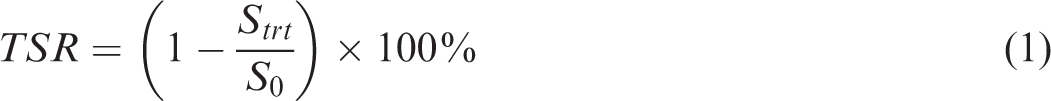Abstract
This paper presents an experimental study on the effect of potassium hydroxide concentration and reaction time on coal properties during desulphurization of high sulphur Indian coal. Experimental results showed that maximum of 36.79% of total sulphur can be removed from coal by potassium hydroxide leaching with 20% potassium hydroxide concentration and 24 h contact time at atmospheric condition. Total sulphur reduction was contributed by 45.03–49.01% pyritic sulphur, 24.10–33.73% sulphate sulphur and 20.93–30.62% organic sulphur depending on potassium hydroxide concentration of 5–20% and reaction time of 6–24 h. Experimental result shows that ash% of coal increased from 8.33 to 14.09% due to accumulation of insoluble potassium aluminosilicate in the coal. Gross calorific value of coal decreased from 6854 to 6084 kcal/kg due to increase of ash content of coal and loss of some combustible matter in leaching process. Physico-chemical characterizations of raw and treated coal samples were carried out by using field emission scanning electron microscope, Fourier transform infrared, petrographic analysis, thermogravimetric analysis and differential thermogravimetry to appraise the product quality. Field emission scanning electron microscope and coal petrographic image infer the occurrence of pitting and cavitation in the coal structure due to removal of pyrite sulphur (1.51–1.21%) from coal which causes escalation of vitrinite (67.65–70.43%) and few amounts of inertinite (18.03–18.86%) and liptinite (1.24–1.27%) as macerals. Thermogravimetric analysis–differential thermogravimetry analysis infers the improvement in combustion characteristics of treated coal by reduction in peak combustion temperature.
Introduction
Emission of sulphur dioxide from thermal power plants is a major environmental problem in the 21st century. Sulphur is present in coal mainly as pyritic, sulphate and organic forms. Pyritic sulphur is present in coal as part of mineral matter whereas the organic sulphur is present as organic compound which is an integral part of the coal matrix. During combustion, these organic sulphur compounds burn along with coal to produce sulphur oxides, whereas pyritic and sulphate sulphur decompose to produce inorganic oxides and SOX (Frigge et al., 2017). As a result, large quantities of coal with high gross calorific value (GCV) cannot be used in thermal power plants. If such high sulphur coals are desulphurized or SOX pollution is controlled, large quantities of coal with high GCV can be used in the combustion process. To overcome this problem associated with high sulphur coal, various techniques like coal desulphurization, SOX emission control, etc. have been proposed by various researchers (Park and Kim, 2006; Wang et al., 2017). Reduction of SOX emission from combustion of coal can be carried out by pre-combustion, during combustion and post-combustion of coal. Some methods such as physical removal of sulphur from coal before combustion, limestone (CaCO3) addition to the combustor to absorb sulphur oxides during combustion and using flue-gas scrubbers to remove sulphur oxides from plant gas emissions after combustion are used in current practice (Park and Kim, 2006; Wang et al., 2017). Among these methods, sulphur removal before combustion seems to be more promising convening of environmental and economic aspects (Ambedkar et al., 2011). Research on desulphurization of pre-combustion coal has been carried out by many researchers (Ambedkar et al., 2011; Park and Kim, 2006; Tian et al., 2016; Wang et al., 2017). Some of the literature survey in which sulphur is removed by chemical treatment (Abdollahy et al., 2006; Longjun et al., 2012; Singh et al., 2013), bio-desulphurization (Cara et al., 2005; Gonsalvesh et al., 2013), oil agglomeration (Sahinoglu and Uslu, 2015), flotation (Abdollahy et al., 2006; Saydut et al., 2008) and ultrasonic treatment (Saikia et al., 2014a, 2014c). All processes include inherent advantages and disadvantages. Considering the mass of coal to be treated for industrial applications, coal desulphurization at elevated temperature, bio-desulphurization, ultrasonic treatment, etc. are not commercially practicable. In this context, desulphurization at ambient temperature and with ordinary equipment involvement are very much advantageous for large-scale applications (Mukherjee, 2003).
On the other hand, most of the studies reported in literature are concerned with sulphur removal efficiency as a substitute for treatment on physico-chemical property rather than ash%, GCV, fuel ratio with combustion performance parameters. Changes in these physico-chemical properties affect the entire utilization capabilities of the treated coal. Due to variations in GCV, coal feed rate in thermal power plant varies and sometimes major changes in coal flow circuit are necessary. Increasing in ash% as in line with the results is an indication of higher solid waste disposal problems arising in plants. Subsequently, changes in ignition properties of coal entirely alter the heat release rate in the boiler. Hence it is very much necessary to evaluate the effects of chemical treatment for desulphurization on the physico-chemical properties of coal.
Large quantities of high sulphur coal reserves are available in Assam and other states in the northeastern region of India (Mukherjee and Borthakur, 2003). Typical Assam coals possess low moisture content, subbituminous in rank, contain high volatile matter (VM) (30–50%), high calorific value (6500–8000 kcal/kg), low ash content (below 15%) and sulphur content (2–7%) (Mukherjee and Borthakur, 2003). High sulphur content in coal inhibits the use as a thermal coal or metallurgical coal, in steel and powder industries. Some researches have been carried out in past by Saikia et al. (2014a, 2014b, 2014c), Mukherjee and Borthakur (2001, 2003, 2004a, 2004b) and Mukherjee et al. (2001) using different combinations of alkali (potassium hydroxide (KOH)–NaOH), ionic liquids, acids (HCl, HNO3 and H2SO4), ultrasound (20–40 kHz) in the presence of H2O2 and other chemicals. Their studies convey that these techniques can be used for desulphurization of Assam coal. Maximum desulphurization efficiency of 53% was reported by Mukherjee and Borthakur (2003) using 16% KOH followed by 10% HCl treatment at 150°C temperature and 8 h reaction time at atmospheric pressure. However, most of the research work was focused mostly on the desulphurization efficiency and changes in coal matrix (characterization using FESEM, FTIR, thermogravimetric analysis–differential thermogravimetry (TGA–DTG), coal petrography, etc.). For commercial implementation of desulphurization of non-cooking coal, effects of desulphurization on properties like VM, FC, ASH, C, N, H, S and O are considered mostly as important parameters which were least priority in previous work. In this context, the present work reports the effects of KOH concentration (varies from 5 to 20%) and contact time (varies from 6 to 24 h) treatment on different physico-chemical properties of coal like proximate analysis, ultimate analysis, GCV and combustion parameters which have a significant impact on applicability of desulphurized thermal coal. Also experimental effort has been given towards the development of easily implementable process like only KOH treatment with stirring at ambient temperature in place of using costly ultrasound energy presence of H2O2. treatment stirring at ambient temperature. Further TGA–DTG analyses have been carried out to see the effects of KOH wash on the burning characteristics of treated coal. Further, petrography analysis, Fourier transform infrared (FTIR) and field emission scanning electron microscope (FESEM) were also done to observe the effects of KOH treatment on coal structure and changes in its morphological properties.
Materials and methods
About 50 kg of coal sample was collected from Makum Coal Field, Assam, India. Sample about 5 kg of coal sample of −212 µm size was prepared by standard sampling method (Mukherjee and Borthakur, 2003). For desulphurization experiment, 20 g of coal sample was mixed with 100 ml of KOH solution (v/v) in different concentrations (5, 10, 15 and 20%) in a 250 ml beaker. The solution was mixed properly at 200 r/min using a magnetic stirrer (Model 5MLH plus; make: REMI, India) for 15 min of each concentration. Simultaneously, the samples were kept at different time intervals (6, 12, 18, 24 h) in ambient condition (25°C) with stirring speed of 200 r/min for studying the effect of soaking time on sulphur removal efficiency. Thereafter, the sample was filtered using Whatman 42 filter paper using conical flask funnel. The filtrate was dried at 60°C in an air oven till constant weight was attained. The raw coal sample and chemical-treated coal sample were subjected to proximate analysis (ASTM D3173), ultimate analysis using CHNS analyser (Model: Vario EL-III) and GCV value using automatic bomb calorimeter (Model: AC-350 LECO). The different forms of sulphur were calculated using Eschka Method (ASTM D2492). Combustion behaviour of coal was analysed using thermogravimetric analyser (Model: STA 449 F3 Jupiter make by NETZSCH) by heating the coal sample in an oxygen environment at constant heating rate of 10°C/min. Petrographic analysis (Leica, Model DM 2700) was carried out by following BIS Standard, 9127 (Part 3) and the reflectance of maceral was analysed by IS 9127 (Part 5). FTIR spectroscopy (Perkin Elmer Spectra 2, USA) and FESEM image capture through SUPRA’55 MonoCL4 model. Reduction of sulphur was calculated using the following equation
Result and discussions
Characterization of raw coal sample
Table 1 shows the results obtained from proximate analysis of coal. From Table 1 it can be observed that selected coal having 8.33% ash, 42.19% volatile matter and 49.48% fixed carbon with GCV of 6854 kcal/kg. Obtained values of FSI (free swelling index) and caking index are of 1.5 and 5 which are characteristics of thermal coal. Overall fuel ratio of coal is 1.17 as dry basis. Table 2 shows the ultimate analysis results of coal having high sulphur content (4.92%) of which 0.83% sulphate, 1.51% pyritic and remaining organic are present. Calculated value of hydrogen/carbon (H/C) ratio is 0.07 and oxygen content is 12.96%.
Proximate analysis and some quality parameters of samples on dry basis.
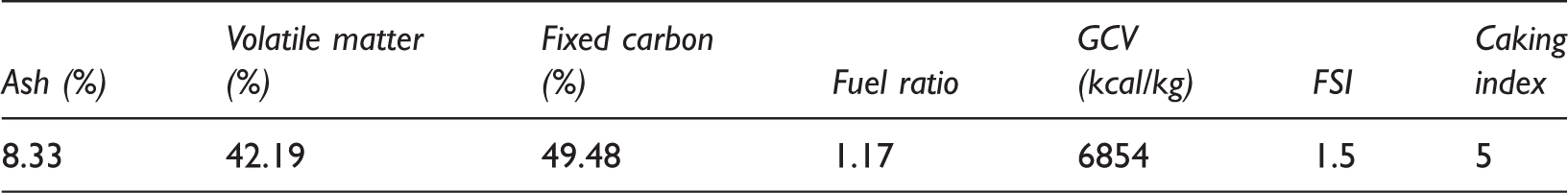
FSI: free swelling index; GCV: gross calorific value.
Ultimate analysis and fuel ratio of samples on dry basis.
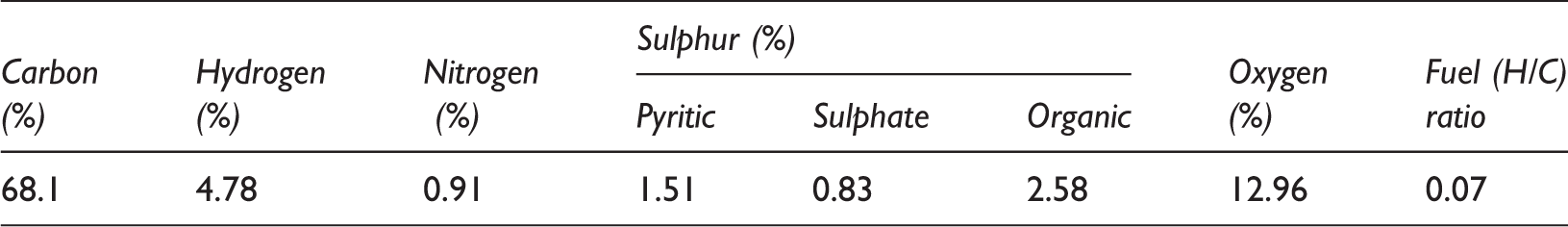
H/C: hydrogen/carbon.
Effect of KOH concentration on ultimate analysis of treated coal
Coal was treated at different concentrations of KOH (5, 10, 15 and 20%) for different contact times of 6, 12, 18 and 24 h to calculate the effect of KOH concentration and contact time on sulphur removal efficiency. Ultimate analysis was carried out to analyse the elemental change (C, H, N, S and O) in coal sample. Results obtained from these experiments are summarized in Table 3. From Table 3 it can be inferred that with increase in KOH concentration and contact time, sulphur removal increases. For example, with 6 h contact time, KOH concentration increases from 5 to 20% and sulphur decreased from 4.92 to 3.5%. Similarly for 20% KOH concentration with increase in contact time from 6 to 24 h sulphur reduced from 3.50 to 3.11%. Decrease in sulphur with increase in contact time may be attributed to breaking of C–S bond, attacking the KOH concentration (Charutawai et al., 2003). The decrement in sulphur content with increment in concentration can be attributed to the solubilization of the sulphates, conversion of pyritic and some organic functional groups like thiols, disulphide, etc. present in the coal to soluble salts (Zhou, 1989). The reduction in sulphur content was found to be 20.12, 24.19, 27.44 and 36.79% with increase in KOH concentration from 5, 10, 15 and 20%, respectively.
Ultimate analysis of treated samples at room temperature.
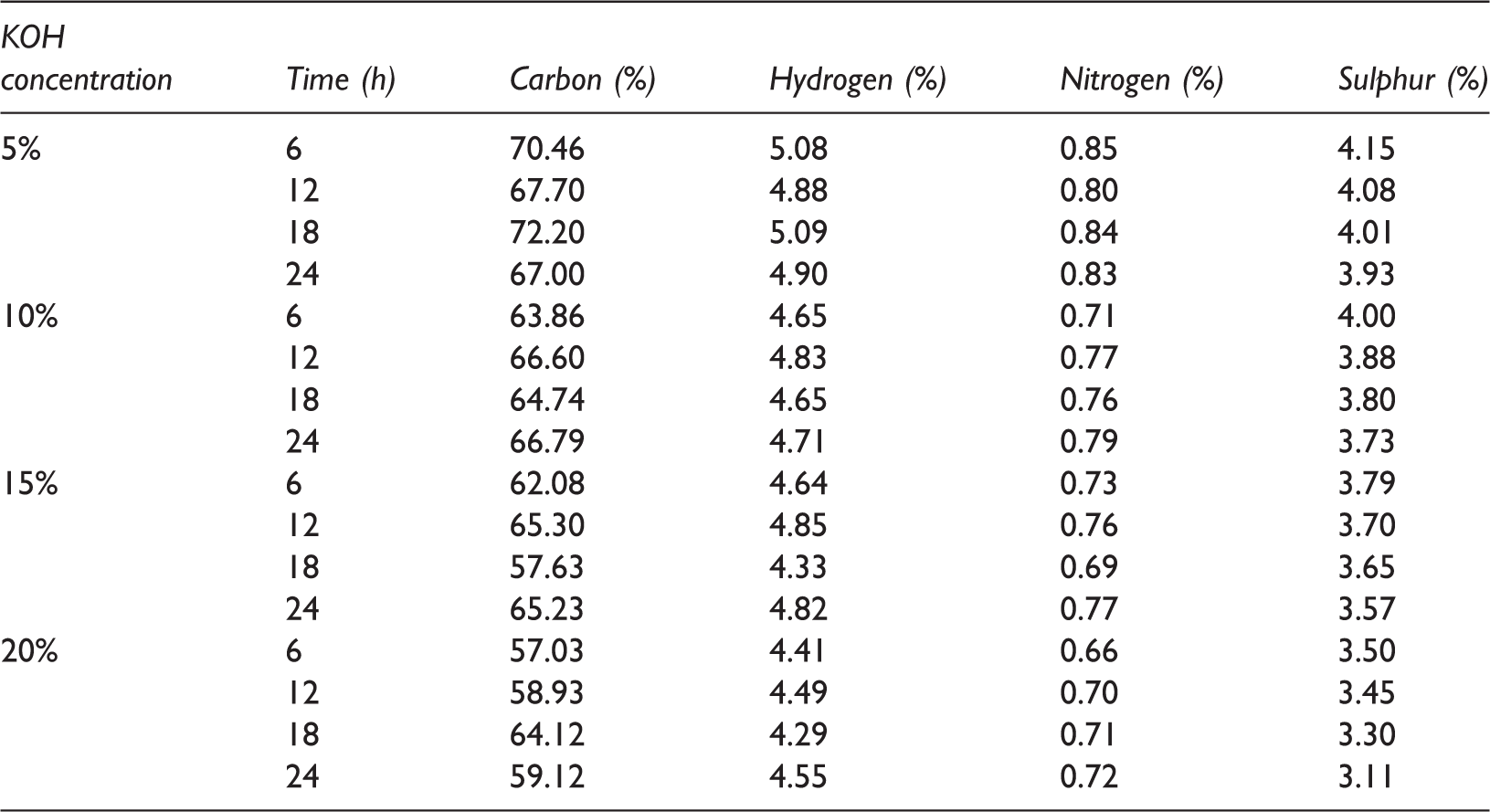
KOH: potassium hydroxide.
Effect of KOH treatment on sulphur removal
The effect of reaction time and KOH concentration on desulphurization is shown in the previous section (Table 3). From Table 3 it can be observed that sulphur content removal increases from 4.15 to 3.50% with increase in KOH concentration from 5 to 20% for 6 h contact time. Similarly for 10% KOH concentration sulphur removal increases from 4.0 to 3.73% with increase in contact time from 6 to 24 h. However, the removals of sulphate, pyritic and organic sulphur are shown in Figure 1.
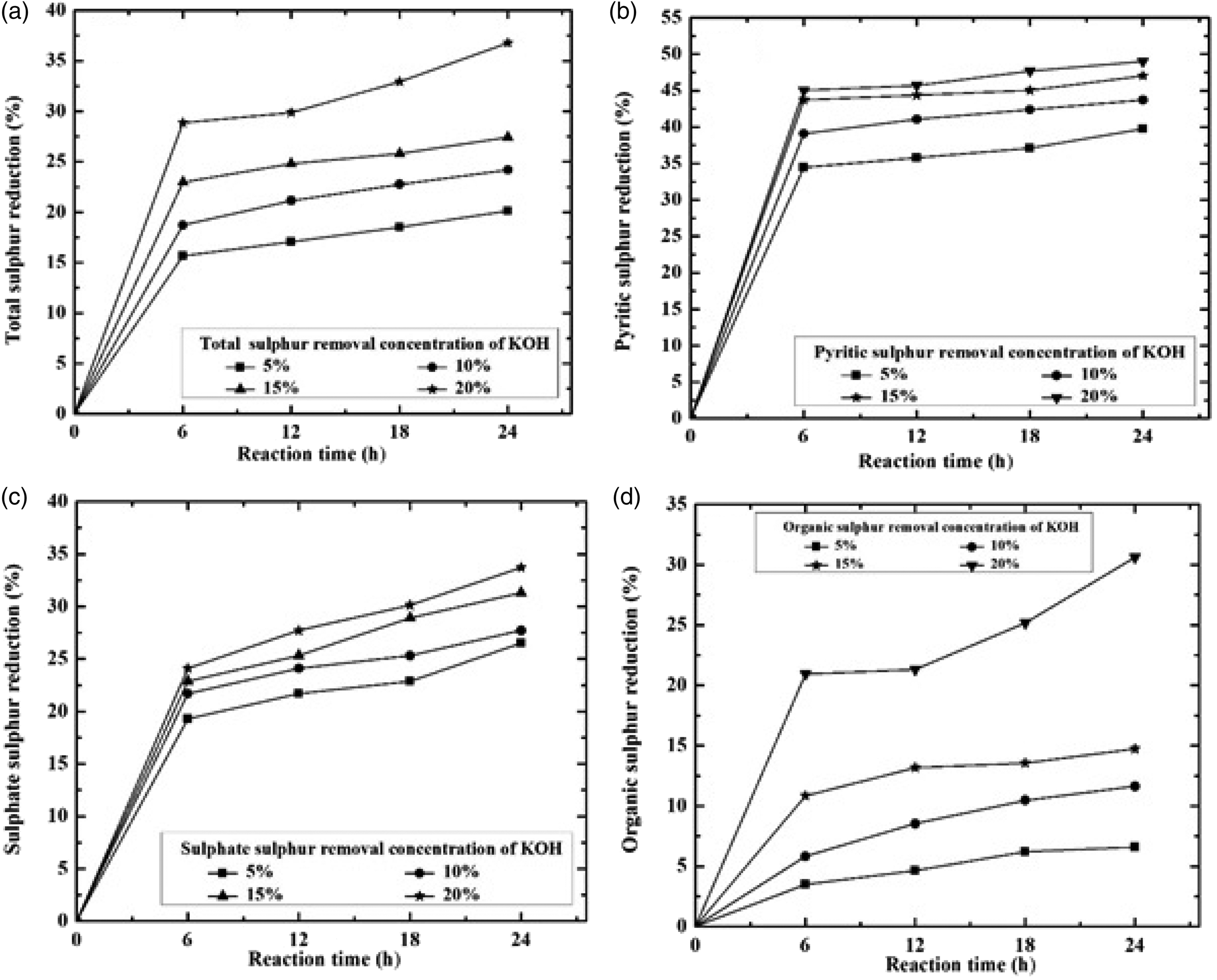
Total sulphur (a) and form of sulphur, (b) pyritic, (c) sulphate and (d) organic sulphur removal as a function of different KOH concentration (%) and reaction time (h). KOH: potassium hydroxide.
The chemical reactions between pyritic sulphur and organic sulphur in coal with KOH treatment take place as follows (Ratanakandilok et al., 2001)
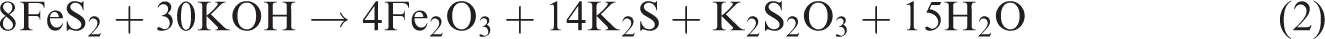
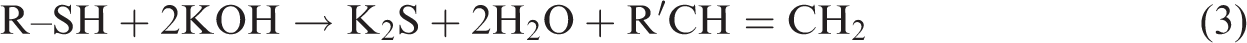
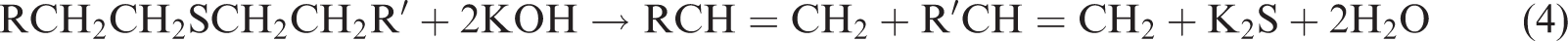
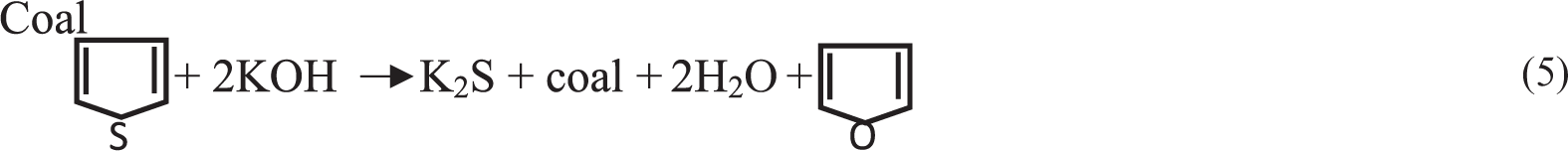
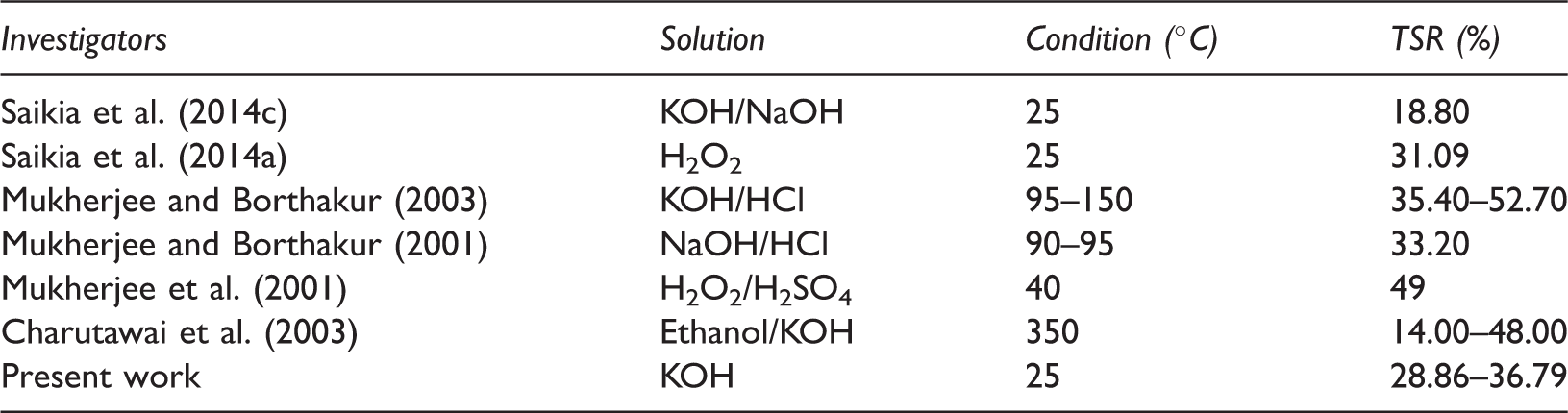
From Figure 1(a) it can be observed that with increase in reaction time and KOH concentration this reduces the sulphur content. For 10% KOH solution, with increase in contact time from 6 to 24 h, total sulphur reduction (TSR) decreases from 18.70 to 24.19%. Simultaneously for 24 h contact time, with increase in KOH concentration from 5 to 20%, the TSR increases from 20.12 to 36.79%. Increase in TSR with KOH concentration was attributed to the solubilization of the sulphates, conversion of pyritic and organic functional groups like thiols, disulphide, etc. present in the coal to soluble salts (Zhou, 1989). Increase in sulphur reduction with contact time may be attributed to carbon–sulphur bond breaking, attacking KOH concentration (Charutawai et al., 2003). However, it can be observed that contact time has the largest effect on desulphurization irrespective of KOH concentration on sulphur reduction. This was due to depolymerization of coal that caused a minor portion of the organic matter to be extracted. From Figure 1(b) it can be observed that with increase in reaction time and KOH concentration, pyritic sulphur reduction (PSR) takes place. For 10% KOH solution, with increase in contact time from 6 to 24 h, PSR decreases from 39.07 to 43.71%. Similarly for 24 h contact time, with increase in KOH concentration from 5 to 20%, PSR increases from 39.74 to 49.01%. Increase in PSR with KOH concentration was attributed to solubilization of the sulphates, conversion of pyritic and some organic functional groups like thiols, disulphide, etc. present in the coal to soluble salts (Zhou, 1989). Increase in PSR with contact time may be attributed to breaking of C–S bond, attacking the KOH concentration (Charutawai et al., 2003). Similar condition is observed for other sulphur (sulphate sulphur Figure 1(c) and organic sulphur Figure 1(d)) which decreased continuously from 6 to 24 h, 20% KOH concentration is 24.1–33.73 and 20.93–30.62%, respectively. Comparison of TSR from this work and previous works is shown in Table 4 at ambient temperature and high temperature conditions. The result achieved at mild conditions compared well with those obtained at low and high temperature with 28.86–36.79% sulphur removal in all cases.
Comparison of total sulphur reduction (TSR) from works by some investigators.
KOH: potassium hydroxide.
Effect of time and concentration on H, C, N and O content
During chemical reaction with KOH concentration, the elemental composition of coal (H, C, N, O) changes. Figure 2 shows the effect of reaction time and KOH concentration on ultimate analysis of coal. For hydrogen (Figure 2(a)), it is observed that the maximum increments (6.09% at 18 h and 5% KOH concentration) with 18 h and 20% KOH concentration give the maximum decrease in hydrogen content (10.25%). For carbon content (Figure 2(b)), it is observed that maximum increments (5.68% at 18 h and 5% KOH concentration) with 6 h and 20% KOH concentration give the maximum decrease (16.26%) in carbon content. This decrease in hydrogen and carbon content has negative impact on coal as it decreases the calorific value. For nitrogen (Figure 2(c)), it is observed that 6 h and 20% KOH concentration gives the maximum decrement in nitrogen content (27.47%) to feed coal. For oxygen (Figure 2(d)), it is observed that 18 h and 5% KOH concentration gives the maximum decrease in oxygen content (36.19%) and maximum increment of 43.82% (6 h, 20% KOH concentration).
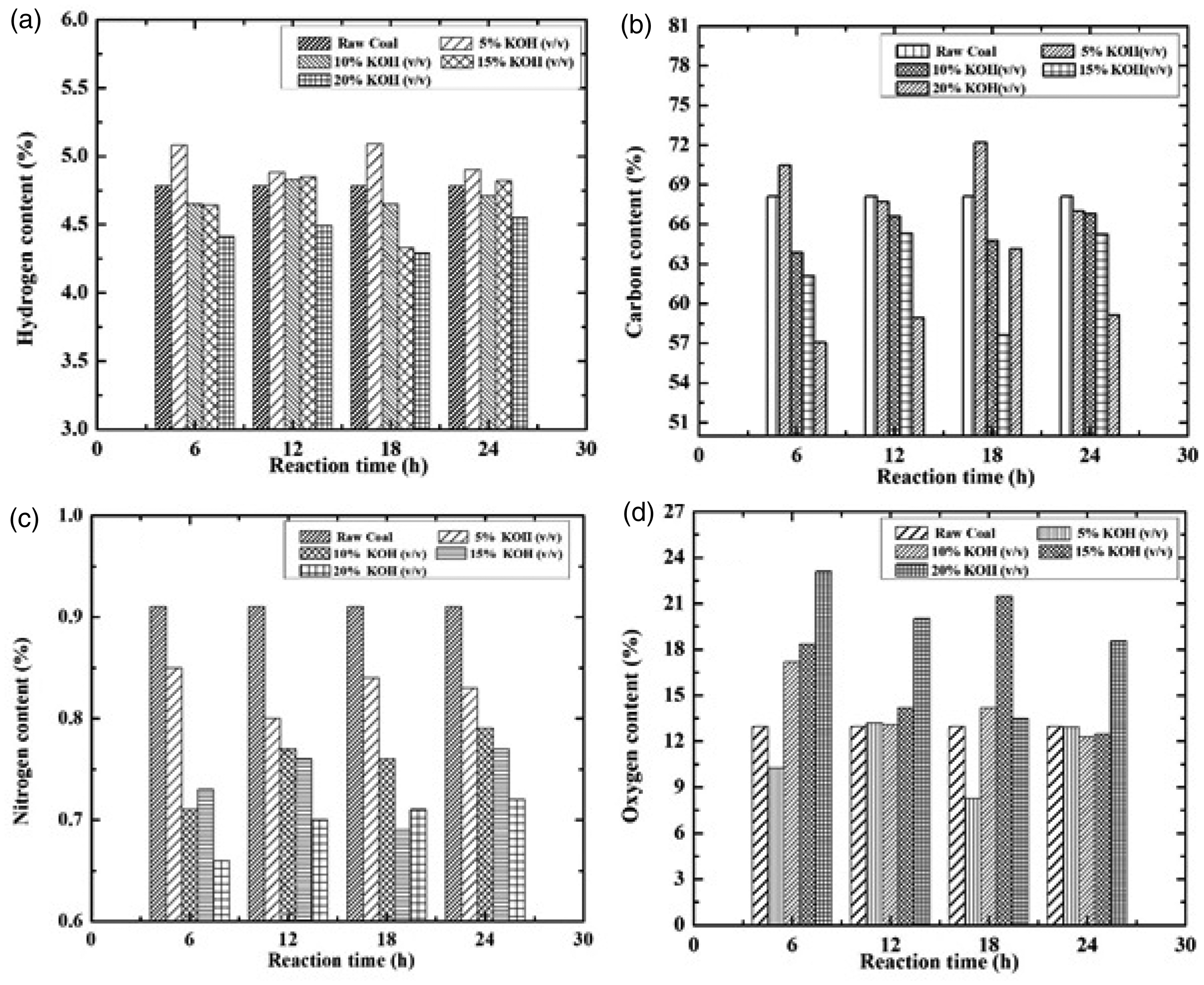
Effect of contact time (h) and KOH concentration (%) on carbon, hydrogen, nitrogen and oxygen content of coal. KOH: potassium hydroxide.
Effect on H/C ratio and fuel ratio
Variation of H/C ratio for coal samples (raw coal and treated coal), along with increase in contact time and KOH concentration can be observed from Figure 3. For 20% KOH solution, the H/C ratio increases from 0.067 to 0.078 with contact time of 6–24 h. Similarly, for 24 h contact time it increases from 0.073 to 0.078 with 5–20% KOH concentration and H/C ratio achieved maximum at 20% KOH concentration. This increase of H/C ratio of fuel during combustion with a decrease in heteroelements like sulphur, nitrogen and metals. Specific gravity of a pure hydrocarbon decreases as H/C ratio increases. Similarly increases in hydrogen content raise the GCV value.
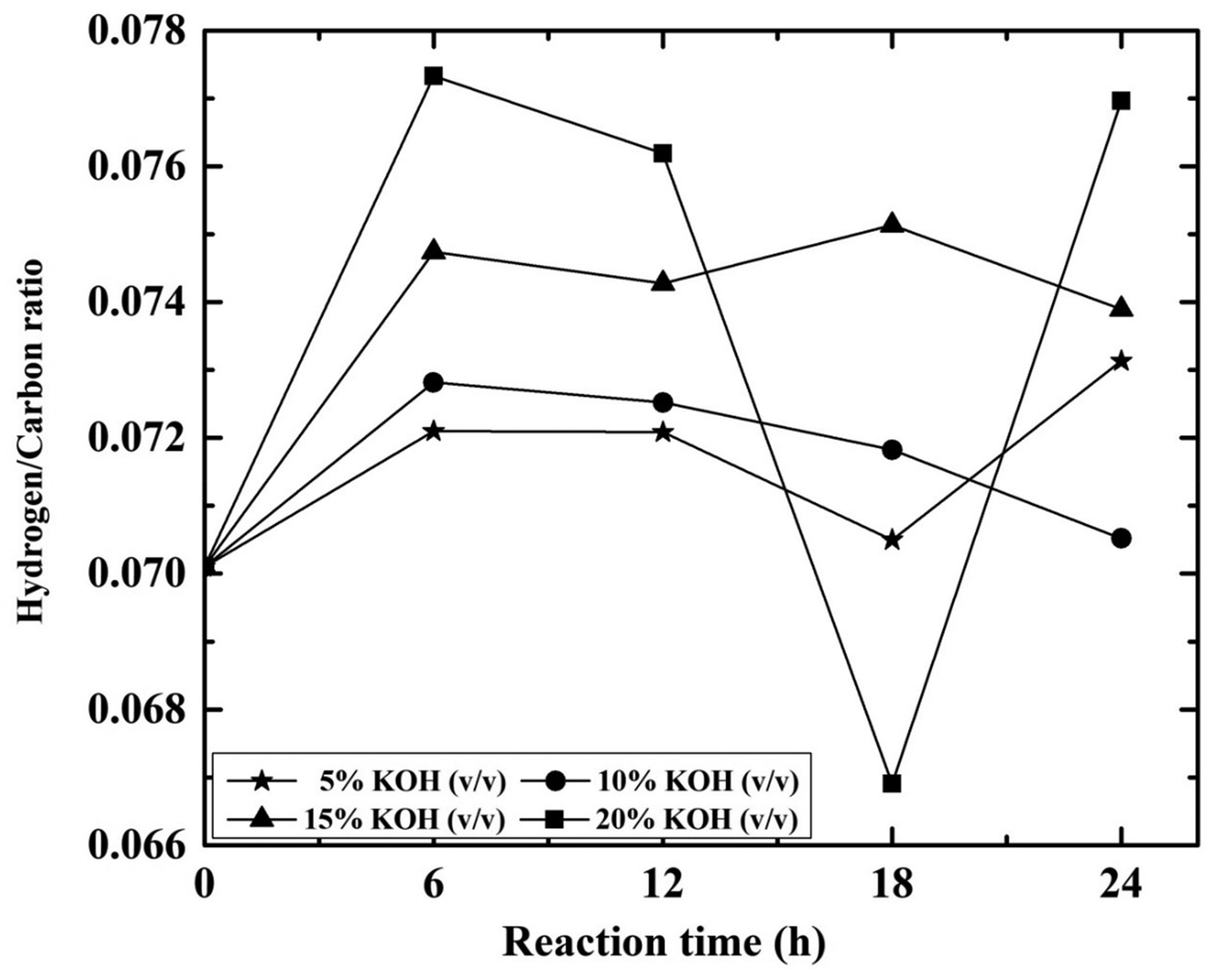
Effect of different KOH concentration (%) and reaction time (h) on H/C* ratio.*H/C: hydrogen/carbon ratio; KOH: potassium hydroxide.
The fuel ratio (FC/VM) explains the effects of process parameters like contact time and KOH concentration as shown in Figure 4. For 10% KOH solution, the fuel ratio increases from 1.18 to 1.27 with contact time 6–24 h but it is maximum at 6 h. Similarly, for 6 h contact times it increases from 1.208 to 1.267 with 5–20% KOH concentration and the fuel ratio achieved maximum at 10% KOH concentration. As the fuel ratio increases, NOx formation decreases during combustion and increases unburned carbon losses. For combustion of high sulphur coal, the fuel ratio should be high (Kurose et al., 2004).
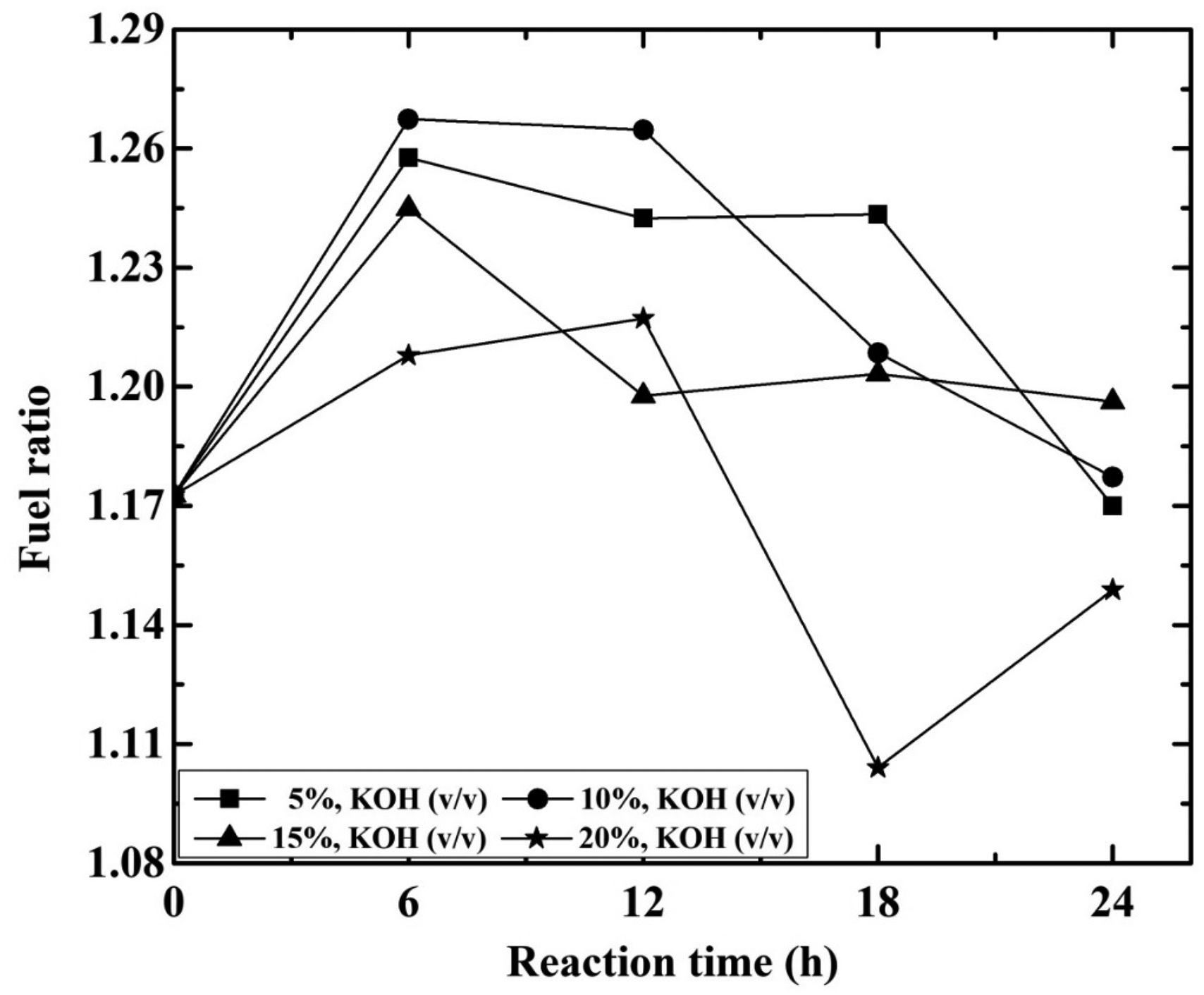
Effect on fuel ratio for different KOH concentration (%) treated samples at reaction time (h). KOH: potassium hydroxide.
Effect of KOH treatment on coal quality parameters
Effect on mineral matter (ash%)
During research, desulphurization process has several coal parameters like ash%, VM% and FC% change. The variations of ash% through desulphurization process are shown in Figure 5. From Figure 5, it has been observed that the ash content of coal is growing with increase in contact time and KOH concentration. For 10% KOH solution, the ash% is increased from 9.60 to 11.69% with increase in contact time from 6 to 24 h. Similarly for 24 h contact time the ash% increased from 10.42 to 13.96% at 5–20% KOH concentration. The increase in ash% with the increase in contact time and KOH concentration was due to reaction of KOH with both carbonaceous part (combined FC and VM) as well as mineral matter part (Ratanakandilok et al., 2001). The reaction of KOH and carbonaceous part results in the generation of some amounts of water soluble compounds which eventually comes out of coal. As a result, coal reduces its carbonaceous content and GCV loss occurs. On the other hand, alkaline KOH solution also reacts with SiO2/Al2O3 or other SiO2/Al2O3 bearing minerals present in coal fines (as a mineral matter it is better reduced with size decrement) to form insoluble silicates or aluminates (Breck, 1973; Singh et al., 2013). To prevent such accumulations of insoluble silicates or aluminates, some authors used alkali followed by acid treatment for reduction of mineral matter in treated coal. The increase in contact time and KOH concentration show negative impact on ash content of coal (Ratanakandilok et al., 2001) due to coal degradation and the remaining potassium content.
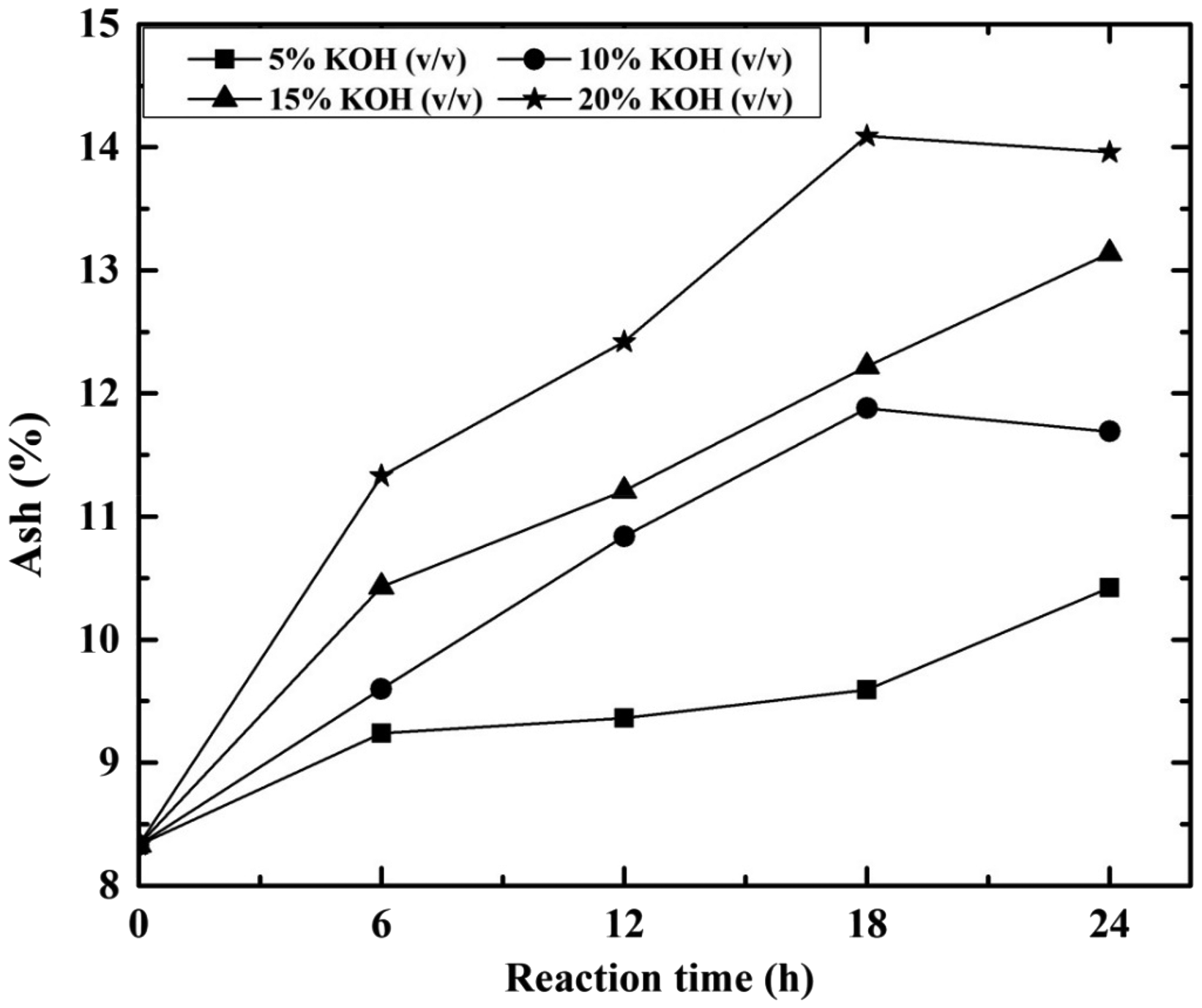
Effect of KOH concentration (%) and reaction time (h) on ash%. KOH: potassium hydroxide.
Effect on GCV
In the present study, the influence of contact time and KOH concentration on GCV of high sulphur coal during desulphurization is presented in Figure 6. The GCV decreases with feed coal (6854 kcal/kg). For 5% KOH solution, the GCV decreases from 6690 to 6528 kcal/kg with increase in contact time from 6 to 24 h. Similarly for 24 h contact time and 5–20% KOH concentration, the GCV is reduced from 6528 to 6084 kcal/kg. The decrease in calorific value revealed that coal energy value was adversely affected instead of enhancement (Mishra et al., 2014). In addition, hydrogen and carbon contents play a significant role in GCV analysis. Further increase in ash content simultaneously decreases the calorific value.
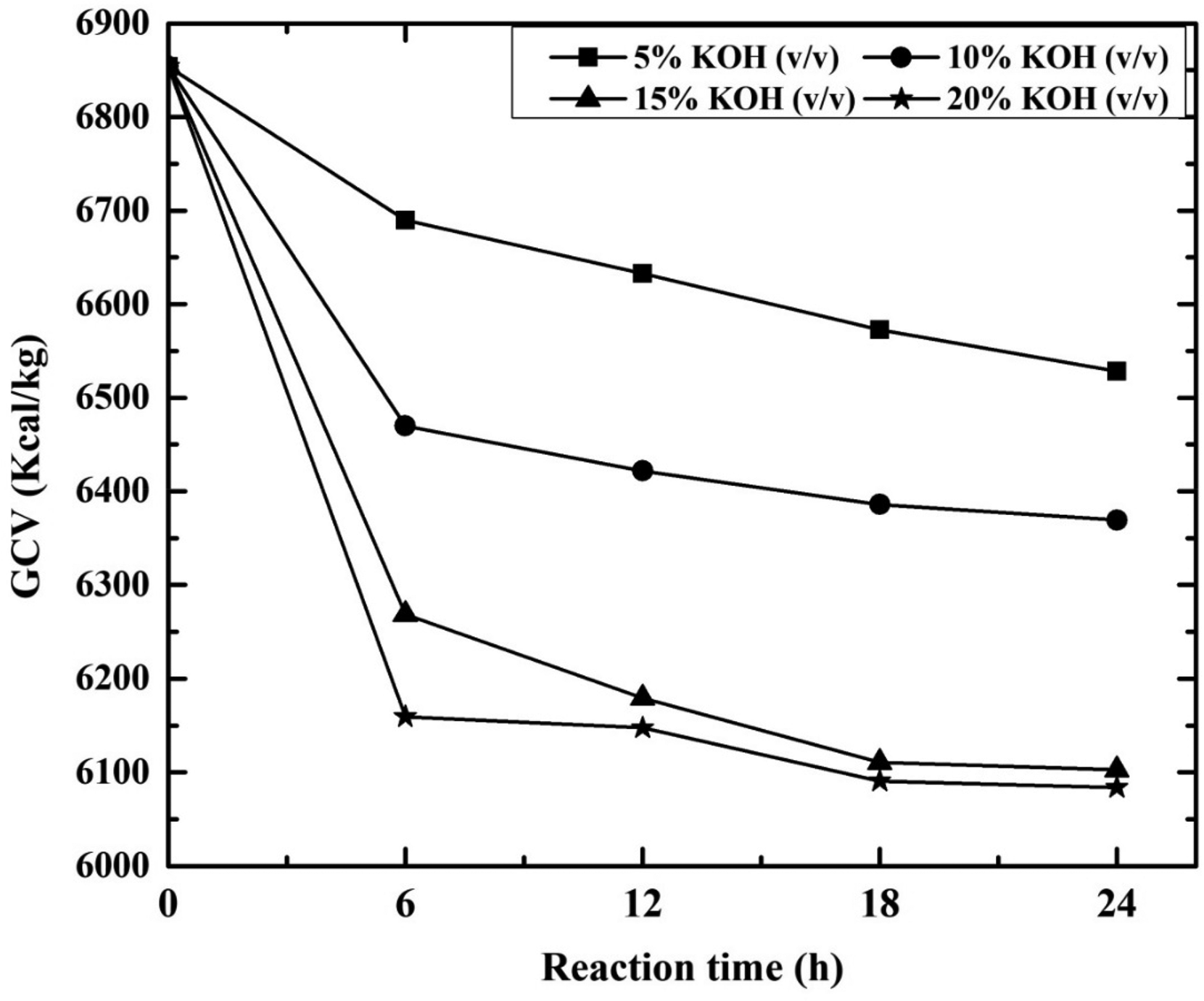
Effect of different KOH concentration (%) on GCV with reaction time (h). KOH: potassium hydroxide.
Effect on organic sulphur removed and heating value loss
Changes with heat values occurs due to enrichment of mineral matter during the chemical treatment and removal of organic matter from coal. Total removed organic sulphur with respect to heating value loss is shown in Figure 7. After leaching process for 10% KOH solution, the heat value losses and organic sulphur removed from 5.61 to 7.07 and 5.81 to 11.63%, respectively, at 6–24 h contact time. Similarly for 24 h contact time the heat value losses and organic sulphur removed from coal range 4.76–11.24 and 6.59–30.62%, respectively, at 5–20% KOH solution. GCV losses also occur due to combustible material loss by dissolution or oxidation of coal samples during leaching process (Karaca and Ceylan, 1997). The results in Figure 7 indicate that, despite some combustible material loss, the heat value of treated coal is relatively decreased compared to original values of sample.
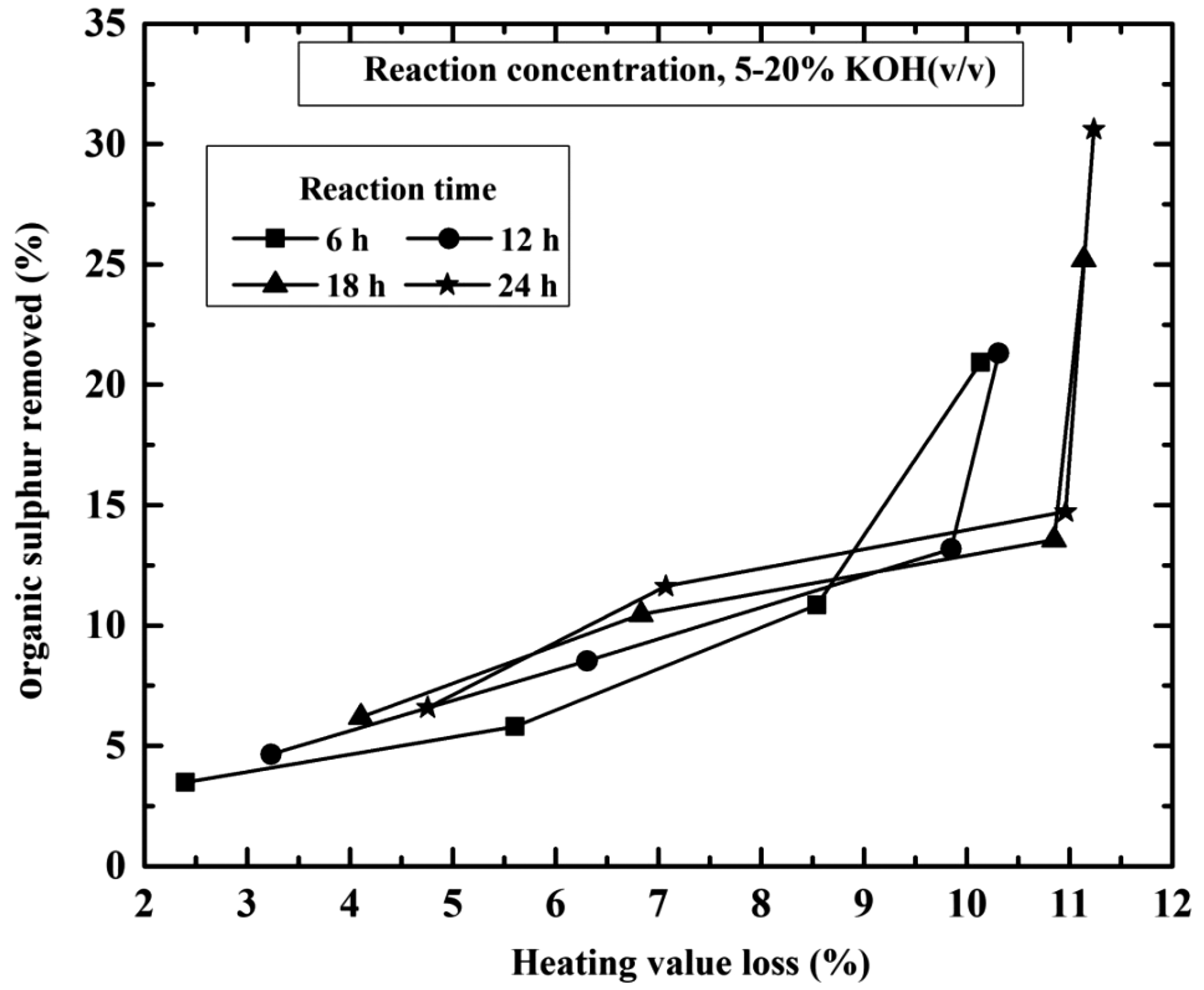
Effect of organic sulphur removal percentage with respect to (%) loss of heat value at different reaction times (h) and KOH concentration (%). KOH: potassium hydroxide.
Combustion characteristics (TGA–DTG) of coal samples
The thermal analysis curves of TGA and DTG of the raw and treated coal samples were represented in Figure 8. TGA plot shown in Figure 8(a) indicates minimal changes in weight loss in the temperature region 30–150°C, signifying low moisture content. Above temperature 150–350°C, similar weight gain is observed for all the samples. This is probably due to adsorption of O2 gas in porous structure of the sample. For raw coal and 5% KOH concentration, O2 absorption is more compared to 10% KOH, 15% KOH and 20% KOH signifying high concentration KOH 10, 15 and 20% coal to be less porous. Lowering of diffusion rate and O2 adsorption in the high concentration sample is due to reduced porous structure in the samples. Weight loss due to VM release starts after 300°C for all the samples. After 320°C, rapid weight loss was obtained indicating the initiation of combustion process. After 500°C raw coal weight loss became negligible signifying no more combustibles left to burn and completion of combustion process. However, 20% KOH sample shows end of combustion after 530°C and thereafter weight loss becomes negligible. This is due to high burnout temperature and reduced porous structure of the sample which results in the delay of combustion process.
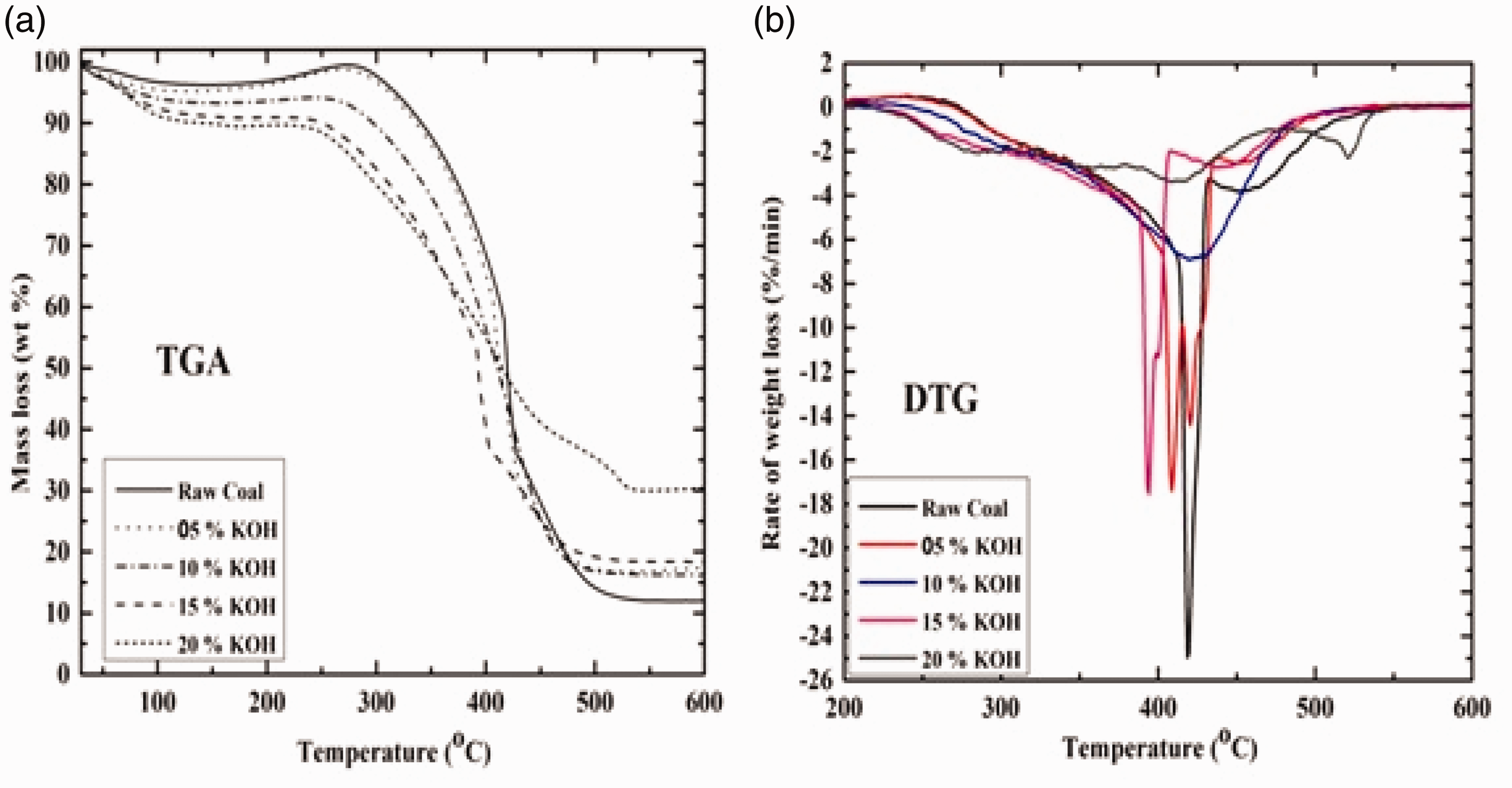
TGA (a) and DTG (b) spectra of raw and treated coal samples at different reaction times (h) and KOH concentration (%). DTG: differential thermogravimetry; KOH: potassium hydroxide; TGA: thermogravimetric analysis.
Characteristics temperature during combustion process can be determined by DTG analysis as shown in Figure 8(b). From Figure 8(b) a distinct variation in the combustion profile of raw sample and high concentration sample (5, 10, 15 and 20%) is observed. From Figure 8(b) it can be inferred that raw coal and 20% KOH sample start burning at lower temperature around 250°C, whereas sample concentration with 5% KOH, 10% KOH and 15% KOH starts burning at high temperature (260–290°C). However, there is a decrease in ignition temperature (Ti) with increase in KOH concentration. This variation can be ascribed due to larger presence of VM release in high concentration KOH (10, 15 and 20%). Higher release of VM content initiates easy combustion and burning of sample. Presence of single peak for raw coal and 15% KOH indicate stagewise smooth combustion with removal of water, loss of high and low molecular hydrocarbons from the sample. Burnout temperatures for all samples lie in the range of 470–530°C. However, with increase in KOH concentration, burnout temperature decreases with high combustion rate and takes less time for complete combustion. Whereas 20% KOH concentration sample shows a high burnout temperature 530°C owing to higher resistance to O2 adsorption and lower heat release which leads to residual unburnt carbon loss.
Analysis of coal petrography
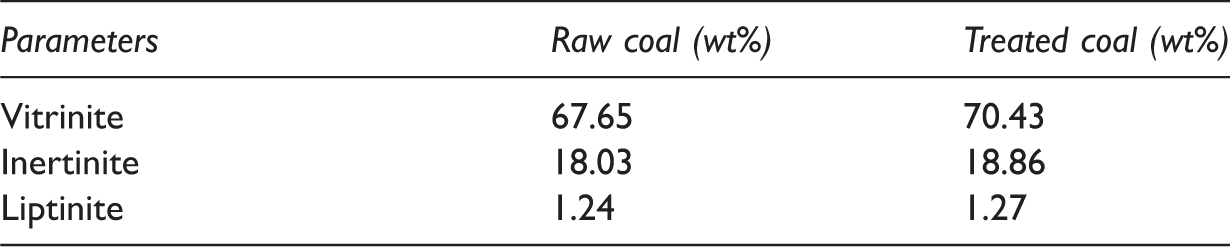
Microscopic analysis of coal infers the presence of different maceral components which affects the reactivity of coal during its utilization. The petrographic images of raw coal and KOH-treated coal (20% KOH concentration and 24 h contact time) are shown in Figure 9. From Figure 9 it can be observed that pyrites sulphur reduced after KOH treatment which is in accordance with the result reported in ‘Effect of KOH treatment on sulphur removal’ section and Figure 1. Results obtained from petrographic analysis showed (Table 5) coal having 67.65, 18.03 and 1.24% of vitrinite, inertinite and liptinite, respectively. Vitrinite content gets improved which is from 67.65 to 70.43% while both inertinite and liptinite varied from 18.03 to 18.86% and 1.24 to 1.27% at maximum condition. Result shows that KOH treatment has positive impact on coal quality by increasing the vitrinite content (3.95%) and also shows the few increment of inertinite and liptinite. This increment was decreased by the pyritic sulphur from the coal. Recent developments in coal microscopy have allowed determining its constituent macerals, thereby helping to understand their effects on coal conversion and reactivity parameters (Gupta, 2007).

Polished sections of raw coal (a) and KOH-treated coal samples (b, c and d).
Petrographic analysis of raw coal and treated coal (20% KOH, 24 h).
KOH: potassium hydroxide.
Analysis of FTIR
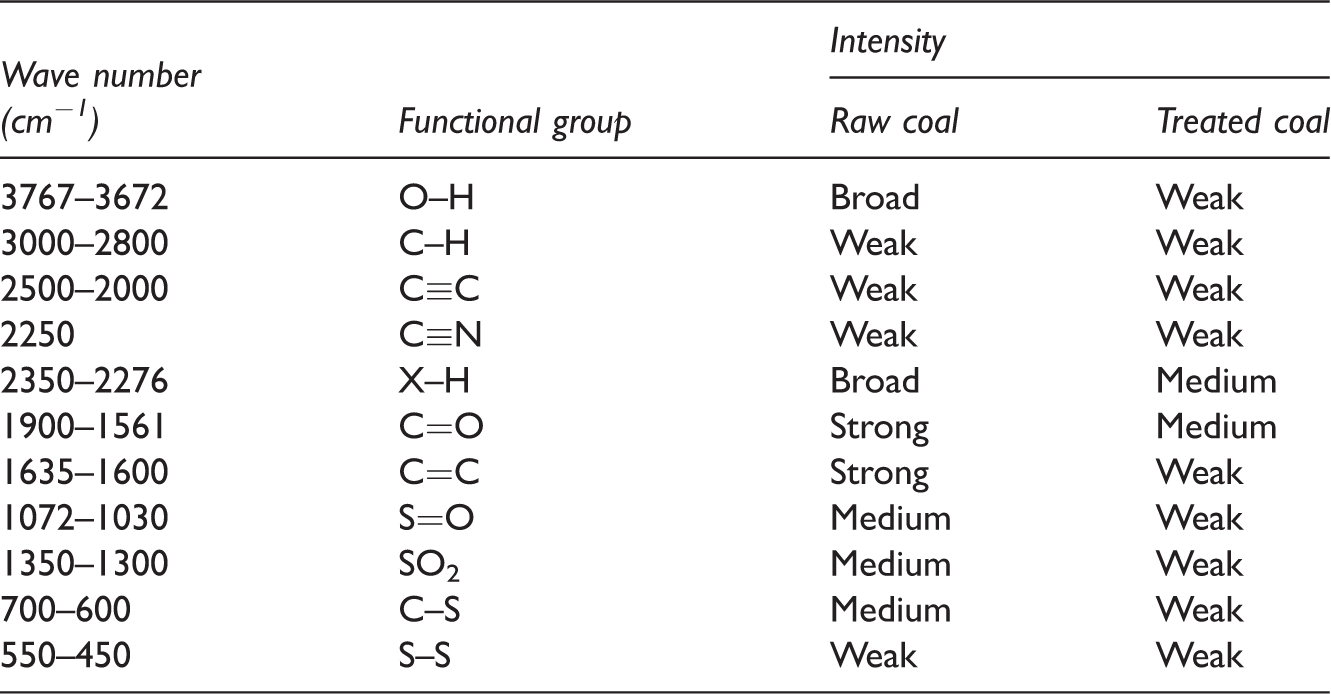
The diffusion reflectance of FTIR spectra of the coal samples was recorded through infrared instrument (Perkin Elmer Spectra 2, USA) in the range of 400–4000 cm−1. The characteristic infrared bands of the coal samples (Table 6) reported the deduction of sulphur and mineral matter in desulphurization process of samples (Figure 10). The bands at 528–535 cm−1 are present due to the clay and silicates existence in the raw and treated samples. These bands are in the form of medium intensity of the samples. Bands found in the range of 700–600 and 550–450 cm−1 are due to the presence of C–S, S–S stretching, respectively. The presence of these bands is due to the existence of sulphides or mercaptans. The intensity of these bands is medium and weak of raw samples and after treatment it became weak for both samples, respectively. The bands in the range of 1300–1350 cm−1 are due to the presence of SO2 stretching and also show a band range of 1072–1030 cm−1 due to the S=O stretching present in quartz and kaolinite. Intensity of these peaks decreases after desulphurization. The X-H stretching bands present in the in the range of 2350-2276 cm−1 shows the compact intensity of coal from broad to medium due to the removal of deleterious metals. The bands between 1900 and 1561 cm−1 are due to the presence of C=O and aromatic C=C stretching vibrations (1635–1600 cm−1) caused by strong and weak absorption of raw and treated samples, due to decarboxylation of the matrix. Triple bond C≡C stretching absorbs between 2500 and 2000 cm−1, while nitrile group C≡N in the range of 2250-2000 cm−1. These bands are generally having weak intensity strength found in raw and treated coal samples, respectively. Bands from 3000 to 2800 cm−1 are due to the presence of C–H stretching (methylene asymmetric C–H stretching). The intensity of peaks of these bands is weak and then medium. Finally, O–H stretching bands occur in the range of 3767–3672 cm−1. The intensity of peaks is found around to be broad and weak in comparison to raw and treated coal; this may be attributed to the increase in OH− when treated with KOH because of the presence of water, even after drying (Stuart, 2004). Weak sulphur bands along with changes in other band structure in treated coal proves the desulphurization of coal in the coal matrix. The proof of the desulphurization of coal increases in the coal matrix.
Characteristic infrared bands for raw and KOH-treated coal samples.
KOH: potassium hydroxide.
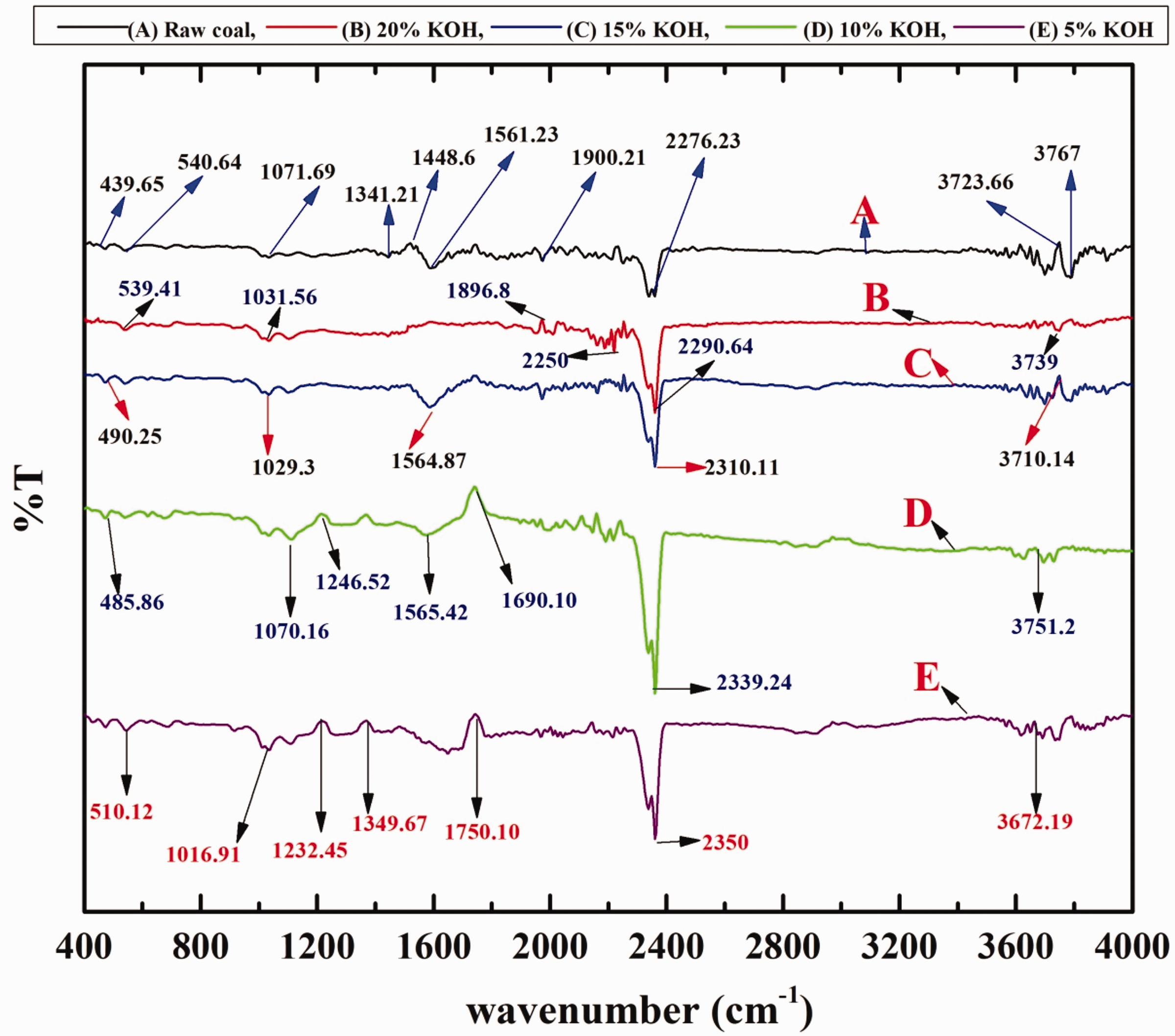
FTIR spectra of raw coal (A) and different KOH concentration (B: 20%, C: 15%, D: 10% and E: 5%) of treated coal samples at different reaction times (h). KOH: potassium hydroxide.
Analysis of FESEM
The analysis of coal particles before and after chemical leaching (chemical treatment using KOH) with a FESEM revealed the microstructural changes which occurred during chemical treatment of the samples. FESEM (make SUPRA’55; Model: MonoCL4, USA) shows the photographs of qualitative changes on exterior surface of the pyrite or coal particles. The SEM morphology of the raw coal and treated coal sample (24 h contact times and 20% KOH concentration) is shown in Figure 11, from the pitting in surfaces, crack formation and breakages of coal particles after treatment of coal. The changes in treated coal structure were due to cavitation of KOH treatment. Cracks formation takes place due to the reaction of KOH with the sulphur compound as well as aluminosilicate in the reaction. Cavitation and extra particles are formed due to the deposition of SiO2/Al2O3 which increases the ash% immediately as reported in ‘Characterization of raw coal sample’ section. Some coarse partials and holes are also seen due to the removal of organic groups and mineral matter from treated coal surface.

SEM morphology of raw sample (a) and KOH-treated coal sample (b).
Conclusions
High sulphur coal collected from Makum Coal Field, Assam, India is treated with different KOH concentration (5–20%) and different contact times (6–24 h) have been experimented to verify effects of the sulphur removal efficiency. Experimental results show that KOH alone removed 28.86–36.79% total sulphur from the coal and form of sulphur such as pyritic sulphur, sulphate sulphur and organic sulphur from 45.03 to 49.01, 24.10 to 33.73 and 20.93 to 30.62%, respectively, at applied conditions. During the removal of sulphur, ash content is increasing in the range of 8.33–11.69% by adding some mineral matter in the leaching process. GCV of coal was found to decrease from 6854 to 6084 kcal/kg due to increase of ash content of coal and loss of some combustible matter in leaching process. The H/C ratio increases up to 0.077 from 0.070% and decreases up to from 0.070 to 0.067% because of variation in KOH concentration. It also has been noticed that 0.91% of nitrogen is converted into 0.66% for the entire experiments; it is also beneficial for us. Result obtained from pre and post treatment of high sulphur coals infers the removal of pyrite sulphur (from 1.51 to 1.21%) from coal matrix which causes escalation of vitrinite (67.65-70.43%), few amounts of inertinite (18.03-18.86%) and liptinite (1.24-1.27%) as macerals. Combustion behaviour obtained from experimental TGA–DTG analysis shows that the burning profiles vary with different KOH concentration and 20% KOH concentration at 24 h contact time with ignition temperature 252°C, peak temperature 409°C is found to give the best result desirable at required condition. FTIR studies abridged the prosperous deterioration of sulphur and increase of combustion characteristic of coal. The bands found in the range 700–600 cm−1 are due to the presence of C–S stretching. After KOH treatment the formation of cavitation, cracking and pitting formation on the coal surface shows the FESEM morphological study. These changes on the coal surface after treatment may be due to deduction of some minerals matter and organic groups. The comparatively low desulphurization of Makum coal is associated to its high hydrophobicity.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.