Abstract
Biomass is one of the renewable energy and material sources. Agricultural biomass wastes are in top list in terms of quantity and uniformity. The stalks, leafs, and peels of them have taken considerable attention for various purposes. The biomass and wastes in recycling of matter and recuperation of chemicals with thermochemical conversion techniques are an efficient way in environmental perspective. The alliaceous plant reaches huge amount and its peels take attention in terms of difficulty of recycling with potential valuable compounds like its pulp. Here the pyrolysis of this garlic peel wastes was accomplished to obtain various valuable solid and liquid products that were analyzed with miscellaneous methods (thermogravimetric analysis/differential thermal analysis, gas chromatography/mass spectrometry, and scanning electron microscope). Three basic zones were appeared in thermal analysis for pyrolysis process. The valorization of these wastes to obtain precious chemicals and combustible compounds equivalent to petroleum products was illustrated by this way. Also carbonaceous compounds have been sequestered in solid and liquid forms by this way. The main fuel additives, methanol was obtained in remarkable amount (22.5%) from the liquid products. Also, porous material was produced from the solid products.
Keywords
Introduction
Taking care of the environment means enabling human activity without disturbing the natural cycles. Nevertheless, it is clear that nature has also been restructured to some extent by mankind. This is called extrinsic environment that is shaping the medium with external molding patterns resulting in various artifacts. This may be beneficial to natural environment like controlling the plant’s growth for the purpose of leveling the accumulation of carbon dioxide gas. The environmental impact of carbon-based wastes has various dimensions; energy and material production, environment, and human health protection. These can be summarized as sustainable development. The sustainability family terms take great attention in many perspectives. These are environmental protection, energy production, and material processing in context with sustainable development worldwide. The many types of cycles whether natural or not are in the centers of these considerations with ultimate zero stable wastes. Concerning this, the biomass is unique substance containing carbonaceous compounds having stored sunlight. The biomass is inevitably important alternative renewable sources for energy and chemical raw materials since they are formed by photon emitted by the sun together with mainly carbon dioxide and water, which is called photosynthesis. It is also carbon sinks with all types ranging from the grasses to trees including photo planktons. Therefore, even the minute advances in this topic may eventually result in great progress and should be welcomed (Makkonen et al., 2015). And they are also readily differentiated from other renewable sources in terms of being not only energy sources but also chemicals (Madhua et al., 2016). Thus, the renewable sources and so biomass have taken great attention in last decades (McKendry, 2002). The main objectives in this type of studies are evaluation of the potential risk of environmental pollution of the agricultural wastes, probably recycling of these wastes, determination of the factors of the evaluation processes, and reporting these to the policy makers to increase the widespread impact.
The main resources have been oil, coal, and natural gas along with nuclear energy and by-products of photosynthesis. Recently, as a renewable source, biomasses have attracted much attention for their promising applications in the preparation of carbonaceous compounds (Sun et al., 2013; Wu et al., 2003). The agricultural plants, biomass, and their residues have been the sources of wood-containing celluloses and lignin; starch, protein, and oil, which are bioenergy resources. It is known that the recycling and utilization of agricultural wastes are a difficult issue worldwide that can be improved by governmental legislation (Wang et al., 2016). Biomass can be categorized in four groups; forestry, agricultural, aquatic, and livestock. In this condition, they are sources of both energy and matter (Saygin et al., 2014). And, they can be directly incinerated in power plants or can be added to solid fuels to obtain heat and electricity. Alternatively, they can be converted into precious and valuable materials by various means especially with thermochemical conversion techniques. And in this perspective, they are readily differentiated from other renewable energy sources. Agricultural wastes without considering their amounts are very important due to the interconnectedness and interdependences of the countries for sustainable development (Bhatnagar et al., 2015). The high amounts of the biomass, the coarse peels, stalks, shells, and cobs etc. are often discarded as waste through incineration or landfill processing, causing severe environmental pollution and resource wasting. In front of us, there are two roads. Either we will choose the right way and evaluate the biomass concisely and confidentially by regenerating environmentally safe products or the other unaware way where the uncontrolled process converts these into some extent of unknown materials. The direct combustion of biomass has long been used for domestic and recent industrial purposes. Unfortunately, the burning products have been recognized as an important source of trace gases and aerosols pollutants in local, regional, and global atmospheric environment. These burning emissions have become an important issue since 1970, however, the gap between theories and practices is far from the mutual interferences (Zhang et al., 2013). The production of charcoal from wood, which is a type of biomass is known for centuries by such pyrolysis process (Hawash et al., 2017).
The pyrolysis is one of the thermochemical conversion technique in which biomass is exposed to high temperature, resulting in solid, liquid, and gas products (Caglar, 2009; Paradela et al., 2009). Environmental protection and sustainable development issues are readily rendered by these studies beside the economical and industrial concerns. The solid products of pyrolysis process basically comprise carbon black powder, which can be used as an adsorbent for abatement of toxics such metal ions, phenols, and dyes (Hameed and Ahmad, 2009; Raj et al., 2013). The liquids can be regarded as equivalent to raw petroleum, which can be converted into precious materials or fuels after the refining process (Demirbas, 2007; Kar and Keles, 2013; Pognani et al., 2011). The evolving gaseous material is called hydrogen-rich gas (Caglar, 2001). The distribution of phases and products in thermochemical process shows variations according to process conditions and feedstock nature (Okoroigwe et al., 2015). The pyrolysis conditions, fast intermediate and slow ones, the reactor design, additives, and of course ultimate pyrolysis temperature make considerable variation in products. The solid part has been generally used as soil conditioner and amendment. Also, it aids the plant for carbon sequestrations (Fu et al., 2012; Windeatt et al., 2014). Apart from this, the fine texture of biomass may also result in porous and nonporous material that can be used for various purposes such as anode for lithium ion batteries, filters, and fillers (Caglar et al., 2016; Sun et al., 2013). Especially the porosity of the remaining solid products is affected by the release of volatiles and chemical (Bian et al., 2016).
The liquid phase can be sources of useful industrial chemicals by the results of the gas chromatography (GC)/mass spectrometry (MS) analysis of the bio oils. The liquid phase compromises energy-bearing molecules, chemical raw feedstock, and polymerizable reactant monomers (Yang et al., 2015). Some of the polymeric materials such as polylactic acid can be synthesized from naturally monomers. These are derived from plants called as bioplastics, which are the alternatives to the traditional petroleum-based plastics (Matuana et al., 2009; Ruíz-Cabello et al., 2016).
There are lots of rhetoric explanations about the content of gaseous part. The copiousness of flammable compounds such as low molecular hydrocarbons and hydrogen has been mentioned (Caglar, 2001; Chen et al., 2015). Also, syngas can be produced from the remaining residue of plants (Saygin et al., 2014).
The agricultural plant, garlic belongs to Liliaceae family (Allium sativum). Its worldwide production is 25 million tons annually (en.wikipedia). It is an important ingredient of the many foods and drugs. And its pulp has been extensively studied in medicine and gastronomy. However, its by-products peels and stalks are not that much touched. The peel has low apparent density and covers great volume. It cannot be utilized as natural fertilizer since it easily flies with winds before intermingling with soil, and making briquette from it is also a hard duty (Hameed and Ahmad, 2009). Garlic peels (GPs) self-extinguish when ignited due to sulfur content, which forms heavy oxides, inhibiting further oxygen diffusion to the burning medium. Garlic production is locally high in the Kastamonu province of Turkey. The approximate production of garlic in this province is 150,000 tons annually and approximately one-fifth of it is peel and leafs (Hacıseferoğulları et al., 2005). The garlic has a bulb section under earth and a stalk during growing. The photograph of garlic is shown after harvesting (Figure 1). The garlic bulb has two layers of peels. The one is called inner peel (a very thin skin of garlic clove) and the second is the outer peel (thick shell).
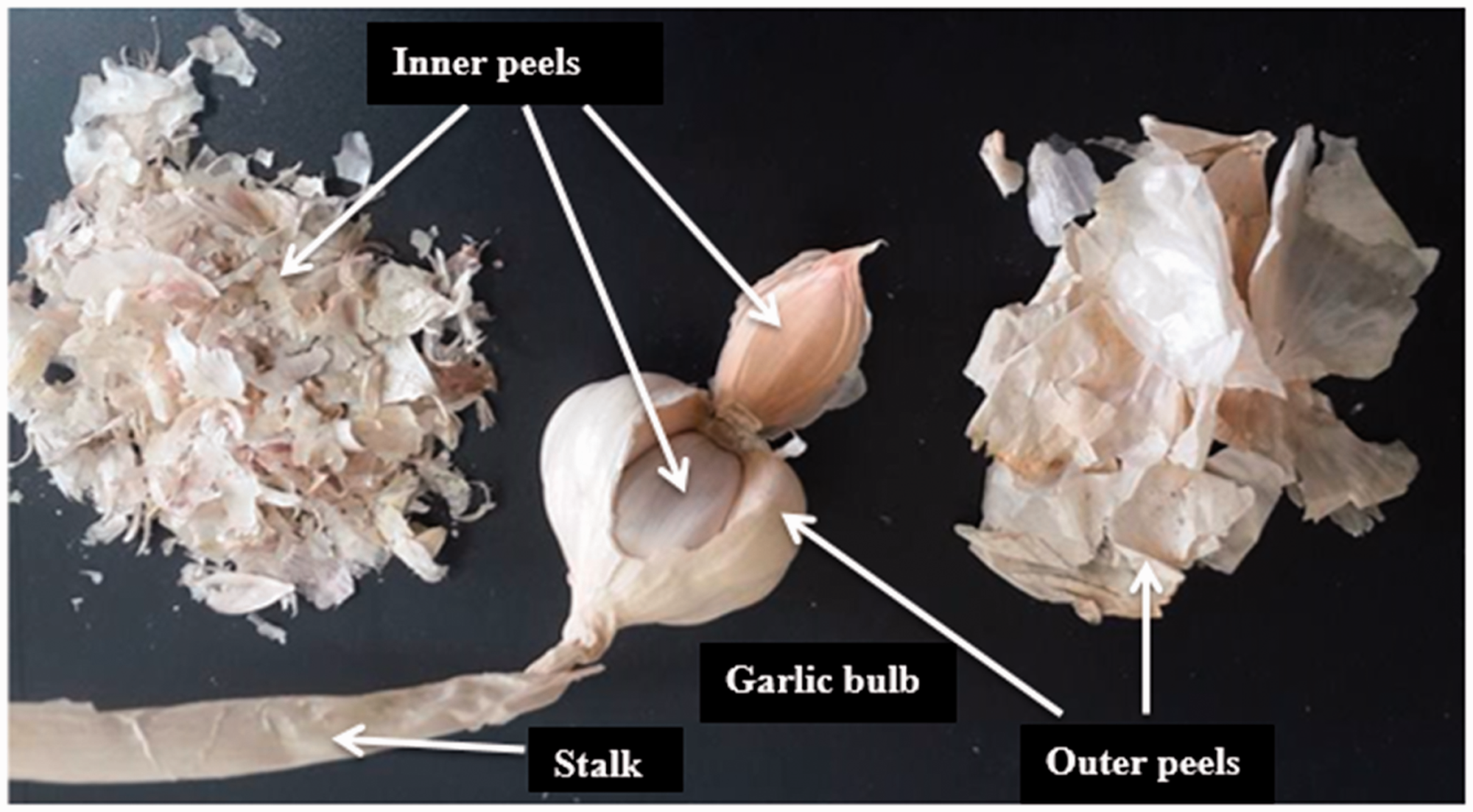
The components of garlic: bulb, stalk, inner, and outer peels.
They can be used as a solid material in its natural or processed forms. Natural garlic skin has been used for extended preservation of food (Hershko et al., 1998). Also, it was used as an adsorbent of heavy metals ions especially Pb2+ in solutions and phenols, major pollutant of water (Chowdhury et al., 2012; Liang et al., 2013; Muthamilselvi et al., 2015). The cellulose fibers extracted from it were used as reinforcing fillers for bio-nanocomposite (Reddy and Rhim, 2014).
Thermally processed peels can also be used an adsorbent material. In addition, the extract of GP was presented as an alternative pesticide to reduce the usage of chemical counterparts (Thomas and Callaghan, 1999). The products of onion peel pyrolysis have been used for flotation in coal-mining process (Fecko et al., 2011). The garlic and onion are in the same family biologically.
In the study, an efficient way is introduced in valorization of the GP wastes, which can be regarded as environmental pollutant material due to inefficient incineration, difficulty in recycling, and elemental content. The elemental, proximate analysis, and higher heating values of GP were determined. In order to decide pyrolysis conditions, the main weight loss and transition temperatures were determined by thermogravimetric analysis (TGA) and differential thermal analysis (DTA). Pyrolysis process of GP was executed in predetermined conditions. The solid, liquid, and gas products were separately collected. The structure and morphology of the solid products called as char were examined with scanning electron microscope (SEM) at various magnifications. The liquid products were analyzed with GC/MS and the compounds were given in percentages. All of the above discussion shows that the volarization of GP by pyrolysis is unique choice otherwise GPs were considered to be environmentally pollutant material.
Materials and method
Materials
The GP known as environmentally pollutant agricultural residue were collected from private garlic peeling machine as biomass waste. This machine works in two stages. In the first step, after the separation of the stalk of the garlic, the outer peel of garlic bulb was obtained by blow of air. And the collected peels here are named as garlic outer peel (GOP). In the second step, the peel of garlic tooth was obtained and named as garlic inner peel (GIP). The GOP and GIP samples were separately stored ambient conditions. The former one (GOP) has thicker peel than the latter GIPs. Both, they have coarse nature and shine white reddish color with pungent scent. Before executing the characterization and pyrolysis experiments, they have to be ready by sewing and grounding into granules by Wiley mill. Then the pre-GP materials were screened to pass through a screen of 40 mesh aperture resulting in powders. These powders were dried at 30°C in oven for 24 h and stored in glass bags (Aydinli and Caglar, 2010).
Method
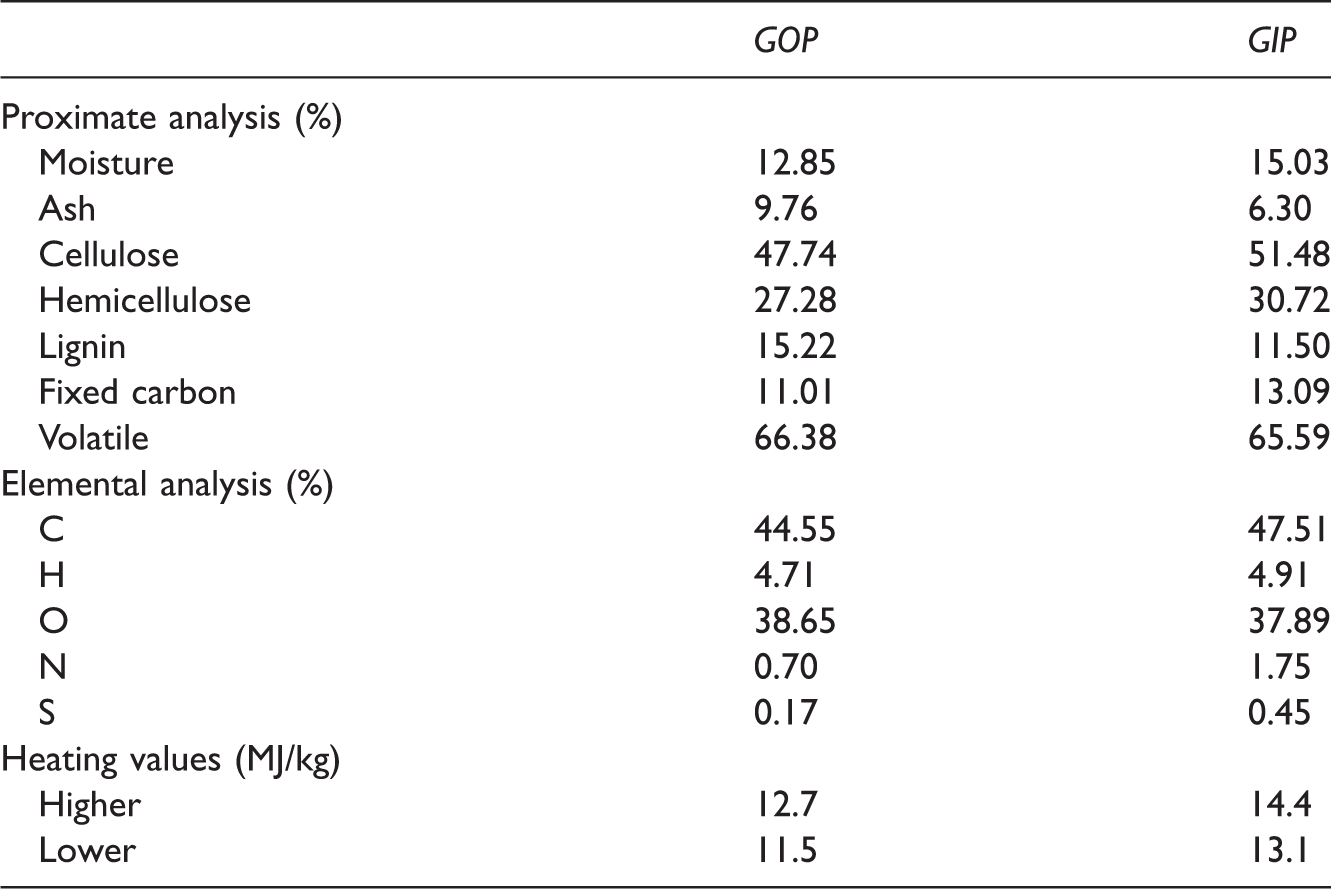
The elemental analysis of the samples was made by standard CHNS/O analyzer. The result of the proximate analyses was given in combination of TGA–DTA analysis under nitrogen atmosphere and National Central Laboratory results. Higher heating measurements were determined by using a bomb calorimeter. And the results of above analysis are given in Table 1.
The proximate and elemental analysis of garlic peels along with heating values.
GIP: garlic inner peel; GOP: garlic outer peel.
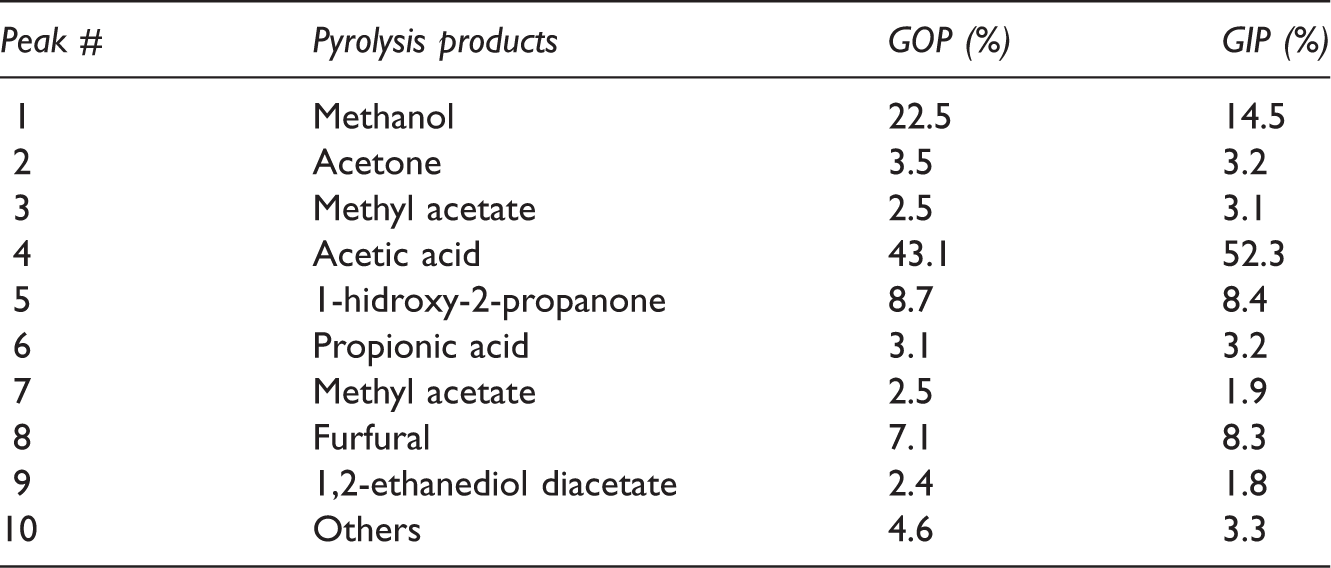
Pyrolysis of peels: The pyrolysis of 1.0 g GOP and GIP samples for every run was accomplished in tubular reactor, which is inserted in vertical position in preheated furnace. The pyrolysis in this condition can be called as flash pyrolysis. In flash pyrolysis, the samples at ambient conditions were inserted in the reactor, the whole system was then transferred into preheated furnace at various temperatures to accomplish pyrolysis process. A broad range of temperature is selected (350, 400, 450, 500, 550, 600, 650, and 700°C) to cover pyrolysis correctly. Approximately in 3 minutes, reactor temperature reaches the ultimate pyrolysis temperature. The pyrolysis of biomass was completed in 15 minutes. The details and setup were given in Figure 2. The solid products were scratched from the reactor and weighed absolutely, and then saved for further examining. The solid product obtained from the pyrolysis of GOP is named as GPOP and from the GIP is named as GPIP in sequence. The liquid amounts were found with simultaneous adding of collected ones and the weight differences of the connecting tube before and after pyrolysis process. The clean portion of liquid products was collected in tubes for further analysis. As it is known that liquid product of pyrolysis process is called as tar which also contains some solid particles. Therefore, after settlement of the tar, the clean part of the liquid products was taken for GC-MS analysis. The easy part was being to find the gas amounts, which were calculated by difference. The gas products were not collected and thus discarded. The content of liquid products was analyzed after some time of the execution of pyrolysis process by separate GC/MS instrument. The resulting GC/MS spectrums were compared with standard library spectrum. And the relative amounts of the identified species were comparatively given (Table 2). As an instrument, single quadrupole GC/MS was used.
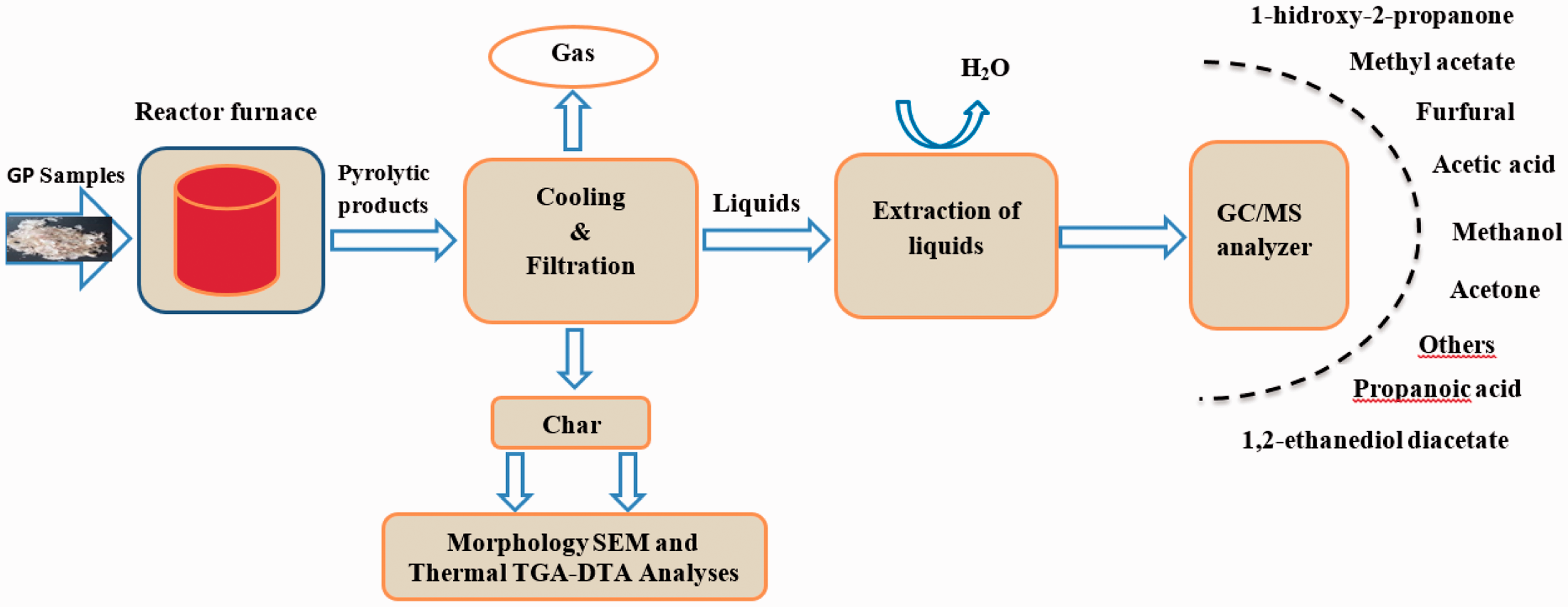
The schematic representation of pyrolysis process and subsequent GC/MS, SEM, and thermal analysis.
The percentages of liquid pyrolysis products of garlic peels by gas chromatography (GC)/mass spectrometry (MS) analysis.
GIP: garlic inner peel; GOP: garlic outer peel.
The combined TGA–DTA analysis of pyrolyzed and raw samples were performed with 5–10 mg analysis starting with ambient conditions to 950°C with temperature increments of 10°C/min and 20°C/min. Also, the structure, morphology, and relative porosity of the materials were determined with SEM after covering surface of the samples with conductive gold at fractured surfaces. The SEM photographs were taken at various magnifications (500×–10,000×).
Results and discussion
The obstacle in evaluation and recycling of residues of garlic which is one of the special, medical, and production increased agricultural plant is investigated readily in order to find an environmentally safe track by thermochemical conversion means.
The differentiating properties of GP
For this purposes, the differentiations of GP from the remaining plants were put forth in basic analysis. It is founded that they contain high amounts of sulfur, which is of course in the form of chemical compounds. The sulfur-containing compounds are resistant to burning due to inner and inter crosslinking ability of sulfur during the oxidative chemical reactions especially in polymeric mediums. Hence the inefficient burning results in low heating values compared to similar to agricultural-cultivated plants.
The proximate and elemental analyses of inner and outer GP along with heating values in original and dry base were presented (Table 1). The inner peel has definitely higher moisture content and controversially distinctly lower ash percentages than the outer peel. Moisture is not desired in pyrolysis process, which indicates of intrinsic hydrophilicity. Ash is the residue of inorganic content of the biomass. Low ash values are preferable in pyrolysis process even though they may have same applications in agricultural and civil engineering applications. The inner peel has distinctly lower ash and higher fixed carbon values than the outer peel. Generally, the average biomass ash value is 4.1% arithmetically. Here, in our case, it is approximately 9% which doubles average agricultural biomass in this concern, which again indicates the effect of sulfur.
The structural stability of plants parts relies on the distributions and weaving of cellulose, hemicellulose, and lignin contents. Again the proximate and elemental analysis results of GP were given (Table 1). Remarkably low lignin percentage characterizes the local GP in comparison to average biomasses. The inner peel has higher content of carbon and nitrogen than the outer peel. The remaining elements namely; H, O, and S have similar values compared to other cultivated garlic in other locations. Liu et al. found that carbon and oxygen content as 37.01 and 47.71% which is contrary to our results (Liu et al., 2013). Heating values of both inner and outer peels are similar to other biomass such as rice husk and saw dust etc. which are again lower than the lots of other biomasses. And the values are very low compared to well-known fuels (lehrafuel.com). Thus, direct incineration of GP is not sufficiently efficient and reliable method for energy production. This directs the studies to other volarization techniques like thermochemical conversions.
Thermal analysis—TGA/DTA
Before pyrolysis it is a routine practice to take the TGA curve and DTA peaks in order to settle the experimental conditions. The garlic samples were heated starting from ambient condition to 900°C at the rate of 20°C/min under nitrogen atmosphere. And as stated before, the main weight loss and transition temperatures of virgin GPs were determined which can be seen (Figures 3 and 4). In addition, to determine the extent to which the pyrolysis accomplished, the experimental solid products were also subjected to thermal analysis (Figures 5 and 6). It is understood that there are three main regions (a, b, and c) which can be attributed to moisture (a), cellulose and hemicellulose (b) and remaining lignin (c) which are the basic contents of biomass matrix (moisture, cellulose, hemicellulose, and lignin) in the thermal curves (Aydinli and Caglar, 2013; Brebu and Vasile, 2010). The important weight loss temperatures width and transitions were summarized (Table 3). The main difference between GOP and GIP is the missing or insignificant region a for GIP. This means that more stable material GIP covers the cloves of garlic. Generally, the transitions and weight loss temperatures give simultaneous signals, which means separate and distinct special materials exist in the GP. Since the analysis was made under the nitrogen atmosphere, the solid remaining converges to the nil beyond the 1000°C due to reactivity initiation of carbon with nitrogen. Again the somehow missing and moderately insignificant region of a and b for GPOP and GPIP indicate the differences between the small scale (in milligrams) and pilot scale (grams to kilograms) pyrolysis processes wherein the former pyrolysis can be completed easily, but in the latter, there can be some parts that are not pyrolyzed. Therefore, to design porous material in macro level, the morphology changes by the effect of variables such as temperatures should be handled carefully to catch the ideal structure and porosity.
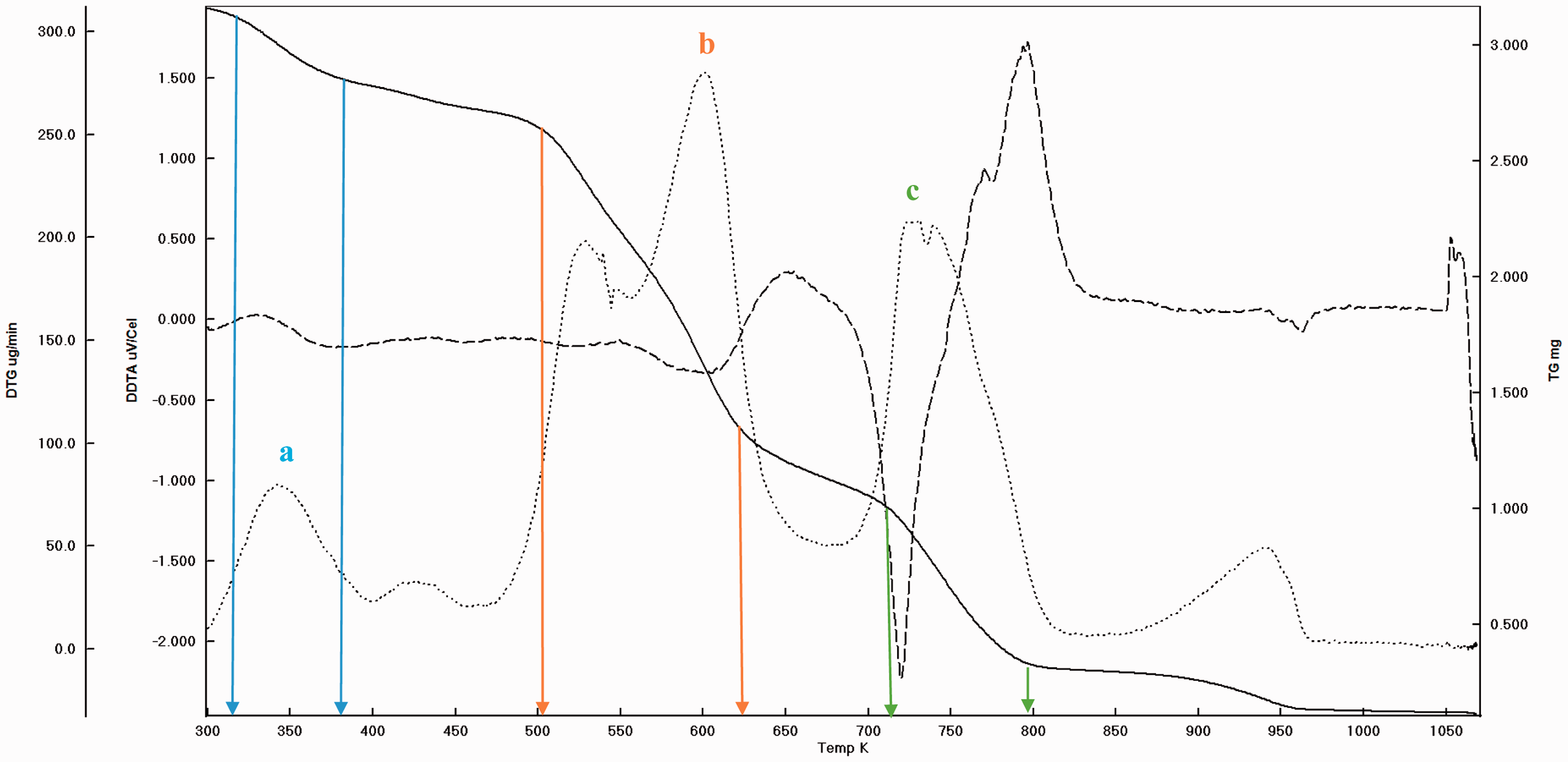
The weight loss and transition temperature curves of GOP by TGA–DTA.
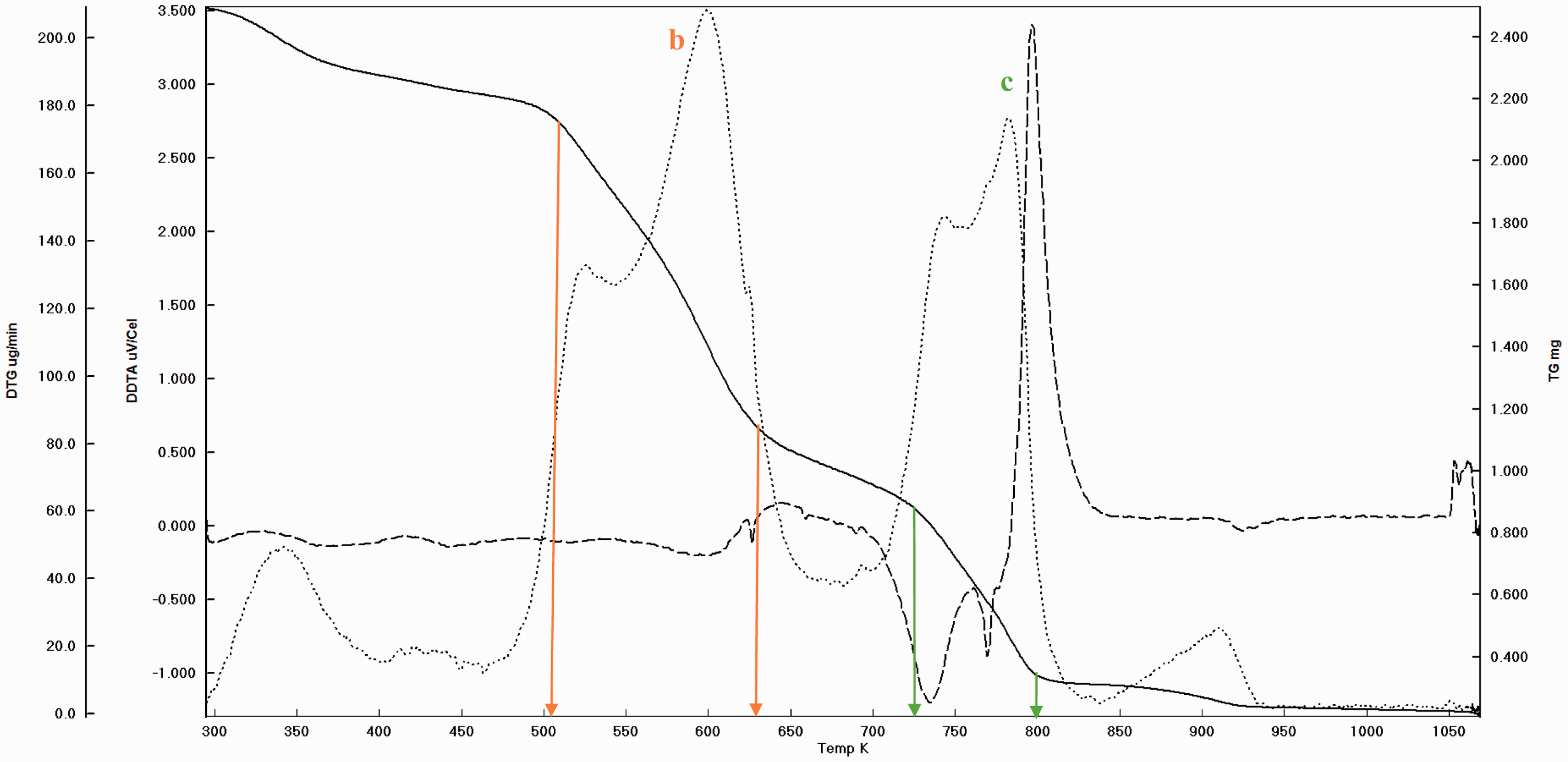
The weight loss and transition temperature curves of GIP by TGA–DTA.
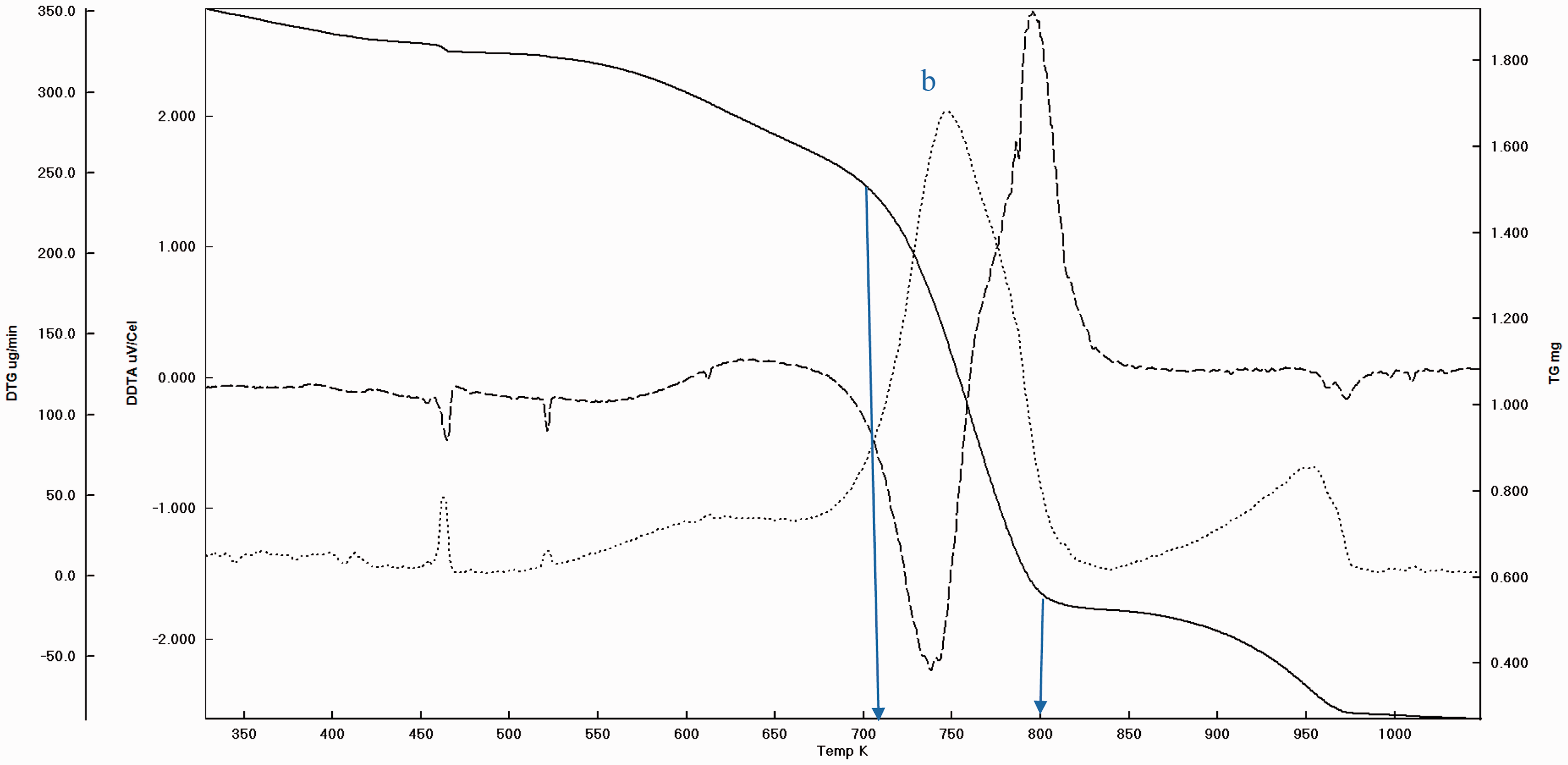
The weight loss and transition temperature curves of GPOP by TGA–DTA.
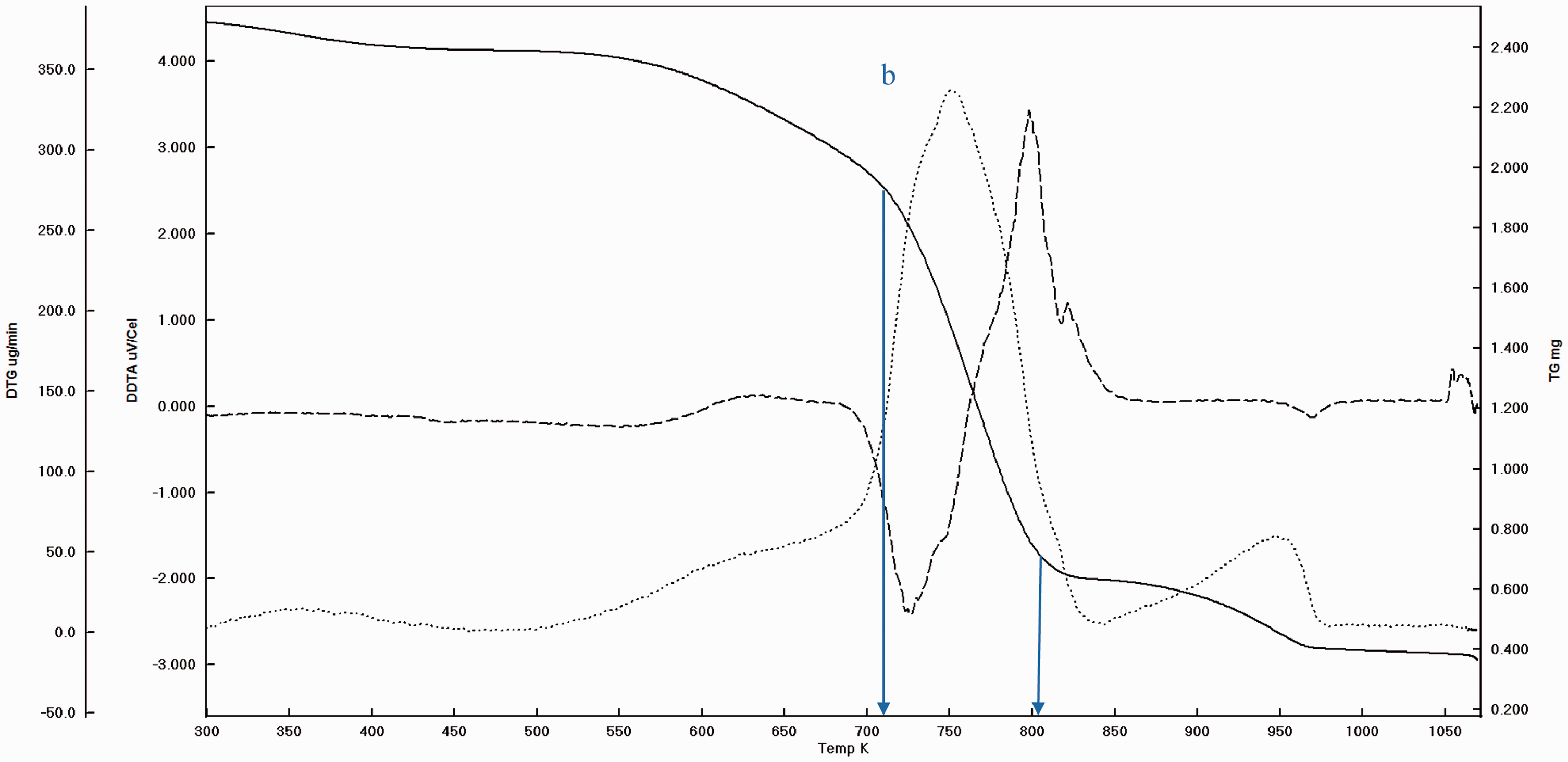
The weight loss and transition temperature curves of GPIP by TGA–DTA.
The numeric values derived from DTA, TGA, and DTG curves.

DTA: differential thermal analysis; TGA: thermogravimetric analysis; DTG: differential thermogravimetri; TG: thermogravimetry.
aNot calculated. bInsignificant.
Basic pyrolysis outputs of GPs
The solid, liquid, and gas product amounts of the pyrolysis of the peels were determined gravimetrically and they are shown in Figures 7 and 8. Although, it is well known that the classical view of percentages of pyrolysis products (solid, liquid, and gas) versus increasing temperature is this; the solid amount decreases, the gas amount increases and the liquid amount reaches a maximum with increasing temperature, some interesting points have been observed in this study. Normally, the maxima for liquid product amounts for GOP and GIP have been reached at moderate temperatures, 600°C and 650°C, respectively. However, the solid products have leveled up or increased somehow at 650°C, contrary to normal decreasing level of solid amounts. This can be due to the continuous and somehow anomalous decomposition rate of lignin especially between 600°C and 700°C (Brebu and Vasile, 2010). At this point, changing decomposition route of lignin may result in increase in especially solid products, moderately liquid products, and severe decrease in gas amounts.
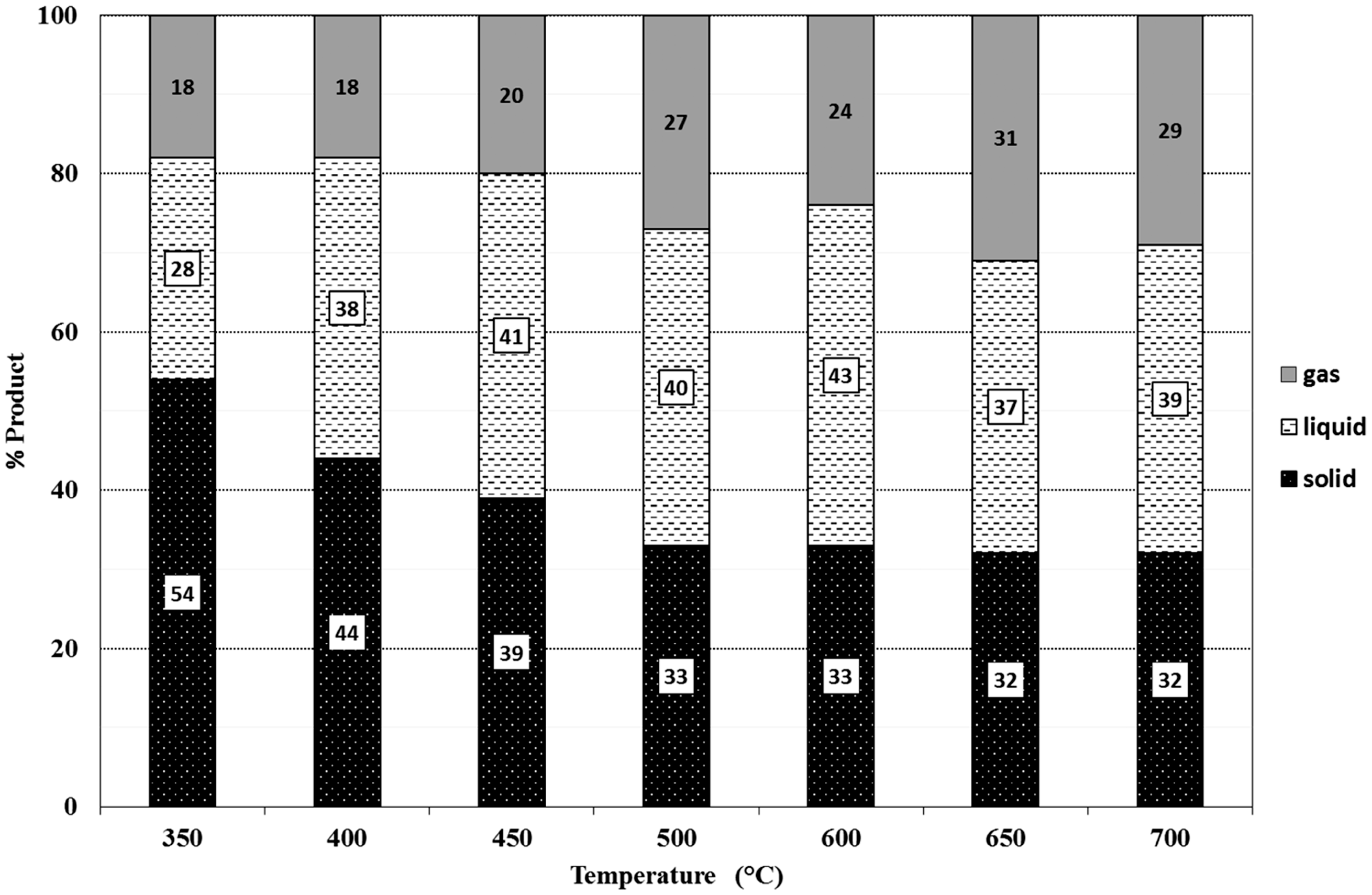
The ratios of pyrolysis solid, liquid, and gas products of GOP.
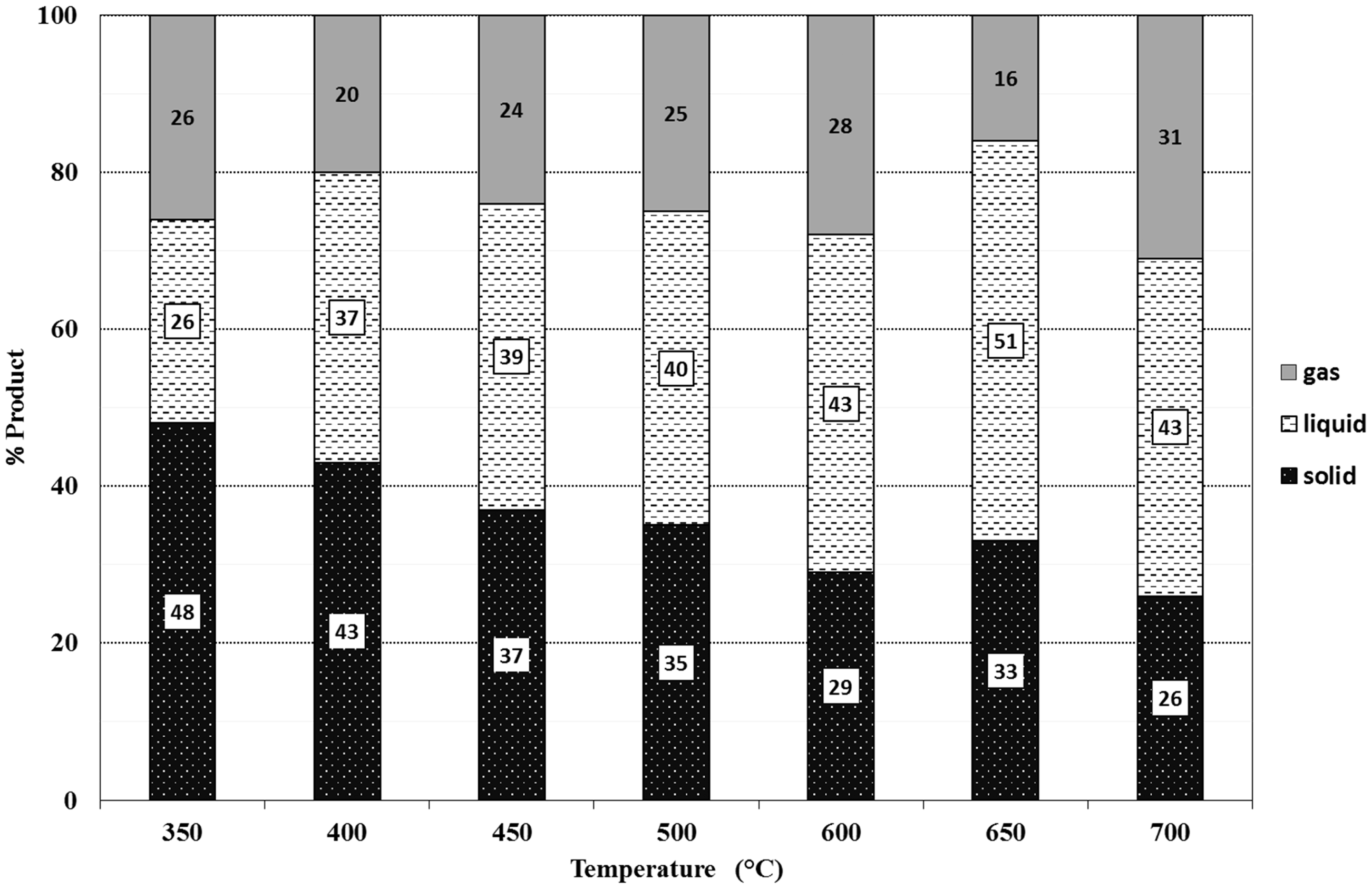
The ratios of pyrolysis solid, liquid, and gas products of GIP.
The liquid products obtained at 550°C were arbitrarily chosen for further GC/MS analysis. The variation of chemical identity with temperature is subject to another further study. Between 450°C to 650°C, the liquid product amounts approximately from the 40% of the total pyrolysis products. That is a good conversion percentage if especially liquid product is dealt with. The unexpected increase in solid product at 650°C for GIP results in decreasing of gas product which accompanies also the increase in liquid product. The increase in solid products which is named as fixed carbon is also important for production of inert stable and porous material in technological applications.
The characterization of pyrolysis liquid products of GPs by GC/MS
The collected pyrolysis liquids were analyzed with GC/MS. These spectrographs of pyrolysis liquids are presented in Figures 9 and 10. The locations of the peaks in time axis show the chemical identity of the molecules and the area covered by them permits the calculation of the relative amount of the compounds. The compounds indicated by the numbers above the peaks and their relative amounts were supplied (Table 2).
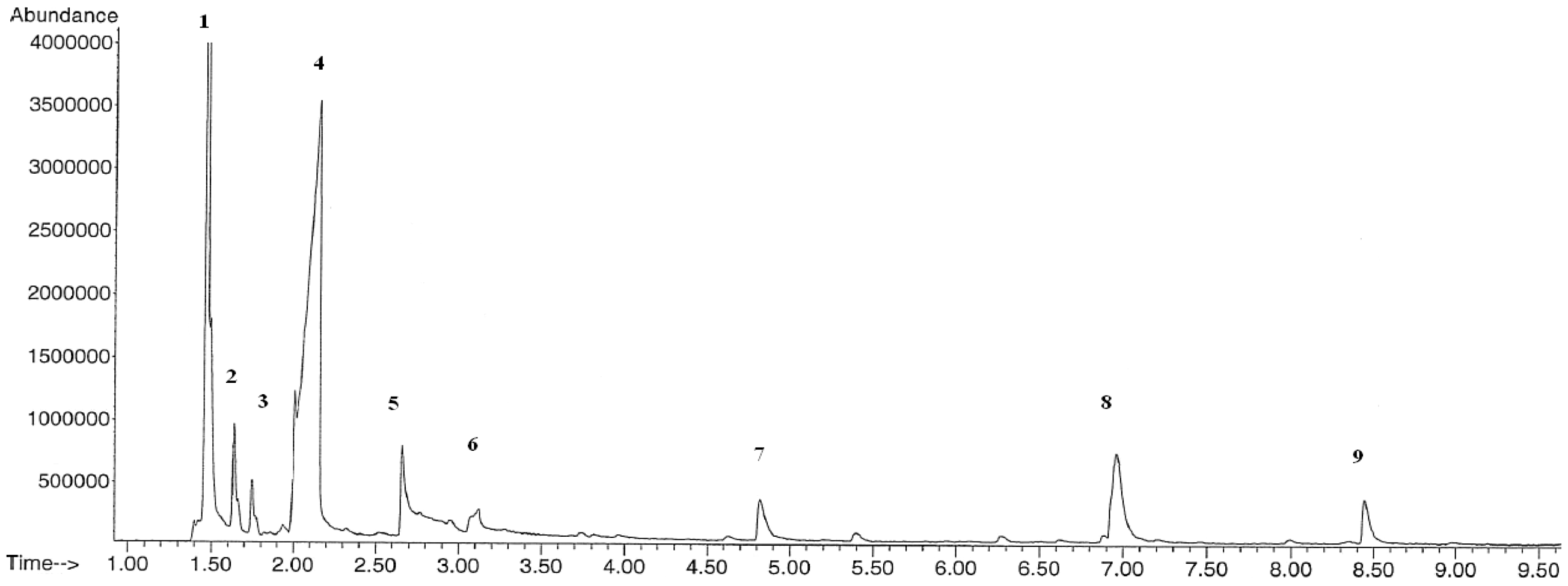
The GC/MS spectrum of the pyrolysis liquid product of GPOP at 550°C.
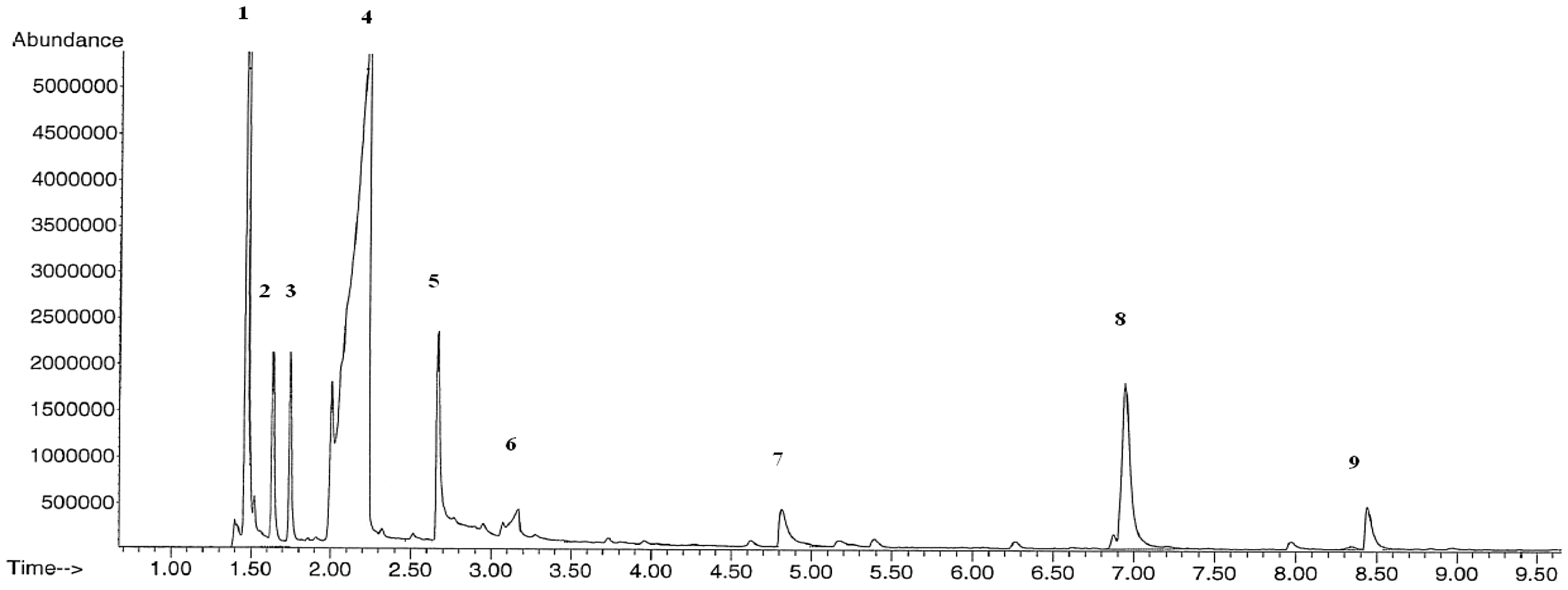
The GC/MS spectrum of the pyrolysis liquid product of GPIP at 550°C.
The GC/MS spectrums have produced well-defined clear peaks. The main products are the acetic acid as usual, methanol, and acetone in our case. The less oxidized molecules are intended ones for regarding energy capacity. The relative abundances of pyrolysis products are similar for GOP and GIP. Acetic acid that is generally a not desired product compromises the half. Methanol comes secondly which can be regarded as fuel.
The analysis of SEM photographs; GPs and pyrolysis solid products
It is well known that the fine texture of woven tissue of plant and animal wastes may result in porous carbon structure after the controlled degradation of this fine structure with removal of other organic elements (N, H, O, and S) by pyrolysis process (Caglar et al., 2016). The produced materials can find high-tech applications such as membrane, tissue scaffolds, and fuel cell technologies. The SEM photographs belonging to virgin inner and outer GPs and their resulting pyrolysis solid products at 550°C were comparatively given (Figure 11) at various magnifications (500×–10,000×). The surface SEM photographs of virgin peels were given (Figure 11(a–d)) at two magnifications. The conduction and distribution tubes of plant tissues have arranged fine structure that can be resulted in porous structure after the execution of pyrolysis process due to the degradation of side chains and low-molecular-weight oligomers. Both surfaces fractured and non-fractured ones of pyrolysis solid products were presented (Figure 11(e–h)). The surface morphology did not show much change compared to virgin ones. Although we could not take the SEM photographs of fractured surfaces of original peels, the ladder-type morphology seen in the structure of fractured surface of pyrolysis solid products means that the pyrolysis process results in porous structure after the removing low–molecular-weight compounds in biomass structure. Resultantly, relatively stable the macro-porous carbonaceous material was produced. The pore size is about 20 μm, which is suitable for many purposes.
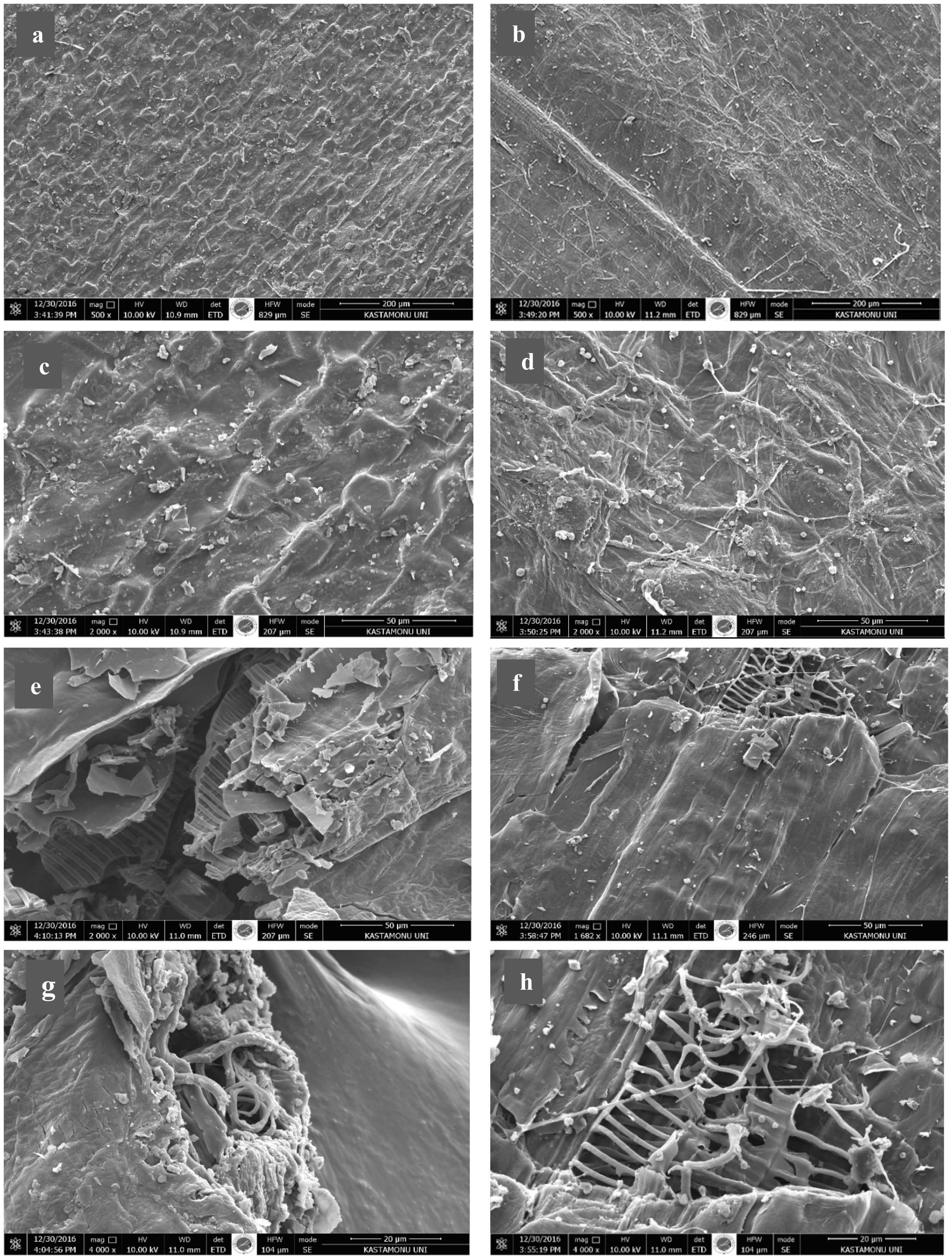
SEM photographs of virgin GOP and GIP before pyrolysis process; (a and b) (500×) and (c and d) (2000×), and after the pyrolysis at 550°C GPOP and GPIP (e and f) (2000×) and (g and h) (4000×) magnifications.
The innocent agricultural plant wastes can sometimes be harmful to environment due to huge clustering from the industrial productions. The famous sulfur-containing garlic wastes of peels and stalks are difficult to incinerate and to use as fertilizer, due to its self-extinguishing property. Thus, the pyrolysis process stands here as a good solution to this problem. And some valuable materials and some insights were obtained from these alliaceous like plants. By this way, an integrated effort to explore an efficient way to promote comprehensive utilization of Alliaceous plant wastes with thermochemical conversion techniques have been introduced. Some valuable solid and liquid products can be obtained and converted into porous material and industrial chemicals, respectively.
Conclusions
The environmental pollutant and hazardous GP wastes were utilized to produce valuable chemicals and materials in terms of recycling attempt. The biomass is the source of both energy and matter. Since they play crucial role in carbon cycle (sequestration and release), both preservation of the environment and efficient use of them must be considered to reduce carbon dioxide accumulation in the atmosphere. The production of garlic is not much compared the other agricultural or industrial crops still it reaches 25 million tons annually. Its production is generally localized and concentrated in suitable agricultural area. Also, the wastes are accumulated in narrow landfill. Therefore, its recycling and conversion studies can be locally very important.
A general scheme was sketched in thermochemical conversion process of agricultural wastes. Thermochemical conversions are efficient in this perspective to find sustainable solutions. The one of the promising process “pyrolysis” is subjected to work in conversion process of GPs into solid, liquid, and gas products. The content of liquid was analyzed by GC/MS analysis and various valuable compounds were detected such as methanol and acetone.
The solid products were examined with scanning electron microscope in order to reveal the structural morphology and porosity of the carbon-based macro-porous material. The pyrolysis process is also interestingly effective method for the production of carbonadoes porous material without disturbing the original fine structure of biological materials, this may lead to new perspectives and technologies in biomimicking the nature.
The basic thermal analysis of the GPs was also performed to find the transition and weight loss temperatures. Distinctly, three zones were identified. This study can be example for thermochemical conversion locally concentrated agricultural plant. And it is important in the perspective of local sustainable development network. Therefore, the anthropogenic effect on environment could be mitigated by this type recycling studies. The reduction of any hazardous material and wastes or their effective recycling should not be thought just as local issue.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Scientific Research Project Fund (KUBAP-01/2012–13) and Central Research Lab. of Kastamonu University, Turkish Republic.
