Abstract
Total organic carbon contents are an important parameter in evaluating hydrocarbon source rocks and it shows the hydrocarbon-generating potential of hydrocarbon source rocks. In this study, the total organic carbon of 16 carbonate rocks with different maturity level was determined by a modified acid treatment protocol of the elemental analyzer method. In the modified method, montmorillonite is used to thicken the residue of acid treatment and remaining acid solution. Then, total organic carbon values of “clay-paste” sample were analyzed by the element analysis method. The results of 16 carbonate rocks show that the total organic carbon values with the new method were greater than the conventional total organic carbon test in a majority of the samples. Additionally, the fraction of acid-soluble organic matters was investigated and it shows no clear relationship of the fraction of acid-soluble organic matters with maturity and total organic carbon values in source rocks. Hence, it indicates that the released acid-soluble organic matters are characterized by complex factors that influence their occurrence and compositions.
Keywords
Introduction
Total organic carbon (TOC) contents have been widely used to assess the hydrocarbon-generating potential of petroleum source rocks (Alsharhan and Salah, 1997; Gentzis, 2013; Mustapha and Abdullah, 2013). Two methods have usually been used to measure TOC contents of source rocks in laboratory: “Rock-Eval”-type pyrolysis–combustion method and combustion (elemental analysis) method. The former method (“Rock-Eval” method) involves two separate stages (pyrolysis and combustion) of temperature-programmed heating and can provide information on the amount of free hydrocarbons (S1), hydrocarbon generated by thermal cracking (S2), CO2 produced during pyrolysis (S3), and of hydrocarbon generated by the oxidation of residual organic carbon (S4). This method has been widely applied to source rocks to evaluate their kerogen types and maturity (Clementz et al., 1979; Espitalie et al., 1977; McCarthy et al., 2011; Peters, 1986). The TOC values can also be calculated from these parameters (S1, S2, S3, and S4) acquired during the pyrolysis–combustion steps (Behar et al., 2001). However, some carbonate minerals in carbonate-rich source rocks decompose at relatively low temperatures. For example, natural siderite starts to decompose at about 465°C (Alkaç and Atalay, 2008) and hydromagnesite decomposes completely at temperatures ranging from 400 to 800°C (Liu et al., 2012). Therefore, the CO2 produced by some carbonate minerals at those low temperatures may lead to an overestimation of TOC values. Apart from this, the parameters (S1, S2, S3, and S4) of source rocks with low maturity are characterized by well-defined single peaks. Hence, “Rock-Eval”-type pyrolysis–combustion method is more suitable for carbonate rocks with low maturity. However, these parameters of carbonate rocks with high maturity are characterized by small, multiple peaks. This causes a problem for “Rock-Eval” software to calculate peak areas accurately, thus leading to errors in the calculations of the pyrolysis parameters and TOC values.
In the combustion (elemental analysis) method, the value of TOCs can be obtained from the amount of CO2 that was produced by the oxidation of all types of organic carbons in samples with excess O2. This elemental analysis method has been used to determine TOC contents of various geologic materials, including soil, sediments, and sedimentary rocks. First, carbonate minerals have to be removed by acid treatment prior to combustion to determine TOC contents (Hedges and Stern, 1984; Heron et al., 1997; Kennedy et al., 2005; Komada et al., 2008; Petersen et al., 2008; Phillips et al., 2011; Verardo et al., 1990). For example, the analytical protocol proposed by Petroleum Exploration Speciality Standardization Committee of China includes the following steps: (1) sediments and rock samples are ground to ≤0.2 mm and 0.01–1 g of a sample is weighed with the accuracy of 0.0001 g; (2) excessive hydrochloric acid (volume ratio of analytically pure hydrochloric acid and water is 1:7) is added to the samples slowly and then heated to 60–80°C at least for 2 h; (3) after treating with acid, the sample is filtered with a Nutsche filter and the residue sample is rinsed with water until the filtrate solution becomes neutral; and (4) the sample is dried in a drying oven at temperatures ranging from 60 to 80°C before the TOC value of the sample can be tested by an elemental carbon analyzer. In this standard analytical protocol, the acid solution, which is generated in carbonate digestion, is discarded. However, Reborts et al. (1973) tested TOC contents in acid solutions that were produced by treating modern carbonate sediments from Florida Bay of U.S. They found that the organic matter contents in the acid solutions can be as high as 44% of the TOC. Froelich (1980) analyzed the modern carbonate sediments from the Pacific and Atlantic Oceans and found that the TOC values in generated acid solutions increase with the increasing carbonate abundances. The TOC values of the acid solutions can be as high as 45% of TOC in the carbonate sediments with low TOC values and high carbonate abundances. Therefore, discarding the acid solution generated in the pretreatment of the elemental analysis method for TOC analysis leads to underestimation of TOC contents, particularly for source rocks with low TOC contents. It makes it difficult to evaluate hydrocarbon source rocks and study their hydrocarbon-generating potential objectively. To overcome this problem in the rinse method for TOC measurements, an in situ acid treatment of individual soil or sediment samples placed in tin or aluminum cups has been developed, either fumigating the samples with acid–vapor within a closed container (Harris et al., 2001) or pipetting acid solution into the cup (Verardo et al., 1990). After acid treatment, the samples in the cups were dried in an oven, so that organic matter dissolved in the acid largely, if not all, remains inside the cups. Although successful in general (Brodie et al., 2011; Larson et al., 2008), these in situ acid solution methods are not well suited for samples of sedimentary rocks with high carbonate contents, particularly of more acid-resistant carbonates (siderite, dolomite). Liu et al. (2016) have developed a modified acid treatment protocol of the elemental analyzer method to measure accurately TOC contents of carbonate-rich source rocks such as carbonaceous shales. In this method, montmorillonite was used to thicken the residue of acid treatment and remaining acid solution. Then, TOC values of the “clay-paste” sample were analyzed by the element analysis method. In this approach, the organic matter in acid solution, which is discarded in conventional measurement, is included in the “clay-paste.” Even though the modified approach is of complex experimental procedure, it can obtain the integral organic matter contents in source rocks. In this study, carbonaceous rocks with different maturity level were collected from Ordos Basin of China and Cretaceous Eagle Ford Formation in Texas of U.S.A. Those samples were analyzed with the modified acid treatment protocol of the elemental analyzer method in order to investigate the fraction of acid–solution organic matters in source rocks and its possibly occurring states.
Samples and methods
Natural carbonaceous rock samples
Natural samples include six carbonaceous outcrop samples from Cretaceous Eagle Ford Formation in Texas, U.S.A. with lower maturity (Ro: 0.6–1.2%). The carbonate content of carbonaceous rock samples in Cretaceous Eagle Ford Formation ranges from 60 to 90% (Hunt and McNichol, 1984). Ten core samples of carbonate rocks were collected from Ordovician Majiagou Formation of Ordos Basin of China, with Ro in the range of 2.07–2.68% (Liu et al., 2009).
Montmorillonite as a thickening agent
Montmorillonite has a characteristic of water absorption (Tanaka et al., 2007) and organic carbon dissolved in solutions and clay can form organic–mineral complexes (Greenland, 1971). Therefore, montmorillonite may be used to thicken the mixture of acid-treated sediment samples and remaining acid solutions into a homogeneous paste.
The montmorillonite (standard mineral sample) needs to be processed as follows to remove possible traces of carbon: (1) The montmorillonite samples were ground to 1 mm. (2) The montmorillonite was immersed in distilled water and dispersed by ultrasonic. (3) Hydrochloric acid (concentration: 3%) was added to remove carbonates and the remaining solution in the montmorillonite suspension was decanted. (4) Hydrogen peroxide (concentration: 30%) was added to the montmorillonite suspension and heated to 40°C in a water bath in order to remove trace organic carbon. (5) The suspension was washed to neutral by distilled water. (6) After the suspension was stirred for 8 h at room temperature, the upper 10 cm overlying suspension was decanted and recovered (Gao and Zuo, 2007). Distilled water was added and this step repeated. (7) All the recovered upper suspension was centrifuged to allow clay minerals to settle. (8) The collected montmorillonite was dried at 60°C and ground to powder. (9) The montmorillonite powder was dried at 400°C for 4 h in order to remove interlayer organic matters.
New method of TOC test
The new method of TOC test follows the method proposed by Liu et al. (2016). In this study, all samples were analyzed in the Key Laboratory of Petroleum Resources Research, Institute of Geology and Geophysics, Chinese Academy of Science at Lanzhou. The details were described as follows. About 1 g of the samples of the natural and synthetic rocks were ground to 100 meshes and transferred to a conical beaker. Excessive hydrochloric acid (concentration: 37.5%) was added to the conical beaker at room temperature for 2 h, repeating the process until no bubbles occur. After thickening with montmorillonite by stirring, the acid solution and residue can be homogenized. The weight of empty conical beaker, conical beaker with sample, and conical beaker with paste are m1, m2, and m3, respectively. The result of carbon element analysis is n% and the TOC value of sample can be calculated by the following equation. The blank montmorillonite was measured and it has been corrected in results
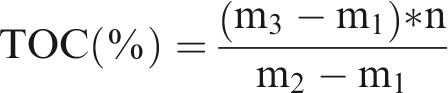
The TOC value of the paste was determined by a Varo Pyro cube element analyzer coupled to an IsoPrime 100 IRMS. The sample was introduced and combusted in reaction tube of the Varo Pyro cube element analyzer. The temperature of reaction tube was 900°C. The CO2 was carried by helium and introduced into an IsoPrime 100 IRMS to analyze δ13C isotopic ratios. The measurement precision is estimated to be ±0.5‰ for δ13C with respect to VPDB.
The TOC contents of the samples were determined by a LECO CS-344 Carbon and Sulfur Analyzer; for the analysis process refer to National standard of P.R. China (GB/T 19145-2003).
Results and discussion
Comparison of TOC measurement results of carbonaceous source rocks in different approaches
The TOC contents of the low maturity carbonaceous rocks, which were collected from Eagle Ford Formation, were analyzed with both conventional “acid-rinse” and new “clay-paste” methods (Table 1). The TOC test with the new method was repeated twice and was compared with the results of the conventional analytical method (Figure 1). The TOC values determined by the new “clay-paste” method are generally greater than those of the conventional method except for MG11, owing to the fact that the organic matter in acid solution was not rinsed off. The δ13C of TOC ranges from −36.7 to −26.5%, suggesting negligible, if any, contributions from inorganic carbonate carbons.
Results of conventional TOC analysis and new method of low maturity carbonate rocks collected from Cretaceous Eagle Ford Formation in Texas, USA.
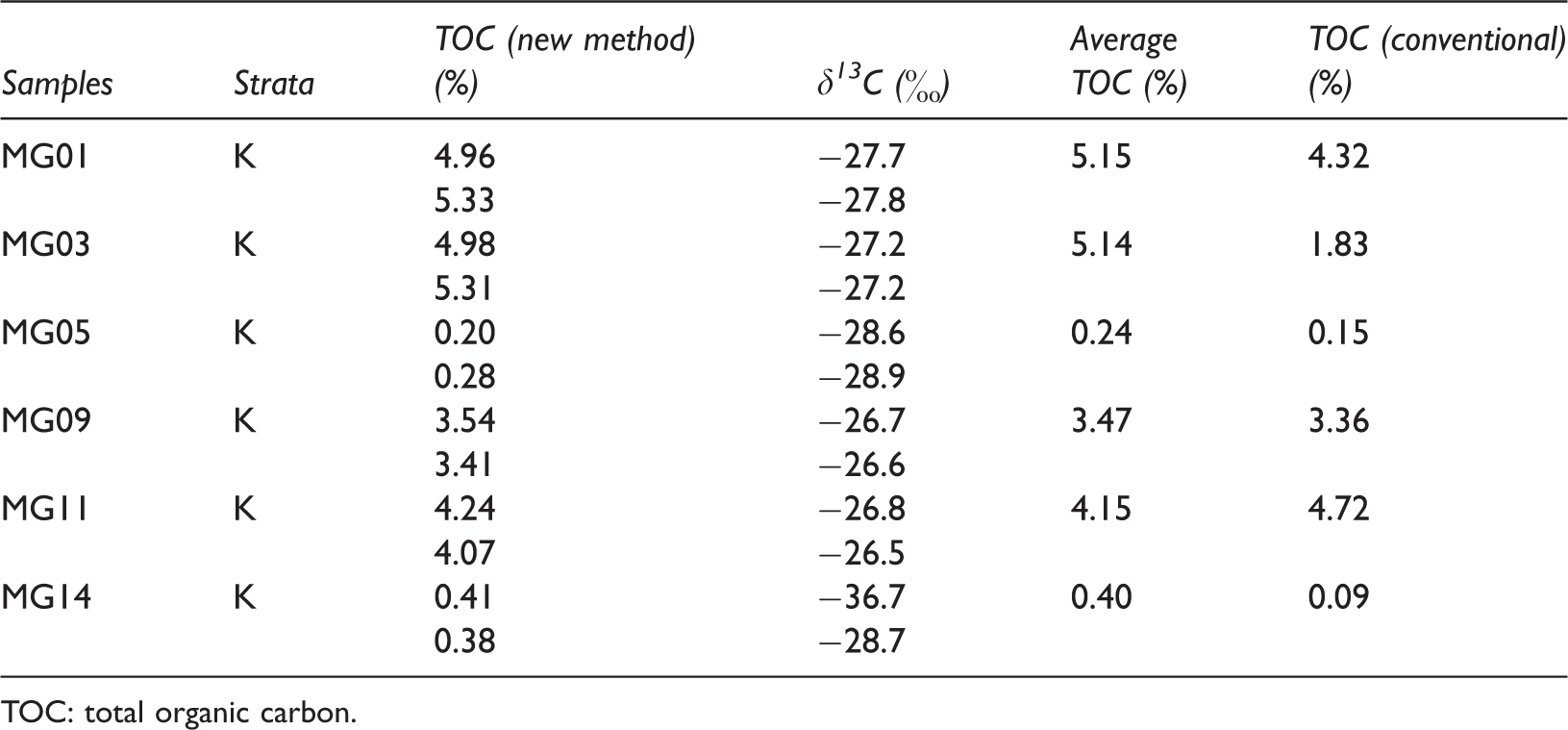
TOC: total organic carbon.
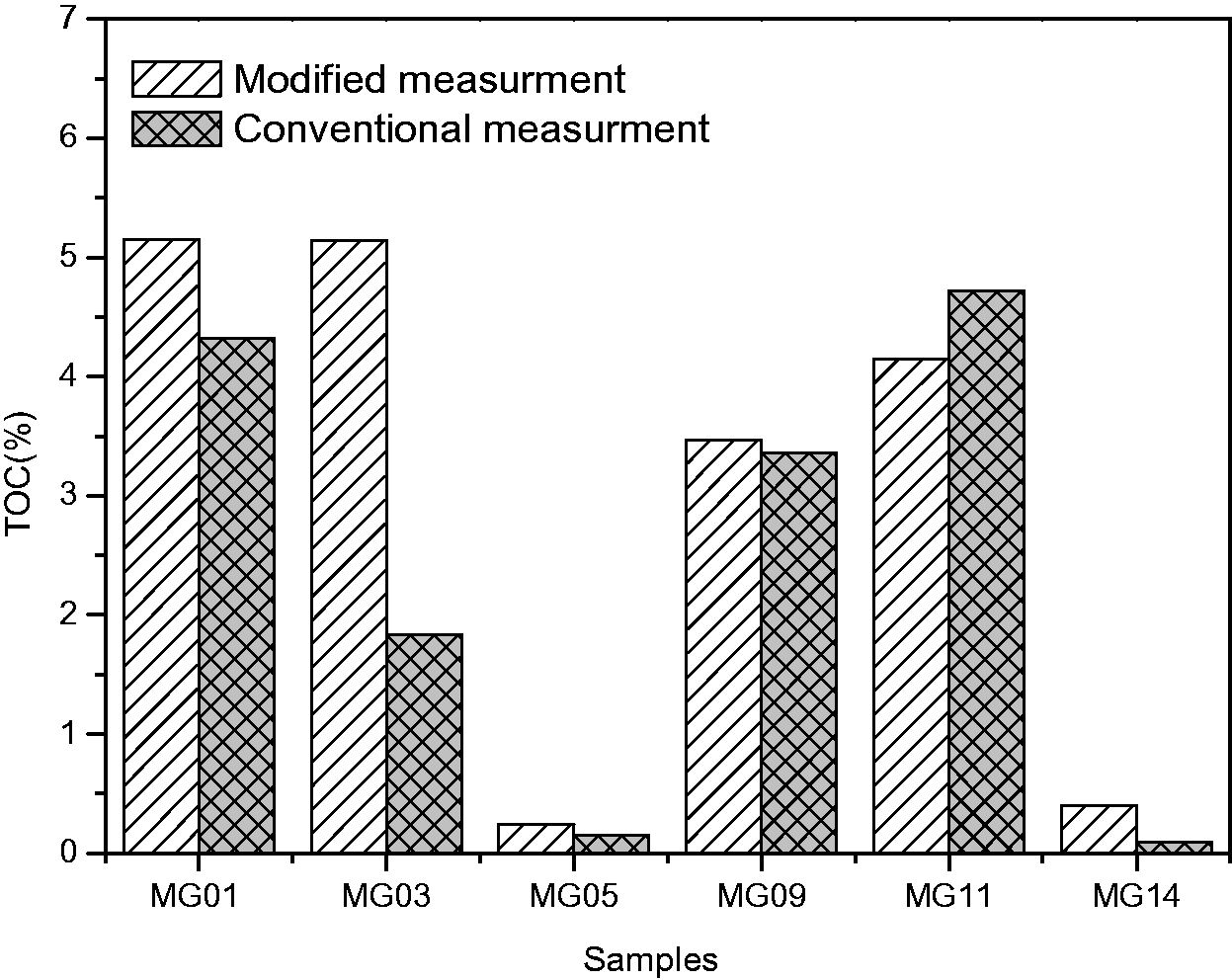
Comparison of TOC values with conventional and modified method of high maturity carbonate from Cretaceous Eagle Ford Formation in Texas, USA. TOC: total organic carbon.
The high maturity carbonaceous source rocks collected from the Ordos Basin in China were also analyzed for their TOC contents with the conventional “acid-rise” and new “clay-paste” methods (Table 2 and Figure 2). The data show that the new method yielded TOC values much greater than those of the conventional method, except for three samples (D67-3, XF5-5, and DB8-8) which have very similar TOC contents by both the methods.
Results of conventional TOC analysis and new method of high maturity carbonate rocks collected from Ordos Basin, China.
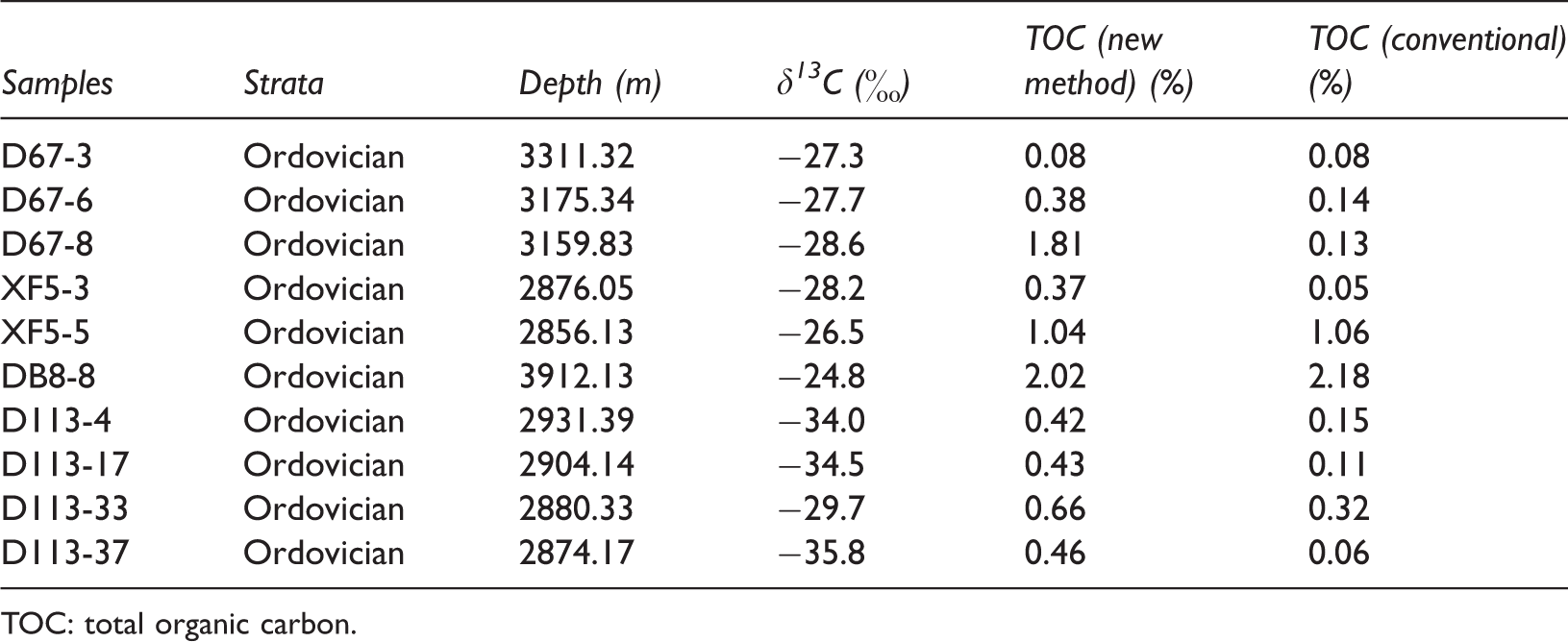
TOC: total organic carbon.
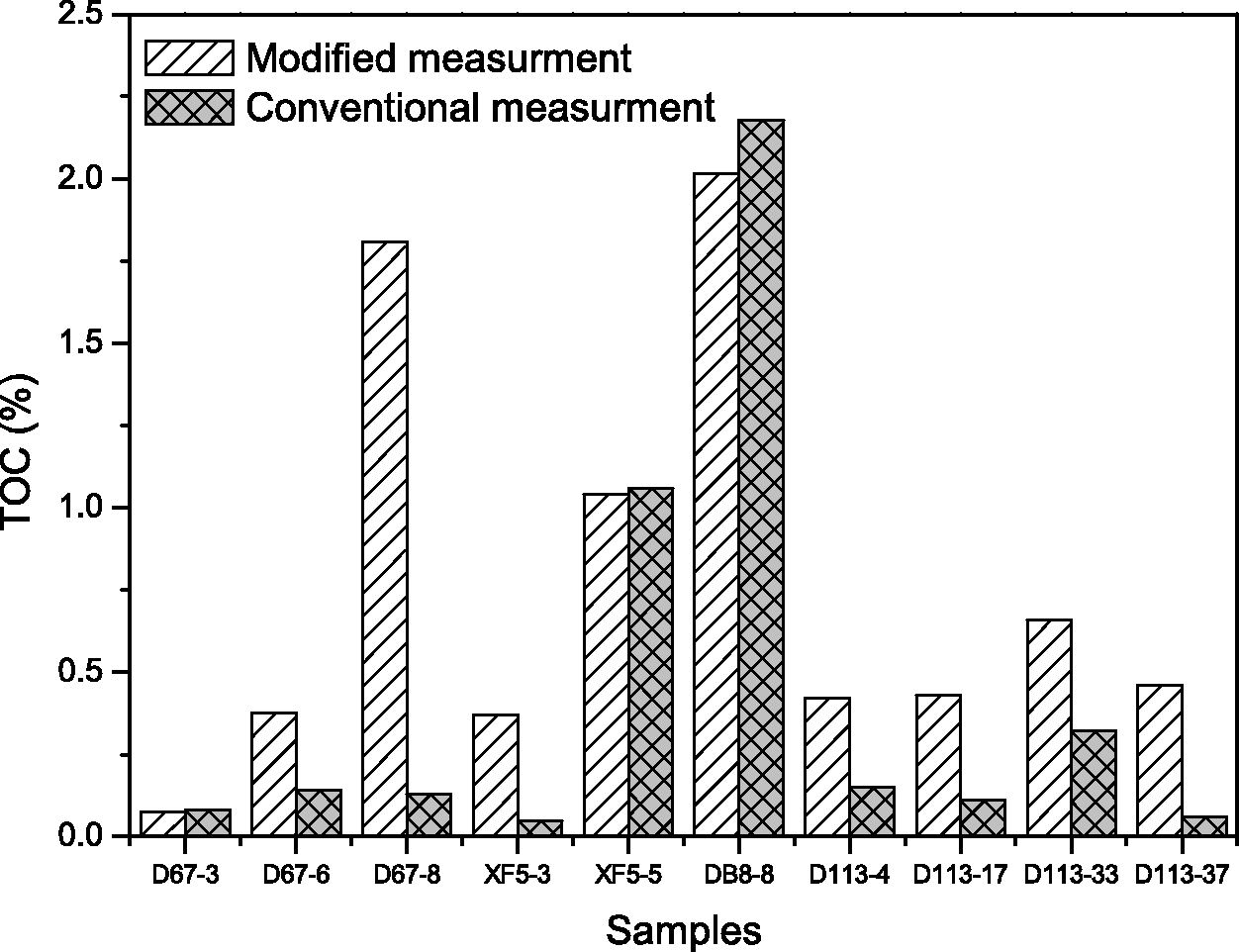
Comparison of TOC values with conventional analytical method and new method of high maturity carbonate from Ordos Basin, China. TOC: total organic carbon.
Some organic matter is released from carbonate with acidolysis and dissolved in acid solution. However, the acid solution is included in the modified approach it is not included in the conventional method. Hence, the TOC results in the modified method are greater than that in conventional method for majority samples.
The fraction of acid-soluble organic matters and its geological significance
The acid solution is included in the measurement scope of the modified method. Hence, the difference of the results between the modified and conventional measurements represents the fraction of acid-soluble organic matters in source rocks. The relationship between the fraction of acid-soluble organic matters and conventional TOC values for both low- and high-maturity carbonaceous rocks (Eagle Ford and Ordos) shows no obvious correlation (Figure 3). It shows that the fraction of acid-soluble organic matter in the source rocks is greater than TOC of conventional measurement in some samples, meanwhile same samples are characterized by low acid-soluble organic matters. The measurement results of those three samples (D67-3, XF5-5, and DB8-8) by the two approaches are almost equal, indicating that the acid-soluble organic matters in those three samples are of limited proportion in TOC.
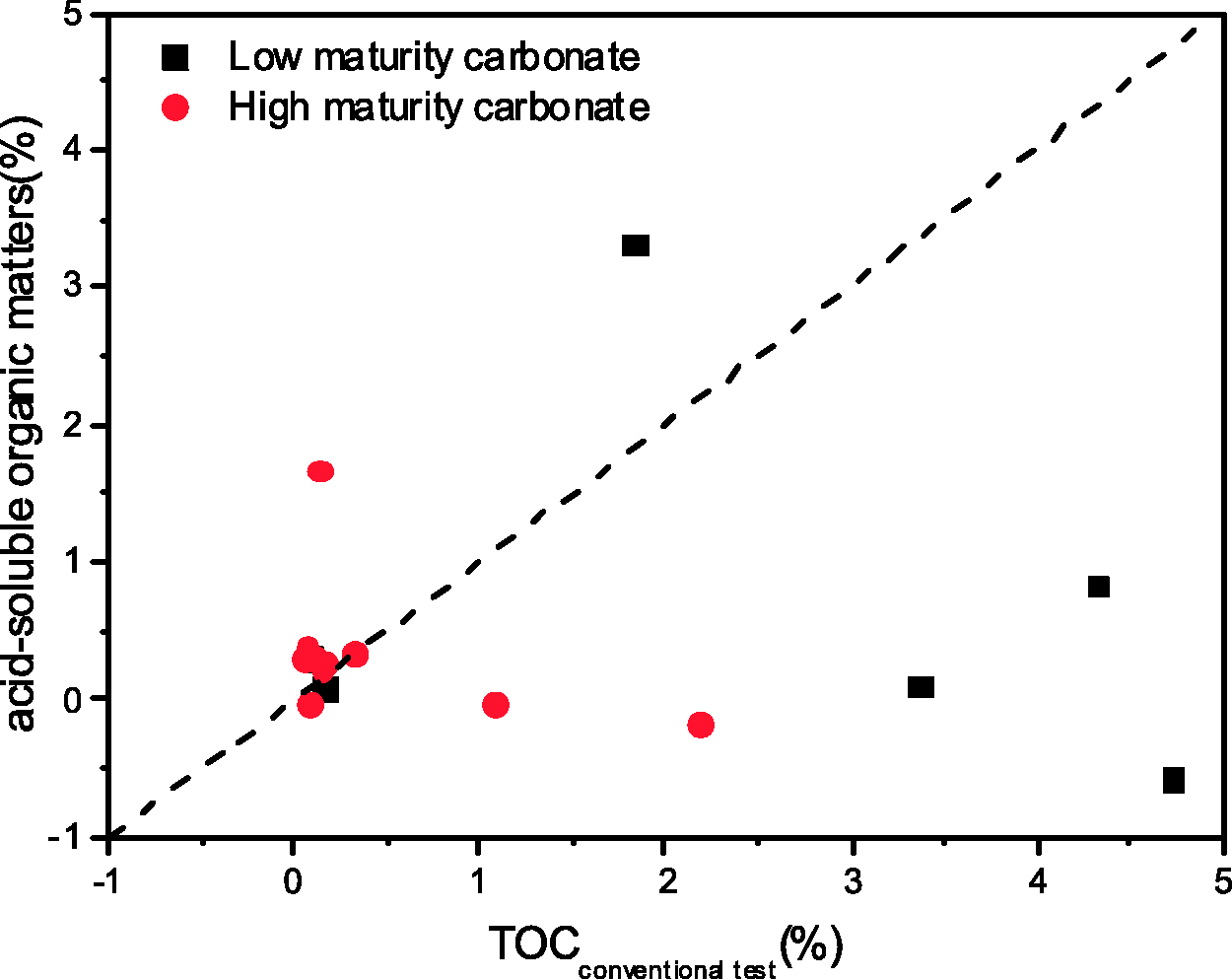
Plot of acid-soluble organic matters versus TOCconventional test in both low maturity carbonate of Cretaceous Eagle Ford Formation in Texas, USA and Ordos Basin of China. TOC: total organic carbon.
The long chain organic acids and asphaltene fractions were found to be adsorbed on the surfaces of the carbonate minerals (Pons-Jiménez et al., 2014; Subramanian et al., 2016; Suess, 1970; Zullig and Morse, 1988). Meanwhile, organic matters can be included in carbonate during formation process (Wang and Cheng, 1997). However, acid treatment can destroy the structure of the carbonate minerals and release the organic matters combined with carbonates and in carbonate inclusion. Hydrophilic organic matters at free and carbonate combined states can be dissolved in acid solution. Previous study indicates that the hydrophilic organic matters may include hydrophilic organic acids, alcohols, aldehydes, phenols, and ketones (Wang and Cheng, 2000). The results of measurements of acid-soluble organic matters suggest that the fractions of acid-soluble organic matter in those source rocks depend on many factors, including depositional environments and burial history among others.
Implications for the evaluation of hydrocarbon source rocks
The marine carbonate formations in China are characterized by deep burial depths, high maturity, and low TOC values (Borjigin et al., 2006a, 2006b). A series of giant gas fields have been discovered in the Ordos Basin of China. The reservoirs of Jingbian gas field are Ordovician weathered crusts (Li et al., 2008; Liu et al., 2009), and Daniudi, Yulin, Wushenqi, and Sulige gas fields had been discovered within Carboniferous–Permian (C–P) formations (Cai et al., 2005; Dai et al., 2005; Liu et al., 2009). It is widely acknowledged that the gas in Jingbian gas fields is a mixture of coal-generated gases derived from the C–P formations and oil-associated gases derived from Lower Paleozoic marine carbonate rocks. However, a dominant gas source is still controversial. Some believe that the gas is mainly derived from the C–P formations (Dai et al., 2005; Hu and Zhang, 2011;Wang et al., 2015, 2017). The others hold the view that the gas is mainly derived from the Lower Paleozoic carbonate rocks (Liu et al., 2009; Xu et al., 1996). The indicators utilized in those studies include carbon isotope compositions of methane (δ13C1) and ethane (δ13C2), hydrogen isotope of methane (δD1), and biomarker compounds. However, investigation of the natural gas source by interpreting those indicators presents inconsistent results, revealing the risk of utilizing isotopic approaches in studying complex gas reservoirs.
Few studies were concerned with the hydrocarbon source rocks and the applicability of TOC analytical methods of these carbonate source rocks with high maturity and low TOC values. It is acknowledged that the carbonate rocks of the Lower Paleozoic are highly matured with type II1 kerogen (Li et al., 1990). However, the TOC values of the Lower Paleozoic carbonate rocks determined with the conventional method range from 0.2 to 0.35% (Guan et al., 1993), which is lower than the limit TOC values of carbonate hydrocarbon source rocks. Undoubtedly, the Lower Paleozoic carbonate rocks have not been considered as hydrocarbon source rocks in the previous studies.
Table 2 and Figure 2 show the TOC values of 10 carbonate core samples from the Ordos Basin, determined with both conventional and new “clay-paste” methods. The new method shows that a majority of the Ordos samples have higher TOC values compared with the conventional TOC test. The new method TOC values of some samples are greater than 0.5%, which is lower than the limit TOC values of carbonate hydrocarbon source rocks (Chen et al., 2012). The results suggest that TOC contents of those samples have been underestimated and more organic matter likely exist in the carbonate rocks before the generation of hydrocarbon. Even though it cannot be concluded that the carbonate rocks of Lower Paleozoic of Ordos Basin can act as hydrocarbon source rocks and have the potential to form gas reservoirs by the modified TOC measurements, it reveals that acid-soluble organic matters are ignored in the previous studies. Therefore, neglecting the acid-soluble fraction in the TOC analysis underestimates the hydrocarbon generation potential of carbonaceous rock sequences in Chinese and other sedimentary basins.
Conclusions
The comparison of TOC values of the carbonate-rich source rocks determined by the conventional “acid-rinse” and new “clay-paste” methods indicates that the acid solution contains organic matters and the “acid-rinse” method for carbonate-rich rocks underestimates the TOC values due to the fact that the fraction of acid-soluble organic matters are not included in the conventional method. Therefore, the TOC values determined by the conventional “acid-rise” TOC analysis may lead to serious underestimation of hydrocarbon-generating potential. Meanwhile, the results of TOC analysis of low to high maturity carbonaceous source rocks with low TOC values by the new “clay-paste” method showed no clear relationship of the fraction of acid-soluble organic matters with maturity and TOC values. It indicates that many factors, including depositional environments and burial history, may influence the fractions of acid-soluble organic matter in carbonate source rocks. Therefore, the fraction of acid-soluble organic matters cannot be neglected in evaluating the hydrocarbon-generating potential of carbonate-rich source rocks. Thus, accurate measurements of TOC values are needed to evaluate properly hydrocarbon generation potentials of carbonate-rich sources rocks in petroleum-producing sedimentary basin in China and elsewhere.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Foundation of Sinopec Key Laboratory of Petroleum Accumulation Mechanisms, Foundation of State Key Laboratory of Shale Oil and Gas Enrichment Mechanisms and Effective Development, West Light Foundation of The Chinese Academy of Sciences, National Nature Science Foundation of China (Grant No. U1663202), U.S. Department of Energy Geosciences program (DE-SC0016271) and the Key Laboratory Project of Gansu Province (Grant No. 1309RTSA041).
