Abstract
The dissolution of gypsum rock is of significance to study because it affects the formation of hydrocarbon reservoirs, cap rocks and evaporite deposits. However, the characteristics and mechanism of the dissolution process remain unclear. Here, we present data from experiments performed to address this issue. The experiments simulate various geological conditions, including different diagenetic stages of burial under different fluid types. The diagenetic stages include: 30°C and 0.3 MPa for the epidiagenetic stage; 60°C and 13 MPa for the early diagenetic stage; 100°C and 27 MPa for the middle diagenetic stage; and 150°C and 43 MPa for the late diagenetic stage. The different fluid types include pure water representing continental water, seawater, 0.3 wt.% CO2 solution representing meteoric water, and a 0.2 wt.% acetic acid solution representing organic fluid. We also carried out the experiments on limestones and dolomites, because these rocks also occur in saline water sedimentary systems with gypsum rocks. Experimental results show that lithology, fluid type and temperature–pressure conditions can all affect dissolution. In terms of lithology, gypsum rocks dissolve more easily than limestones and dolomites. Fluid type has little effect on the dissolution of gypsum rock, and gypsum is soluble in all four types of fluids. In contrast, limestones and dolomites are almost insoluble in pure water and seawater, but show clear dissolution in CO2 and acetic acid solutions. The data indicate that gypsum rock has a dissolution peak close to the early diagenetic stage. In contrast, limestones and dolomites have dissolution peaks in the CO2 solution at the early–middle diagenetic stage, and do not show a peak in the acetic acid solution under surficial temperature–pressure conditions. The dissolution rates of limestone and dolomite show different trends with increasing temperature and pressure: limestone dissolution rates decline whereas dolomite dissolution rates increase. Therefore, we infer that the physicochemical properties of a rock are important drivers of dissolution.
Introduction
Gypsum rock is one of the main types of evaporite deposit, along with anhydrite and half water gypsum (Stawski et al., 2016; Warren, 2006; Zhu, 2008). Gypsum rock is an important cap rock because it has an extremely dense structure and low porosity and permeability (Beydoun, 1998; Chen et al., 2005; Yao, 2007; Zhu et al., 2014). It is also a significant nonmetallic mineral, with a wide range of industrial applications. Thus, the study of gypsum rock is of significance both in basic geology and in mineral deposit and petroleum geology, of which its dissolution has been a research highlight for several decades (Baruah et al., 2000; Burgoscara et al., 2016; Domínguez-Villar et al., 2017; Feng et al., 2017; Jeschke et al., 2001; Mbogoro et al., 2011; Xin et al., 2012) because the rocks are vulnerable to be dissolved and can further affect the formation of hydrocarbon reservoirs, cap rocks and evaporite deposits. Previous research indicates that over 200 mg of gypsum rock is soluble in 100 g water at room temperatures. In contrast, the solubility of limestone and dolomite, which are commonly co-deposited with gypsum rock in saline sedimentary environments and have a great influence on the accumulation of oil and gas (Liu et al., 2017; Ma et al.,2016; Wu et al., 2017; Xiao et al., 2017), is an order of magnitude less, at only 12 mg per 100 g of water. Therefore, the dissolution of gypsum rock is one of the key factors to consider when evaluating its distribution and preservation, and the wider implications of its sealing properties.
Geological conditions that drive dissolution have been investigated in previous studies, such as a supergene water environment and the acid–base property of soil (Gutiérrez et al., 2008; Johnson, 2008; Liguori et al., 2008; Lu et al., 2002; Lu and Zhang, 2008; Raines and Dewers, 1997; Sumner et al., 1986). However, few studies have considered the dissolution of gypsum rock compared with carbonate rocks (Amrhein et al., 1985; Fan et al., 2007; Huang et al., 1993; Pokrovsky et al., 2005; Sjöberg and Rickard, 1984; Yang et al, 1995a, 1995b; Zhu, 1990). Furthermore, most studies on gypsum dissolution are descriptive in nature (Cooper and Gutiérrez, 2013; Guerrero et al., 2004; Zhu et al., 2006).
Therefore, to provide new understanding on this critical issue, we perform simulation experiments on the dissolution in this study. A dissolution ratio is calculated and the morphology of the rock is compared before and after the dissolution reaction. The simulated geological conditions include a range of temperature–pressure conditions, intended to represent different stages of burial diagenesis (30°C and 0.3 MPa, epidiagenetic stage, around 0 km; 60°C and 13 MPa, early diagenetic stage, around 1.2 km; 100°C and 27 MPa, middle diagenetic stage, around 2.5 km; 150°C and 43 MPa, late diagenetic stage, around 4.2 km), and different types of dissolution fluid (pure water representative of continental formation water, seawater, 0.3 wt.%CO2 solution representative of meteoric water, and 0.2 wt.% acetic acid solution representative of organic fluid). In addition to gypsum rock, we also carried out the experiments on limestones and dolomites, which also occur in saline water sedimentary systems. This aims to compare the dissolution characteristic of gypsum rocks and carbonate rocks in paralleling experiments. The experiments allow us to explore the characteristics and mechanisms of dissolution more thoroughly.
Samples and methods
Gypsum rocks were collected from the Middle Triassic Zhouchongcun Formation (T2z) in the Hanshan–Hexian area, Anhui Province, China, which is the largest gypsum mining area in China (Xie et al., 2005). Limestones and dolomites were collected from the Lower Triassic Qinglong Formation (T1q) in the adjacent Guichi–Susong area.
The dissolution simulation experiments were performed using the reservoir dissolution simulation experiment instrument, developed by the Wuxi Research Branch of the Petroleum Exploration and Production Research Institute, SINOPEC (Figure 1; Fan et al., 2011). Briefly, the instrument controls the experiment temperature through a tube furnace. A well-plate hosting six wells allows sample tubes to be connected to the instrument. The discharge of each sample tube is controlled by micro-pore and control valves at the bottom of the tubes. The tube furnace heats the autoclave from the outside, with a maximum temperature of 200°C. All six sample tubes were maintained within the same pressure system, controlled by adjusting the inlet velocity.
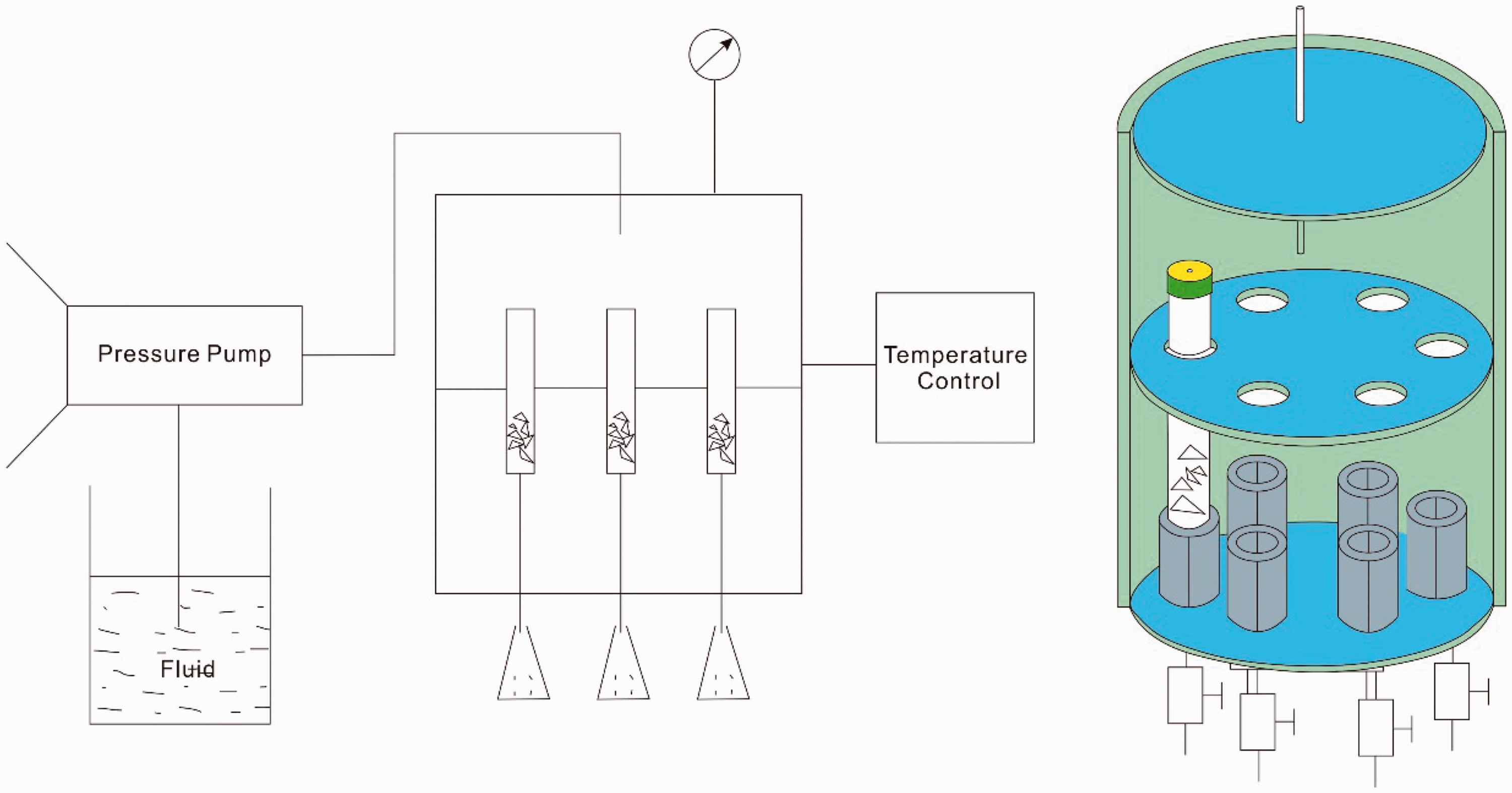
Schematic diagram of the instrument used for simulation experiments of dissolution in this study (modified from Fan et al., 2011).
During the experiments, 10 g samples of gypsum rock, limestone and dolomite particles were first introduced to the respective sample tubes, and the tubes were placed in a reaction kettle. The particle diameter is approximately 5 mm. Samples were then exposed to various fluid types under different temperature–pressure conditions designed in advance. The flow velocity was 2 mL/min. After the experiments, the residual samples in the tubes were removed for weighing and the dissolution ratio at the reaction equilibrium was calculated. The residual samples were then observed under a scanning election microscope (SEM) to interpret the dissolution morphology.
Two kinds of SEM were used to study the residual samples: a tungsten filament scanning electron microscopy and a field emission scanning electron microscopy (FESEM). The FESEM enabled greater magnification than tungsten filament scanning electron microscopy and therefore provided a clearer view of the dissolution morphology. The details of the tungsten filament SEM were as follows: model JSM-6490; 5 × to 300,000 × magnification; secondary electron detector with 3.0 nm resolution, backscattered electron detector with 4.0 nm resolution; acceleration voltage of 300 V to 30 kV; and Oxford INCA energy 350 EDS. The details of the field emission SEM were as follows: Carl Zeiss Supra 55 model; 12 × to 1,000,000× magnification; 15 kV at 0.8 nm, 1 kV for 1.6 nm and 30 kV for 2 nm resolution; acceleration voltage: 0.02–30 kV; Oxford Aztec X-Max 150 EDS.
Results
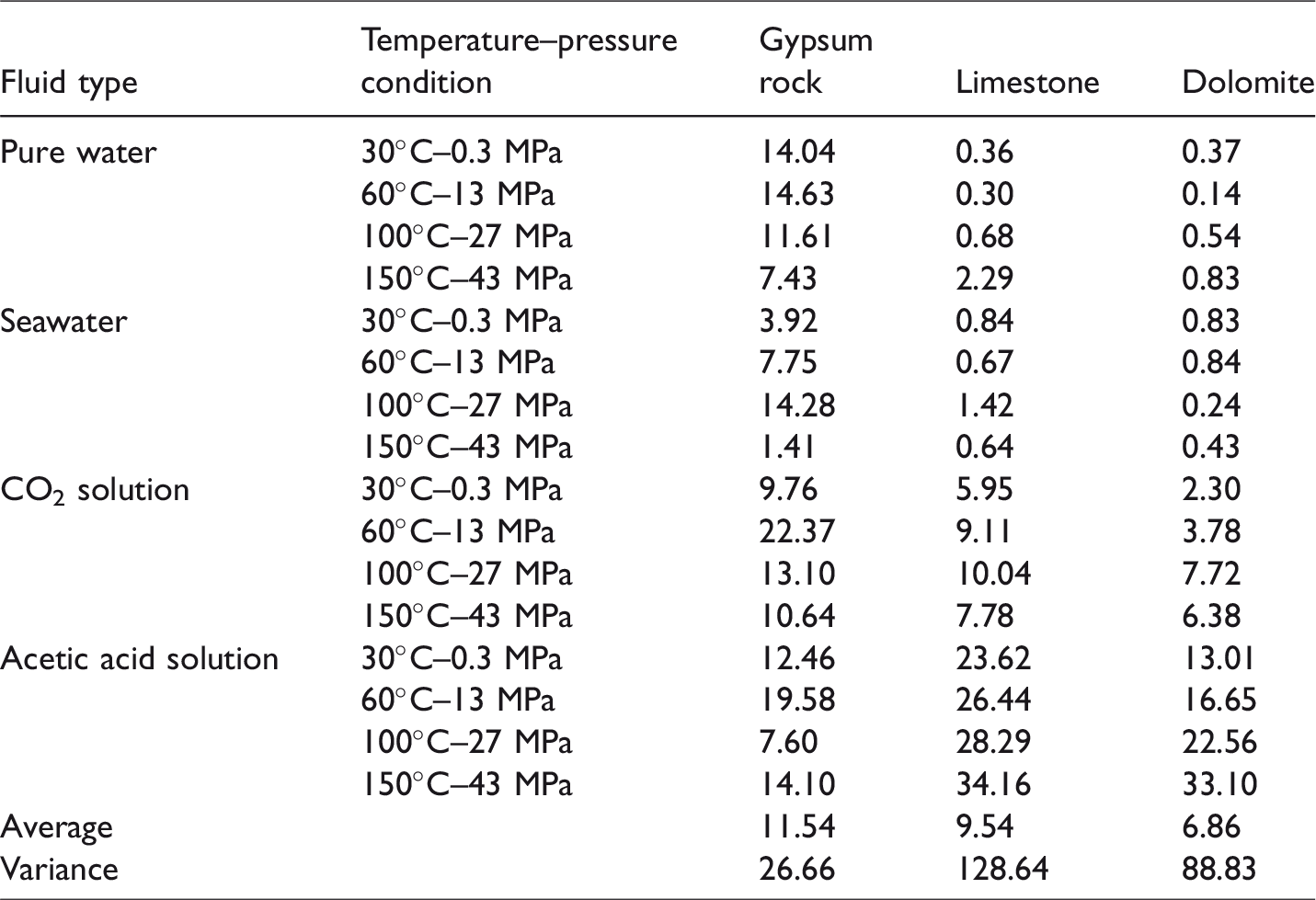
The experiments used in the study, as outlined above, include three kinds of lithology, four kinds of fluid types and four temperature–pressure conditions, which result in a total of 48 experiments. The results of the experiments are listed in Table 1.
Results of dissolution experiments for gypsum, limestone and dolomite (dissolution ratio, %).
Control of lithology on dissolution
Different lithology have different dissolution characteristics. As shown in Table 1, the dissolution ratio of gypsum rock is 1.41–22.37%, with an average of 11.54% and a variance of 26.66. The dissolution ratio of the limestone is 0.30–34.16%, with an average of 9.54% and a variance of 128.64. The dissolution ratio of the dolomite is 0.14%–33.10%, with an average of 6.86% and a variance of 88.83. Thus, there is little difference between the average dissolution ratios of the three lithology, but their variances vary widely. This, therefore, indicates that the characteristic dissolution of the three lithology differs with the external conditions. The smaller variance of gypsum rocks suggests that the external conditions have little influence on the dissolution of gypsum, and its dissolution ratio remains within a relatively narrow range. In contrast, the variance of limestone and dolomites is several times higher, indicating that external conditions have a greater influence on the dissolution of these rock types. The specific conditions affecting the dissolution of the three lithology are discussed later.
Control of fluid type on dissolution
Fluid type can be an important factor affecting dissolution. As shown in Table 1 and Figure 2, the dissolution ratio of gypsum rock in pure water ranges from 7.43% to 14.63%, which is approximately dozens of times higher than the dissolution ratios of limestone and dolomite (0.14–2.29%). In seawater, the dissolution ratio of gypsum rock is 1.41–14.2%, which is also approximately dozens of times higher than the dissolution ratios of limestone and dolomite (0.24–1.42%). In CO2 solution, the dissolution ratio of gypsum rock is 9.76–22.37%, which is only one to two times higher than the dissolution ratios of limestone and dolomite (2.30–10.04%). However, in acetic acid solution the dissolution ratios of limestone (23.62–34.16%) and dolomite (13.01–33.01%) are about two times higher than that of gypsum rock (7.60–19.58%).
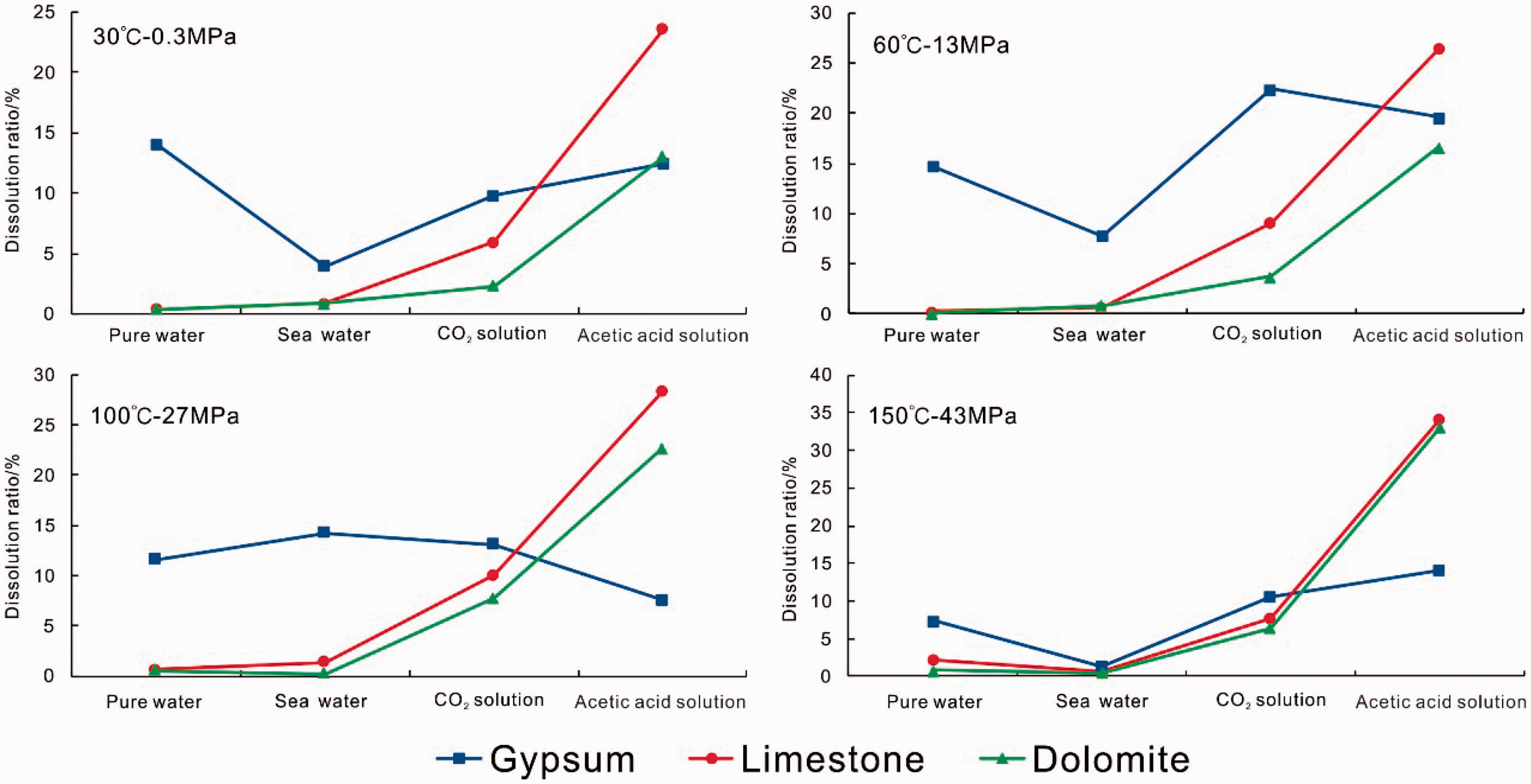
Dissolution ratios of the three lithology (gypsum, limestone and dolomite) under the same temperature and pressure conditions, but with exposure to different types of fluids.
Figure 2 indicates that fluid type has relatively less effect on the dissolution of gypsum rock than that of limestone and dolomite. Overall, compared with the pronounced changes of the dissolution rate of carbonate rocks in different fluids, the change of the gypsum rock is relatively smooth, e.g. the changes in 30°C and 0.3 MPa are 3.92–14.04% and 0.36–23.62% for gypsum rocks and carbonate rocks, respectively. Furthermore, the influence of fluid type on dissolution ratio cannot be fully neglected, especially in seawater. For example, the dissolution rate is about 15% in pure water, but 5% in seawater (Figure 2(a)).
Gypsum rock is readily soluble in all of the fluid types and its dissolution ratios are generally greater than 7%. This indicates that gypsum rock will readily dissolve in the presence of any unsaturated fluid. In contrast, fluid type has a greater influence on limestone and dolomite dissolution ratios. The degree of dissolution gradually increases from non-acid fluid (pure water and seawater) to weak acid fluid (CO2 solution, pH = 5.6) to strong acid fluid (acetic acid solution, pH = 2.4). Limestone and dolomite can only be dissolved under acidic conditions and the dissolution is more pronounced with increasing acid strength. The dissolution of limestone and dolomite is therefore reasonably high in meteoric water and organic fluid. The dissolution mechanism is discussed in detail later (see ‘Discussion’ section).
Fluid components are another important factor affecting dissolution, especially in seawater. For example, the dissolution rate is about 15% in pure water, but 5% in seawater (Figure 2(a)). The seawater used in this study is collected from Qingdao, East China Sea, which has been widely used in previously similar experiments (Fan et al., 2007, 2011). Main components of the seawater include SO42−, Ca2+ and Mg2+, with the abundance being 2370.31, 387.01 and 1136.13 mg/L, respectively. These components would have an effect on the dissolution, but has little effect on the judgment of qualitative results. The aim of this study is to compare the dissolution ratio of gypsum rock and carbonate rock in paralleling experiments, and thus the seawater used in gypsum rock and carbonate rock is same. As a result, the components of seawater have little effect on the results of this study. The influence of fluid components on dissolution will be discussed in other separated and focused articles.
Control of temperature and pressure on dissolution
The temperature–pressure conditions are another important factors affecting dissolution. As shown in Figure 3, the dissolution ratio of gypsum rock typically shows a rise followed by a fall for all four kinds of fluid under increasing temperature and pressure. Gypsum rock has dissolution peaks of 14.63% at 60°C and 13 MPa (in pure water), of 14.28% at 100°C and 27 MPa (in seawater), of 22.37% at 60°C and 13 MPa (in CO2 solution), and of 19.58% at 60°C and 13 MPa (in acetic acid solution). The dissolution morphology reflects the progress of dissolution (Figure 4). Under the temperature–pressure conditions at the dissolution peak, after hydration and dissolution, anhydrite shows irregular dissolution pores or ditches on its surface. The pores or ditches can be deep, but the overall morphology of the mineral still remains close to the original shape. The stair-step shape of gypsum remains visible after dissolution.
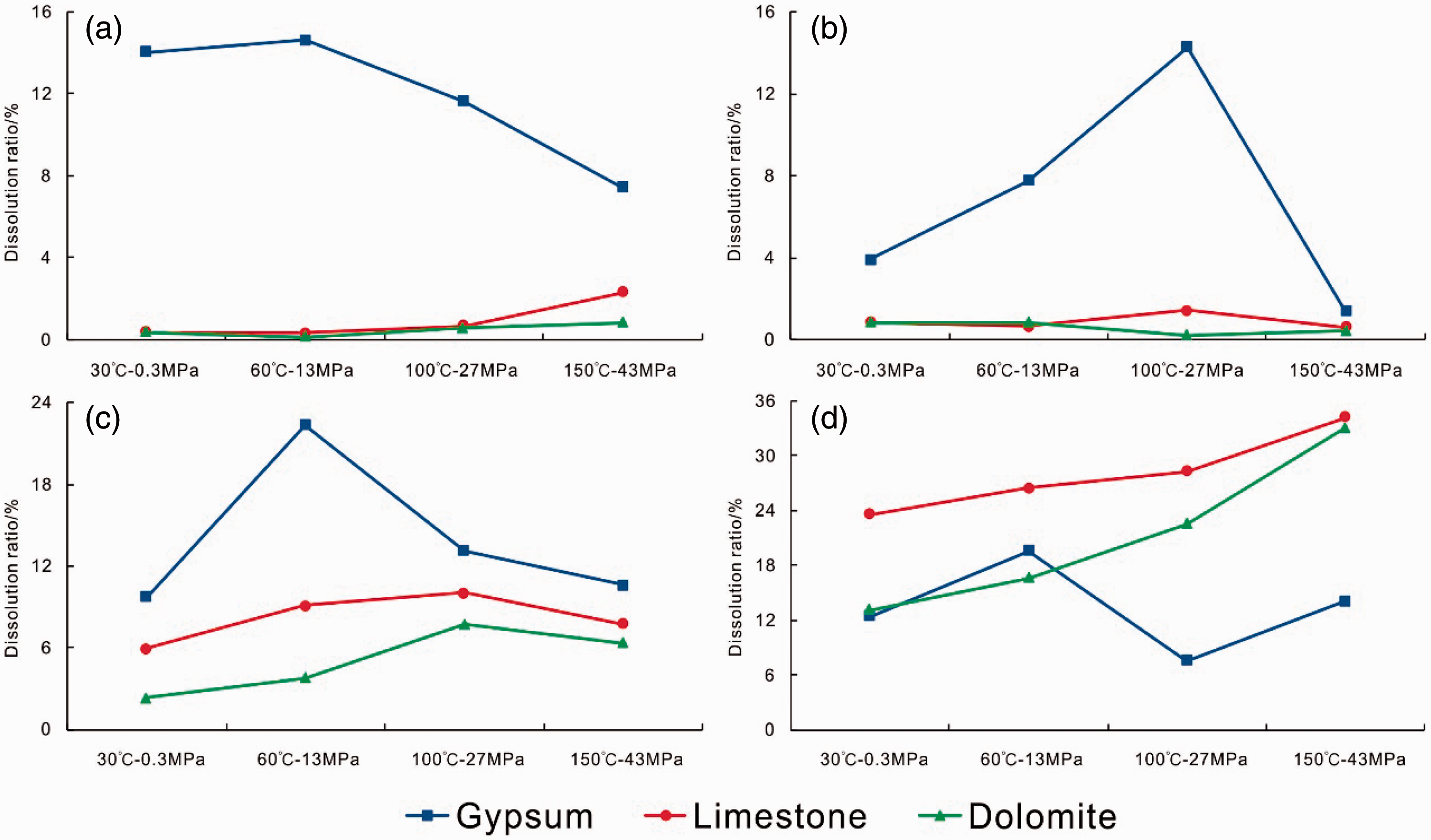
Dissolution ratios of the three lithology in the same fluid over different temperature and pressure conditions. (a) Pure water. (b) Seawater. (c) CO2 solution. (d) Acetic acid solution.

SEM images of gypsum rock at the dissolution peak. (a) Pure water, 60°C and 13 MPa. (b) Seawater, 100°C and 27 MPa. (c) CO2 solution, 60°C and 13 MPa. (d) Acetic acid solution, 60°C and 13 MPa.
The limestone and dolomite behave differently to the gypsum rock, being almost insoluble in pure water and seawater. In CO2 solution, their dissolution ratios first rise and then fall, with a dissolution peak at 100°C and 27 MPa (10.04% for limestone; 7.72% for dolomite). However, there is little difference between the dissolution ratios of limestone at 60°C and 13 MPa (9.11%) and at 100°C and 27 MPa (10.04%). In contrast, the difference between the dissolution ratios of dolomite under these conditions is relatively large (3.78% at 60°C and 13 MPa; 7.72% at 100°C and 27 MPa). Therefore, it can be deduced that the temperature and pressure at the dissolution peak for dolomite are higher than those for limestone: the former is ∼60°C and 13 MPa, while the latter is 100°C and 27 MPa. In acetic acid solution, both limestone and dolomite fail to reach the dissolution peak at the pre-set temperature–pressure conditions. Nevertheless, the rising slope of the dissolution ratio for limestone has a shallower gradient than the slope for dolomite. This suggests that the rate of dissolution of limestone is limited but the dissolution rate of dolomite rises with increasing temperature and pressure.
In summary, external conditions have little influence on the dissolution of gypsum rock, which shows dissolution peaks that increase with temperature and pressure; this is roughly between the early and middle diagenetic stages. In contrast, external conditions have a much stronger influence on the dissolution of limestone and dolomite. Limestone and dolomite are almost insoluble in pure water and seawater, but show a clear dissolution trend in CO2-enriched and acetic acid solutions. Furthermore, the dissolution rate of limestone declines with increasing temperature and pressure, whereas the dissolution rate of dolomite increases.
Discussion
The above results show that lithology, fluid type and temperature–pressure conditions can affect dissolution, and the dissolution rule depends on the conditions. Anhydrite can turn into gypsum after hydration, and then gypsum may dissolve to release Ca2+ and SO42− (see equation (1)). The chemical characteristics of gypsum rock make it soluble in various fluid types. Gypsum rock continues to dissolve in the presence of an unsaturated aqueous solution fluid, regardless of fluid type, making the dissolution of gypsum rock a common occurrence. In comparison to gypsum rock, the dissolution of limestone and dolomite is restricted to certain environmental conditions with specific fluid types (see equation (2)–(7) below); consequently, these rocks possess more complicated dissolution characteristics (Figures 2 and 3)


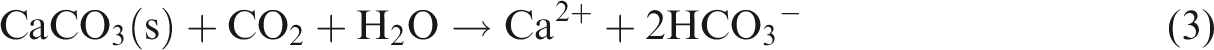
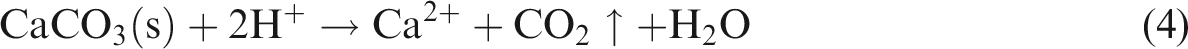
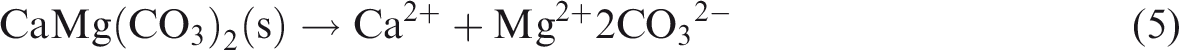

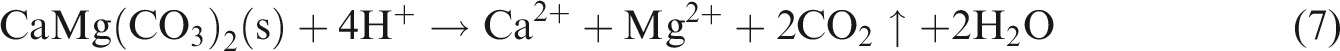
The dissolution peaks for gypsum rock reflect its solubility. In theory, the solubility of anhydrite and gypsum first rises and then falls with increasing temperature, reaching a maximum at 40°C (Table 2). However, these results differ slightly from our experimental results. We find that the majority of the dissolution ratios of gypsum rock reach a peak at 60°C in our experiments. In seawater, the temperature and pressure at the dissolution peak are higher, and the temperature reaches 100°C. This may be due to the Ca2+ in seawater altering the behaviour of gypsum with pressure, ultimately inhibiting the dissolution of gypsum and shifting the dissolution peak. Thus far, this research mainly focuses on the idea that pressure-driven dissolution is a function of changes in gas content. High pressure is beneficial for the dissolution of gas in solution and reduces the system volume: an increase in pressure increases the intermolecular forces of water, which changes the properties of water to some extent, and thereby affects the solubility of gypsum rock. However, few studies have considered how pressure influences the dissolution of solids (Zheng et al., 2009). Here, we find that a pressure of 13 MPa (130 times higher than barometric pressure) and a temperature of 60°C are conducive to gypsum dissolution.
Solubilities of gypsum rock, limestone and dolomite at different temperatures (g/100 g H2O).
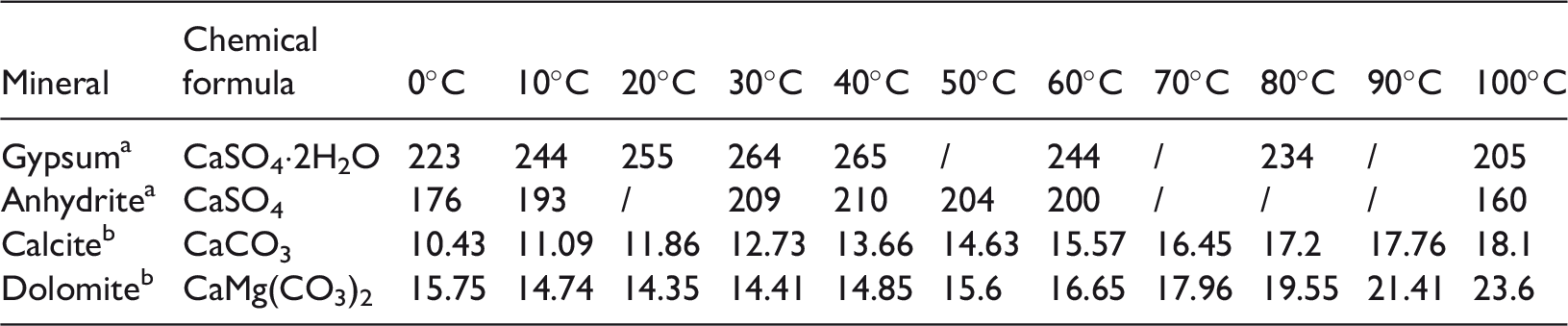
The solubilities of gypsum and anhydrite are from Liu et al. (2002).
The solubilities of calcite and dolomite are from Yan et al. (2009).
Our results indicate that temperature has important effects on dissolution (Table 2). The Gibbs free energy of the dissolution of gypsum rock with changes in temperature can be calculated by the formula provided by Liu et al. (2002) (equation (8)). As shown in Figure 5, the Gibbs free energy for the dissolution of gypsum rock increases with increasing temperature, thereby making the rock more resistant to dissolution. This indicates that gypsum rock is more stable at greater burial depths, which has implications for petroleum and minerals exploration
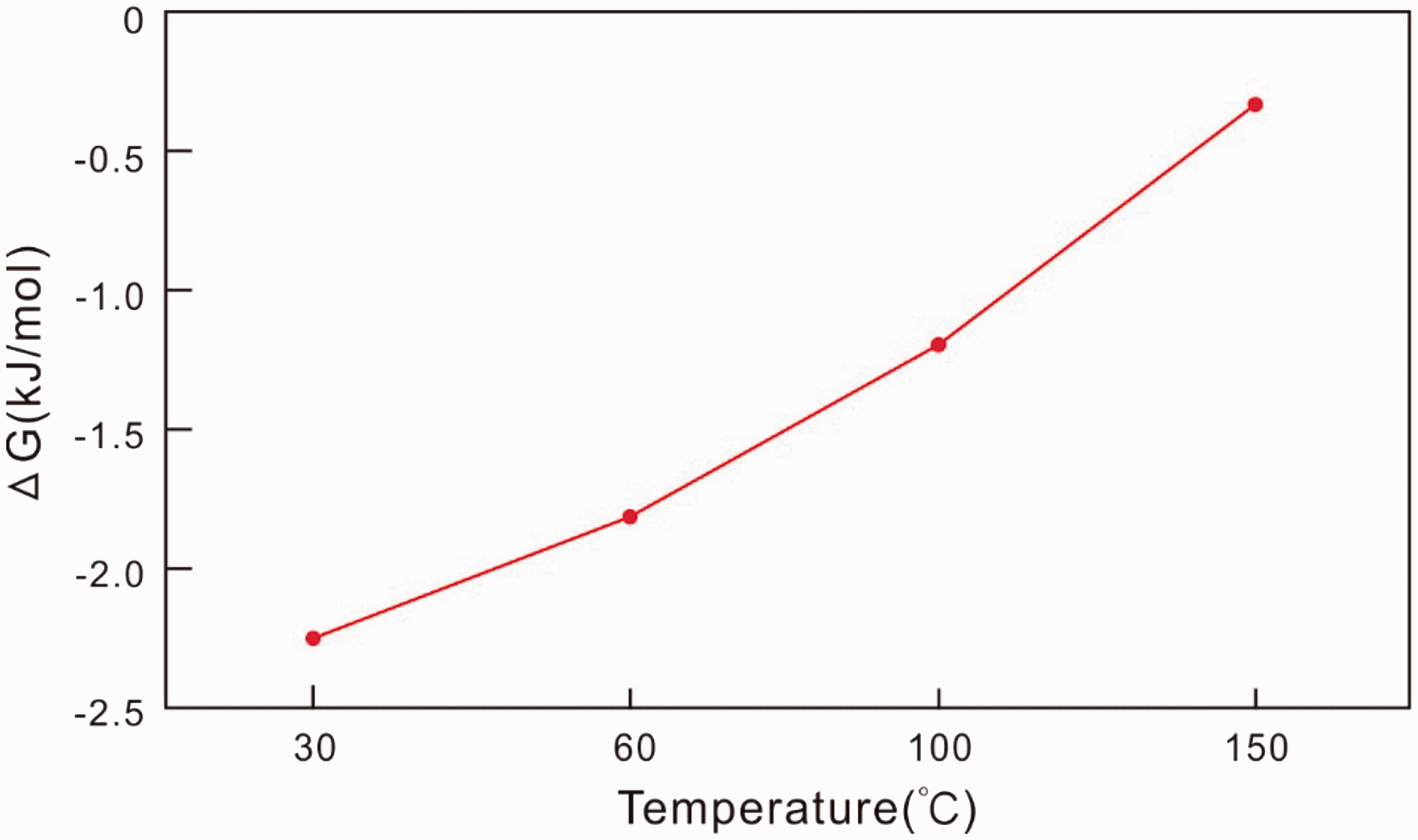
Gibbs free energy for the dissolution of gypsum rock.
For comparison with gypsum rock, the solubilities of limestone and dolomite are also listed in Table 2. The solubility of gypsum rock in 100 g water is greater than 200 mg at room temperature, while the solubility of limestone and dolomite is just 12 mg. Consequently, the dissolution of limestone and dolomite depends on CO2 and acid strength (equations (2)–(7)). During diagenesis, the two main sources of CO2 and acid are: (1) the leaching of meteoric water, which generally occurs in shallow areas unless there are deep fractures; and (2) hydrocarbon generation, which can produce CO2 and organic acid.
The dissolution rate of limestone declines and the dissolution rate of dolomite increases with increasing temperature and pressure (Fan et al., 2011; Pokrovsky et al., 2005, 2009; Wang et al., 2016, 2017; Yan et al., 2009; Yang et al., 1995a, 1995b). Yang et al. (1995b) carried out a dissolution simulation experiment using limestone (dolomite/calcite = 1/1) and dolomite (dolomite/calcite = 3.4/1). In their study, when the temperature and pressure were 75°C and 20 MPa, and 100°C and 25 MPa for the limestone and dolomite, respectively, the dissolution rates for limestone (dolomite/calcite = 1/1) increased by 0.0011 mg/L/h and the dissolution rate of dolomite (dolomite/calcite = 3.4/1) increased by 0.0033 mg/L/h, with every 1°C rise in temperature. Consequently, the effect of temperature and pressure on dolomite dissolution is several times greater than the effect on limestone dissolution. This suggests that the dissolution of dolomite occurs more readily with increasing burial depth. Therefore, it can be deduced that secondary porosity is more important in dolomite than in limestone for driving dissolution at depth. This result has implications for the formation of high-quality carbonate reservoirs at deep levels.
Conclusions and implications
External conditions have little influence on the dissolution of gypsum rock, which shows dissolution peaks that increase with temperature and pressure, roughly between the early and middle stages of diagenesis. In contrast, limestone and dolomite have different characteristics and external conditions have a much greater influence on their behaviour in the presence of fluids. Although they are almost insoluble in pure water and seawater, limestone and dolomite demonstrate a clear dissolution trend in CO2 and acetic acid solutions. Furthermore, with increases in temperature and pressure, the dissolution rate of limestone declines whereas that of dolomite increases.
The dissolution characteristics depend upon the physicochemical properties of rock. The solubility of gypsum rock is relatively high, so it readily dissolves under a range of external conditions, provided there is unsaturated fluid flowing through the rock. However, the solubility of limestone and dolomite is lower, so these rocks are almost insoluble in non-acidic fluid, and their dissolution relies on CO2 and acid being present, which are typically derived from leaching of meteoric water or from hydrocarbon generation and deep fluids.
The Gibbs free energy for the dissolution of gypsum rock increases with increasing temperature; this inhibits dissolution and means that gypsum rock is more readily preserved with increasing burial depth. This finding is significant for exploration for petroleum and minerals at depth. The dissolution rate of limestone declines and the dissolution rate of dolomite increases with increasing temperature and pressure. This observation suggests that the dissolution of dolomite occurs more readily with increasing burial depth. Thus, it may be deduced that secondary porosity is the key factor contributing to the dissolution of dolomite at depth, compared with limestone dissolution. This is significant for understanding the formation of high-quality carbonate reservoirs at deep levels.
In future studies, we suggest a need to link theory with practice, and combine simulation experiments with geological field studies to further our understanding of the factors influencing the dissolution of gypsum rock.
Footnotes
Acknowledgements
We thank Editor-in-Chief Prof. Yuzhuang Sun and anonymous reviewers for their constructive comments, which help to improve the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: jointly funded by the National Science and Technology Major Project of China (Grant No. 2016ZX05002-006-005), and National Natural Science Foundation of China (Grant Nos. 41322017 and 41472100).
