 2016; Yousef et al., 2011; Zhang et al., 2007). Lager et al. (2007), McGuire et al. (2005), and Wolthers et al. (2008) confirmed that LSW injection caused the dissolution of calcite and an increase in pH due to OH– ions generated from the dissolution as follows
In other words, an increase in pH by 1 to 3 was observed as calcite within the core became dissolved through the LSWF experiments. Since there was only one equilibrium point among the oil, brine, and rock, at a specific pH, the change in pH shifted the chemical equilibrium, which could result in precipitation (Mahani et al., 2015). Zhang and Sarma (2012) demonstrated the precipitation of CaSO4 in pores, increasing the pressure difference during the LSW injection.
When ferrous ion (Fe2+) ion is contained in formation water or injection water, Fe2+ ion as well as Ca2+ and Mg2+ can also affect the alteration in wettability. Haugen (2016) experimentally confirmed that Fe2+ in brines had been oxidized to Fe3+, and it had a greater influence on wettability than Ca2+ cation. Also, Fjelde et al. (2017) performed a laboratory experiment to investigate the effect of Fe2+ oxidation and cation bridging by Fe3+ on wettability, and they observed that low concentration of Fe3+ increased the oil wetness of rock surface and altered the wettability to less water-wet. Also, Fe2+ ion can react with anions such as SO42–, HCO3–, and OH–, yielding precipitates of FeSO4, siderite (FeCO3), and Fe(OH)2, respectively. Al-Saiari et al. (2008) measured the quantity of FeCO3 precipitation when the Fe2+ to Ca2+ molar ratio in the FW was varied and identified that 60% of the Fe2+ precipitated. Particularly among the Fe-precipitations, the low solubility of FeCO3 and Fe(OH)2 could directly affect the permeability reduction (Stumm and Lee, 1961).
Fe-minerals are present deep underground; for instance, pyrite (FeS2), which is one of the most common sulfides in sedimentary rocks, can often be found in carbonates that are rich in organic materials (Clavier et al., 1976). When Fe-minerals such as FeS2, troilite (FeS), and FeCO3 exist in carbonates, the increase in pH changes during LSW injection can generally lead to the dissolution of Fe-minerals (Hanor, 1994; Liu and Millero, 2002; Rickard and Luther, 2007).
In the case of the acidic reservoir with a low pH condition, FeS2 can be easily dissolved as mentioned in the following research works. Nicol et al. (2013) performed a laboratory experiment on the dissolution of FeS2 and confirmed that FeS2 dissolved to a greater extent in a solution with a low concentration of SO42– such as formation water. Wolfe et al. (2016) observed through a FeS2 dissolution experiment that FeS2 dissolved into iron and sulfur within a solution at pH 3; iron predominantly existed in the form of Fe2+ ions and sulfur existed as SO42– ions. According to these experimental results, the permeability change could have been caused by the dissolution of FeS2 when it was present in carbonates. As well, EOR effects could be observed due to the generation of SO42– ions via the FeS2 dissolution.
Previous studies have focused on the optimum ion composition leading to the maximum oil recovery from a carbonate reservoir by modifying the concentrations of SO42–, Ca2+, and Mg2+ in LSW. It has been suggested that increasing the concentration of SO42– rather than Ca2+ and Mg2+ was more effective toward improving the recovery of oil (Fathi et al., 2010; Shariatpanahi et al., 2011; Sohal et al., 2016; Strand et al., 2006; Zhang et al., 2006). However, there has not yet been an analytical study with regard to EOR effects in consideration of precipitation and dissolution due to geochemical reactions when LSWF was applied to the carbonate reservoir containing a large quantity of Fe2+ ions in the FW and Fe-minerals in carbonates. Additionally, Fe-minerals have different solubilities depending on the type of mineral; some are insoluble, and thus, the permeability change could have occurred due to the reactions with LSW.
In this study, we analyzed the dissolution and precipitation of Fe-minerals resulting from geochemical reactions during the injection of LSW in the presence of Fe-minerals in carbonates. Also, Fe-mineral dissolution generated SO42– and these ions promoted Fe-precipitation. Since this could lead to a change in permeability, in addition to EOR effects of the LSW injection, there may have been additional effects on improving the oil recovery. Therefore, we intended to explain the relationships between reservoir temperature, SO42– concentration, and oil recovery by analyzing the change in dissolution and precipitation of Fe-minerals depending on the temperature and Fe-mineral content within carbonates.
Governing equation
Darcy’s law governs the flow of fluids in porous media. Multiple components within the aqueous phase can also be attributed to the behavior of fluids, such as dispersion and diffusion. The general partial differential flow equation for the multi-component in the fluids and mineral species is as follows
The material balance equations for multi-component fluid flow have been provided in an adaptive-implicit manner by Collins et al. (1992) and Siu et al. (1989). Nghiem et al. (2011) have also introduced aqueous phase behavior and chemical reactions within compositional simulations. The material balance finite difference equation for the multiple components and species is as follows
In this equation, ql is the source/sink term and Vσi,m is the reaction term for rate-dependent reactions. Symbols αc and βc are the unit conversion factors, and other symbols are described in Appendix.
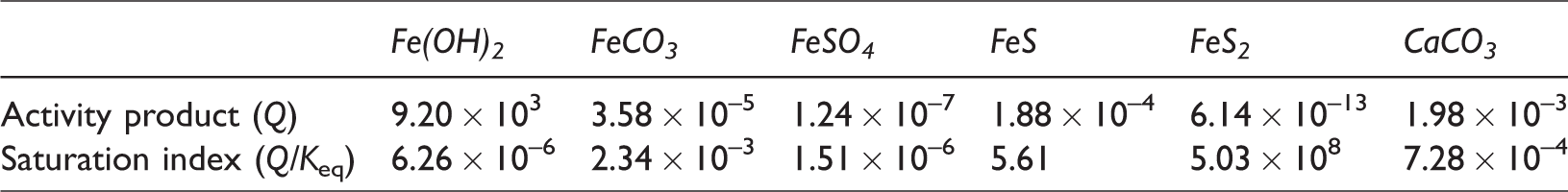
The reaction term (Vσi,m) of i, m in equations corresponds to the rate of precipitation and dissolution as follows
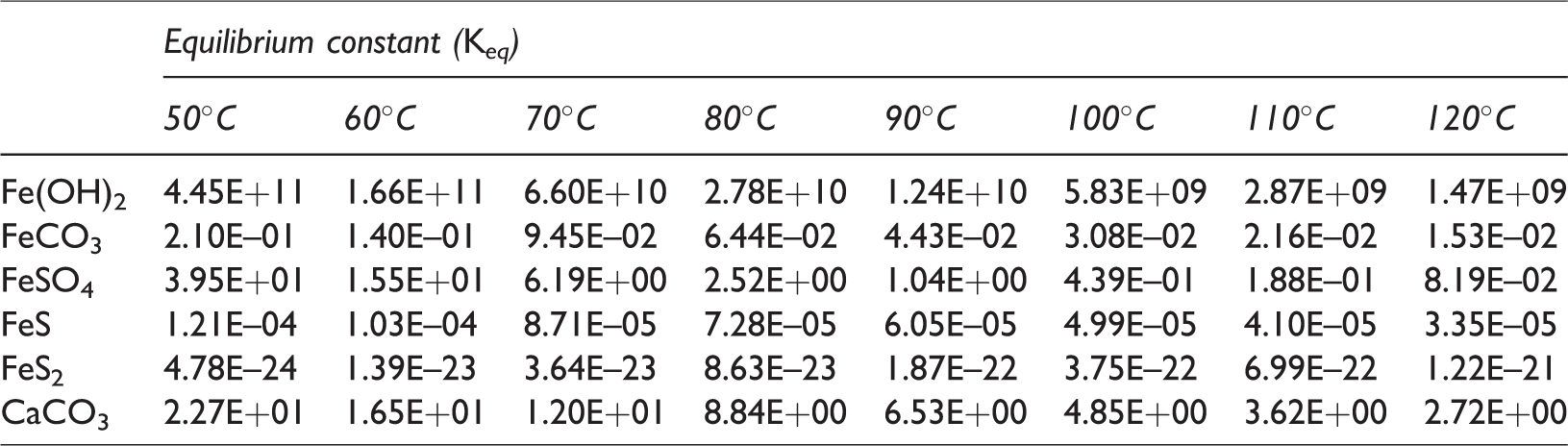
If the reaction rate, ri, is positive ((Qi/Keq,i) < 1), mineral precipitation occurs; if the rate is negative ((Qi/Keq,i) > 1), mineral dissolution occurs, where Ai is the reactive surface area and ki is the rate constant. In this simulation, the initial values of Ai and ki were set as 100 m2/m3 (A0i) and 1.58 × 10−9 mol/m2s (k0i). These values were initially set as the same for six different minerals. As chemical reactions process into the equilibrium state, the values of Ai and ki for different minerals were calculated by the equations shown in Table 1. In the case of ki, since initial value of ki (k0i) is extremely small, the calculated values were almost same for different minerals as listed in Table 1. And also, Qi is the activity product of mineral reaction i, Keq,i is the chemical equilibrium constant for mineral reaction i, and ai denotes the activity of component i. A modified Debye–Hückel model is preferred to calculate the activity determined by the molality, temperature, and ionic strength (Mistry and Lienhard, 2013). The chemical equilibrium constant Keq,i represents the temperature-dependent constant, and tables of the Keq,i values are provided by Delany and Lundeen (1991) and Kharaka et al. (1988). In equation (5), chemical reaction is progressed until the chemical equilibrium, in which Qi is equal to Keq,i (Ma et al., 2015). In this study, chemical reactions for calcite and Fe-minerals with low-salinity injection water containing SO42– are applied and the involved chemical reaction equations are as follows
The reactive surface are (Ai) and the rate constant (ki) after two pore volumes injected at location Ⓐ .

In the above equations, reaction to the right direction denotes dissolution and precipitation for left direction.
Equations of Ai
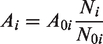
where the reactive surface area at time zero, the mole of mineral per unit grid block bulk volume at current time, and the mole of mineral per unit grid block bulk volume at time zero.
Equations of ki
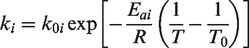
where the rate constant at time zero, the activation energy, the gas constant (8.314 J/mol K), the current temperature, and reference temperature.
In this study, we used a commercial compositional reservoir simulator GEM of Computer Modeling Group Ltd. for compositional and chemical modelling. In this simulator for describing LSWF, the geochemical reacting mechanisms are selectively considered. Meanwhile, all mechanisms are involved simultaneously in the case of experiment. In this aspect, the simulational work is limited comparing to experimental work in describing the geochemical reactions. The flow equations are discretized using the adaptive-implicit method. The adaptive implicit selects a block’s implicitness dynamically during the computation and is useful complex reservoir. The Jacobian matrix is solved by Incomplete LU factorization followed by the generalized minimal residual method iterative method.
Reservoir model
The numerical one-dimensional simulation model was created with 51 grid blocks of the same width and thickness having a length of 1400 ft. As shown in Figure 1, injection well and production well were set to be at both ends of model. The model was carbonate rock composed of 90% calcite. Its pore volume was 605,895 ft3, the initial oil saturation was 88%, and the original oil in place was 70,114 bbl. The porosity and absolute permeability were 0.27 and 30 md. The initial pressure and temperature were set to 3810 psia and 120°C under isothermal conditions. Since the relative permeability is the most critical factor on oil recovery, which has the greatest uncertainty, we applied the relative permeability from the experiments of Yousef et al. (2011) to this study by performing the history matches with the SO42– concentration (Beretta and Gyftopoulos, 2015; Yousef et al., 2011). Figure 2 illustrates the relative permeability curves during the initial and shifted states after wettability alteration by the LSW injection. These curves would be shifted depending on the concentration of SO42– ions in aqueous, which play a major role in detaching oil and altering the wettability. In the production well, the operating pressure was specified at 1500 psia as boundary condition of right-end of the system. In the injection well, the injection rate of 70 bbl/day was specified as boundary condition at the left-end of the system. When the injection pressure was over the upper limit of 4000 psi, this pressure was set as boundary condition. The LSW was injected at the start of production and the injection rate was set to 1–2 ft/day so that the chemical reaction could occur sufficiently. The period of injection and production was 2 years for which two pore volumes were injected.
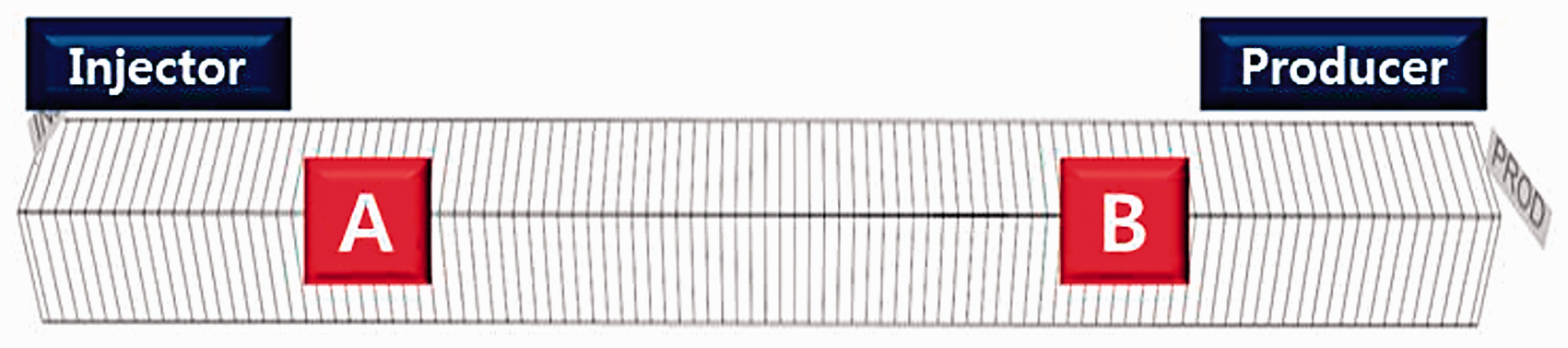
One-dimensional carbonate reservoir system.
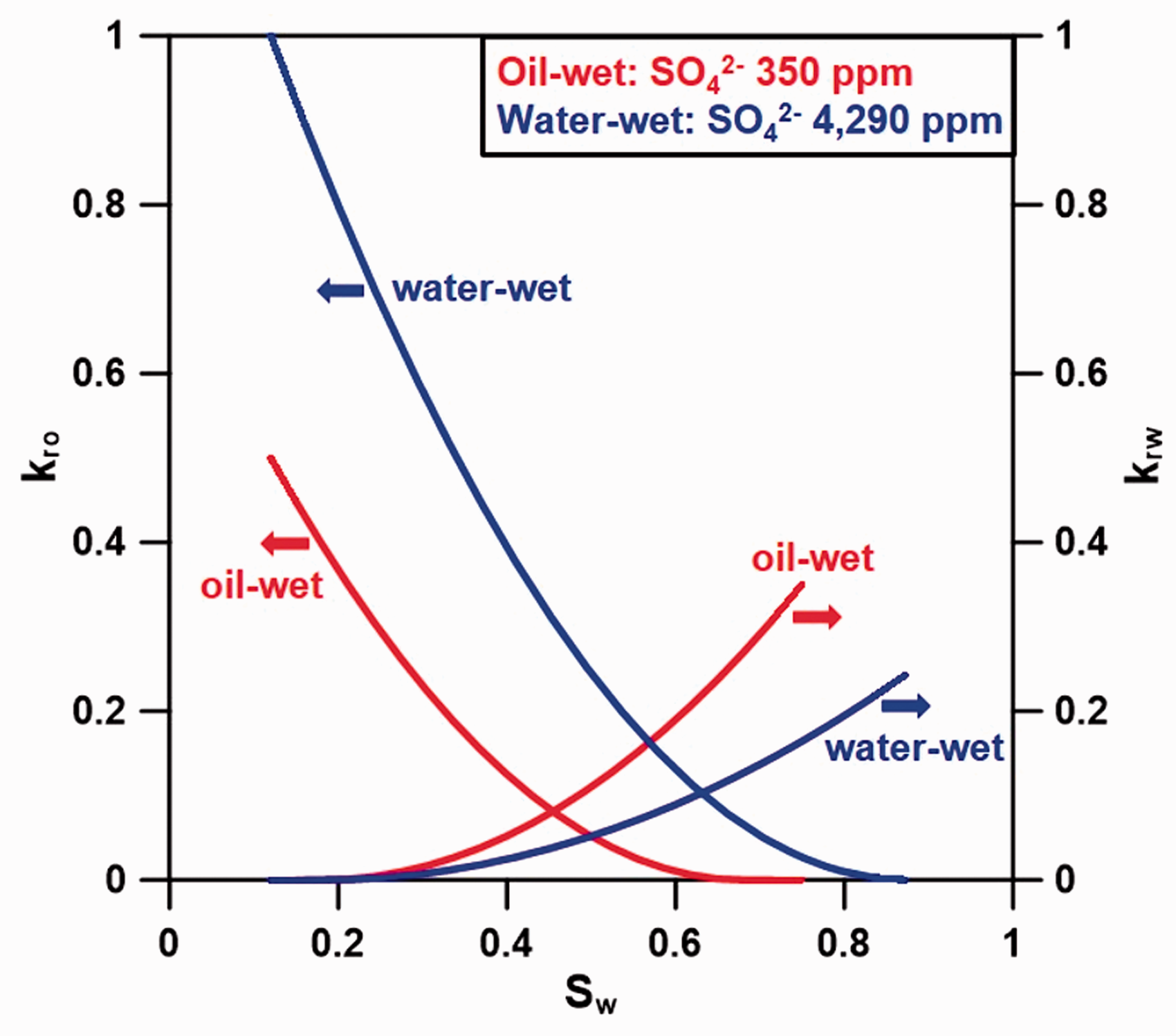
Relative permeability curves used in the simulation when the wettability changes from oil-wet to water-wet (Yousef et al., 2011).
In this paper, in order to construct the simulation model for the application of LSWF to the carbonate reservoir containing Fe2+ ions in the FW and Fe-minerals within rock, 910 ppm of Fe2+ ions were included in the FW, and FeS2, FeS, and FeCO3 were present in the carbonate rock at content of 1.0%, 1.0%, and 0.5%, respectively (Table 2). Table 3 summarizes the ionic compositions and concentrations of the FW and injection water. The data of the FW ware at initial state, which was in equilibrium with the rocks before the injection of LSW. They were based on the ionic compositions of the FW in the Middle East where Fe2+ ions were present amid a low pH of 3.2. The total dissolved solids of the FW were as high as 150,000 ppm; the Ca2+ concentration was especially high due to the dissolution of calcite (CaCO3), which accounted for 90% of the carbonate rock. In the case of the injection water, the seawater composition used in the laboratory coreflooding experiment by Yousef et al. (2011) was taken. Although the salinity of the seawater was twice as high as that of typical ocean seawater, it was a third of the FW salinity, so it was suitable for LSW. The injection water was set to a pH 7.8, with reference to the pH of the Persian Gulf.
Fe-mineral content in carbonate rock and the Fe2+ ion concentration in the formation water.

Input data for ion composition and concentrations (Yousef et al., 2011).
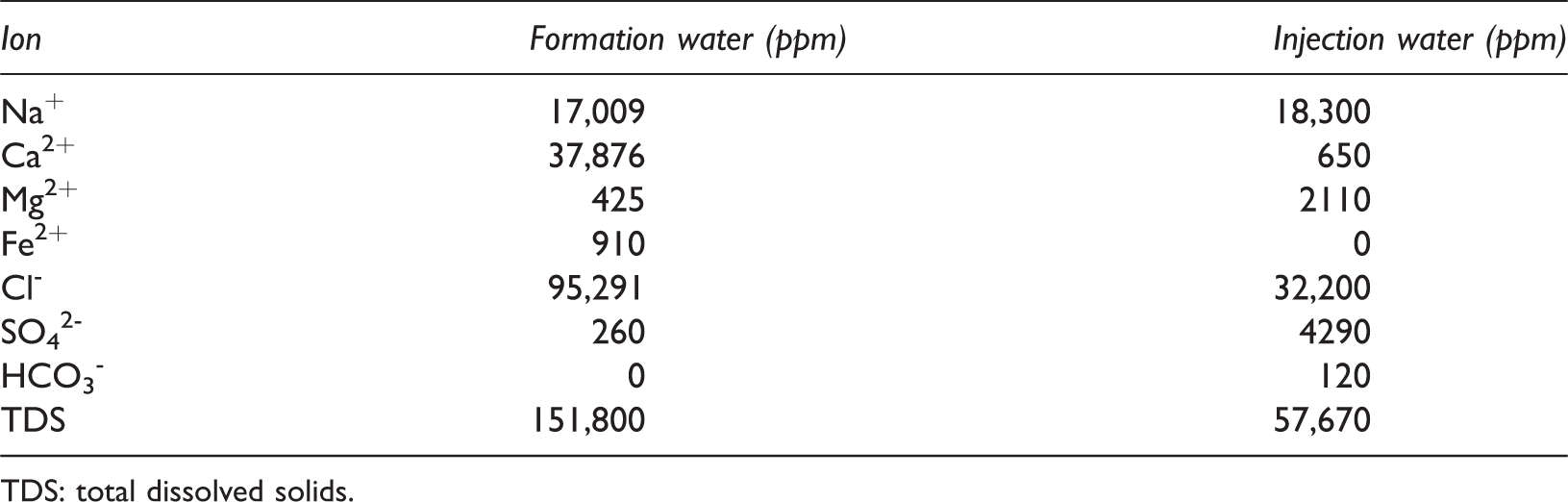
TDS: total dissolved solids.
Results and discussion
pH change analysis
Within this study, a simulation was performed for the application of LSWF in the oil reservoir, where Fe-minerals exist in carbonate rock with Fe2+ ions in the FW as given in Table 2. When LSW was injected into the carbonate reservoir, Fe-mineral dissolution and precipitation occurred as the pH increased, which was the main factor influencing the chemical reactions. Thus, we examined the dissolution and precipitation as a factor of pH change. The initial pH of the FW containing 910 ppm of Fe2+ ions was 3.2, which was relatively low as shown in Figure 3; however, the pH at the location of Ⓐ near the injection well drastically increased to 4.0 as a result of the LSW injection. At the time of one pore volume injected (1 PVI), the mixing effects with the LSW (pH 7.8) prevailed at location Ⓐ, which promoted the dissolution of Fe-minerals (Chen et al., 2012). However, at the location Ⓑ far from the injection well, the pH for 1 PVI remained in the initial state due to the little effect by LSW injection. However, at the time of two pore volume injected (2 PVI), the pH was stabilized to 4.0 at the locations Ⓐ and Ⓑ since the mixing effects were stabilized by the injection of LSW for a long period of time. It could also be considered that the chemical reactions between ions were nearly equilibrated. Therefore, due to the above phenomena, we quantitatively analyzed the dissolution and precipitation of Fe-minerals at location Ⓐ after 2 PV injected with sufficient chemical reactions.
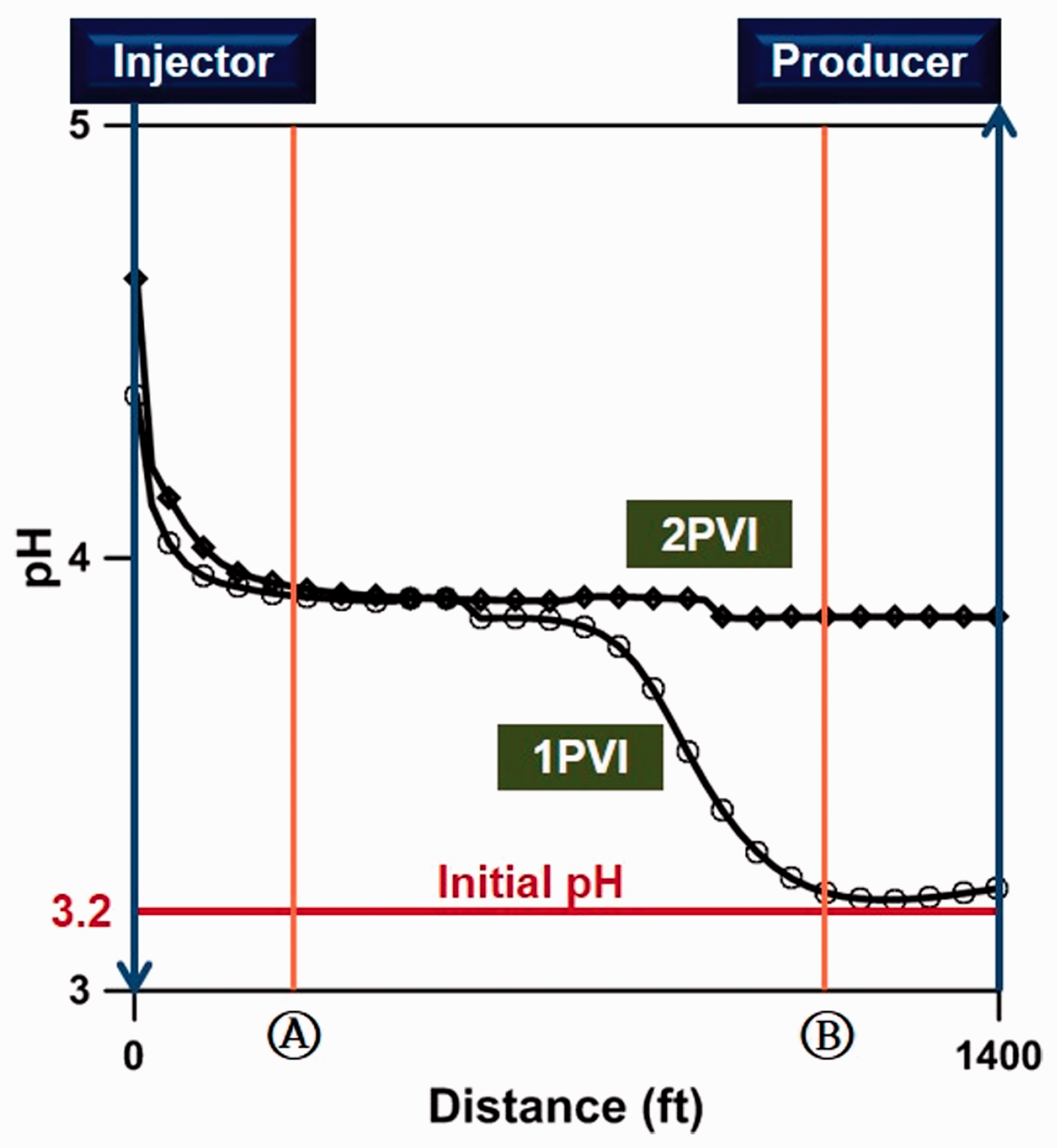
Distribution of pH values after low-salinity water injection.
Precipitation and dissolution of minerals
As LSW was injected into the carbonate reservoir, the chemical equilibrium was broken due to differences in the ionic composition and concentration between the FW and injection water, resulting in the dissolution and precipitation of rock. Figure 4 shows the total dissolution and precipitation quantities of Fe-minerals regardless of type at location Ⓐ. The quantity of Fe-precipitation was 264.4 × 103 gmol and the quantity of Fe-mineral dissolution was 264.8 × 103 gmol at 2 PVI; there was almost no difference in terms of the total volume in this system. The difference was very small, but the quantity of dissolution or precipitation itself was as large as 264 × 103 gmol. Thus, it was necessary to analyze the quantity of precipitation and dissolution in detail for each Fe-mineral. This was because the quantity of precipitation and dissolution caused by chemical reactions between the ionic composition of the FW, the injection water, and the mineral constituents of the carbonate rock were different for each type of Fe-minerals. Also, since the solubility or insolubility characteristics of each Fe-mineral were different, the permeability changed accordingly. The precipitation and dissolution quantities for each Fe-mineral were estimated as can be seen in Figure 5. In this figure, a positive value for the change in mineral moles indicated precipitation, while a negative value denoted dissolution. At the time of 2 PVI, a noticeable increase in the quantity of Fe(OH)2 precipitation and FeS2 dissolution yielded 220 × 103 gmol and 200 × 103 gmol, respectively. These quantities were significantly higher compared to the quantities for the other Fe-minerals, such as FeCO3, FeSO4, FeS, and the carbonate, CaCO3. In the case of Fe(OH)2, it was not contained within carbonate rock but was generated via precipitation; while FeS2 existing within the carbonates was dissolved. The aspect for Fe(OH)2 and FeS2 precipitation or dissolution relative to each injection time is depicted in Figure 6. The reason for the significantly larger quantities of precipitation and dissolution for these two minerals compared to the other minerals could be comprehended from the equilibrium constants for the chemical reactions. The equilibrium constants could be defined as a function of temperature as follows
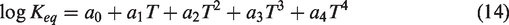
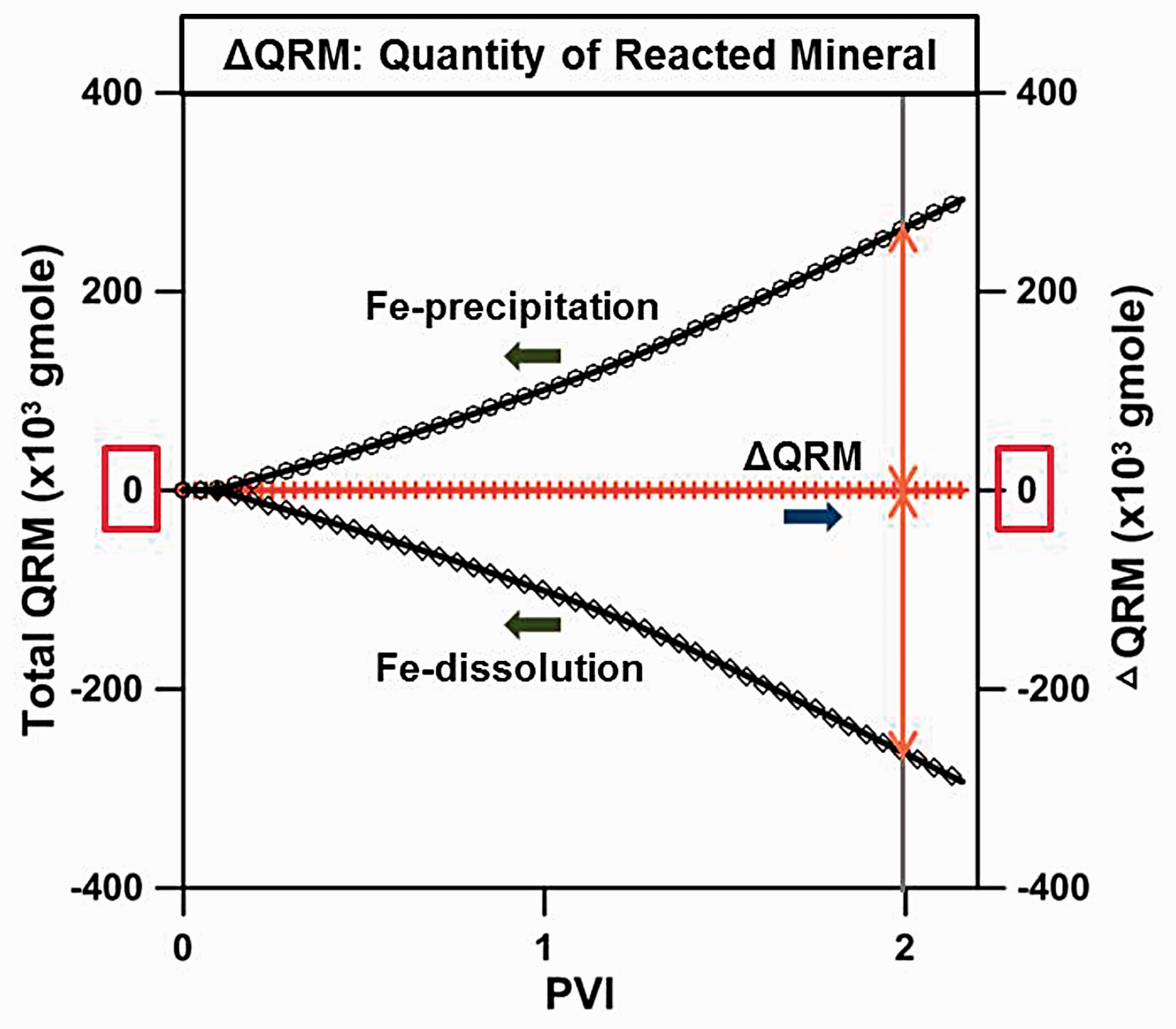
The quantity of reacted minerals for the Fe-precipitation and Fe-dissolution at location Ⓐ.
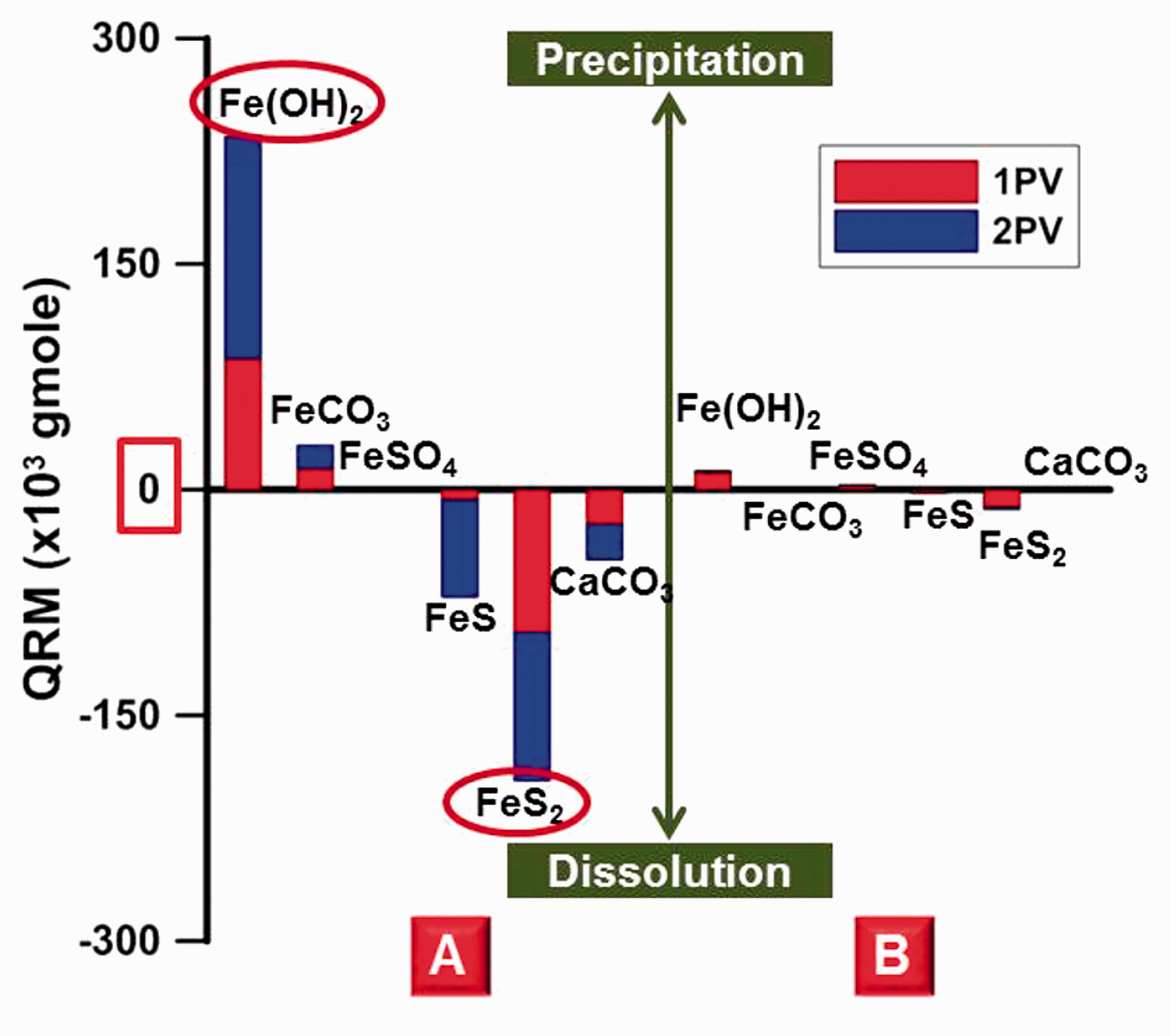
The quantity of precipitation and dissolution for Fe-minerals and calcite at locations Ⓐ and Ⓑ.
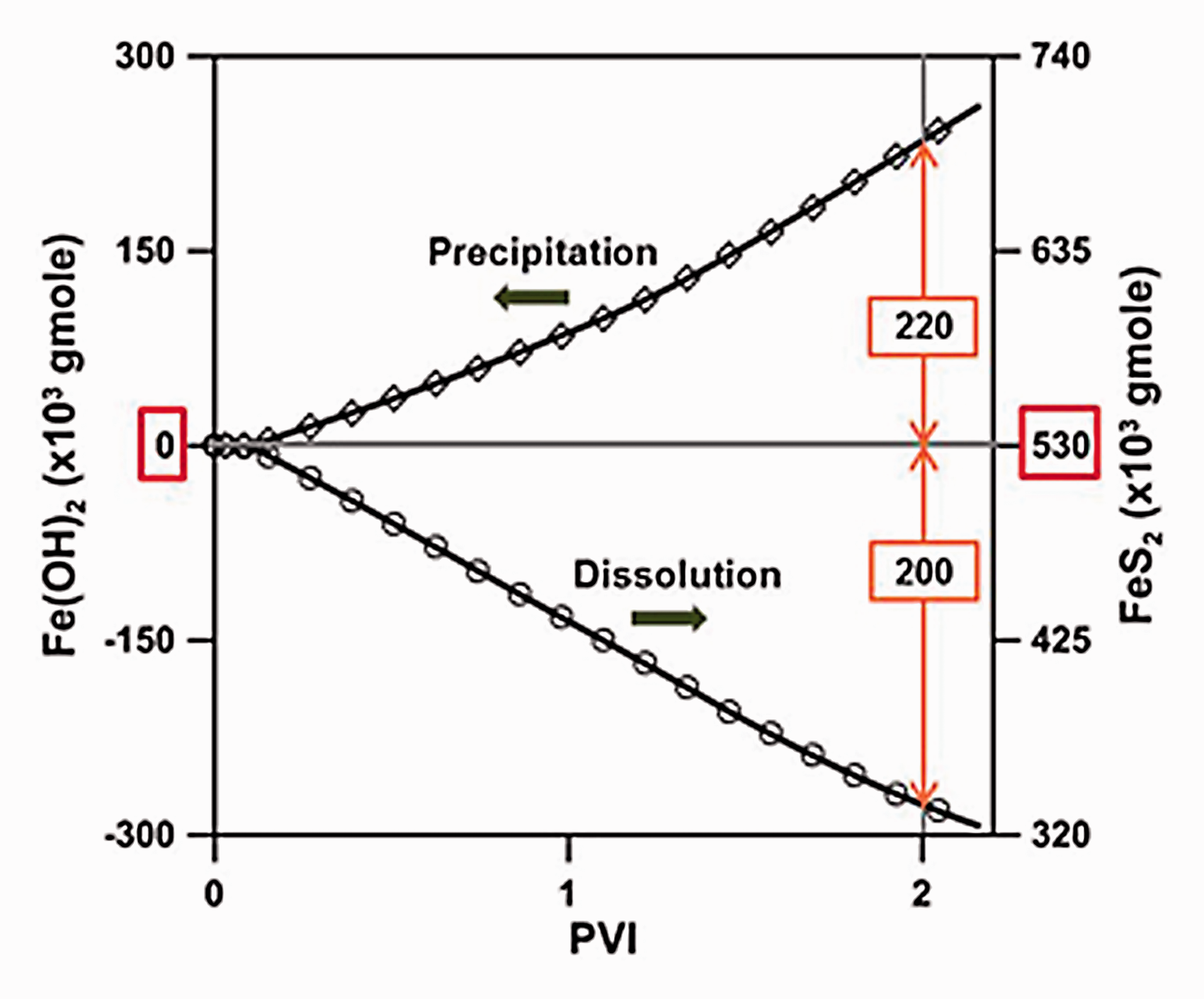
The quantity of reacted minerals for Fe(OH)2 and FeS2 at location Ⓐ.
Chemical equilibrium coefficients (ai) used for calculation of the equilibrium constants (Keq) (Bethke, 1996).

The reaction for the precipitation of Fe(OH)2, possessing an enormously large equilibrium constant, can be seen in equation (9) as being independent of the SO42– ions. On the other hand, FeS2 dissolved when contacting the injection water, resulting in the generation of 0.25 mol of SO42– ions (equation (11)). The FeS2 dissolution is also attributed to the generation of hydrogen sulfide (H2S) which can cause the problems for souring of oil (Al-Kindi et al., 2008). However, in this simulation study, the amount of H2S was not enough to occur the souring problem during the period of production. Also, we focused on the effect of mineral precipitation and dissolution on the recovery of oil. In wettability alteration mechanism, SO42– ions played a major role in enhancing the oil recovery by detaching oil that had attached on the carbonate rock surface forming an oil-wet condition and by causing the alteration in wettability. The initial SO42– concentration of the FW in this study was 260 ppm and that of LSW was 4290 ppm. As LSW was injected into the carbonate reservoir where FeS2 did not exist, the SO42– concentration increased to about 2300 ppm due to simple mixing effects without the additional generation of SO42– ions (Figure 7). However, in the presence of FeS2 in carbonate rock, SO42– ions were released from the dissolution of FeS2 as LSW was injected and the additional SO42– ion concentration reached up to 960 ppm, which played a notably important role with regard to improving the efficiency of the LSWF method. Therefore, to analyze these reactions in more detail, we examined the occurrence of SO42– ions depending on the temperature and content of FeS2, which were the primary factors influencing the above reactions.
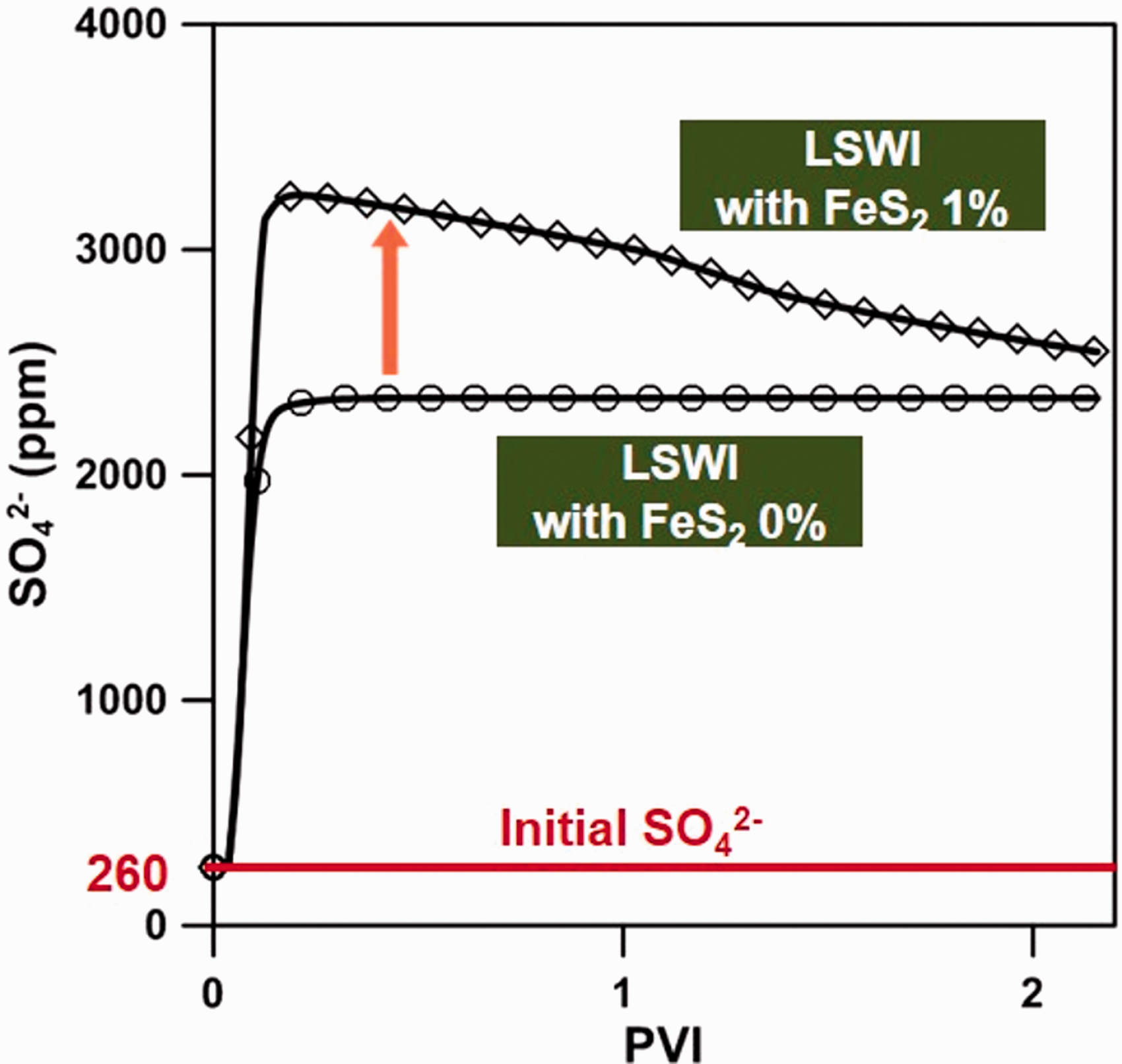
The occurrence of SO42– ions due to the FeS2 dissolution at location Ⓐ.
In order to investigate the effect of the reservoir temperature and FeS2 content on the generation of SO42– from the FeS2 dissolution, we first performed a simulation using Keq in Table 5 for reservoir temperatures from 50 to 120°C in intervals of 10°C. After 2 PV was injected, the quantity of dissolved FeS2 increased by 6.4 times when the temperature was 120°C compared to 50°C, which indicated that higher reservoir temperatures yielded more SO42– ions due to the dissolution of FeS2, thereby improving the EOR efficiency through LSWF. Then, simulations for the effects of FeS2 content were performed when FeS2 was contained within carbonate rock from 0 to 8.5%, over 0.5% intervals. Figure 8 shows the additional SO42– ions from the FeS2 dissolution at the time of 2 PV injected, except for simple mixing effects due to the injection of LSW. As a result, the SO42– concentration was close to the maximum when 7% or more FeS2 existed within the carbonate reservoir of 120 C. Meanwhile, the quantity of generated SO42– at 50 C was less than that at 120 C and SO42– ions did not occur any longer even when the content of FeS2 was 1% or more. Therefore, since the EOR effect due to SO42– ions was limited at certain temperatures and FeS2 content levels, it is necessary to pay attention to the determination of the SO42– concentration for the design of PDI compositions in LSW.
Chemical equilibrium constants (Keq) for various temperatures.
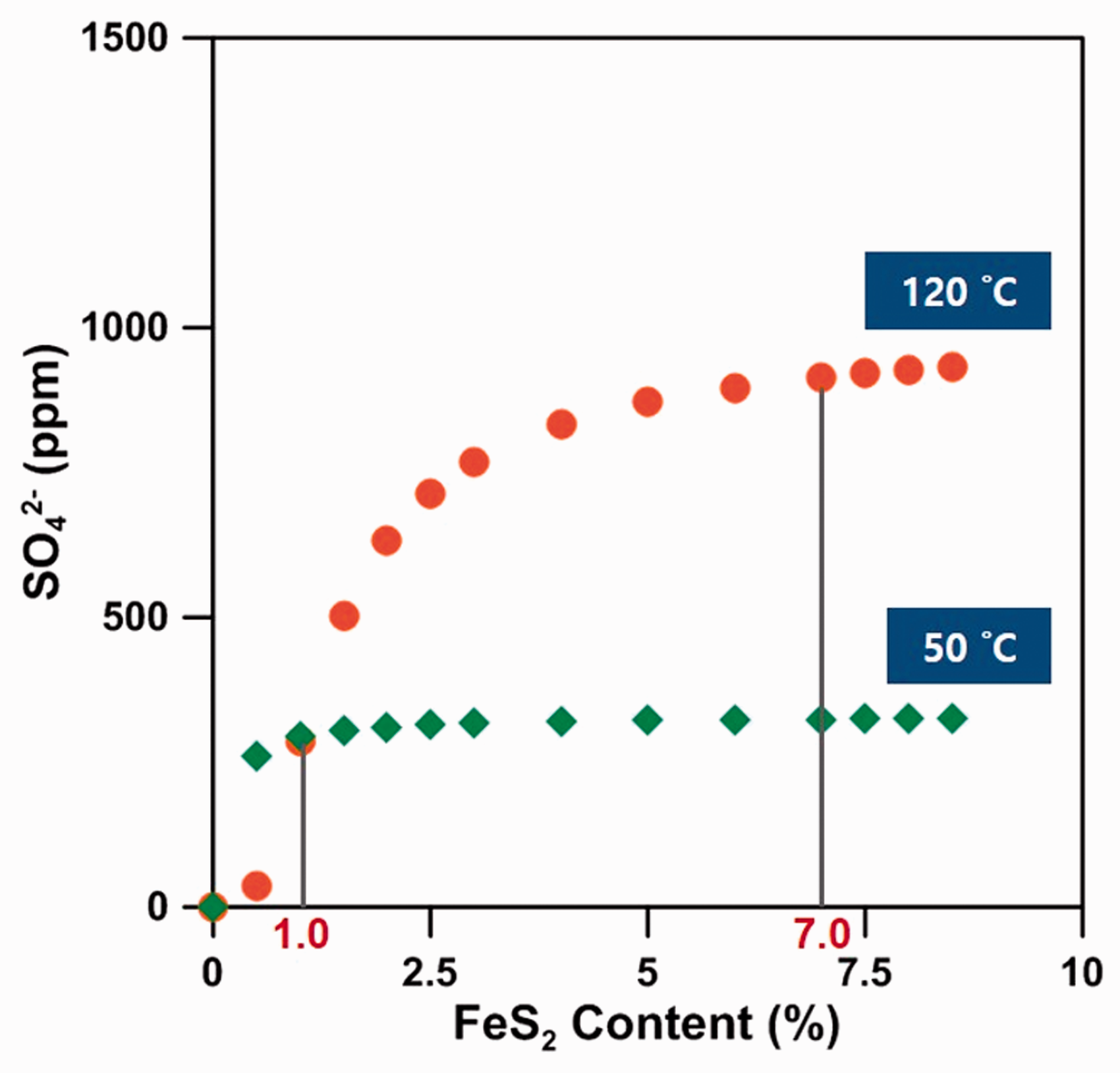
The additional SO42– ions due to the FeS2 dissolution depending on the FeS2 content at location Ⓐ.
Activity product at 120°C after two pore volumes injected.
Permeability change and EOR effect analysis
When LSW was injected into the carbonate reservoir containing Fe-minerals, the precipitation or dissolution that occurred due to chemical reactions caused a permeability change (Figure 9). The simulation results analyzing this phenomenon showed that the pore volumes decreased by 250 ft3 and increased by 270 ft3 due to precipitation and dissolution including calcite dissolution, respectively, after 2 PV injections at location Ⓐ. Among the volumes, the volume reduction due to Fe-precipitation was 250 ft3 and the volume increase due to the dissolution of Fe-minerals was 210 ft3, with the exceptions of the precipitation and dissolution of calcite (CaCO3). This indicated that the Fe-minerals precipitated more easily as opposed to dissolution. The pore volume change due to the dissolution and precipitation of Fe2+ is 40 ft3, which is 6.6 × 10−5 times the model total pore volume 605,895 ft3. This is almost negligible with respect to the total pore volume. Accordingly, the porosity was estimated as 27.09%, in which the change of porosity is also negligible; permeability was calculated using the Kozeny–Carman equation as follows
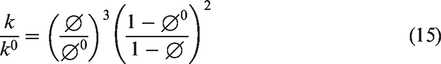
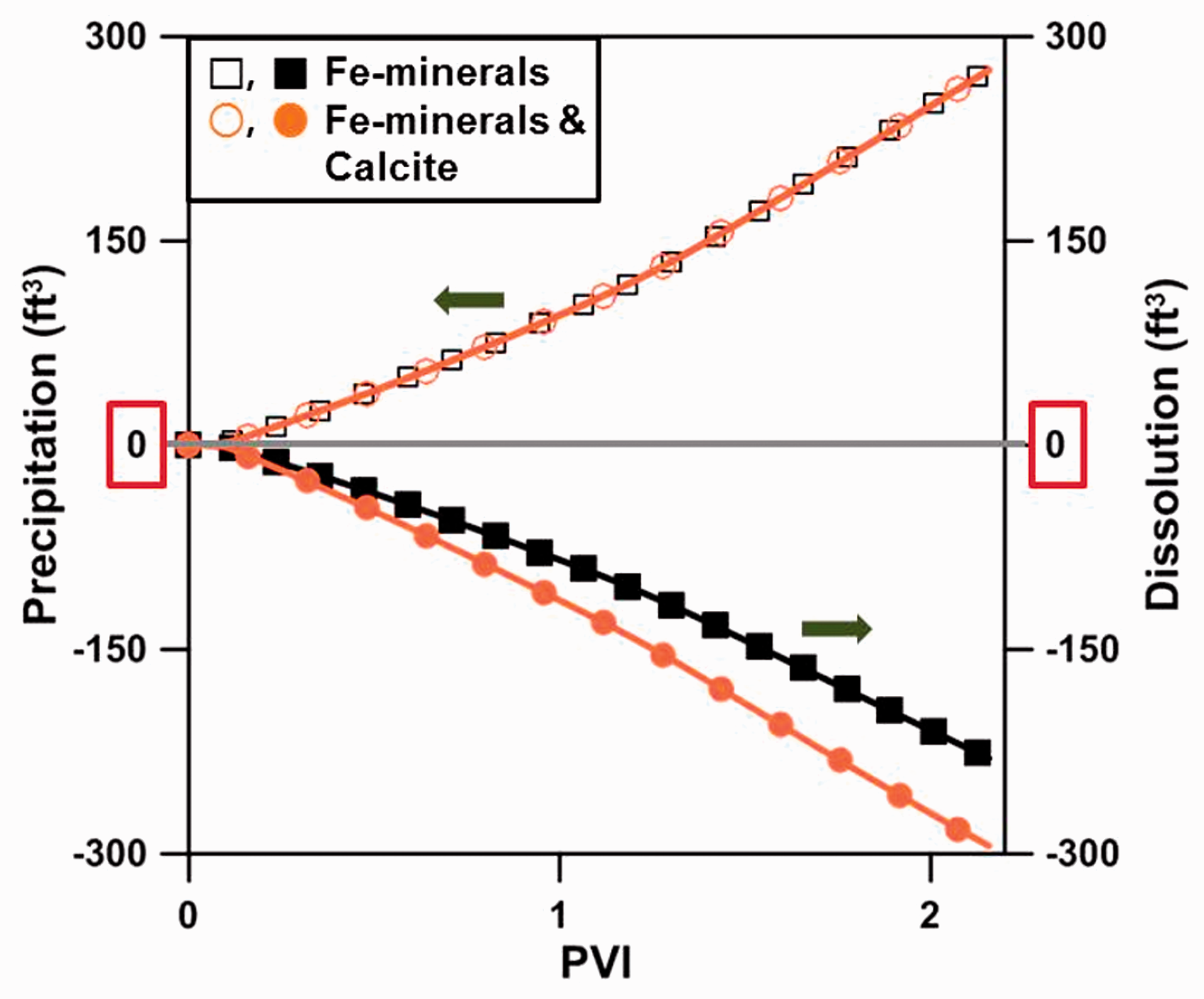
The estimated volumes of precipitation and dissolution at location Ⓐ.
This equation is one of the most widely accepted and simplest model for the permeability–porosity relationship, which provides a link between media properties and flow resistance in pore channels (Carman, 1937; Kozeny, 1927). The estimated permeability depending on the temperature and FeS2 content is summarized in Table 7. As shown in the results, both quantities of precipitation and dissolution were very large; however, there was little difference between the two. Thus, it was confirmed that the permeability did not change from the initial value of 30 md. Also, the permeability did not change even when the reservoir temperature was lowered to 50°C or the FeS2 content was increased to a value as high as 8.5%.
Permeability change at location Ⓐ due to precipitation and dissolution after two pore volumes injected.

Finally, we investigated the EOR effect when LSWF was applied to the carbonate reservoir. In our system, there was almost no difference between the quantities of Fe-precipitation and Fe-mineral dissolution, it seems that the concentration of Fe2+ ion was almost not changed in this system, which would not affect the wettability of rock surface. Additionally, since Fe2+ was contained only in the formation water as 910 ppm, not in injection water, the concentration of Fe2+ was reduced up to 11 ppm by injecting the LSW of 0 ppm. In this aspect, we assumed that the effect of Fe2+ on wettability alteration was considered to be negligible. Therefore, in this paper, we mainly focused on the effect of precipitation and dissolution of Fe-minerals on oil recovery. In the case of carbonate rock containing FeS2 as a Fe-mineral, as FeS2 was dissolved, SO42– ions were generated. These ions detached oil from the rock surface, and consequently, more oil was produced. As the results shown in Figure 10, the oil recovery for carbonates without FeS2 was 69.06%, which was 12.41% greater than 56.65% for conventional WF due to the injection of LSW containing SO42– ions. However, 76.67 and 76.70% were recovered from carbonates containing 1 and 8.5% FeS2. This was much higher than that for conventional WF. Figure 11 shows schematically overall process in the absence and presence of FeS2. In the case of its absence, some SO42– ions in injection water attach on the carbonate rock surface, then oil is detached from rock surface. On the other hand, when FeS2 is contained in carbonate rock, FeS2 is dissolved releasing SO42– ions near the rock surface. Therefore, more SO42– ions can attach on rock surface causing the detachment of more oil. However, additional oil was not recovered for FeS2 contents above 1%. This was because the total aqueous SO42– concentration equilibrated to 3700 ppm after 2 PV injected when the FeS2 content was 1% or more; thus, additional oil was not produced by the SO42– ions. Since the equilibrium SO42– ion concentration was 2300 ppm in the absence of FeS2, this difference in the SO42– concentration led to an additional 7.61% recovery of oil.
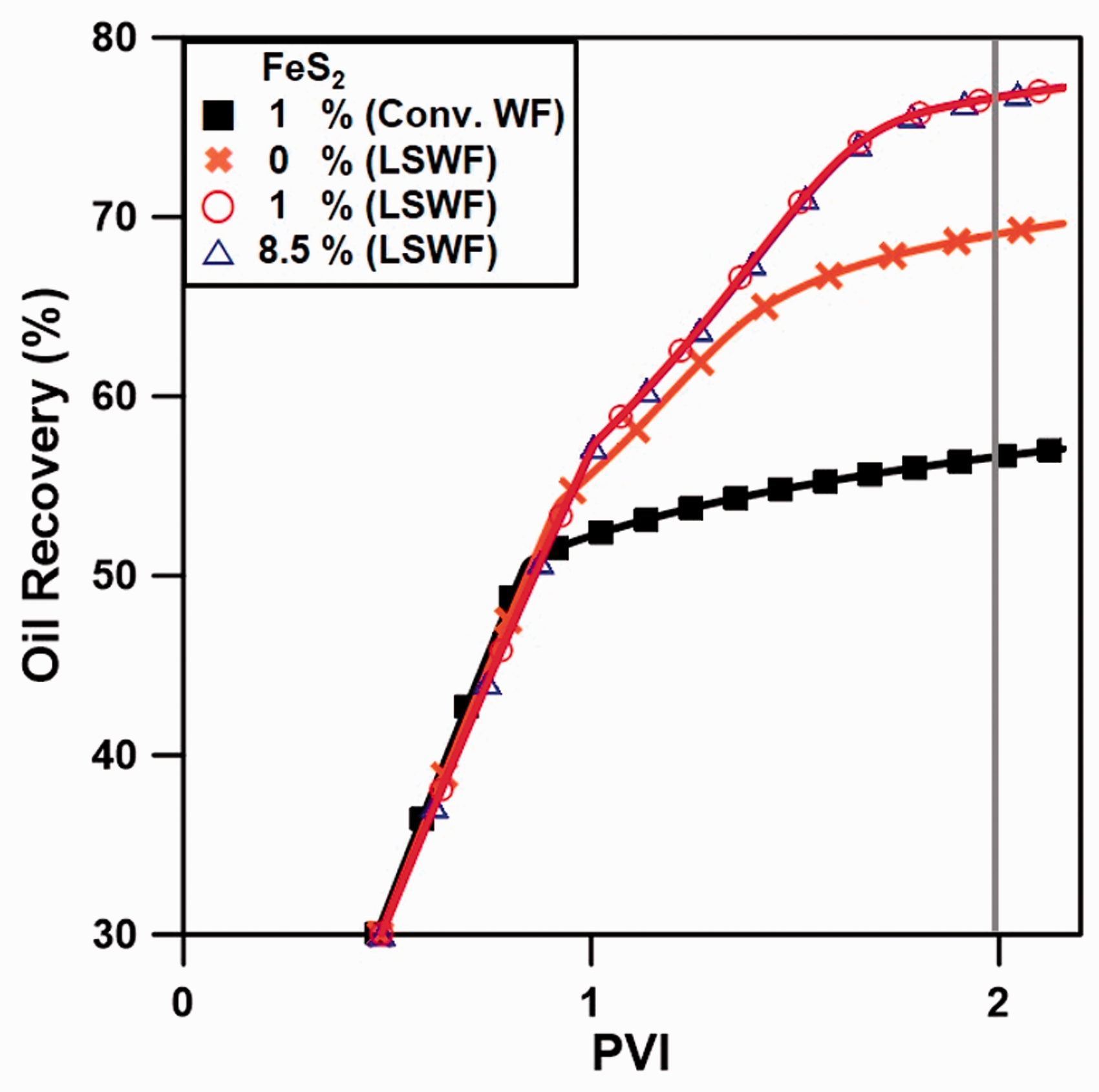
A comparison of the oil recovery depending on the FeS2 content.
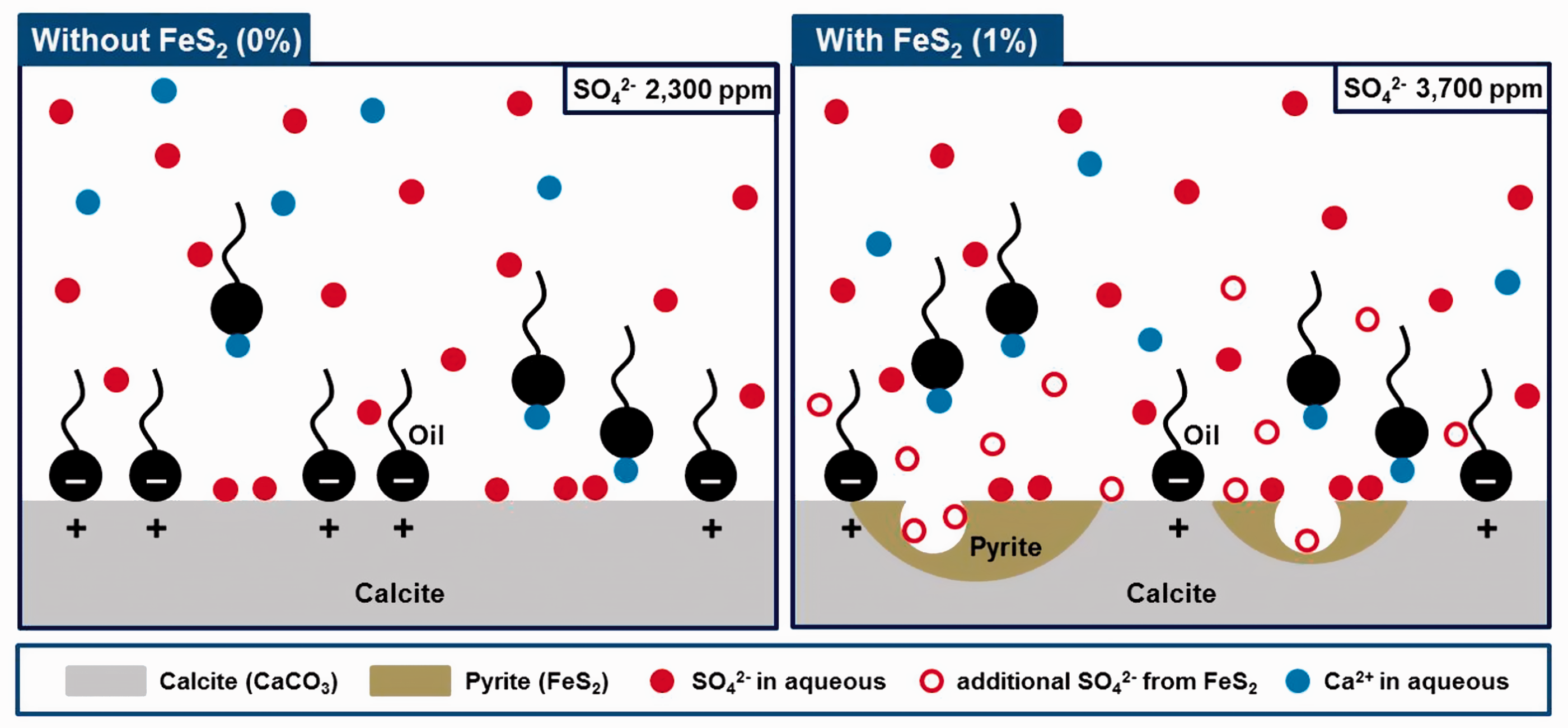
Schematic representation for oil detachment due to additional SO42– ions from FeS2.
Based on the above results, when applying LSWF to the carbonate reservoir containing Fe-minerals, we proposed an oil recovery relationship as a function of the temperature and SO42– concentration in order to determine the optimal SO42– concentration with regard to designing the PDI composition in LSW. A correlation between the oil recovery and SO42– ion concentration, which was the most important PDI in LSW, and the reservoir temperature, which greatly affected the precipitation and dissolution of Fe-minerals, is represented in Figure 12.
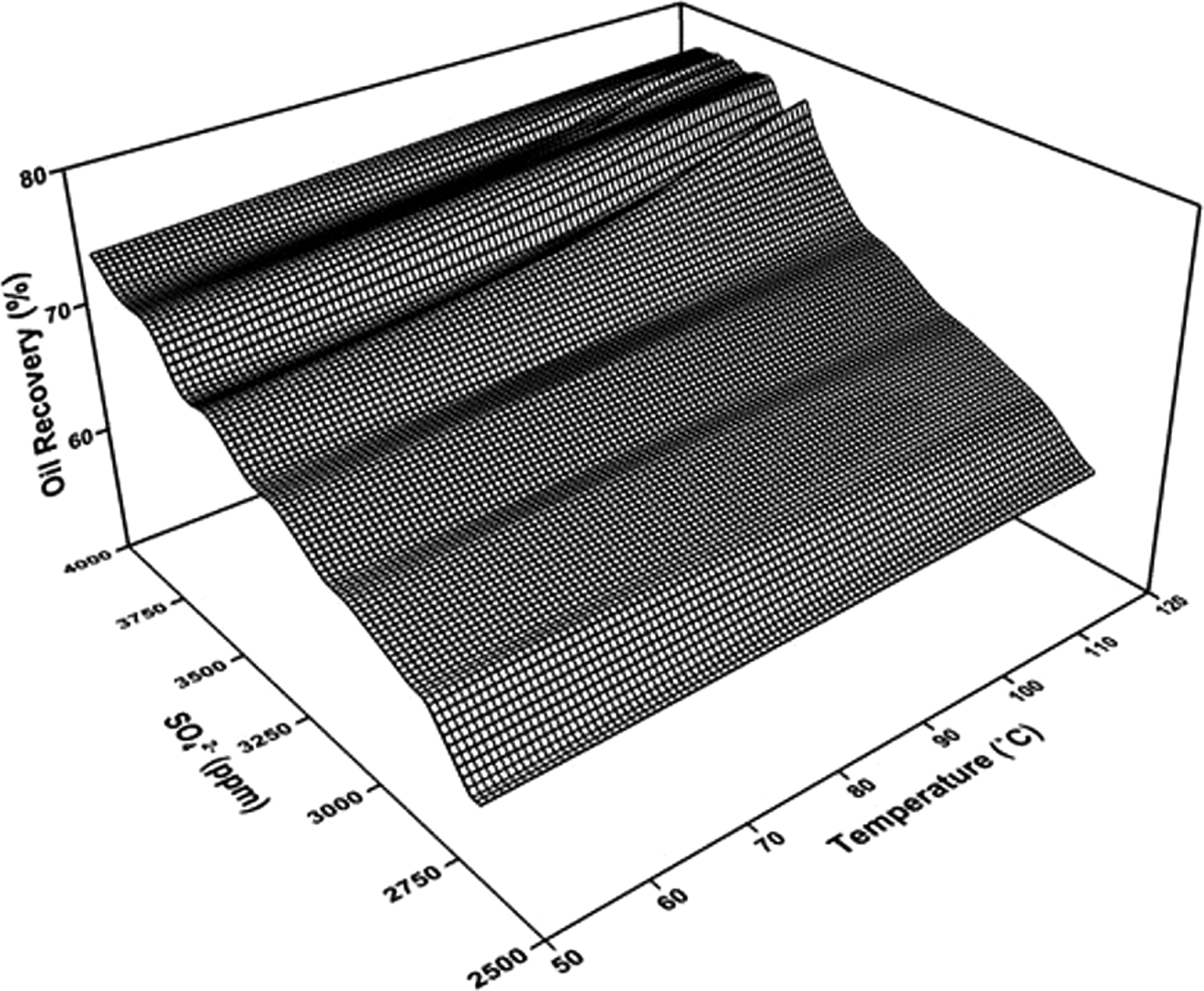
The oil recovery in relation to temperature and SO42–.
Summary and conclusions
In this study, we performed several simulations to analyze the EOR effects focusing on the dissolution of Fe-minerals and Fe-precipitation when LSW containing SO42– was injected into carbonate oil reservoirs.
From the results investigating precipitation and dissolution of Fe-minerals, it was found that the magnitude of precipitation and dissolution due to chemical reactions during LSW injections was enormously large, and the quantities were quite different according to the type of Fe-minerals. Among the Fe-minerals, the amounts of Fe(OH)2 precipitation and FeS2 dissolution were noticeably large in this system compared to FeS, FeSO4, and FeCO3, i.e. Fe(OH)2 not contained in carbonate rock was precipitated, which is insoluble. On the other hand, FeS2 contained in the rock was dissolved. This could be attributed by the saturation index criteria which was calculated by the ratio of activity product to equilibrium constant in the chemical reactions. As mentioned above, since the solubility characteristics of each Fe-mineral are different, it is essential to analyze precipitation or dissolution for each Fe-mineral individually, in which these phenomena yielded a permeability change (increase or decrease). However, in the case of this system, there was almost no difference between precipitation and dissolution in terms of total quantities of Fe-mineral, and consequently permeability was not changed.
From the analysis results for the SO42– ions released from the process of precipitation and dissolution, in the dissolving process of FeS2, the additional SO42– ions were released differently depending on the content of FeS2. However, because the generation of SO42– ion was limited at certain level of FeS2 content in carbonate rock, it is necessary to pay attention to the determination of SO42– concentration for the design of PDI compositions in the injection water. Ultimately, in the investigation of EOR efficiency, oil production was enhanced due to additional SO42– ions generated from the FeS2 dissolution.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant funded as part of the project “Development of IOR/EOR technologies and field verification for carbonate reservoir in UAE” by the Korean Government Ministry of Trade, Industry and Energy (MOTIE) (No. 20152510101980).